X-ray and Hydrogen-bonding Properties of 1-((1H-benzotriazol-1-yl)methyl)naphthalen-2-ol
Abstract
:Introduction

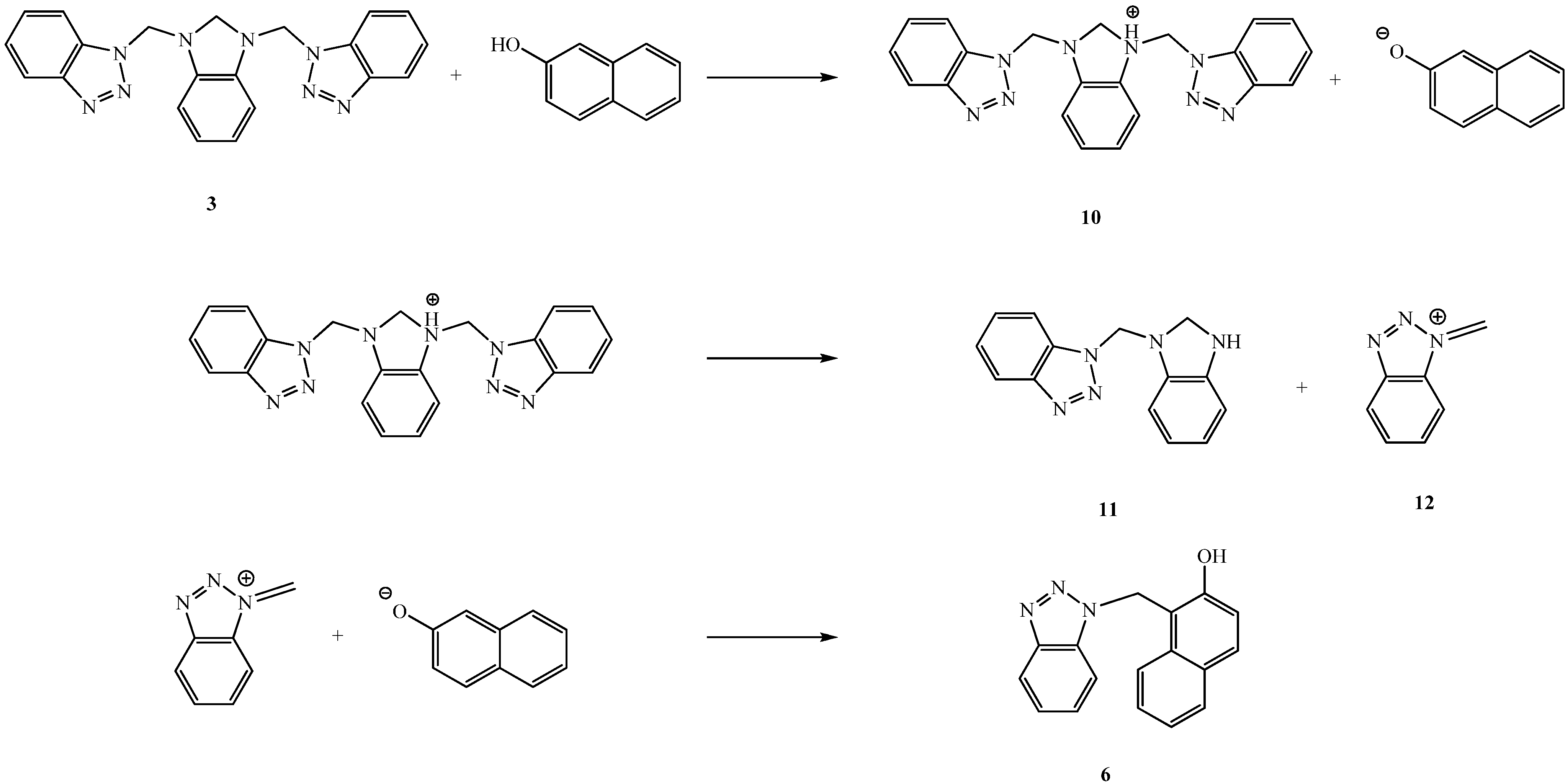
Results and Discussion
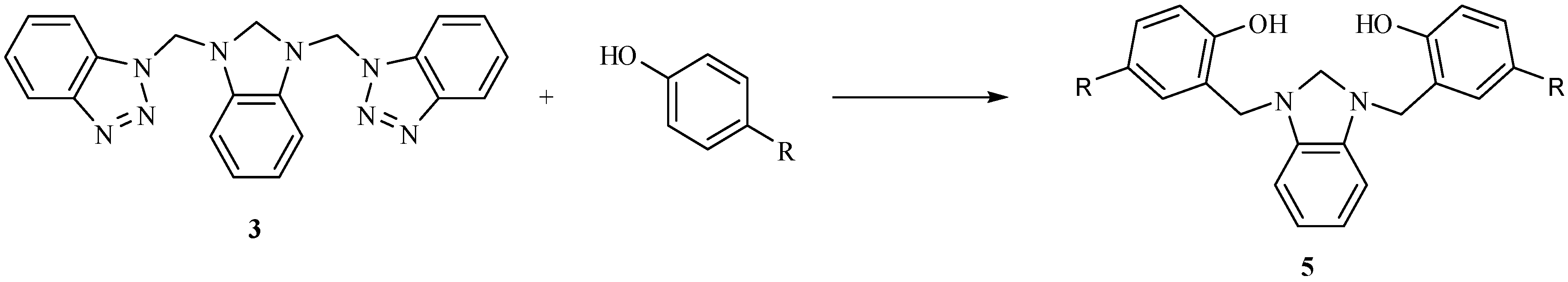
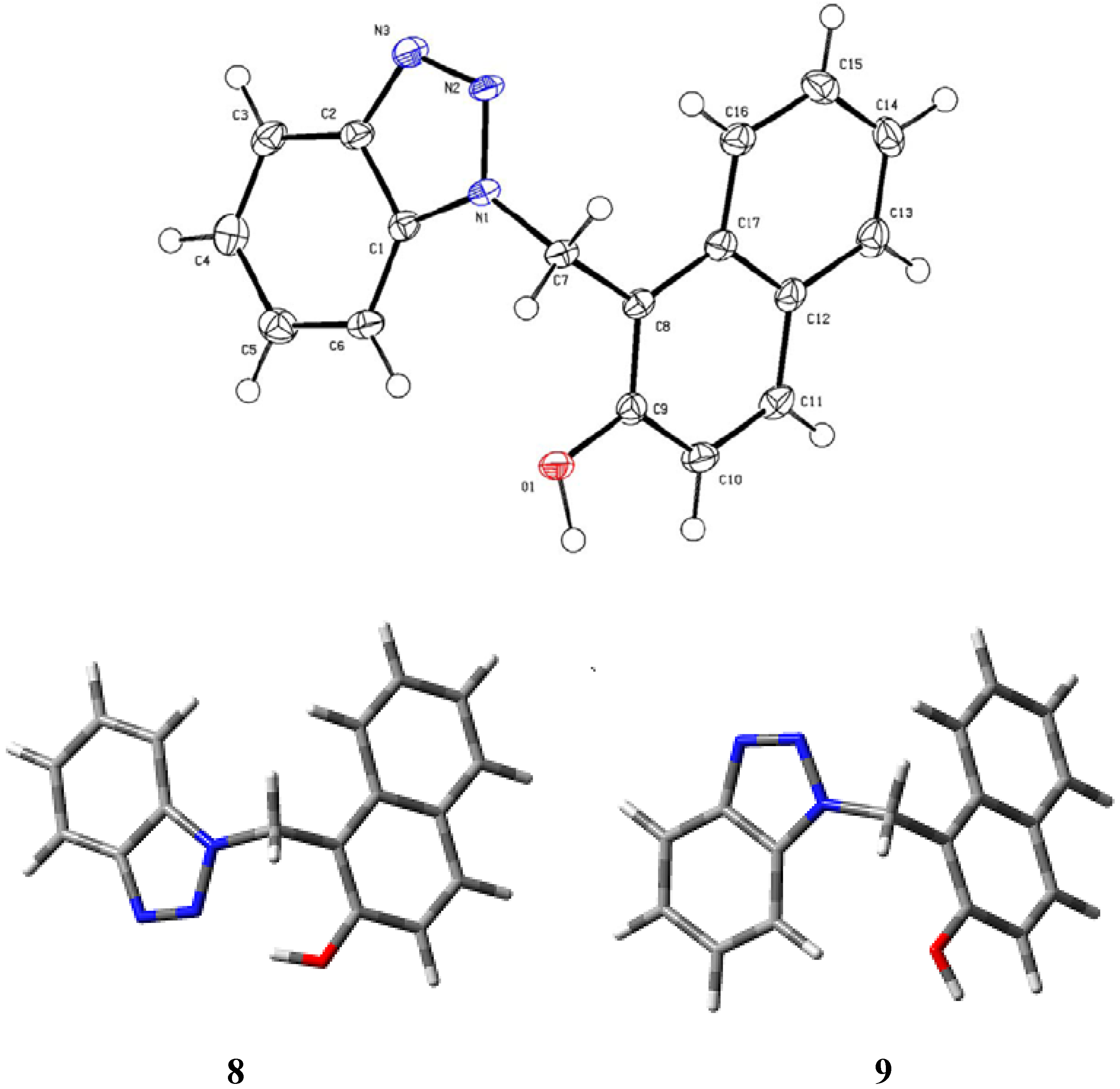
| Bond lengths (Å) | Bond angles (deg) | ||
| O(1)-C(9) | 1.365(2) | N(2)-N(1)-C(7) | 120.64(13) |
| N(1)-N(2) | 1.348(2) | N(2)-N(1)-C(1) | 110.69(14) |
| N(1)-C(7) | 1.471(2) | C(1)-N(1)-C(7) | 128.67(14) |
| N(1)-C(1) | 1.363(2) | N(3)-N(2)-N(1) | 108.36(13) |
| N(3)-N(2) | 1.312(2) | N(2)-N(3)-C(2) | 108.76(14) |
| N(3)-C(2) | 1.377(2) | C(9)-C(8)-C(17) | 119.23(15) |
| C(8)-C(9) | 1.384(2) | C(9)-C(8)-C(7) | 119.82(15) |
| C(8)-C(17) | 1.429(2) | C(17)-C(8)-C(7) | 120.93(15) |
| C(8)-C(7) | 1.505(2) | O(1)-C(9)-C(8) | 118.94(15) |
| C(9)-C(10) | 1.411(2) | O(1)-C(9)-C(10) | 119.94(15) |
| C(10)-C(11) | 1.362(2) | C(8)-C(9)-C(10) | 121.13(16) |
| C(11)-C(12) | 1.418(2) | C(11)-C(10)-C(9) | 120.20(16) |
| C(12)-C(13) | 1.416(2) | C(10)-C(11)-C(12) | 120.96(17) |
| C(12)-C(17) | 1.423(2) | C(13)-C(12)-C(11) | 121.26(16) |
| C(13)-C(14) | 1.365(3) | C(11)-C(12)-C(17) | 119.11(16) |
| C(14)-C(15) | 1.403(3) | C(13)-C(12)-C(17) | 119.61(16) |
| C(15)-C(16) | 1.372(3) | C(14)-C(13)-C(12) | 120.68(18) |
| C(16)-C(17) | 1.420(2) | C(13)-C(14)-C(15) | 120.20(17) |
| C(1)-C(6) | 1.401(2) | C(16)-C(15)-C(14) | 120.53(17) |
| C(1)-C(2) | 1.397(2) | C(15)-C(16)-C(17) | 121.02(17) |
| C(5)-C(6) | 1.372(2) | C(16)-C(17)-C(12) | 117.89(16) |
| C(4)-C(5) | 1.413(3) | C(16)-C(17)-C(8) | 122.84(16) |
| C(3)-C(4) | 1.372(3) | C(12)-C(17)-C(8) | 119.26(15) |
| C(2)-C(3) | 1.404(2) | N(1)-C(7)-C(8) | 112.34(13) |
| N(1)-C(1)-C(2) | 104.29(15) | ||
| N(1)-C(1)-C(6) | 132.97(16) | ||
| C(2)-C(1)-C(6) | 122.68(16) | ||
| Structure | d(Å) | Angle | ||
|---|---|---|---|---|
| 6a | 0.99(2) | 1.77(2) | 2.7618(19) | 176(2) |
| 7b | 0.85(3) | 1.85(4) | 149(3) | |
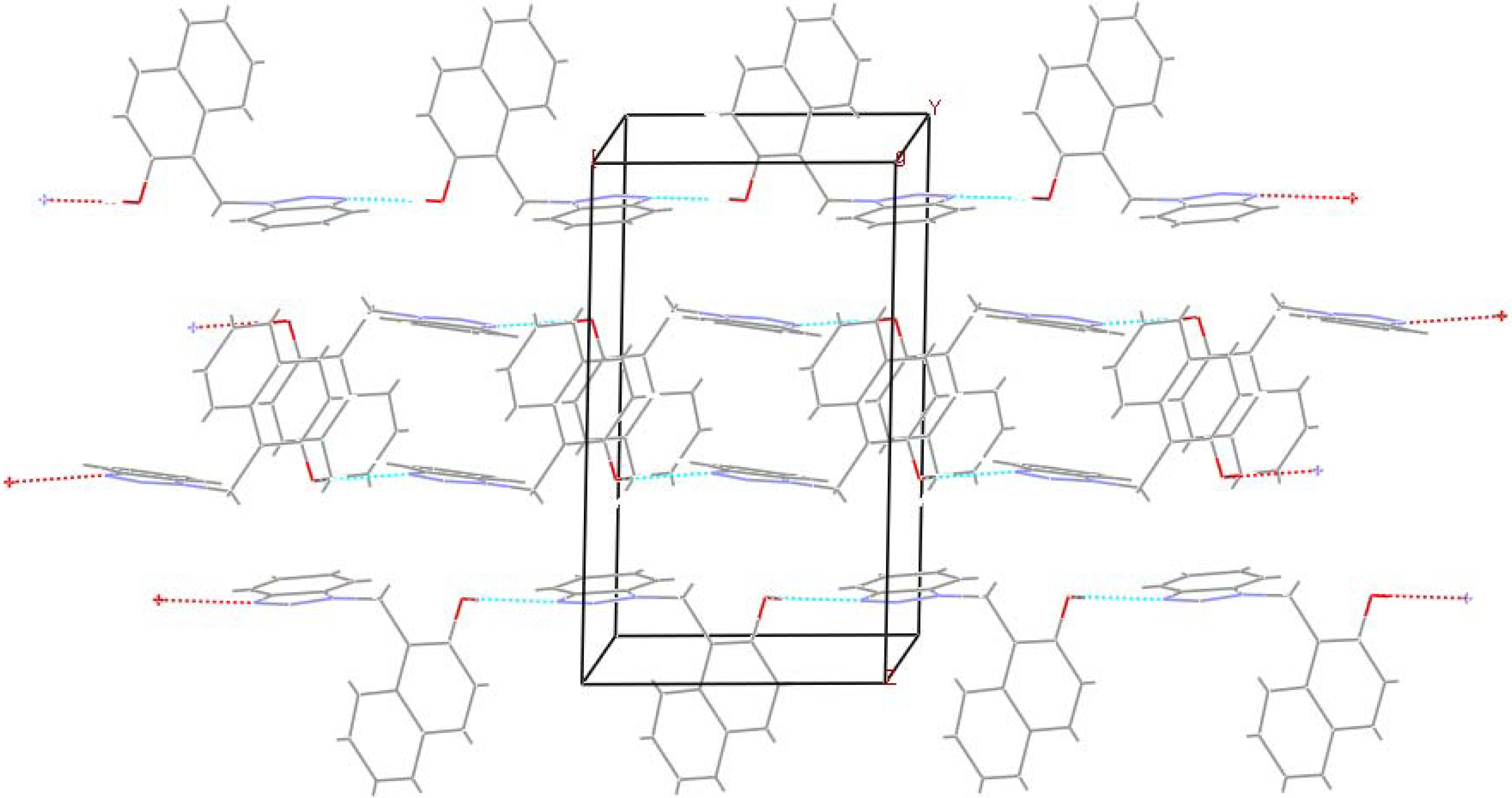
Experimental
General
Synthesis
X-ray crystallography
| Empirical formula | C17H13N3O |
|---|---|
| Temperature | 100(2) K |
| Formula weight | 275.30 |
| Wavelength | 0.7107 Å |
| Crystal system | Monoclinic |
| Space group | P21/c |
| Unit cell dimensions | a = 11.7934(9) Å |
| b = 14.3002(14) Å | |
| c = 8.4444(8) Å | |
| β = 106.243(5)° | |
| Volume | 1367.3(2) Å3 |
| Z | 4 |
| Calculated density | 1.337 g/cm3 |
| Absorption coefficient | 0.09 mm-1 |
| F(000) | 576 |
| Crystal size | 0.36 x 0.12 x 0.07 mm |
| θ range for data collection | 2.3 - 23.9o |
| Limiting indexes | 0 ≤ h ≤ 14, -17 ≤ k ≤ 0, -10 ≤ l ≤ 10 |
| Reflections collected/unique | 9100/2696 [R(int) = 0.0634] |
| Completeness to θ = 30.50 | 100% |
| Final shift / error, max and avg | 0.002, 0.000 |
| Refinement method | Full-matrix least-squares on F2 |
| Data / restraints / parameters | 2696/0/194 |
| Goodness of fit on F2 | 1.038 |
| Final R indices [I >2sigma (I)] | R1 =4.61%, wR2 = 10.58% |
| R indices (all data) | R1 = 6.96%, wR2 = 11.41% |
| Largest peak and hole | 0.30 and -0.31 e-Å-3 |
Theoretical calculations
Acknowledgements
References and Notes
- Katritzky, A.R.; Manju, K.; Singh, S.K.; Meher, N.K. Benzotriazole mediated amino-, amido, alkoxy- and alkylthio-alkylation. Tetrahedron 2005, 61, 2555–2581. [Google Scholar] [CrossRef]
- Katritzky, A.R.; Rogovoy, B.V. Benzotriazole: an ideal synthetic auxiliary. Chem. Eur. J. 2003, 9, 4586–4593. [Google Scholar] [CrossRef]
- Katritzky, A.R.; Lan, X.; Yang, J.Z.; Denisko, O.V. Properties and synthetic utility of N-substituted benzotriazoles. Chem. Rev. 1998, 98, 409–548. [Google Scholar] [CrossRef]
- Katritzky, A.R.; Lan, X. Benzotriazole-mediated arylalkylation and heteroarylalkylation. Chem. Soc. Rev. 1994, 23, 363–373. [Google Scholar] [CrossRef]
- Katritzky, A.R.; Zhang, Z.; Lang, H.; Lan, X. o-(α-benzotriazolylalkyl)phenols: Novel precursors for the preparation of 1,1-bis(2-hydroxyaryl)alkanes. J. Org. Chem. 1994, 59, 7209–7213. [Google Scholar] [CrossRef]
- Katritzky, A.R.; Zhang, Z.; Lan, X.; Lang, H. o-(α-Benzotriazolylalkyl)phenols: Novel precursors for the preparation of ortho-substituted phenols via intermediate o-quinone methides. J. Org. Chem. 1994, 59, 1900–1903. [Google Scholar] [CrossRef]
- Katritzky, A.R.; Lan, X. o-(α-benzotriazolylalkyl)phenols: Novel precursors of o-quinone methides. Synthesis 1992, 8, 761–764. [Google Scholar] [CrossRef]
- Rivera, A.; Maldonado, M.; Núñez, M.E.; Joseph-Nathan, P. Nucleophilic substitution at the aminalic carbon of some macrocyclic polyaminals. Heterocycl. Commun. 2004, 10, 77–80. [Google Scholar]
- Katritzky, A.R.; Rachwal, S.; Hitchings, G.J. Benzotriazole: A novel synthetic auxiliary. Tetrahedron 1991, 47, 2683–2732. [Google Scholar] [CrossRef]
- Katritzky, A.R.; Belyakov, S.A.; Sorochinsky, A.E.; Steel, P.J.; Schall, O.F.; Gokel, G.W. Novel syntheses of N-pivot lariat diaza-crown ethers from 4,13-bis(benzotriazolylmethyl)-4,13-diaza-1,7,10,16-tetraoxacyclooctadecane. J. Org. Chem. 1996, 61, 7585–7592. [Google Scholar] [CrossRef]
- Katritzky, A.R.; Abdel-Fattah, A.A.A.; Tymoshenko, D.O.; Belyakov, S.A.; Ghiviriga, I.; Steel, P.J. Amino(hetero)arylmethylation of phenols with N-[r-amino(hetero)arylmethyl]-benzotriazoles. J Org. Chem. 1999, 64, 6071–6075. [Google Scholar]
- Katritzky, A.R.; Lan, X.; Lam, J.N. o-(α-benzotriazolylalkyl)phenols: Versatile intermediates for the synthesis of substituted phenols. Chem. Ber. 1991, 124, 1809–1817. [Google Scholar] [CrossRef]
- Etter, M.C.; MacDonald, J.C.; Bernstein, J. Graph-set analysis of hydrogen-bond patterns in organic crystals. Acta Cryst. B. 1990, 46, 256–262. [Google Scholar] [CrossRef]
- Cai, Y.-P.; Li, G.-B.; He, G.-P.; Su, C.-Y.; Xu, A.-W. Zhang, 1,4-Bis(1H-benzotriazol-1-ylmethyl)benzene tetrahydrate. Acta Cryst. E. 2004, 60, o2062–o2064. [Google Scholar] [CrossRef]
- Zonta, C.; Crisma, M.; De Lucchi, O.; Motterle, R.; Serafini, S. Benzotriazole complexes with amines and phenol: Cooperativity mediated by induction effects in the crystal state. Org. Lett. 2006, 8, 1577–1579. [Google Scholar] [CrossRef]
- Fluegge, A.P.; Waiblinger, F.; Stein, M.; Keck, J.; Kramer, H.E.A.; Fischer, P.; Wood, M.G.; DeBellis, A.D.; Ravichandran, R.; Leppard, D. Probing the intramolecular Hydrogen Bond of 2-(2-hydroxyphenyl)benzotriazole in Polar Environment: A photophysical study of UV absorber efficiency. J. Phys. Chem. A. 2007, 111, 9733–9744. [Google Scholar] [CrossRef]
- Woessner, G.; Goeller, G.; Kollat, P.; Stezowski, J.J.; Hauser, M.; Klein, U.K.A.; Kramer, H.E.A. Photophysical and photochemical deactivation processes of ultraviolet stabilizers of the (2-hydroxyphenyl)benzotriazole class. J. Phys. Chem. 1984, 88, 5544–5550. [Google Scholar] [CrossRef]
- Tomas, F.; Abboud, J.L.M.; Laynez, J.; Notario, R.; Santos, L.; Nilsson, S.; Catalan, J.; Claramunt, R. M.; Elguero, J. Tautomerism and aromaticity in 1,2,3-triazoles: the case of benzotriazole. J. Am. Chem. Soc. 1989, 111, 7348–7353. [Google Scholar] [CrossRef]
- Desiraju, G.R. Supramolecular synthons in crystal engineering-A new organic synthesis. Angew. Chem. Int. Ed. 1995, 34, 2311–2327. [Google Scholar] [CrossRef]
- Macrae, C.F.; Edgington, P.R.; McCabe, P.; Pidcock, E.; Shields, G.P.; Taylor, R.; Towler, M.; Van de Streek, J. Mercury: visualization and analysis of crystal structures. J. Appl. Cryst. 2006, 39, 453–457. [Google Scholar] [CrossRef]
- Borisenko, K.B.; Bock, C.W.; Hargittai, I. Molecular geometry of benzaldehyde and salicylaldehyde: A gas-phase electron diffraction and ab Initio molecular orbital investigation. J. Phys. Chem. 1996, 100, 7426–7434. [Google Scholar] [CrossRef]
- Steiner, T. The hydrogen bond in the solid state. Angew. Chem. Int. Ed. 2002, 41, 48–76. [Google Scholar] [CrossRef]
- Volovodenko, A.P.; Trifonov, R.E.; Ostrovskii, V.A. Basicity of Benzotriazole. Russ, J. Org. Chem. 2000, 36, 1357–1359. [Google Scholar]
- Biggs, A.I.; Robinson, R.A. The ionization constants of some substituted anilines and phenols: a test of the Hammett relation. J. Chem. Soc. 1961, 388–393. [Google Scholar] [CrossRef]
- Burckhalter, J.H.; Leib, R.I. Amino- and chloromethylation of 8-quinolinol. Mechanism of preponderant ortho substitution in phenols under Mannich Conditions. J. Org. Chem. 1961, 26, 4078–4083. [Google Scholar] [CrossRef]
- Bruker. APEX2 Bruker AXS Inc.: Madison, WI, USA, 2004.
- Bruker. Program for Absorption Correction for Area Detectors; Bruker AXS Inc.: Madison, Wisconsin, USA, 2001. [Google Scholar] or Sheldrick, G.M. Program for Empirical Absorption Correction of Area Detector Data; University of Göttingen: Göttingen, Germany, 1996. [Google Scholar]
- Altomare, A; Burla, M.C.; Camalli, M.; Cascarano, G. SIR97: a new tool for crystal structure determination and refinement. J. Appl. Cryst. 1999, 32, 115–119. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SHELX-97 (SHELXS 97 and SHELXL 97), Programs for Crystal Structure Analyses; University of Göttingen: Göttingen, Germany, 1998. [Google Scholar]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Montgomery, J.A., Jr.; Vreven, T.; Kudin, K.N.; Burant, J.C.; Millam, J.M.; Iyengar, S.S.; Tomasi, J.; Barone, V.; Mennucci, B.; Cossi, M.; Scalmani, G.; Rega, N.; Petersson, G. A.; Nakatsuji, H.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Klene, M.; Li, X.; Knox, J.E.; Hratchian, H.P.; Cross, J.B.; Bakken, V.; Adamo, C.; Jaramillo, J.; Gomperts, R.; Stratmann, R.E.; Yazyev, O.; Austin, A.J.; Cammi, R.; Pomelli, C.; Ochterski, J.W.; Ayala, P.Y.; Morokuma, K.; Voth, G.A.; Salvador, P.; Dannenberg, J.J.; Zakrzewski, V.G.; Dapprich, S.; Daniels, A.D.; Strain, M.C.; Farkas, O.; Malick, D.K.; Rabuck, A.D.; Raghavachari, K.; Foresman, J.B.; Ortiz, J.V.; Cui, Q.; Baboul, A.G.; Clifford, S.; Cioslowski, J.; Stefanov, B.B.; Liu, G.; Liashenko, A.; Piskorz, P.; Komaromi, I.; Martin, R.L.; Fox, D.J.; Keith, T.; Al-Laham, M.A.; Peng, C.Y.; Nanayakkara, A.; Challacombe, M.; Gill, P.M. W.; Johnson, B.; Chen, W.; Wong, M.W.; Gonzalez, C.; Pople, J.A. Gaussian 03; Gaussian, Inc.: Wallingford, CT, USA, 2004. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Rivera, A.; Duarte, Y.; González-Salas, D.; Ríos-Motta, J.; Zaragoza, G. X-ray and Hydrogen-bonding Properties of 1-((1H-benzotriazol-1-yl)methyl)naphthalen-2-ol. Molecules 2009, 14, 1234-1244. https://doi.org/10.3390/molecules14031234
Rivera A, Duarte Y, González-Salas D, Ríos-Motta J, Zaragoza G. X-ray and Hydrogen-bonding Properties of 1-((1H-benzotriazol-1-yl)methyl)naphthalen-2-ol. Molecules. 2009; 14(3):1234-1244. https://doi.org/10.3390/molecules14031234
Chicago/Turabian StyleRivera, Augusto, Yorley Duarte, Diego González-Salas, Jaime Ríos-Motta, and Guillermo Zaragoza. 2009. "X-ray and Hydrogen-bonding Properties of 1-((1H-benzotriazol-1-yl)methyl)naphthalen-2-ol" Molecules 14, no. 3: 1234-1244. https://doi.org/10.3390/molecules14031234
APA StyleRivera, A., Duarte, Y., González-Salas, D., Ríos-Motta, J., & Zaragoza, G. (2009). X-ray and Hydrogen-bonding Properties of 1-((1H-benzotriazol-1-yl)methyl)naphthalen-2-ol. Molecules, 14(3), 1234-1244. https://doi.org/10.3390/molecules14031234




