Antioxidant Phenolic Substances of Turkish Red Wines from Different Wine Regions
Abstract
:Introduction
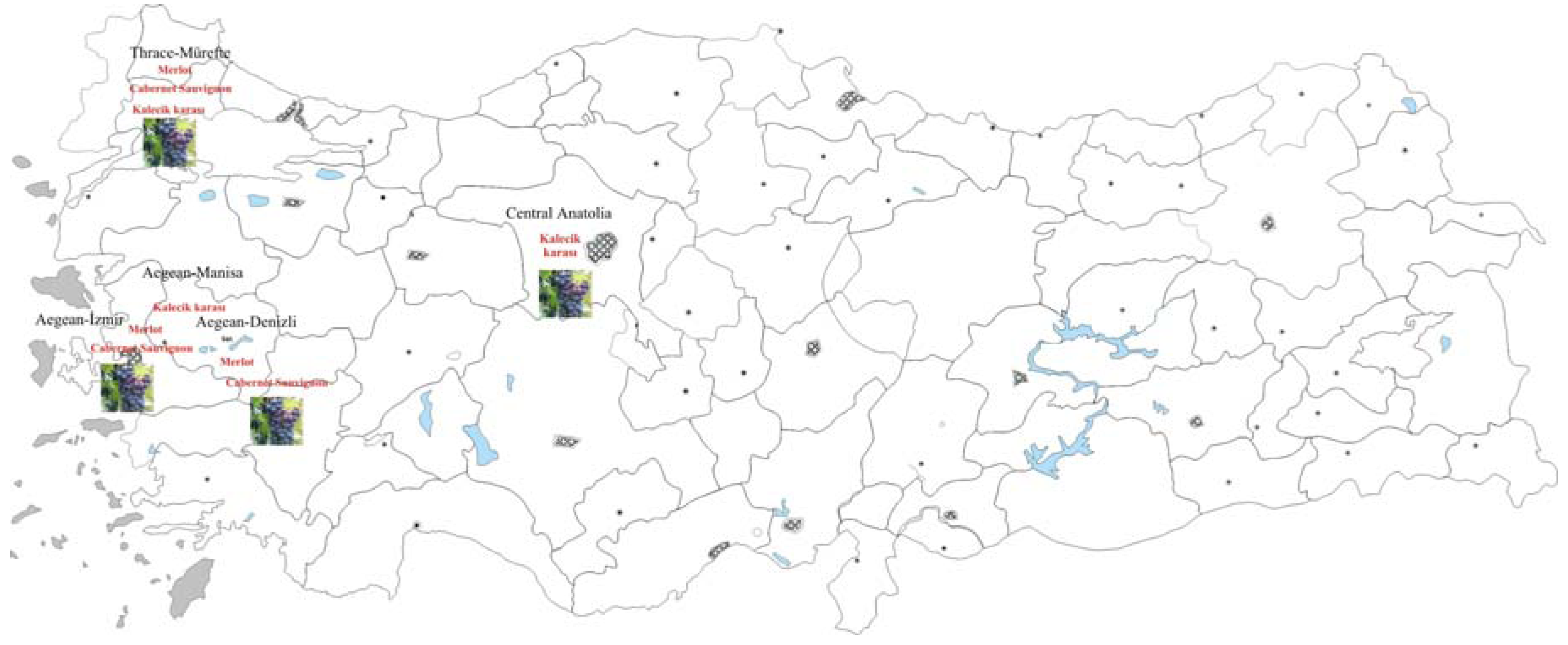
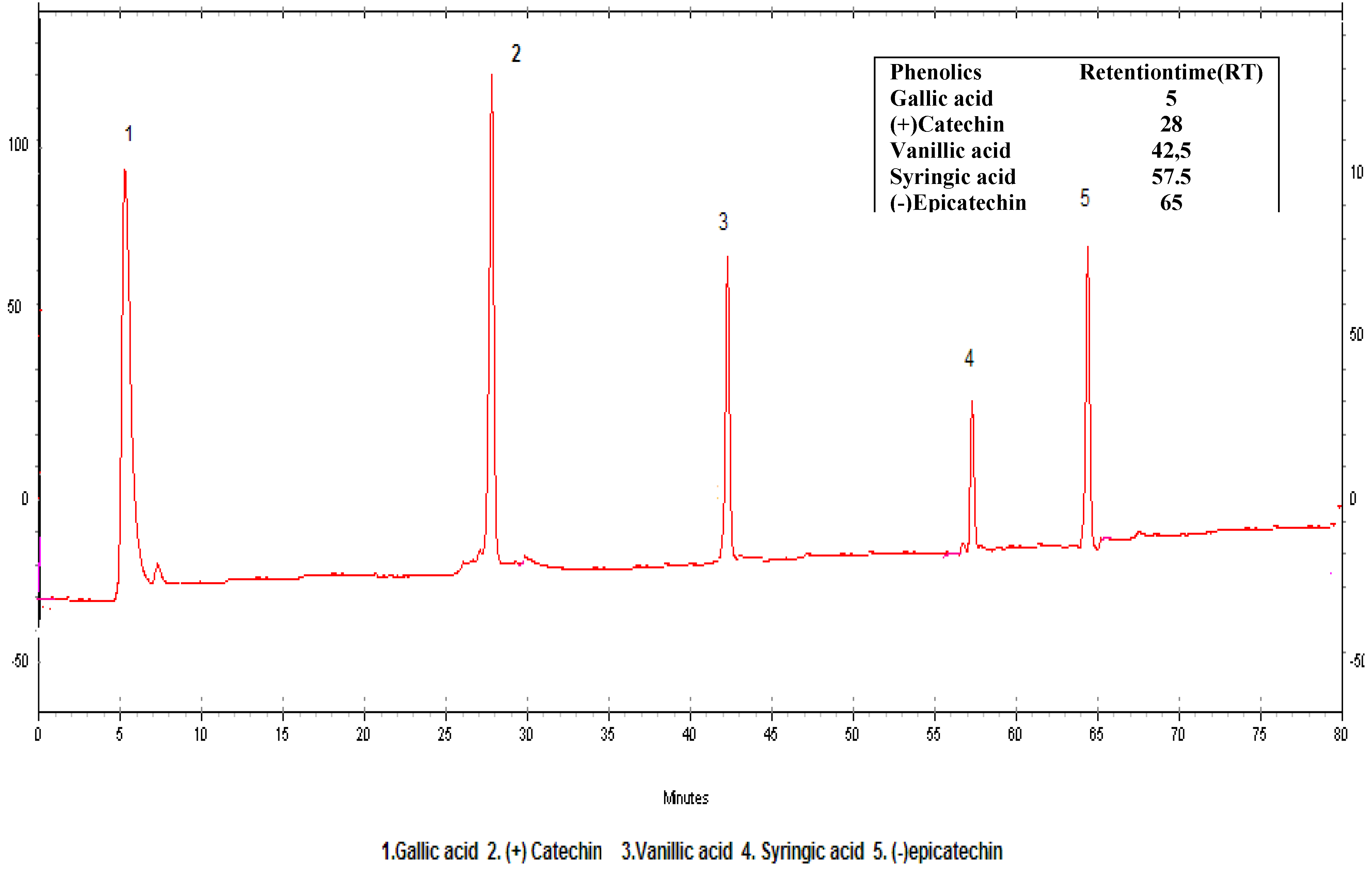
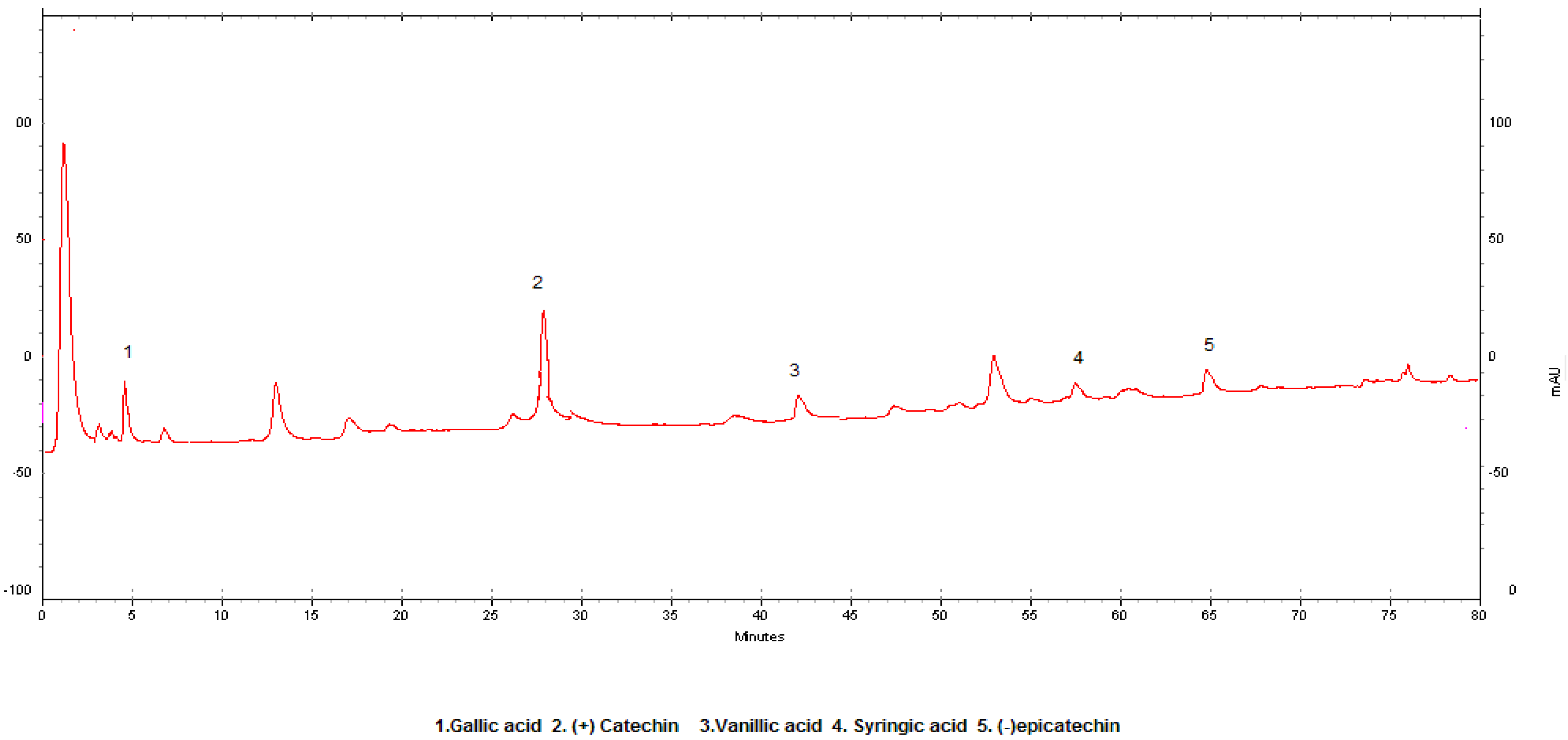
Results and Discussion
| Compound | LOD (mg/L) | LOQ(mg/L) | A | b | R2 | RSD |
|---|---|---|---|---|---|---|
| 1. Gallic acid | 0.025 | 0,078 | 64.021 | -5800 | 0.9996 | 1.2 |
| 2.(+)-Catechin | 0.075 | 0,200 | 31.572 | 765.06 | 0.9987 | 1.1 |
| 3.Vanilic acid | 0.050 | 0,150 | 68.958 | 4807.2 | 0.9999 | 3.7 |
| 4.Syringic acid | 0.010 | 0,035 | 93.157 | 2492.6 | 0.9997 | 3.2 |
| 5.(-)Epicatechin | 0.095 | 0,285 | 24.128 | 768.80 | 0.9998 | 0.8 |
| Variety | Origin of
Region | n | Gallic acid | (+)- Catechin | Vanilic acid | Syringic acid | (-)- Epicatechin |
|---|---|---|---|---|---|---|---|
| Kalecik karası | Central Anatolia | 3 | 40.85 ± 0.5452 c | 31.98 ±0.148 a | 4.66 ±0.0525 a | 4.39 ± 0.0225 c | 26.61 ± 0.1050 b |
| Kalecik karası | Thrace-Mürefte | 3 | 45.48 ± 0.1380 c | 33.98 ±0.148 a | 5.22 ± 0.110 a | 4.71 ± 0.095 b | 29.48 ± 0.3403 b |
| Kalecik karası | Aegean-Denizli | 3 | 42.71± 0.4430 c | 32.78 ± 0.143a | 4.99 ± 0.0329a | 4.52 ± 0.074 b | 27.98 ± 0.3402 b |
| Merlot | Thrace-Mürefte | 3 | 52.12 ± 0.023 b | 19.38 ± 0,04 c | 4.85 ± 0.0410 a | 4.82 ± 0.020 b | 27.10 ± 0.210 b |
| Merlot | Aegean-İzmir | 3 | 52.11 ± 0.021 b | 20.29 ± 0.122 c | 4.66 ± 0.051 b | 5.09 ± 0.0825 a | 26.37 ± 0.0850 c |
| Merlot | Aegean-Manisa | 3 | 51.99 ± 0.020b | 19.53 ± 0.122c | 4.73 ± 0.0410 b | 4.93 ± 0.0713b | 26.98 ± 0.0671 c |
| Cabernet Sauvignon | Thrace-Mürefte | 3 | 61.22 ± 1.090 a | 25.730 ± 0.600 b | 4.77 ±0.0500 a | 5.12 ± 0.0125 a | 51.91 ± 0.418 a |
| Cabernet Sauvignon | Central Anatolia | 3 | 61.56 ±0.187a | 20.23 ± 0.135c | 4.97 ± 0.0432a | 5.21 ± 0.0234a | 50.66 ± 0.387a |
| Cabernet Sauvignon | Aegean-Denizli | 3 | 68.430±0.05a | 24.351±0.130 b | 4.99 ±0.0275 a | 5.012±0.148 a | 58.93 ± 0.0250 a |
| Variety | Region | ACª
(mmol/L) | Total phenol
(mg/L GAE) |
|---|---|---|---|
| Kalecik karası | Central Anatolia | 15.8 | 1070 |
| Kalecik karası | Thrace-Mürefte | 16.3 | 1130 |
| Kalecik karası | Aegean-Denizli | 18.7 | 1420 |
| Merlot | Thrace-Mürefte | 15.8 | 1510 |
| Merlot | Aegean-İzmir | 17.6 | 1480 |
| Merlot | Aegean-Manisa | 17.1 | 1720 |
| Cabernet Sauvignon | Thrace-Mürefte | 18.1 | 2320 |
| Cabernet Sauvignon | Aegean-İzmir | 22.6 | 2410 |
| Cabernet Sauvignon | Aegean-Denizli | 19.3 | 2390 |
Experimental
Wine samples
Antioxidant capacities
Total Phenolics
Standards and Reagents
HPLC analysis
Statistical analysis
References
- Soleas, G.J.; Diamandis, E.P.; Goldberg, D.M. Wine as a biological fluid: history, production, and role in disease prevention. J. Clin. Lab. Anal. 1997, 11, 287–313. [Google Scholar] [CrossRef]
- Anli, E.; Vural, N.; Demiray, S. Trans-resveratrol and other phenolic compounds in Turkish red wines with HPLC. J. Wine Res. 2006, 17, 117–125. [Google Scholar] [CrossRef]
- Aviram, M.; Fuhrman, B. Wine flavonoids protect against LDL oxidation and atherosclerosis. Ann. NY Acad. Sci. 2002, 957, 146–161. [Google Scholar] [CrossRef]
- Landrault, N.; Poucheret, N.; Ravel, P.; Gasc, F.; Cros, G.; Teissedre, P.L. Antioxidant capacities and phenolics levels of French wines from different varieties and vintages. J. Agric. Food Chem. 2001, 49, 3341–3348. [Google Scholar] [CrossRef]
- Jang, M.; Cai, L.; Udenai, G.O.; Slowing, K.W.; Thomas, J.F.; Beecher, C.W.W.; Fong, H.H.S.; Farnswort, N.R.; Douglas Kinhorn, A.; Metha, R.G.; Moon, R.J.; Pezzuto, J.M. Cancer chemopreventive activity of resveratrol, a natural product derived from grapes. Science 1997, 257, 218–220. [Google Scholar]
- Tedesco, I.; Russo, M.; Russo, P.; Iacamino, G.; Russo, G.L.; Carraturo, A.; Faruolo, C.; Kop, P. Resveratrol, a phytoestrogen found in red wine. A possible explanation for the conundrum of the “French paradox”? J. Endocrinol. 1998, 138, 619–620. [Google Scholar]
- Ribéreau-Gayon, P.; Glories, Y.; Maujean, A.; Dubourdieu, D. The chemistry of wine Stabilization and Treatments. In Handbook of Enology; John Wiley and Sons Ltd.: West Sussex, England, UK, 2005; Vol. 2, pp. 157–162. [Google Scholar]
- Faustino, R.S.; Sobrattee, S.; Edel, A.L.; Pierce, G.N. Comparative analysis of the phenolic content of Chilean, Canadian and American merlot red wines. Mol. Cell. Biochem. 2003, 249, 11–19. [Google Scholar] [CrossRef]
- Gambelli, L.; Sanatorini, G.P. Polyphenol content of Italian red wine of different geographical areas. J. Food Comp. Anal. 2004, 17, 613–618. [Google Scholar] [CrossRef]
- Plumb, G.W.; de Pascaul-Teresa, S.; Santos-Buelga, C.; Cheynier, V.; Williamson, G. Antioxidant properties of catechins and proanthocyanides: effect of polymerisation, galloylation and glycosylation. Free Radic. Res. 1998, 29, 351–358. [Google Scholar] [CrossRef]
- Soleas, G.J; Grass, L.; Josephy, P.D.; Goldberg, D.M.; Diamandis, E.P. A comparison of the anticarcinogenic properties of four red wine polyphenols. Clin. Biochem. 2002, 35, 119–124. [Google Scholar] [CrossRef]
- Kerry, N.L.; Abbey, M. Red wine and fractionated phenolic compounds prepared from red wine inhibit low density lipoprotein oxidation in vitro. Atherosclerosis 1997, 1, 93–102. [Google Scholar] [CrossRef]
- Blackhurst, D.M.; Marais, A.D. Concomitant consumption of red wine and polyunsaturated fatty acids in edible oil does not influence the peroxidation status of chylomicron lipids despite increasing plasma catechin concentration. Nutri. Metab. Cardiovasc. Dis. 2006, 16, 550–558. [Google Scholar] [CrossRef]
- González-Manzano, S.; Rivas-Gonzalo, J. C.; Santos-Buelga, C. Extraction of flavan-3-ols from grape seed and skin into wine using simulated maceration. Anal. Chim. Acta 2004, 513, 283–289. [Google Scholar] [CrossRef]
- Anonymous. ETS Laboratories Technical Bulletin, 2000.
- Romero, I.; Páez, A.; Ferruelo, A.; Luján, M.; Berenguer, A. Polyphenols in red wine inhibit the proliferation and induce apoptosis of LNCaP cells. BJU Int. 89 2002, 950–954. [Google Scholar]
- Chantal, C.M.A.; Arnaud, B.; Bianca, C.H.L.; Kim, D.; Theo, J.C.V.B.; Marc, F. H.; Erik, A.L.B. Gallic Acid Antagonizes P-Selectin–Mediated Platelet–Leukocyte Interaction. Circulation 2005, 111, 106–112. [Google Scholar]
- Kampa, M.; Alexaki, V.I.; Notas, G.; Nifli, A-P.; Nistikaki, A.; Hatzoglou, A.; Bakogeorgou, E.; Kouimtzoglou, E.; Blekas, G.; Boskou, D.; Gravanis, A.; Castanas, E. Breast Cancer Res. 2004, 6, R63–R74. [CrossRef]
- Anli, R.E. History of wine culture in Anatolia during the Hittite Period. In Symposio Internacional de Historia e Civilizaçao da Vinha e do Douro; Porto-Lamego-Vila Real: Portugal, 2001; 10-12 Set. 15h, Sala 2,5; p. 2000. [Google Scholar]
- Goldberg, D.M.; Tsang, E.; Karumanchiri, A.; Diamandis, E.P.; Ng, E. Method to essay the concentration of biological interest in wines. Anal. Chem. 1996, 68, 1688–1694. [Google Scholar] [CrossRef]
- Frankel, E.N.; Waterhouse, A.L.; Teissedre, P.L. Principal phenolic phytochemicals in selected California wines and their antioxidant activity in inhibiting oxidation of human low-density lipoproteins. J. Agric. Food Chem. 1995, 43, 890–894. [Google Scholar] [CrossRef]
- Miller, N.C.; Rice-Evans, J.; Davies, M.J.; Gopinathan, V.; Milner, A. A novel method for measuring antioxidant capacity and application to monitoring the antioxidant status in premature neonates. Clin. Sci. 1993, 84, 407–412. [Google Scholar]
- Singleton, V. L.; Rossi, J. R. Colorimetry of total phenolics with phosphomolybdic–phosphotungstic acid. Am. J. Enol. Vitic. 1965, 16, 144–158. [Google Scholar]
- Proestos, C.; Bakogiannis, A.; Psarianos, C.; Koutinas, A.A.; Kanellaki, M.; Komaitis, M. High performance liquid chromatography analysis of phenolic substances in Greek wines. Food Control. 2005, 16, 319–323. [Google Scholar] [CrossRef]
- Sokal, R.R.; Rolf, F.J. Biometry. The principles and Practice of Statistics in Biological Research, 3rd Ed. ed; WH Freeman & Co: San Francisco, USA, 1995; pp. 334–337. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ertan Anli, R.; Vural, N. Antioxidant Phenolic Substances of Turkish Red Wines from Different Wine Regions. Molecules 2009, 14, 289-297. https://doi.org/10.3390/molecules14010289
Ertan Anli R, Vural N. Antioxidant Phenolic Substances of Turkish Red Wines from Different Wine Regions. Molecules. 2009; 14(1):289-297. https://doi.org/10.3390/molecules14010289
Chicago/Turabian StyleErtan Anli, R., and Nilüfer Vural. 2009. "Antioxidant Phenolic Substances of Turkish Red Wines from Different Wine Regions" Molecules 14, no. 1: 289-297. https://doi.org/10.3390/molecules14010289
APA StyleErtan Anli, R., & Vural, N. (2009). Antioxidant Phenolic Substances of Turkish Red Wines from Different Wine Regions. Molecules, 14(1), 289-297. https://doi.org/10.3390/molecules14010289




