Examination of Imprinting Process with Molsidomine as a Template
Abstract
:Introduction
Results and Discussion
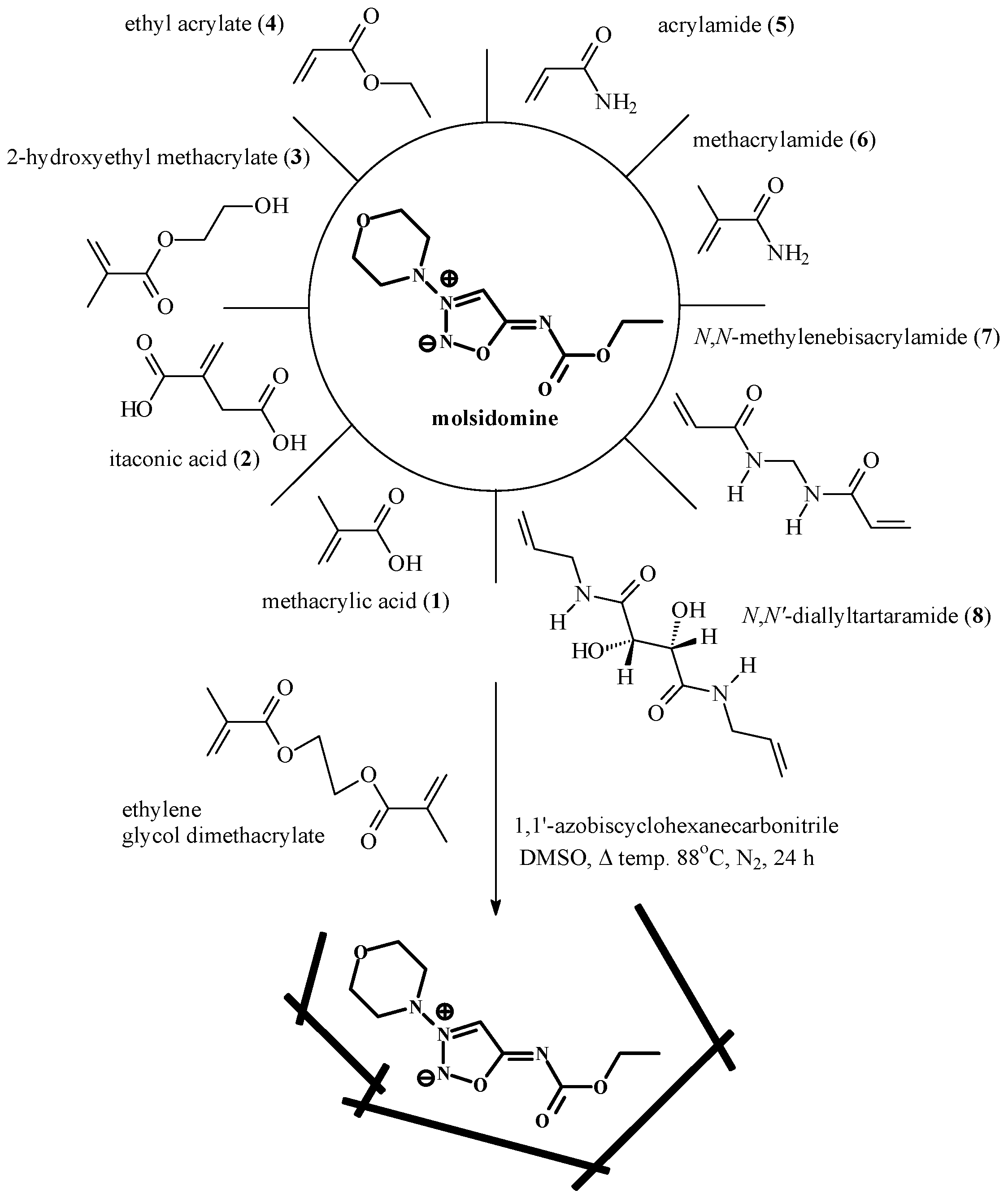
Choice of functional monomer
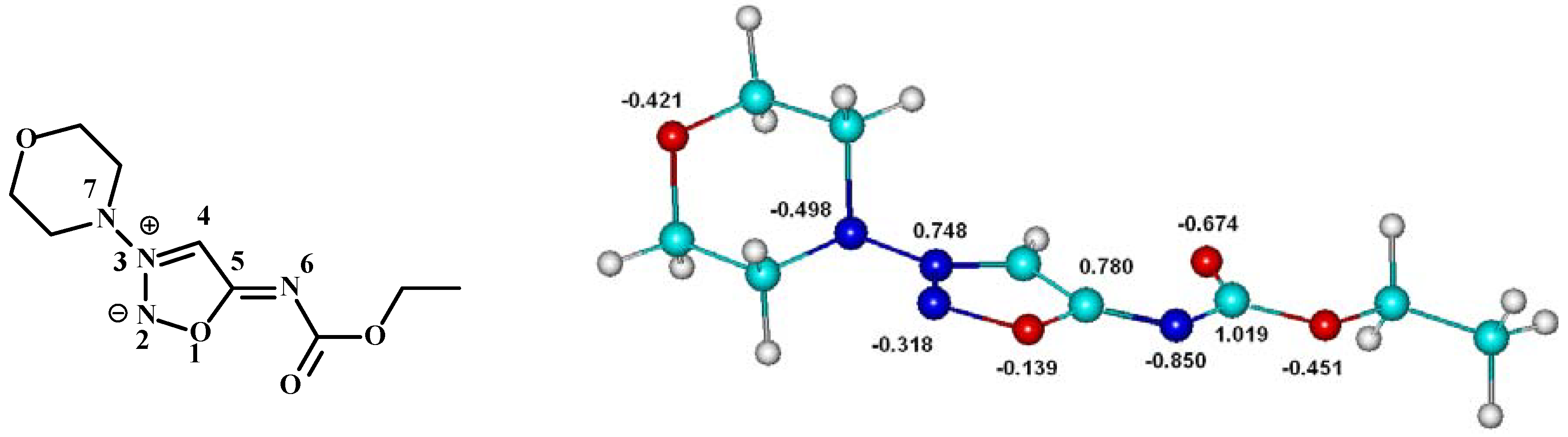
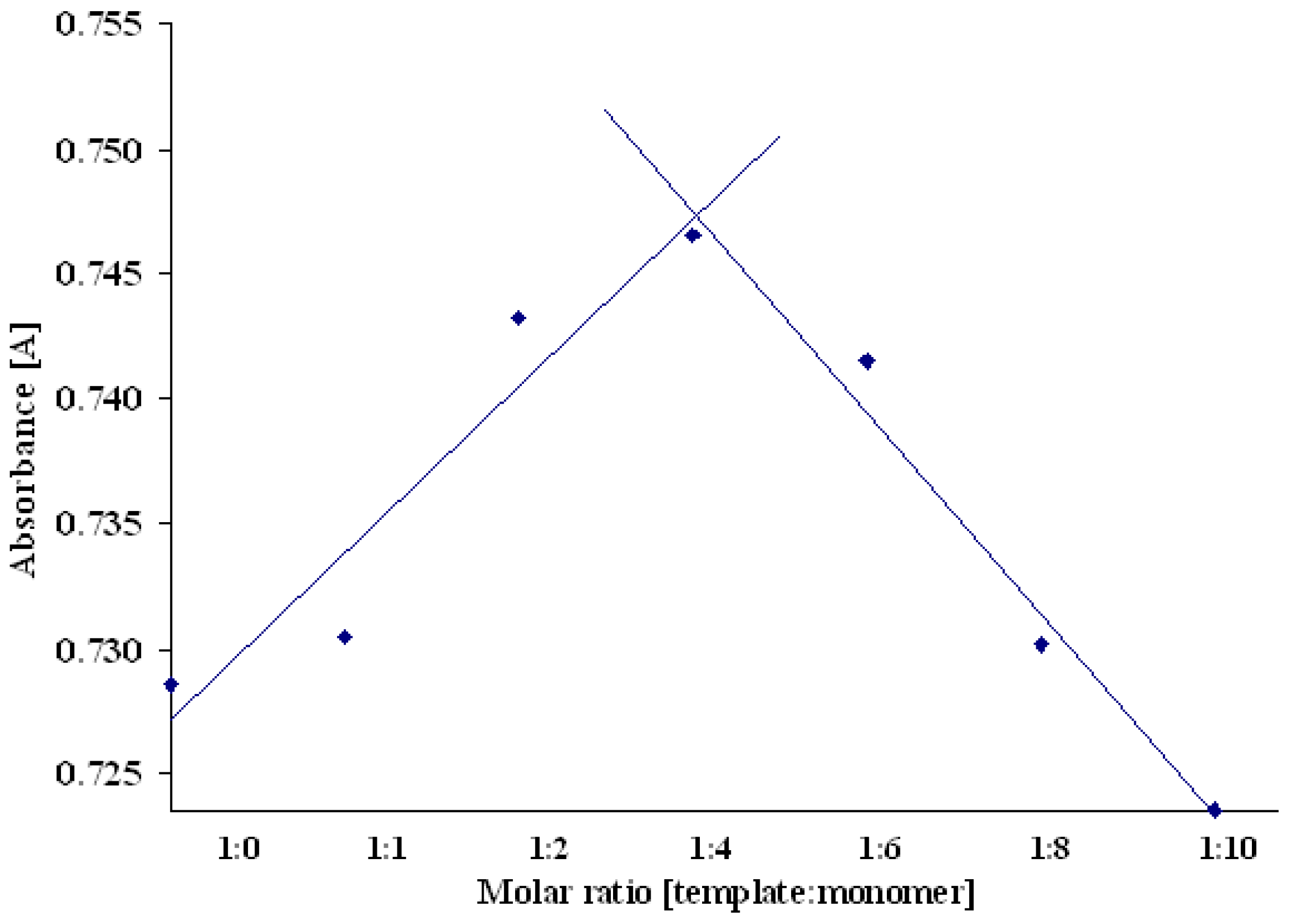
Composition of the prepolymerization complex
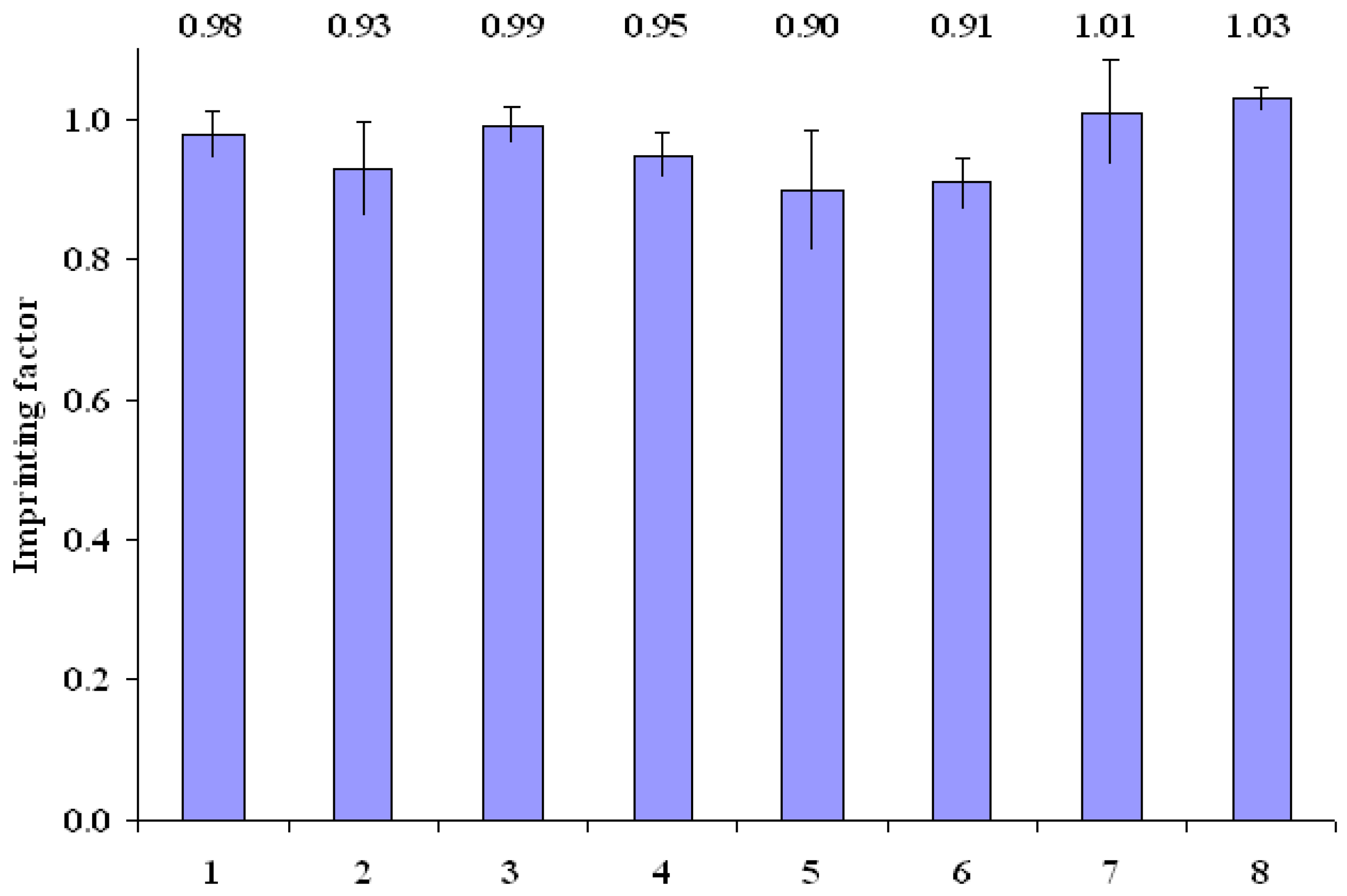
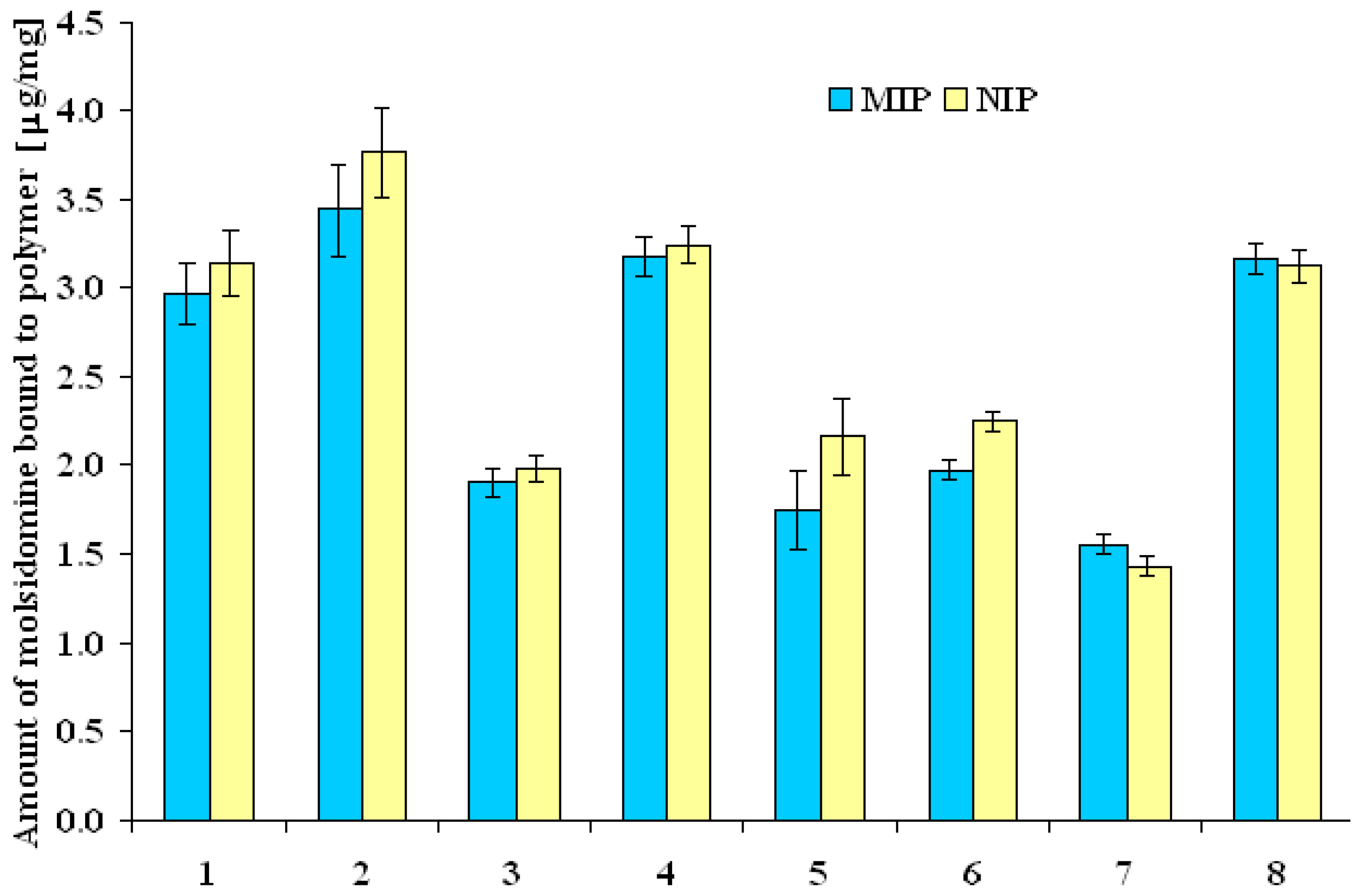
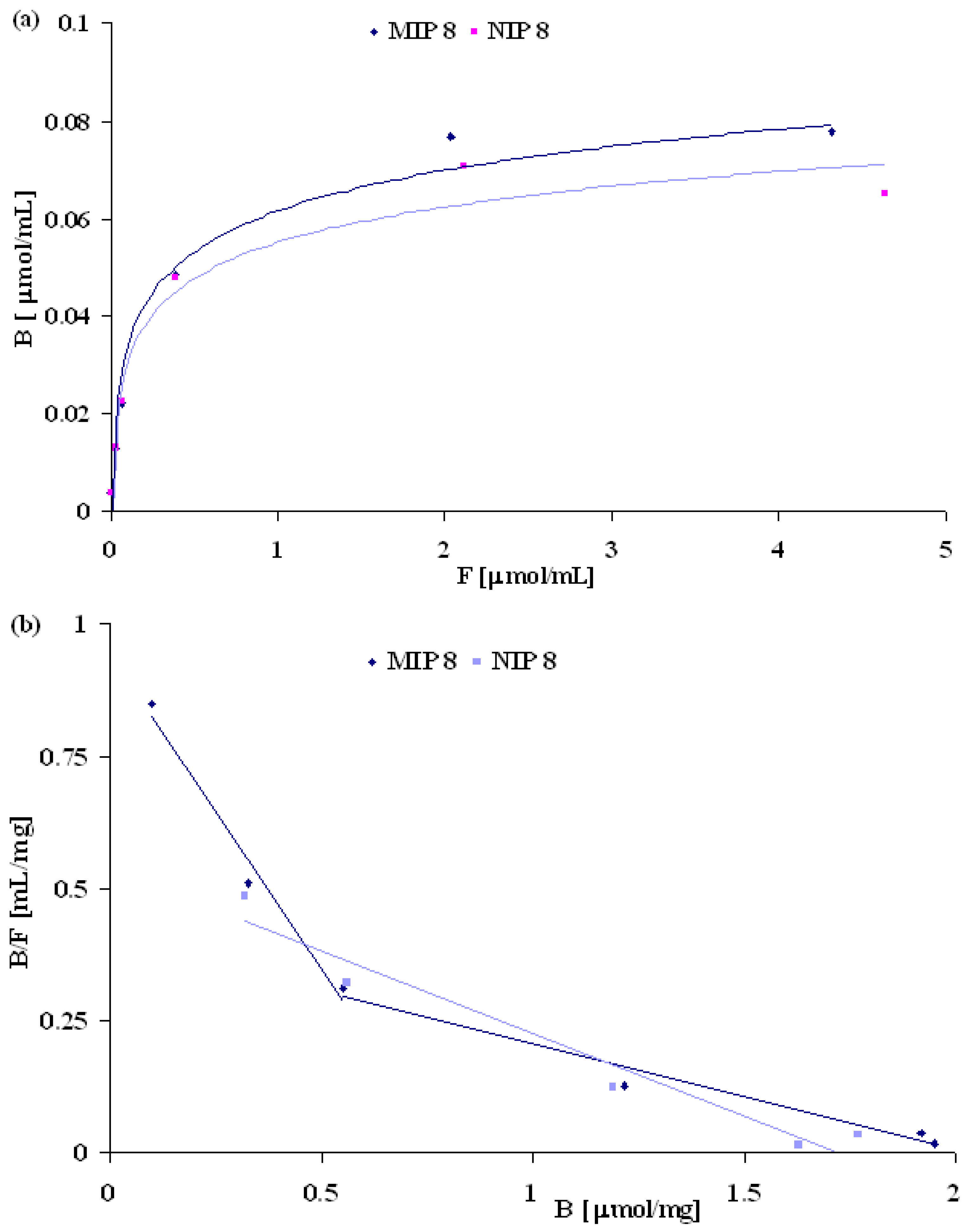
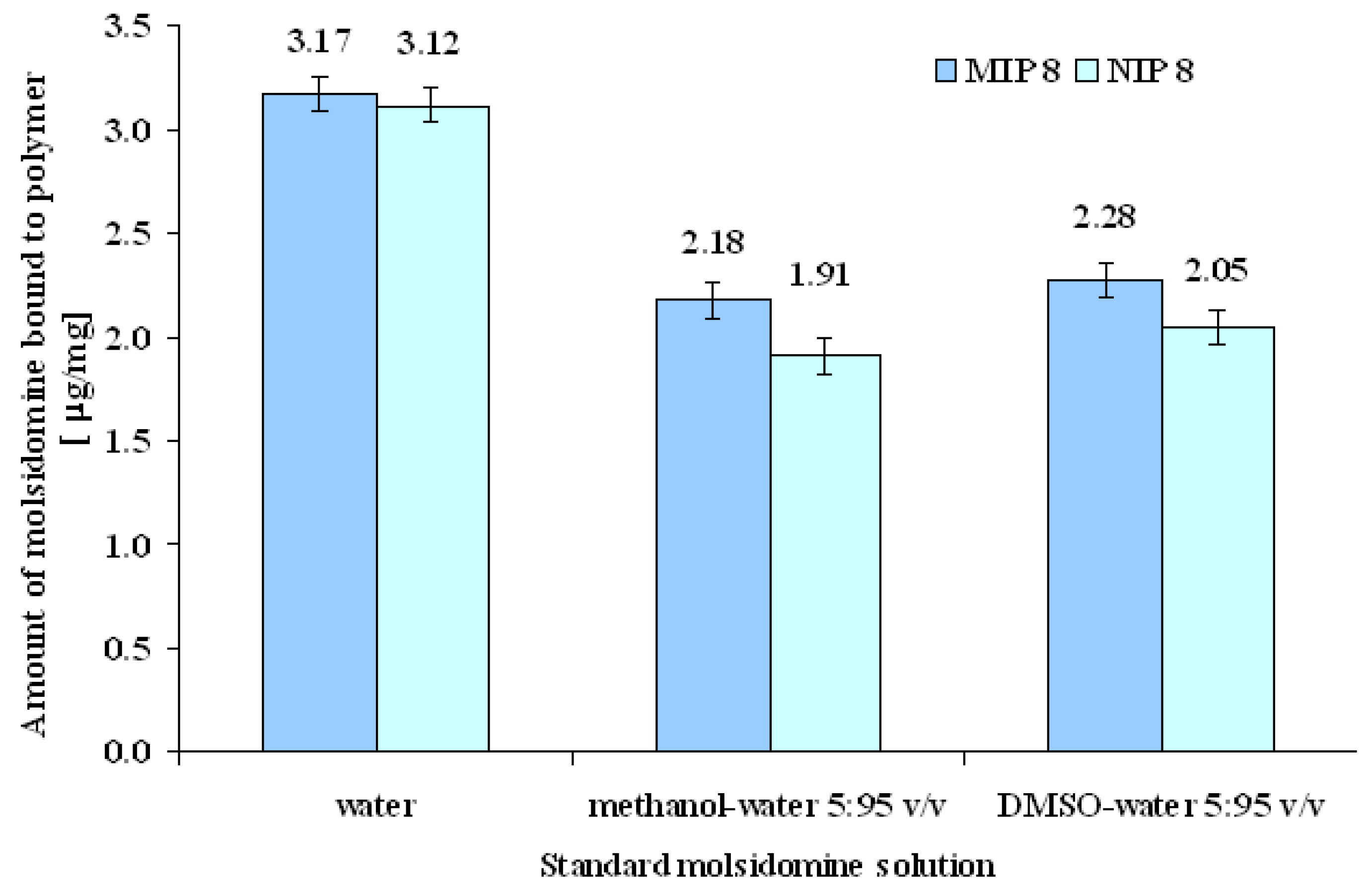
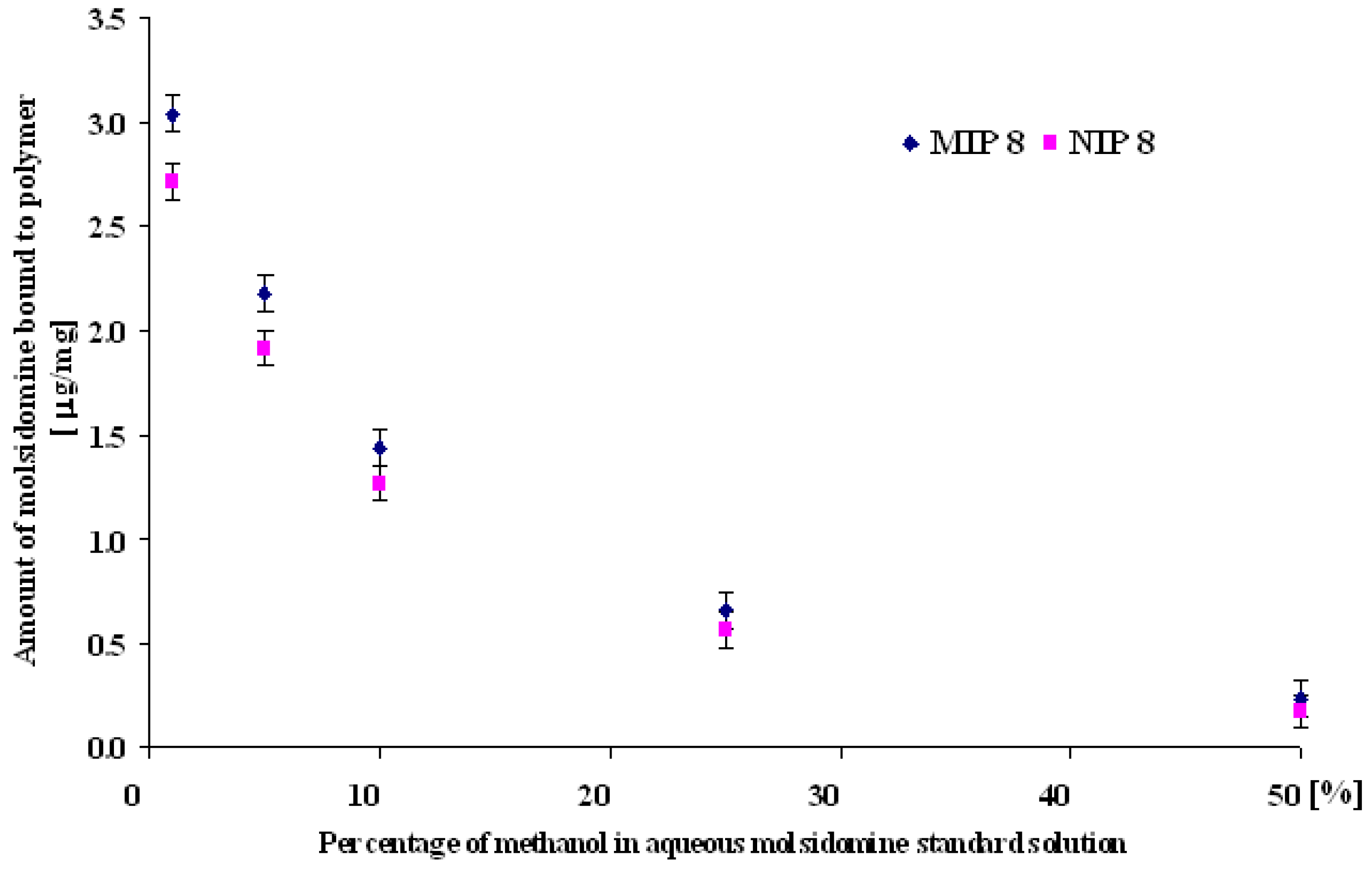
MIP and NIP binding properties
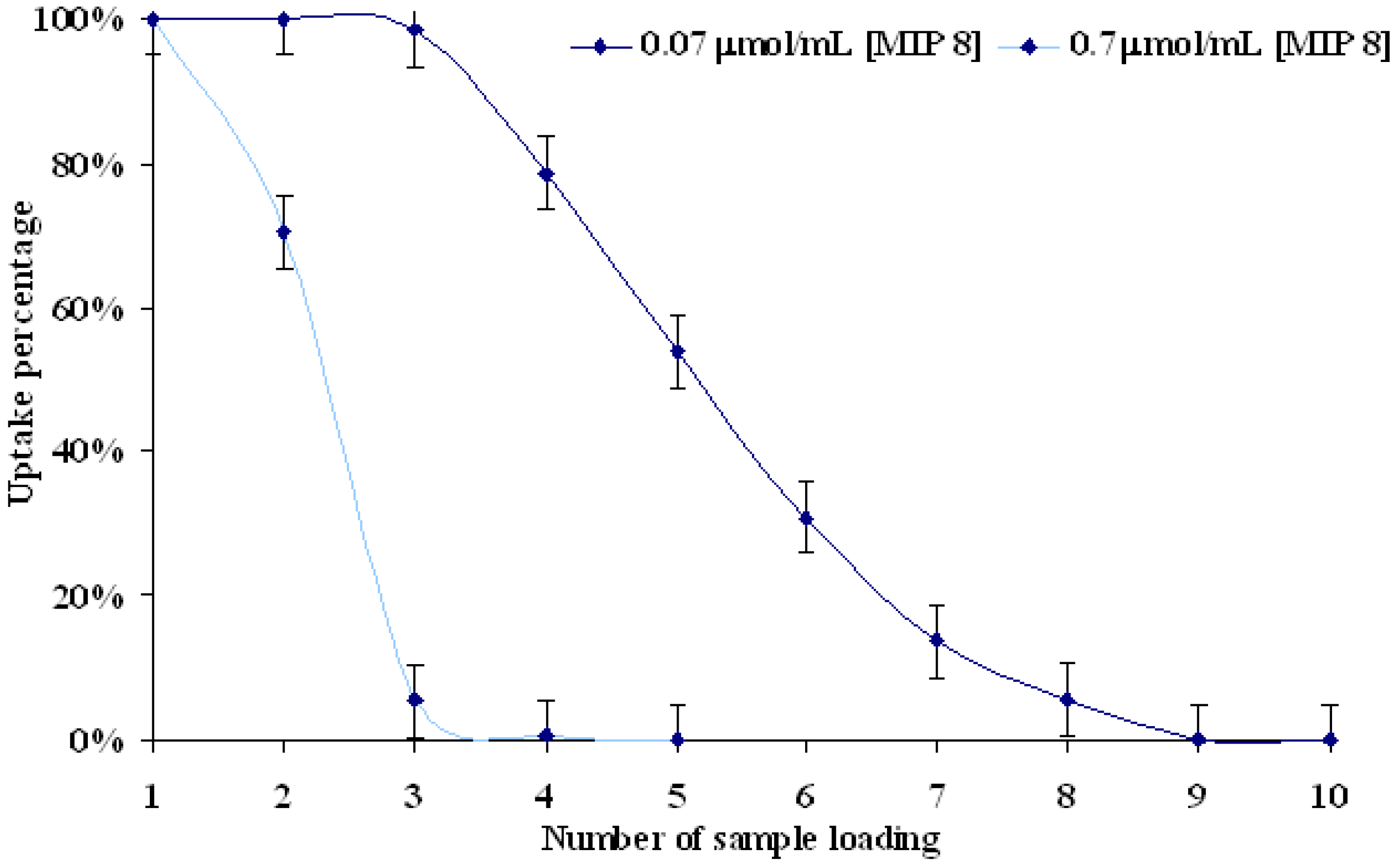
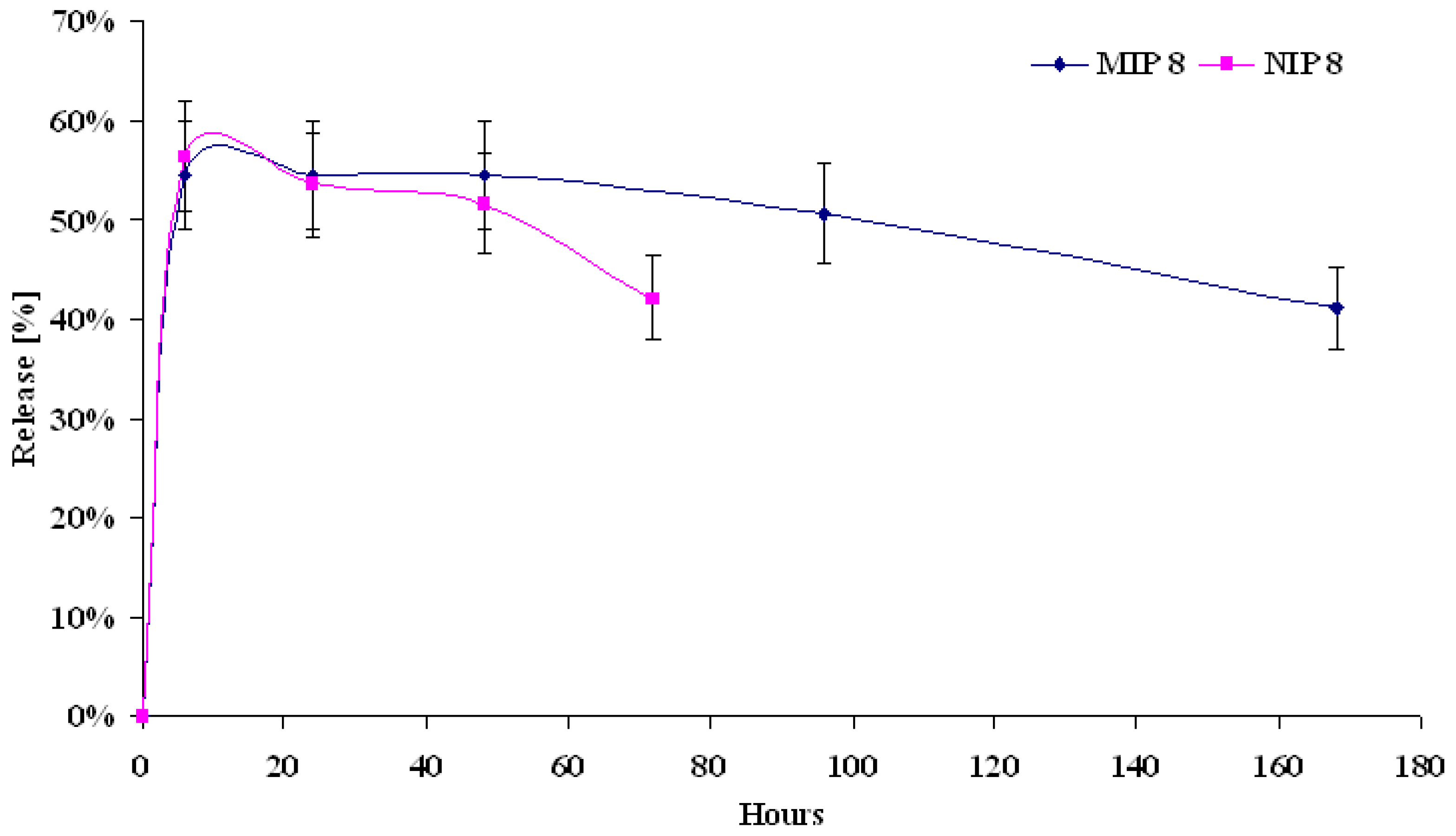
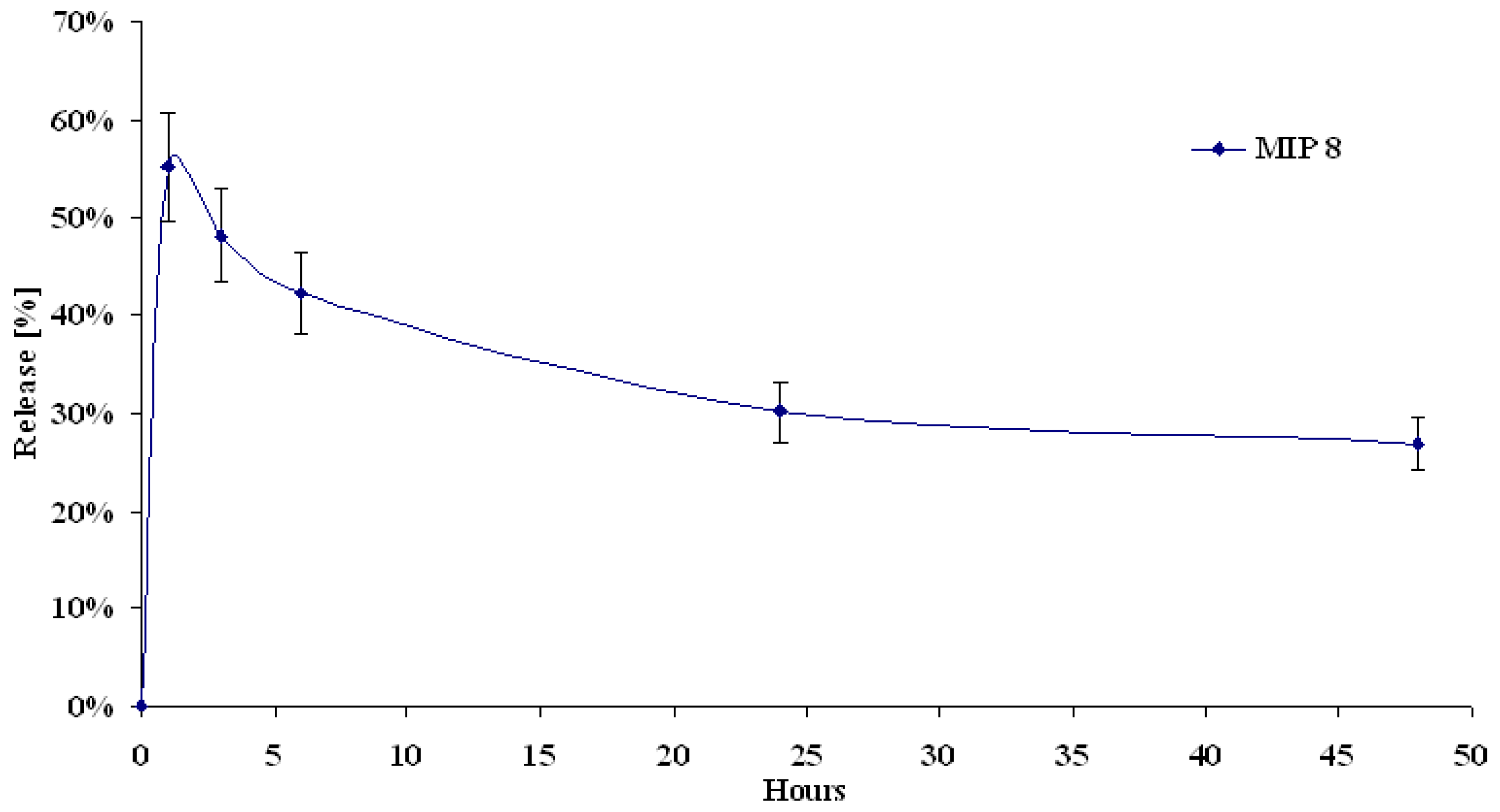
MIP and NIP release properties
Conclusions
Experimental
General
Stock solution of molsidomine
Preparation of molecularly imprinted and control polymers
Evaluation of polymers
References
- Komiyama, M.; Takeuchi, T.; Mukawa, T.; Asanuma, H. Molecular Imprinting: From Fundamentals to Applications; Wiley-VCH: Weinheim, Germany, 2003. [Google Scholar]
- Qiao, F.X.; Sun, H.W.; Yan, H.Y.; Row, K.H. Molecularly imprinted polymers for solid-phase extraction. Chromatographia 2006, 64, 625–634. [Google Scholar] [CrossRef]
- Haupt, K. Imprinted polymers – tailor-made mimics of antibodies and receptors. Chem. Commun. 2003, 171–178. [Google Scholar] [CrossRef]
- Alexander, C.; Davidson, L.; Hayes, W. Imprinted polymers: artificial molecular recognition materials with applications in synthesis and catalysis. Tetrahedron 2003, 59, 2025–2057. [Google Scholar] [CrossRef]
- Alvarez-Lorenzo, C.; Concheiro, A. Molecularly imprinted polymers for drug delivery. J. Chromatogr. B 2004, 804, 231–245. [Google Scholar] [CrossRef] [PubMed]
- Sellergren, B.; Allender, C.J. Molecularly imprinted polymers: A bridge to advanced drug delivery. Adv. Drug Deliv. Rev. 2005, 57, 1733–1741. [Google Scholar] [CrossRef] [PubMed]
- Puoci, F.; Iemma, F.; Cirillo, G.; Picci, N.; Matricardi, P.; Alhaique, F. Molecularly imprinted polymers for 5-fluorouracil release in biological fluids. Molecules 2007, 12, 805–814. [Google Scholar] [CrossRef] [PubMed]
- Bayer, C.L.; Peppas, N.A. Advances in recognitive, conductive and responsive delivery systems. J. Control. Release 2008, 132, 216–221. [Google Scholar] [CrossRef] [PubMed]
- Jantarat, C.; Tangthong, N.; Songkro, S.; Martin, G.P.; Suedee, R. S-Propranolol imprinted polymer nanoparticle-on-microsphere composite porous cellulose membrane for the enantioselectively controlled delivery of racemic propanolol. Int. J. Pharm. 2008, 349, 212–225. [Google Scholar] [CrossRef] [PubMed]
- Streel, B.; Ceccato, A.; Peerboom, C.; Zimmer, C.; Sibenaler, R.; Maes, P. Determination of molsidomine and its active metabolite in human plasma using liquid chromatography with tandem mass spectric detection. J. Chromatogr. A 1998, 819, 113–123. [Google Scholar] [CrossRef]
- Schönafinger, K. Heterocyclic NO prodrugs. Il Farmaco 1999, 54, 316–320. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Montgomery, J.A., Jr.; Vreven, T.; Kudin, K.N.; Burant, J.C.; Millam, J.M.; Iyengar, S.S.; Tomasi, J.; Barone, V.; Mennucci, B.; Cossi, M.; Scalmani, G.; Rega, N.; Petersson, G.A.; Nakatsuji, H.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Klene, M.; Li, X.; Knox, J.E.; Hratchian, H.P.; Cross, J.B.; Adamo, C.; Jaramillo, J.; Gomperts, R.; Stratmann, R.E.; Yazyev, O.; Austin, A.J.; Cammi, R.; Pomelli, C.; Ochterski, J.W.; Ayala, P.Y.; Morokuma, K.; Voth, G.A.; Salvador, P.; Dannenberg, J.J.; Zakrzewski, V.G.; Dapprich, S.; Daniels, A.D.; Strain, M.C.; Farkas, O.; Malick, D.K.; Rabuck, A.D.; Raghavachari, K.; Foresman, J.B.; Ortiz, J.V.; Cui, Q.; Baboul, A.G.; Clifford, S.; Cioslowski, J.; Stefanov, B.B.; Liu, G.; Liashenko, A.; Piskorz, P.; Komaromi, I.; Martin, R.L.; Fox, D.J.; Keith, T.; Al-Laham, M.A.; Peng, C.Y.; Nanayakkara, A.; Challacombe, M.; Gill, P.M.W.; Johnson, B.; Chen, W.; Wong, M.W.; Gonzalez, C.; Pople, J.A. Gaussian 03, Revision B.02; Gaussian, Inc.: Pittsburgh, PA, USA, 2003. [Google Scholar]
- Improta, R.; Santoro, F.; Barbier, C.; Giordano, F.; Del Re, G. On the geometry of 3-amino-sydnones. J. Mol. Struct. (THEOCHEM) 1998, 433, 291–299. [Google Scholar] [CrossRef]
- Moss, G.P.; Smith, P.A.S.; Tavernier, D. Glossary of class names of organic compounds and reactive intermediates based on structure. Pure Appl. Chem. 1995, 67, 1307–1375. [Google Scholar] [CrossRef]
- Laudy, D.; Tetrat, F.; Truant, E.; Blach, P.; Fourmentin, S.; Surpateanu, G. Development of a competitive continuous variation plot for the determination of inclusion compounds stoichiometry. J. Incl. Phenom. Macrocycl. Chem. 2007, 57, 409–413. [Google Scholar]
- Striegler, S.; Dittel, M. Evaluation of new strategies to prepare templated polymers with sufficient oligosaccharide recognition capacity. Anal. Chim. Acta 2003, 484, 53–62. [Google Scholar] [CrossRef]
- Svenson, J.; Karlsson, J.G.; Nicholls, I.A. 1H Nuclear magnetic resonance study of the molecular imprinting of (-)nicotine: template self-association, a molecular basis for cooperative ligand binding. J. Chrom. A 2004, 1024, 39–44. [Google Scholar] [CrossRef]
- Breton, F.; Delepee, R.; Jegourel, D.; Deville-Bonne, D.; Agrofoglio, L.A. Selective adenosine-5’-monophosphate uptake by water-compatible molecularly imprinted polymer. Anal. Chim. Acta 2008, 616, 222–229. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Schüssler, W.; Sengl, M.; Niessner, R.; Knopp, D. Selective trace analysis of diclofenac in surface and wastewater samples using solid-phase extraction with a new molecularly imprinted polymer. Anal. Chim. Acta 2008, 620, 73–81. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Contact the authors. |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Luliński, P.; Maciejewska, D. Examination of Imprinting Process with Molsidomine as a Template. Molecules 2009, 14, 2212-2225. https://doi.org/10.3390/molecules14062212
Luliński P, Maciejewska D. Examination of Imprinting Process with Molsidomine as a Template. Molecules. 2009; 14(6):2212-2225. https://doi.org/10.3390/molecules14062212
Chicago/Turabian StyleLuliński, Piotr, and Dorota Maciejewska. 2009. "Examination of Imprinting Process with Molsidomine as a Template" Molecules 14, no. 6: 2212-2225. https://doi.org/10.3390/molecules14062212
APA StyleLuliński, P., & Maciejewska, D. (2009). Examination of Imprinting Process with Molsidomine as a Template. Molecules, 14(6), 2212-2225. https://doi.org/10.3390/molecules14062212



