Catalytic Asymmetric 1,4-Additions of β-Keto Esters to Nitroalkenes Promoted by a Bifunctional Homobimetallic Co2-Schiff Base Complex
Abstract
:1. Introduction
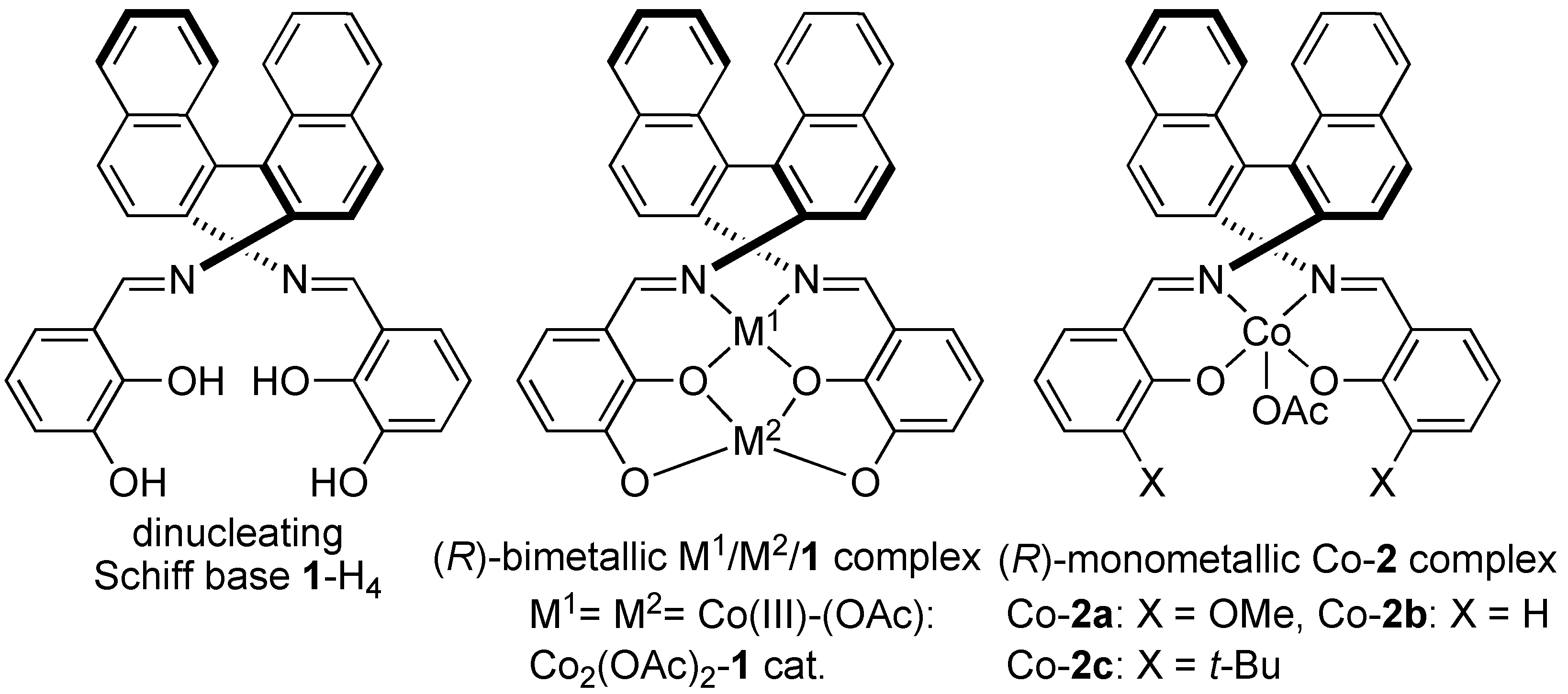
2. Results and Discussion
2.1. Homobimetallic Co2-Schiff Base Complex-catalyzed Asymmetric 1,4-Addition to Nitroalkenes
 | ||||||||
|---|---|---|---|---|---|---|---|---|
| Entry | M 1 | M 2 | cat. (mol %) | time (h) | solvent (M) | Dr b | % yield b | % ee |
| 1 | Cu | Sm-OiPr | 10 | 36 | THF (0.4) | 3:1 | >99 | 23 |
| 2 | Pd | La-OiPr | 10 | 36 | THF (0.4) | 4:1 | >99 | 14 c |
| 3 | Ni | Ni | 10 | 36 | THF (0.4) | 6:1 | 71 | 74 |
| 4 | Co-OAc | Co-OAc | 10 | 6 | THF (0.4) | 26:1 | 86 | 95 |
| 5 | Mn-OAc | Mn-OAc | 10 | 36 | THF (0.4) | 3:1 | 61 | 32 |
| 6 | Cu | Cu | 10 | 36 | THF (0.4) | 9:1 | 6 | 12 c |
| 7 | Zn | Zn | 10 | 36 | THF (0.4) | 2:1 | 90 | 8 |
| 8 d | Co-OAc | Co-OAc | 2.5 | 9 | THF (2.0) | 18:1 | 94 | 93 |
| 9 e | Co-OAc | Co-OAc | 2.5 | 8 | neat | 25:1 | >99 | 97 |
 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Entry | R 1 | 3 | 4 | cat. (x mol %) | time (h) | solvent (y M) | 5 | Dr b | % yieldc | % ee |
| 1 | Ph | 3a | 4a | 2.5 | 8 | neat | 5aa | 25:1 | >99 | 97 |
| 2 | 4-Cl-C6H4 | 3b | 4a | 2.5 | 4 | neat | 5ba | >30:1 | 94 | 98 |
| 3 | 4-Br-C6H4 | 3c | 4a | 2.5 | 5 | neat | 5ca | >30:1 | 95 | 98 |
| 4 | 3-Br-C6H4 | 3d | 4a | 2.5 | 4 | neat | 5da | 30:1 | 88 | 97 |
| 5 | 2-Br-C6H4 | 3e | 4a | 2.5 | 14 | neat | 5ea | 27:1 | 77 | 94 |
| 6 | 4-MeO-C6H4 | 3f | 4a | 2.5 | 17 | neat | 5fa | 9:1 | 93 | 94 |
| 7 | 4-Me-C6H4 | 3g | 4a | 2.5 | 10 | neat | 5ga | 22:1 | 93 | 96 |
| 8 | 2-furyl | 3h | 4a | 2.5 | 3 | neat | 5ha | >30:1 | 93 | 92 |
| 9 | PhCH2CH2 | 3i | 4a | 2.5 | 7 | neat | 5ia | >30:1 | 96 | 95 |
| 10 | 4-Cl-C6H4 | 3b | 4b | 2.5 | 24 | neat | 5bb | >30:1 | 75 | 98 |
| 11d | Ph | 3a | 4c | 10 | 36 | neat | 5ac | 3.3:1 | 73 | 96 |
| 12 | 4-Cl-C6H4 | 3b | 4a | 0.2 | 24 | THF (20) | 5ba | >30:1 | 87e | 99 |
| 13 | 4-Cl-C6H4 | 3b | 4a | 0.1 | 48 | THF (20) | 5ba | 16:1 | 98 | 95 |
2.2. Mechanistic Studies of Homobimetallic Co2(OAc)2-Schiff Base Complex
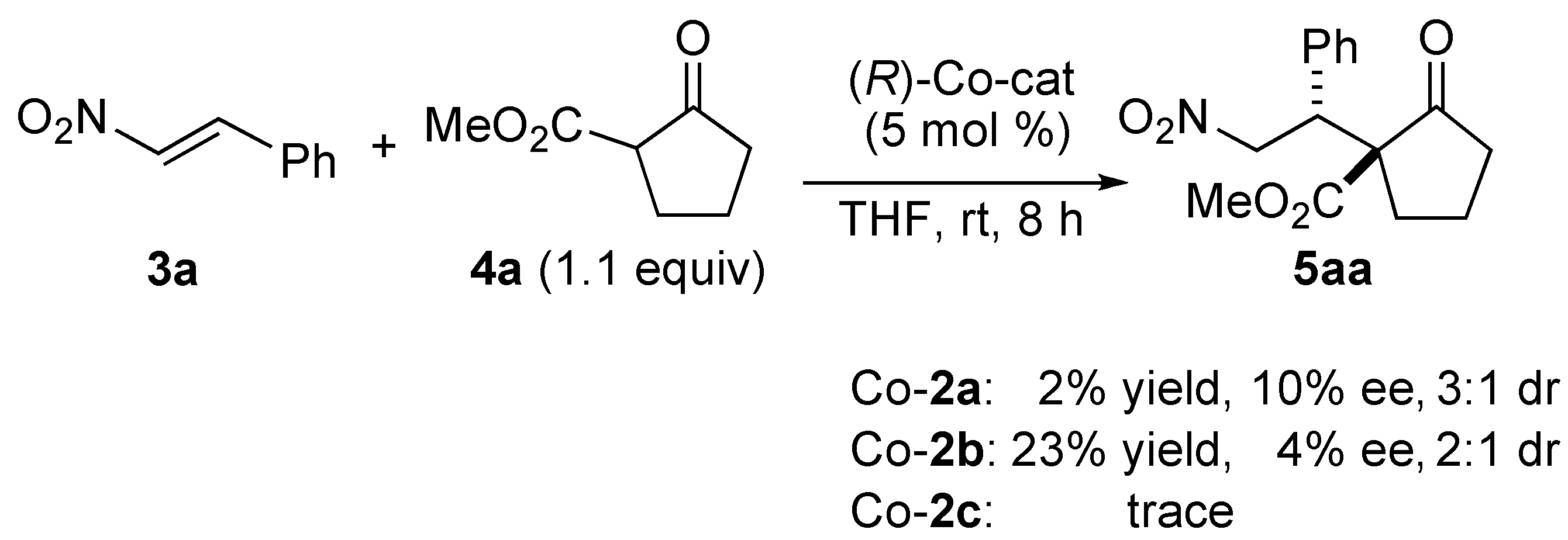
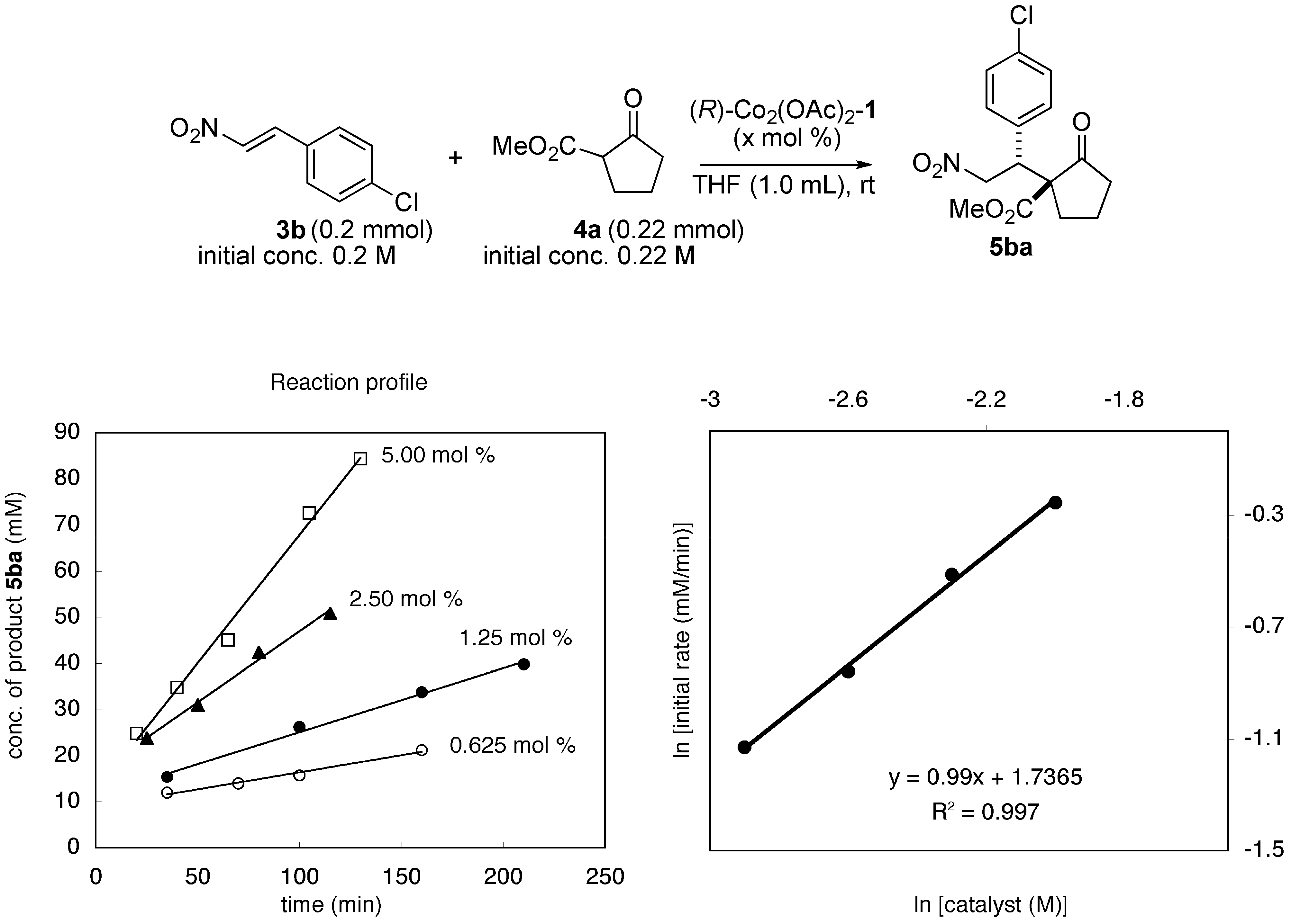

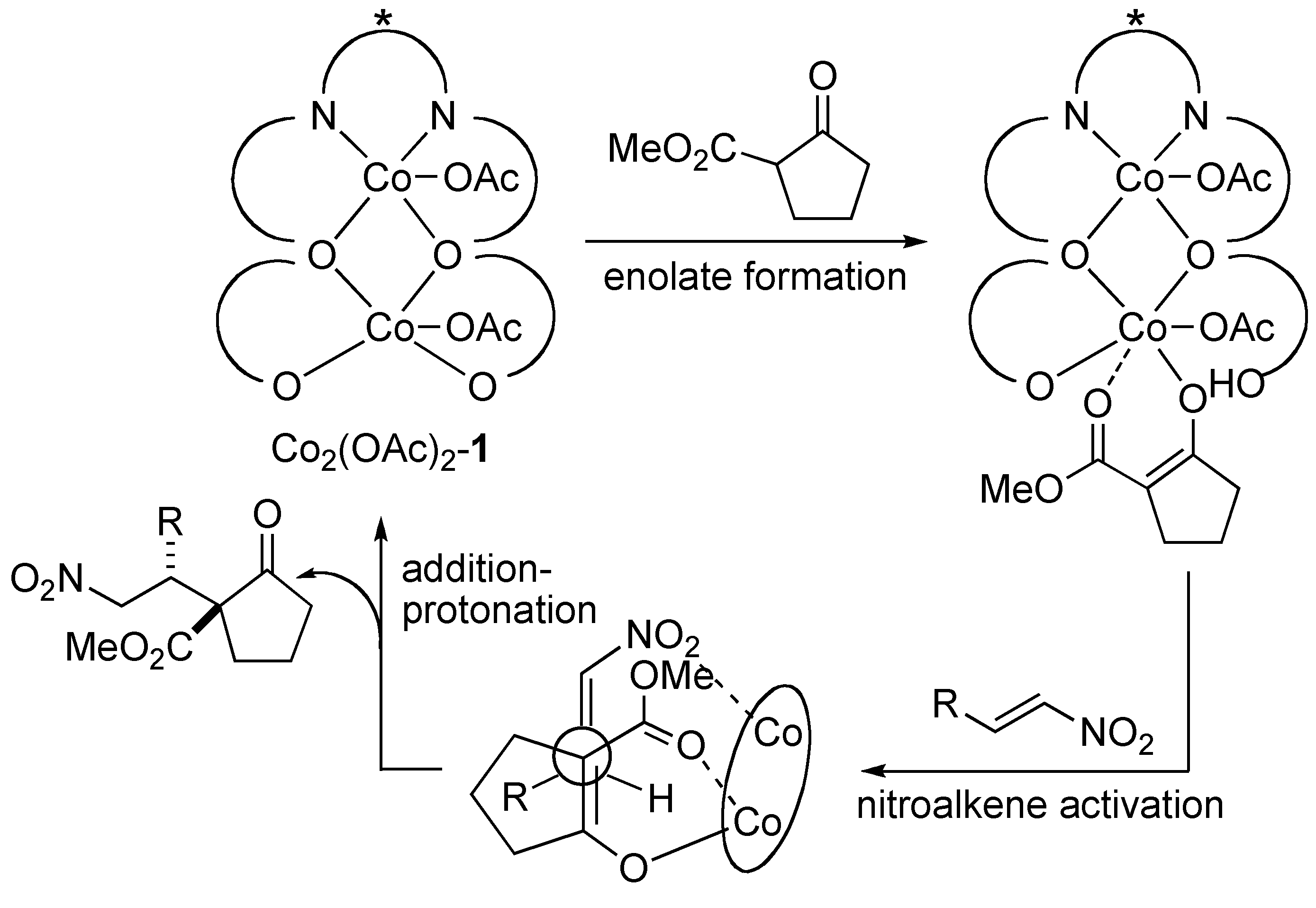
3. Experimental
3.1. General
3.2. Preparation of Co(III)2(OAc)2-Schiff Base 1 Complex
3.3. General Procedure for Catalytic Asymmetric 1,4-Additions of β-Keto Esters to Nitroalkenes under Solvent-Free Conditions
4. Conclusions
Acknowledgements
- Sample Availability: Samples of the compounds Co2(OAc)2-1 and Ni2-1 are available from the authors.
References and Notes
- Shibasaki, M.; Kanai, M.; Matsunaga, S.; Kumagai, N. Recent progress in asymmetric bifunctional catalysis using multimetallic systems. Acc. Chem. Res. 2009, 42, 1117–1127. [Google Scholar] [CrossRef]
- Matsunaga, S.; Shibasaki, M. Multimetallic bifunctional asymmetric catalysis based on proximity effect control. Bull. Chem. Soc. Jpn. 2008, 81, 60–75. [Google Scholar] [CrossRef]
- Mukherjee, S.; Yang, J.W.; Hoffmann, S.; List, B. Asymmetric enamine catalysis. Chem. Rev. 2007, 107, 5471–5569. [Google Scholar] [CrossRef]
- Taylor, M.S.; Jacobsen, E.N. Asymmetric catalysis by chiral hydrogen-bond donors. Angew. Chem., Int. Ed. 2006, 45, 1520–1543. [Google Scholar] [CrossRef]
- Yamamoto, H.; Futatsugi, K. “Designer acids”: combined acid catalysis for asymmetric synthesis. Angew. Chem., Int. Ed. 2005, 44, 1924–1942. [Google Scholar] [CrossRef]
- Katsuki, T. Unique asymmetric catalysis of cis-β metal complexes of salen and its related Schiff-base ligands. Chem. Soc. Rev. 2004, 33, 437–444. [Google Scholar]
- Cozzi, P.G. Metal-salen Schiff base complexes in catalysis: practical aspects. Chem. Soc. Rev. 2004, 33, 410–421. [Google Scholar]
- Jacobsen, E.N. Asymmetric catalysis of epoxide ring-opening reactions. Acc. Chem. Res. 2000, 33, 421–431. [Google Scholar] [CrossRef]
- Handa, S.; Gnanadesikan, V.; Matsunaga, S.; Shibasaki, M. syn-Selective catalytic asymmetric nitro-Mannich reactions using a heterobimetallic Cu-Sm-Schiff base complex. J. Am. Chem. Soc. 2007, 129, 4900–4901. [Google Scholar] [CrossRef]
- Handa, S.; Nagawa, K.; Sohtome, Y.; Matsunaga, S.; Shibasaki, M. A heterobimetallic Pd/La/Schiff base complex for anti-selective catalytic asymmetric nitroaldol reactions and applications to short syntheses of β-adrenoceptor agonists. Angew. Chem., Int. Ed. 2008, 47, 3230–3233. [Google Scholar] [CrossRef]
- Sohtome, Y.; Kato, Y.; Handa, S.; Aoyama, N.; Nagawa, K.; Matsunaga, S.; Shibasaki, M. Stereodivergent catalytic doubly diastereoselective nitroaldol reactions using heterobimetallic complexes. Org. Lett. 2008, 10, 2231–2234. [Google Scholar] [CrossRef]
- Mihara, H.; Xu, Y.; Shepherd, N.E.; Matsunaga, S.; Shibasaki, M. A heterobimetallic Ga/Yb-Schiff base complex for catalytic asymmetric α-addition of isocyanides to aldehydes. J. Am. Chem. Soc. 2009, 131, 8384–8385. [Google Scholar]
- Chen, Z.; Morimoto, H.; Matsunaga, S.; Shibasaki, M. A bench-stable homodinuclear Ni2-Schiff base complex for catalytic asymmetric synthesis of α-tetrasubstituted anti-α,β-diamino acid surrogates. J. Am. Chem. Soc. 2008, 130, 2170–2171. [Google Scholar] [CrossRef]
- Chen, Z.; Yakura, K.; Matsunaga, S.; Shibasaki, M. Direct catalytic asymmetric Mannich-type reaction of β-keto phosphonate using a dinuclear Ni2-Schiff base complex. Org. Lett. 2008, 10, 3239–3242. [Google Scholar] [CrossRef]
- Xu, Y.; Lu, G.; Matsunaga, S.; Shibasaki, M. Direct anti-selective catalytic asymmetric Mannich-type reactions of α-ketoanilides for the synthesis of γ-amino amides and azetidine-2-amides. Angew. Chem., Int. Ed. 2009, 48, 3353–3356. [Google Scholar] [CrossRef]
- Mouri, S.; Chen, Z.; Matsunaga, S.; Shibasaki, M. Direct catalytic asymmetric aldol reaction of β-keto esters with formaldehyde promoted by a dinuclear Ni2-Schiff base complex. Chem. Commun. 2009, 5138–5140. [Google Scholar]
- Kato, Y.; Chen, Z.; Matsunaga, S.; Shibasaki, M. Catalytic asymmetric synthesis of nitrogen-containing gem-bisphosphonates using a dinuclear Ni2-Schiff base complex. Synlett 2009, 1635–1638. [Google Scholar]
- Mouri, S.; Chen, Z.; Mitsunuma, H.; Furutachi, M.; Matsunaga, S.; Shibasaki, M. Catalytic asymmetric synthesis of 3-aminooxindoles: enantiofacial selectivity switch in bimetallic vs. monometallic Schiff base catalysis. J. Am. Chem. Soc. 2010, 132. [Google Scholar] [CrossRef]
- Chen, Z.; Furutachi, M.; Kato, Y.; Matsunaga, S.; Shibasaki, M. A stable homodinuclear biscobalt(III)-Schiff base complex for catalytic asymmetric 1,4-additions of β-keto esters to alkynones. Angew. Chem. Int. Ed. 2009, 48, 2218–2220. [Google Scholar]
- Kato, Y.; Furutachi, M.; Chen, Z.; Mitsunuma, H.; Matsunaga, S.; Shibasaki, M. A homodinuclear Mn(III)2-Schiff base complex for catalytic asymmetric 1,4-additions of oxindoles to nitroalkenes. J. Am. Chem. Soc. 2009, 131, 9168–9169. [Google Scholar]
- Annamalai, V.; DiMauro, E.F.; Carroll, P.J.; Kozlowski, M.C. Catalysis of the Michael addition reaction by late transition metal complexes of BINOL-derived salens. J. Org. Chem. 2003, 68, 1973–1981. [Google Scholar] [CrossRef]
- Gao, J.; Woolley, F.R.; Zingaro, R.A. Catalytic asymmetric cyclopropanation at a chiral platform. Org. Biomol. Chem. 2005, 3, 2126–2128. [Google Scholar] [CrossRef]
- Yang, M.; Zhu, C.; Yuan, F.; Huang, Y.; Pan, Y. Enantioselective ring-opening reaction of meso-epoxides with ArSeH catalyzed by heterometallic Ti−Ga−salen system. Org. Lett. 2005, 7, 1927–1930. [Google Scholar] [CrossRef]
- Li, W.; Thakur, S.S.; Chen, S.-W.; Shin, C.-K.; Kawthekar, R.B.; Kim, G.-J. Synthesis of optically active 2-hydroxy monoesters via-kinetic resolution and asymmetric cyclization catalyzed by heterometallic chiral (salen) Co complex. Tetrahedron Lett. 2006, 47, 3453–3457. [Google Scholar]
- Mazet, C.; Jacobsen, E.N. Dinuclear {(salen)Al} complexes display expanded scope in the conjugate cyanation of α,β-unsaturated imides. Angew. Chem., Int. Ed. 2008, 47, 1762–1765. [Google Scholar] [CrossRef]
- Hirahata, W.; Thomas, R.M.; Lobkovsky, E.B.; Coates, G.W. Enantioselective polymerization of epoxides: a highly active and selective catalyst for the preparation of stereoregular polyethers and enantiopure epoxides. J. Am. Chem. Soc. 2008, 130, 17658–17659. [Google Scholar]
- Wu, B.; Gallucci, J.C.; Parquette, J.R.; RajanBabu, T.V. Enantioselective desymmetrization of meso-aziridines with TMSN3 or TMSCN catalyzed by discrete yttrium complexes. Angew. Chem., Int. Ed. 2009, 48, 1126–1129. [Google Scholar] [CrossRef]
- Berner, O.M.; Tedeschi, L.; Enders, D. Asymmetric Michael additions to nitroalkenes. Eur. J. Org. Chem. 2002, 1877–1894. [Google Scholar]
- Tsogoeva, S.B. Recent advances in asymmetric organocatalytic 1,4-conjugate additions. Eur. J. Org. Chem. 2007, 1701–1716. [Google Scholar] [CrossRef]
- Christoffers, J.; Koripelly, G.; Rosiak, A.; Rössle, M. Recent advances in metal-catalyzed asymmetric conjugate additions. Synthesis 2007, 1279–1300. [Google Scholar]
- Ji, J.; Barnes, D.M.; Zhang, J.; King, S.A.; Wittenberger, S.J.; Morton, H.E. Catalytic enantioselective conjugate addition of 1,3-dicarbonyl compounds to nitroalkenes. J. Am. Chem. Soc. 1999, 121, 10215–10216. [Google Scholar] [CrossRef]
- Watanabe, M.; Ikagawa, A.; Wang, H.; Murata, K.; Ikariya, T. Catalytic enantioselective Michael addition of 1,3-dicarbonyl compounds to nitroalkenes catalyzed by well-defined chiral Ru amido complexes. J. Am. Chem. Soc. 2004, 126, 11148–11149. [Google Scholar] [CrossRef]
- Evans, D.A.; Mito, S.; Seidel, D. Scope and mechanism of enantioselective Michael additions of 1,3-dicarbonyl compounds to nitroalkenes catalyzed by nickel(II)−diamine complexes. J. Am. Chem. Soc. 2007, 129, 11583–11592. [Google Scholar] [CrossRef]
- Li, H.; Wang, Y.; Tang, L.; Wu, F.; Liu, X.; Guo, C.; Foxman, B.M.; Deng, L. Stereocontrolled creation of adjacent quaternary and tertiary stereocenters by a catalytic conjugate addition. Angew. Chem. Int. Ed. 2005, 44, 105–108. [Google Scholar] [CrossRef]
- Okino, T.; Hoashi, Y.; Furukawa, T.; Xu, X.; Takemoto, Y. Enantio- and diastereoselective Michael reaction of 1,3-dicarbonyl compounds to nitroolefins catalyzed by a bifunctional thiourea. J. Am. Chem. Soc. 2005, 127, 119–125. [Google Scholar]
- Malerich, J.P.; Hagihara, K.; Rawal, V.H. Chiral squaramide derivatives are excellent hydrogen bond donor catalysts. J. Am. Chem. Soc. 2008, 130, 14416–14417. [Google Scholar] [CrossRef]
- Zhang, Z.-H.; Dong, X.-Q.; Chen, D.; Wang, C.J. Fine-tunable organocatalysts bearing multiple hydrogen-bonding donors for construction of adjacent quaternary and tertiary stereocenters via a Michael reaction. Chem. Eur. J. 2008, 14, 8780–8783. [Google Scholar]
- Luo, J.; Xu, L.W.; Hay, R.A.S.; Lu, Y. Asymmetric Michael addition mediated by novel Cinchona alkaloid-derived bifunctional catalysts containing sulfonamides. Org. Lett. 2009, 11, 437–440. [Google Scholar]
- Chen, F.-X.; Shao, C.; Liu, Q.; Gong, P.; Liu, C.-L.; Wang, R. Asymmetric Michael addition of trisubstituted carbanion to nitroalkenes catalyzed by sodium demethylquinine salt in water. Chirality 2009, 21, 600–603. [Google Scholar] [CrossRef]
- Jiang, X.; Zhang, Y.; Liu, X.; Zhang, G.; Lai, L.; Wu, L.; Zhang, J.; Wang, R. Enantio- and diastereoselective asymmetric addition of 1,3-dicarbonyl compounds to nitroalkenes in a doubly stereocontrolled manner catalyzed by bifunctional Rosin-derived amine thiourea catalysts. J. Org. Chem. 2009, 74, 5562–5567. [Google Scholar] [CrossRef]
- Yu, Z.; Liu, X.; Zhou, L.; Lin, L.; Feng, X. Bifunctional guanidine via an amino amide skeleton for asymmetric Michael reactions of β-ketoesters with nitroolefins: a concise synthesis of bicyclic β -amino acids. Angew. Chem., Int. Ed. 2009, 48, 5195–5198. [Google Scholar] [CrossRef]
- Almasi, D.; Alonso, D.A.; Gómez-Bengoa, E.; Nájera, C. Chiral 2-aminobenzimidazoles as recoverable organocatalysts for the addition of 1,3-dicarbonyl compounds to nitroalkenes. J. Org. Chem. 2009, 74, 6163–6168. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Furutachi, M.; Chen, Z.; Matsunaga, S.; Shibasaki, M. Catalytic Asymmetric 1,4-Additions of β-Keto Esters to Nitroalkenes Promoted by a Bifunctional Homobimetallic Co2-Schiff Base Complex. Molecules 2010, 15, 532-544. https://doi.org/10.3390/molecules15010532
Furutachi M, Chen Z, Matsunaga S, Shibasaki M. Catalytic Asymmetric 1,4-Additions of β-Keto Esters to Nitroalkenes Promoted by a Bifunctional Homobimetallic Co2-Schiff Base Complex. Molecules. 2010; 15(1):532-544. https://doi.org/10.3390/molecules15010532
Chicago/Turabian StyleFurutachi, Makoto, Zhihua Chen, Shigeki Matsunaga, and Masakatsu Shibasaki. 2010. "Catalytic Asymmetric 1,4-Additions of β-Keto Esters to Nitroalkenes Promoted by a Bifunctional Homobimetallic Co2-Schiff Base Complex" Molecules 15, no. 1: 532-544. https://doi.org/10.3390/molecules15010532
APA StyleFurutachi, M., Chen, Z., Matsunaga, S., & Shibasaki, M. (2010). Catalytic Asymmetric 1,4-Additions of β-Keto Esters to Nitroalkenes Promoted by a Bifunctional Homobimetallic Co2-Schiff Base Complex. Molecules, 15(1), 532-544. https://doi.org/10.3390/molecules15010532




