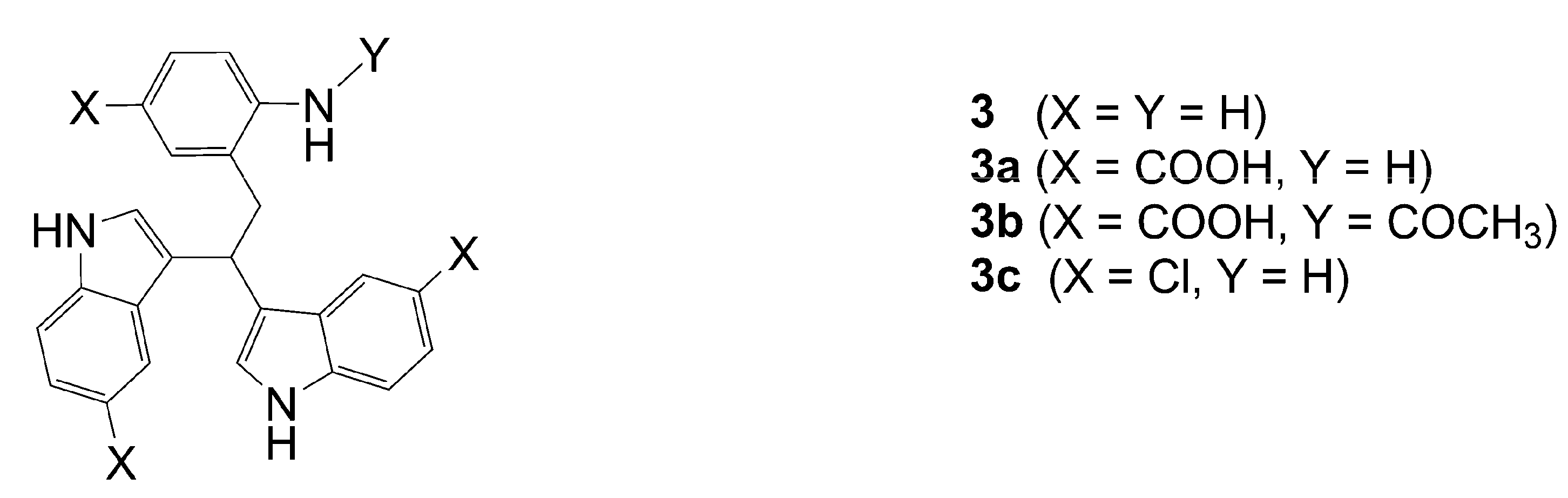Characterization data
2,3-Dihydro-2,3'-biindole-5,5'-dicarboxylic acid (2a): HRMS: M+H+ (C18H15N2O4) 323.1019, calculated 323.1032; M-H- (C18H13N2O4) 321.0861, calculated 321.0876; Elemental analysis: found, %: N 8.2, C 64.9, H 4.8; calculated for 3C18H14N2O4·2H2O, %: N 8.38, C 64.67, H 4.62.
1H-NMR (400 MHz, DMSO-d6, 25°C): δ 3.03 (dd, J = 16.0, 8.8 Hz, 1H, H-3), 3.43 (dd, J = 16.0, 9.2 Hz, 1H, H-3), 5.28 (t, J = 9.2 Hz, 1H, H-2), 6.50 (d, J = 8.0 Hz, 1H, H-7), 7.36 (d, J = 2.0 Hz, 1H, H-4), 7.53 (d, J = 8.4 Hz, 1H), 7.58 (m, 1H), 7.61 (dd, J = 8.0, 1.6 Hz, 1H, H-6), 7.81 (dd, J = 8.4, 1.6 Hz, 1H), 7.96 (m, 1H), 11.26 (d, J = 1.6 Hz, 1H, H-1), 11.79 (d, J = 2.0 Hz, 1H, H-1’), 12.3 (m, 2H, 2COOH)
3,3'-[2-(2-Amino-5-carboxyphenyl)ethane-1,1-diyl]bis(1H-indole-5-carboxylic acid (3a): HRMS: M+H+ (C27H22N3O6) 484.1504, calculated 484.1508; M-H- (C27H20N3O6) 482.1331, calculated 482.1352; Elemental analysis: found, %: N 7.7, C 58.5, H 5.4; calculated for C27H21N3O6·4H2O, %: N 7.56, C 58.37, H 5.26.
1H-NMR (270 MHz, DMSO-d6, 25°C): δ 3.22 (m, 2H, 2H-1’’), 4.93 (m, 1H, H-2’’), 6.58 (d, J = 8.6 Hz, 1H, H-3), 7.31 (d, J = 8.6 Hz, 2H, H-7’, H-7’), 7.33, 7.34 (2m, 2H, H-2’, H-2’), 7.35 (m, 1H, H-4), 7.40 (m, 1H, H-6), 7.59 (dd, J = 8.6 Hz, 1.6 Hz, 2H, H-6’, H-6’), 8.08 (d, J = 1.7 Hz, 2H, H-4’, H-4’), 11.14 (d, J = 1.9 Hz, 2H, H-1’, H-1’).
3-(2-(2-Amino-5-carboxyphenyl)-1-(2-mercaptoethylthio)ethyl)-1H-indole)-5-carboxylic acid (5a): HRMS: M+H+ (C20H21N2O4S2) 417.0957, calculated 417.0943; M-H- (C20H19N2O4S2) 415.0794, calculated 415.0787; Elemental analysis: found, %: N 6.5, C 57.8, H 5.0; calculated for C20H20N2O4S2, %: N 6.73, C 57.67, H 4.84.
1H-NMR (400 MHz, DMSO-d6, 25°C): δ 2.34(m, 1H, SH), 2.5 (m, 4H, 2H-1’’’, 2H-2’’’), 3.22 (m, 2H, 2H-1’’), 4.67(m, 1H, H-2’’), 6.61 (d, J = 8.3 Hz, 1H, H-3), 7.39 (d, J = 8.6 Hz, 1H, H-7’), 7.45 (d, J = 2.2 Hz, 1H, H-2’), 7.46 (dd, J = 8.3 Hz, 2.0 Hz, 1H, H-4), 7.53 (d, J = 2.0 Hz, 1H, H-6), 7.71 (dd, J = 8.6 Hz, 1.6 Hz, 1H, H-6’), 8.43 (d, J = 1.6 Hz, 1H, H-4’), 11.27 (d, J = 2.2 Hz, 1H, H-1’); 13C-NMR (100 MHz, DMSO-d6, 25°C): δ 24.8 (C-2’’’), 35.0 (C-1’’’), 37.2 (C-1’’), 40.0 (C-2’’), 112.0 (C-7’), 116.6 (C-3’), 119.0 (C-5), 121.5 (C-5’), 122.8 (C-4’), 123.1 (C-1), 123.1 (C-6’), 125.9 (C-3a’), 126.0 (C-2’), 129.6 (C-4), 132.8 (C-6), 139.6 (C-7a’), 150.0 (C-2), 168.1 (C-5a), 169.1 (C-5a’).
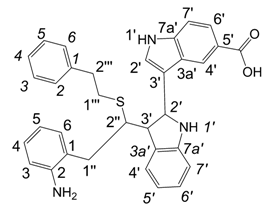
3,3'-{1,1'-[Ethane-1,2-diylbis(sulfane-diyl)]bis[2-(2-amino-5-carboxyphenyl)ethane-1,1-diyl]}bis(1H-indole-5-carboxylic acid) (6a): HRMS: M+H+ (C38H35N4O8S2) 739.1880, calculated 739.1896; M-H- (C38H33N4O8S2) 737.1733, calculated 737.1740; Elemental analysis: found, %: N 5.3, C 47.9, H 4.2; calculated for C38H34N4O8S2·2CF3COOH·5H2O, %: N 5.30, C 47.73, H 4.39.
1H-NMR (270 MHz, DMSO-d6, 25°C): δ 2.3 (m, 4H, 2H-1’’’, 2H-1’’’), 3.13 (m, 4H, 2H-1’’, 2H-1’’), 4.55 (m, 2H, H-2’’, H-2’’), 6.55 (d, J = 8.6 Hz, 2H, H-3, H-3), 7.30, 7.31 (2d, J = 2.3 Hz, 2H, H-2’, H-2’), 7.35 (d, J = 8.6 Hz, 2H, H-7’, H-7’), 7.42-7.64 (m, 2H, H-4, H-4), 7.43-7.66 (m, 2H, H-6, H-6), 7.68 (dd, J = 8.6 Hz, 1.6 Hz, 2H, H-6’, H-6’), 8.38, 8.39 (2d, J = 1.6 Hz, 2H, H-4’, H-4’), 11.18 (d, J = 2.3 Hz, 2H, H-1’, H-1’).
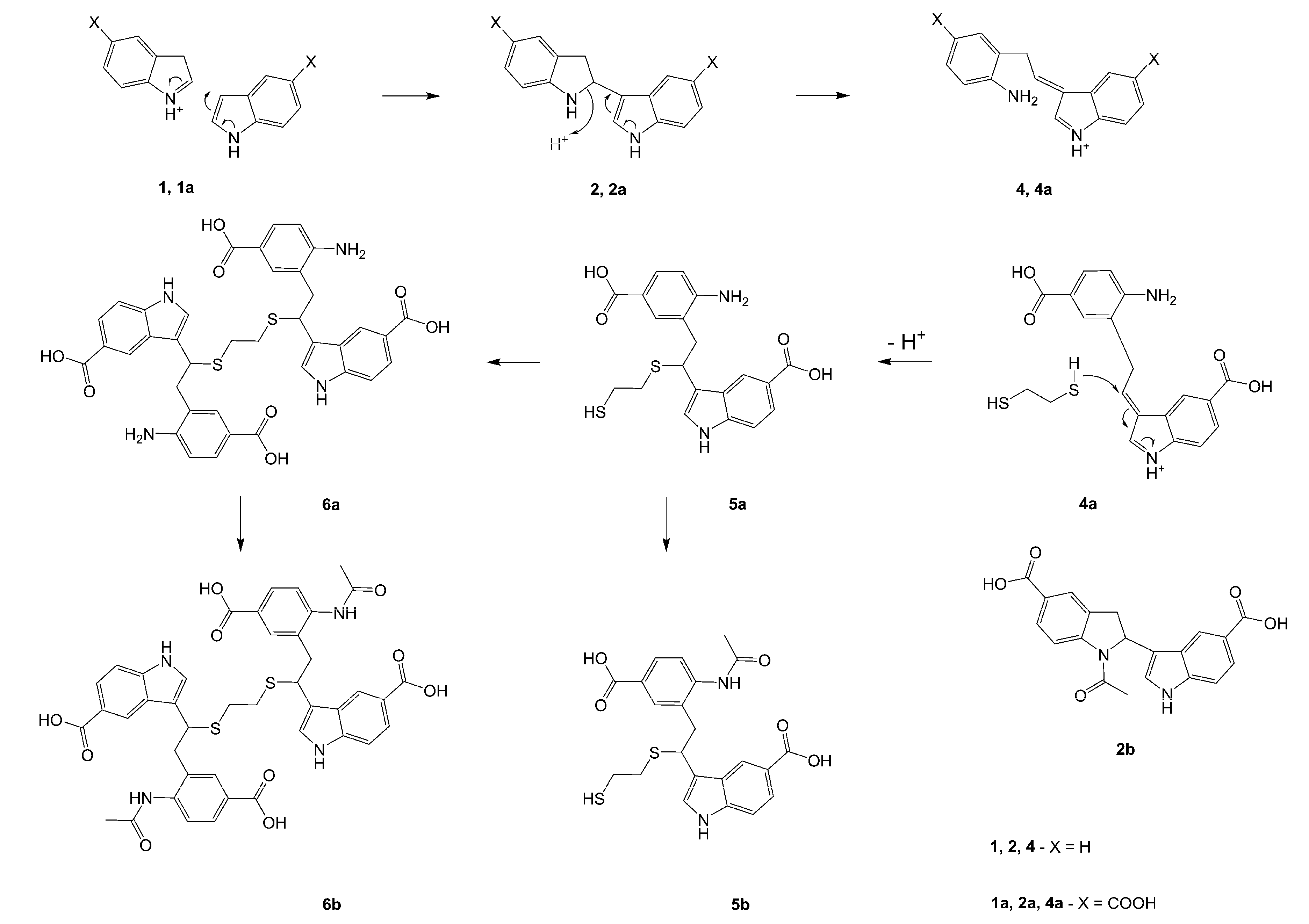
Procedure B: Compounds 2a, 3a, 5a and 6a were also obtained by a modification of Procedure A, whereby the crude product was dissolved in chloroform and applied onto a glass column filled with silica gel (pore size 60Å, 70-230 mesh). The column was eluted with chloroform-methanol mixture, gradually changing its proportions from 20:1 to 1:4, and thereafter the elution was made with pure methanol. Eluate fractions containing pure 3a and 5a were separately pooled and evaporated. White crystalline products were obtained. Isolated yield of 3a was 7%, and that of 5a was 25%. (however, determination of the reaction products by HPLC provided the following yields: 2a: 22%, 3a: 27%, 5a: 28% and 6a: 1.3%).
3a: Elemental analysis. Found, %: N 7.6, C 64.7, H 5.3. Calculated for 3C27H21N3O4·4MeOH, %: N 7.99. C 64.67, H 5.04.
2-(2,2-bis(5-Chloro-1H-indol-3-yl)ethyl)-4-chloroaniline (3c). Compound 3c was obtained from 5-chloroindole using Procedure A. Yield 63 %. HRMS: M+H+ (C24H19Cl3N3) 454.0637, calculated 454.0644; Elemental analysis: found, %: N 7.17, C 54.81, H 3.45; calculated for C24H18Cl3N3·CF3COOH, %: N 7.39, C 54.90, H 3.37.
1H-NMR (400 MHz, DMSO-d6, 25°C): δ 3.36 (d, J = 7.9 Hz, 2H, 2H-1’’), 4.89 (t, J = 7.9 Hz, 1H, H-2’’), 6.78 (m, 1H, H-3), 6.93 (m, 2H, H-4, H-6), 6.99 (dd, J = 8.6 Hz, 2.2 Hz, 2H, H-6’, H-6’), 7.29 (d, J = 8.6 Hz, 2H, H-7’, H-7’), 7.42 (d, 2H, H-2’, H-2’), 7.49 (d, J = 2.2 Hz, 2H, H-4’, H-4’), 10.99 (d, J = 2.4 Hz, 2H, H-1’, H-1’); 13C-NMR (67.5 MHz, DMSO-d6, 25°C): δ 32.0 (C-2’’), 35.7 (C-1’’), 113.4 (C-7’, C-7’), 118.2 (C-3’, C-3’), 118.8 (C-4’, C-4’), 120.1 (C-3), 121.2 (C-6’, C-6’), 123.3 (C-5’, C-5’), 124.9 (C-1, C-2’, C-2’), 126.8 (C-6), 128.2 (C-3a’, C-3a’), 129.8 (C-4), 130.8 (C-5), 135.4 (C-7a’, C-7a’), 140.0 (C-2).
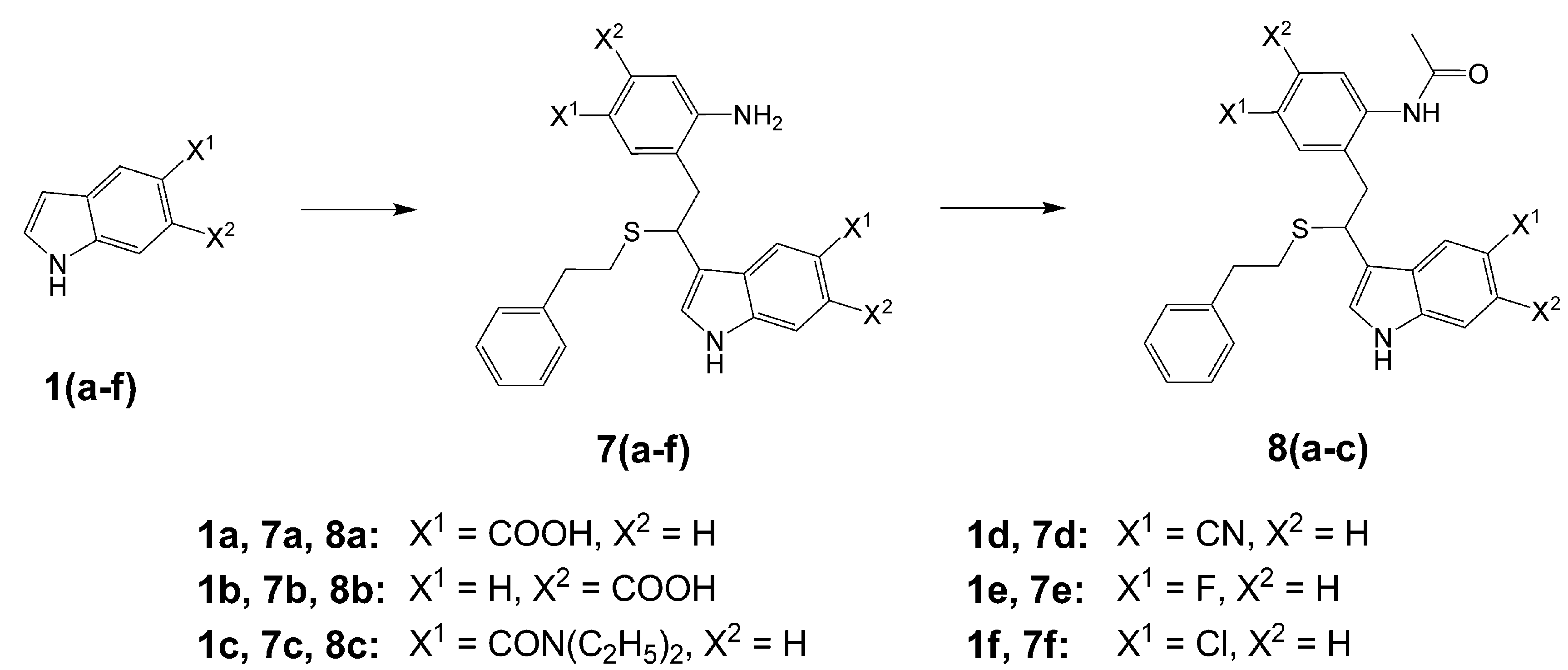
3-[2-(2-Amino-5-carboxyphenyl)-1-(phenethylthio)ethyl]-1H-indole-5-carboxylic acid (7a). This compound was prepared according to Procedure A using indole-5-carboxylic acid and 2-phenylethanethiol as starting materials. Yield 19%; HRMS: M+H+ (C26H25N2O4S) 461.1532, calculated 461.1535; Elemental analysis: found, %: N 5.82, C 65.44, H 5.31; calculated for C26H24N2O4S·H2O, %: N 5.85, C 65.25, H 5.48.
1H-NMR (270 MHz, DMSO-d6, 25°C): δ 2.39 and 2.58 (2m, 4H, 2H-1’’’, 2H-2’’’), 3.13-3.32 (m, 2H, 2H-1’’), 4.66 (t, J = 7.6 Hz, 1H, H-2’’), 6.59 (d, J = 8.5 Hz, 1H, H-3), 7.02 (XX´ part of AA´XX´system, 2H, H-2b, H-6b), 7.08-7.22 (m, 1H, H-4b), 7.08-7.22 (AA´ part of AA´XX´system, 2H, H-3b, H-5b), 7.37 (d, J = 8.6 Hz, 1H, H-7’), 7.45 (m, 2H, H-4, H-6), 7.53 (d, J = 2.3 Hz, 1H, H-2’), 7.69 (dd, J = 8.6 Hz, 1.6 Hz, 1H, H-6’), 8.43 (d, J = 1.6 Hz, 1H, H-4’), 11.28 (br s, 1H, H-1’).
3-[2-(2-Amino-4-carboxyphenyl)-1-(phenethylthio)ethyl]-1H-indole-6-carboxylic acid (7b). Prepared according to Procedure A from indole-6-carboxylic acid and 2-phenylethanethiol. Yield 32%; HRMS: M+H+ (C26H25N2O4S) 461.1548; calculated 461.1535. Elemental analysis: found, %: N 5.26, C 61.88, H 4.74; calculated for 2C26H24N2O4S·CF3COOH·H2O, %: N 5.32, C 61.59, H 4.88.
1H-NMR (400MHz, DMSO-d6, 25°C): δ 2.42 and 2.52-2.63 (2m, 4H, 2H-1’’’, 2H-2’’’), 3.20-3.32 (m, 2H, 2H-1’’), 4.69 (m, 1H, H-2’’), 6.97 (d, J = 8.0 Hz, 1H, H-H-6), 7.02 (XX´ part of AA´XX´system, 2H, H-2b, H-6b), 7.02 (m, 1H, H-5), 7.09-7.14 (m, 1H, H-4b), 7.16-7.20 (AA´ part of AA´XX´system, 2H, H-3b, H-5b), 7.29 (d, J = 1.6 Hz, 1H, H-3), 7.53 (d, J = 2.4 Hz, 1H, H-2’), 7.58 (dd, J = 8.4, 1.6 Hz, 1H, H-5’), 7.77 (d, J = 8.4 Hz, 1H, H-4’), 7.96 (dd, J = 1.6, 0.8 Hz, 1H, H-7’), 11.26 (d, J = 2.8 Hz, 1H, H-1’).
3-{2-[2-Amino-5-(diethylcarbamoyl)phenyl]-1-(phenethylthio)ethyl}-N,N-diethyl-1H-indole-5-carbox-amide (
7c). Compound
7c was prepared according to Procedure A using indole-5-carboxylic acid diethylamide [
24] and 2-phenylethanethiol as starting materials. Yield 30%; HRMS: M+H
+ (C
34H
43N
4O
3S) 571.3101; calculated 571.3106; Elemental analysis: found, %: N 9.47, C 69.97, H 7.37; calculated for 6C
34H
42N
4O
2S·CF
3COOH, %: N 9.50, C 69.92, H 7.21.
1H-NMR (400 MHz, DMSO-d6, 25°C): δ 0.87 (m, 6H, 2CH3), 1.09 (m, 6H, 2CH3), 2.52-2.63 (m, 4H, 2H-1’’’, 2H-2’’’), 3.02 (m, 4H, 2CH2 NEt), 3.22 (m, 2H, H-1’’), 3.31 (m, 4H, 2CH2 NEt), 4.64(m, 1H, H-2’’), 6.69 (d, J = 8.0 Hz, 1H, H-3), 6.77 (d, J = 2.0 Hz, 1H, H-6), 6.91 (dd, J = 8.0, 2.0 Hz, 1H, H-4), 7.00 (XX´ part of AA´XX´system, 2H, H-2b, H-6b), 7.04 (dd, J = 8.4, 1.6 Hz, 1H, H-6’), 7.09-7.13 (m, 1H, 1H-4b), 7.15-7.19 (AA´ part of AA´XX´system, 2H, H-3b, H-5b), 7.33 (d, J = 2.4 Hz, 1H, H-2’), 7.34 (d, J = 8.4 Hz, 1H, H-7’), 7.71 (m, 1H, 1H-4’), 11.08 (d, J = 2.4 Hz, 1H, H-1’).
3-[2-(2-Amino-5-cyanophenyl)-1-(phenethylthio)ethyl]-1H-indole-5-carbonitrile (7d). 7d was prepared according to Procedure A from 5-cyanoindole and 2-phenylethanethiol. Yield 15%; HRMS: M+H+ (C26H23N4S) 423.1674; calculated 423.1643.
1H-NMR (400MHz, DMSO-d6, 25°C): δ 2.43 and 2.52-2.64 (2m, 4H, 2H-1’’’, 2H-2’’’), 3.16 (dd, J = 8.0, 2.0 Hz, 2H, 2H-1’’), 4.70 (t, J = 8.0 Hz, 1H, H-2’’), 6.59 (d, J = 8.8 Hz, 1H, H-3), 7.02 (XX´ part of AA´XX´system, 2H, H-2b, H-6b), 7.02 (m, 1H), 7.12-7.23 (AA´ part of AA´XX´system, 2H, H-3b, H-5b), 7.12-7.23 (m, 1H, H-4b), 7.32 (d, J = 2.0 Hz, 1H), 7.36-7.44 (m, 1H), 7.47 (m, 1H), 7.54 (d, J = 2.4 Hz, 1H), 11.48 (br s, 1H, H-1’).
4-Fluoro-2-[2-(5-fluoro-1H-indol-3-yl)-2-(phenethylthio)ethyl]aniline (7e). 7e was prepared according to Procedure A using 5-fluoroindole and 2-phenylethanethiol as starting materials. Yield 2%; HRMS: M+H+ (C24H23F2N2S) 409.1533; calculated 409.1550.
4-Chloro-2-[2-(5-chloro-1H-indol-3-yl)-2-(phenethylthio)ethyl]aniline (7f). 7f was prepared according to Procedure A from 5-chloroindole and 2-phenylethanethiol. Yield 5%; HRMS: M+H+ (C24H23Cl2N2S) 441.0958; calculated 441.0959; Elemental analysis: found, %: N 6.09, C 65.43, H 4.96; calculated for C24H22Cl2N2S, %: N 6.35, C 65.30, H 5.02.
1H-NMR (400MHz, DMSO-d6, 25°C): δ 2.47 and 2.53 (2m, 2H, 2H-1’’’), 2.62 (m, 2H, 2H-2’’’), 3.14 (m, 2H, 2H-1’’), 4.62 (m, 1H, 1H-2’’), 6.56 (d, J = 8.8 Hz, 1H, H-3), 6.83 (dd, J = 8.8, 2.8 Hz, 1H, H-4), 6.91 (d, J = 2.8 Hz, 1H, H-6), 7.02-7.06 (XX´ part of AA´XX´system, 2H, H-2b, H-6b), 7.02-7.06 (m, 1H, H-6’), 7.12 (m, 1H, H-4b), 7.17-7.22 (AA´ part of AA´XX´system, 2H, H-3b, H-5b), 7.32 (d, J = 8.4 Hz, 1H, H-7’), 7.41 (d, J = 2.4 Hz, 1H, H-2’), 7.73 (d, J = 2.0 Hz, 1H, H-4’), 11.09 (br s, 1H, H-1’).
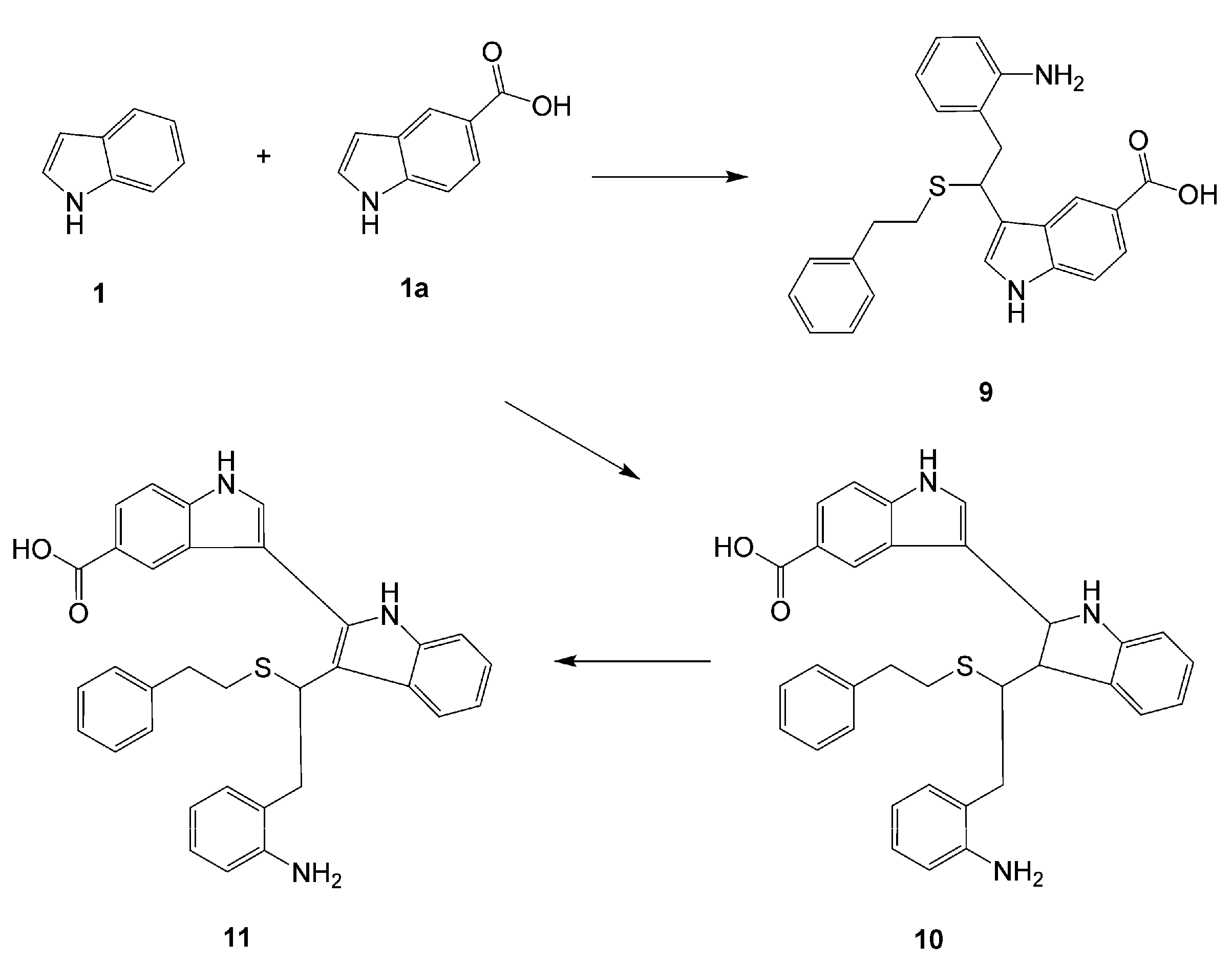
3-[2-(2-Aminophenyl)-1-(phenethylthio)ethyl]-1H-indole-5-carboxylic acid (9). Compound 9 was prepared according to Procedure A using indole, indole-5-carboxylic acid (equimolar quantities) and 2-phenylethanethiol as starting materials. Yield 19%; HRMS: M+H+ 417.1618. C25H25N2O2S; calculated 417.1636. Elemental analysis: found, %: N 5.94, C 64.41, H 5.10; calculated for 2C25H24N2O2S· CF3COOH·H2O, %: N 5.81, C 64.71, H 5.33.
1H-NMR (500 MHz, DMSO-d6, 25°C): δ 2.65 (m, 2H, 2H-2’’’), 3.31, 3.35 (m, 2H, 2H-1’’’), 3.31, 3.35 (m, 2H, 2H-1’’), 4.74 (m, 1H, H-2’’), 6.79 (m, 1H, H-3), 6.96(m, 1H, H-5), 7.03 (m, 1H, H-6), 7.05 (m, 1H, H-4), 7.05 (XX´ part of AA´XX´system, 2H, H-2b, H-6b), 7.14 (m, 1H, H-4b), 7.20 (AA´ part of AA´XX´system, 2H, H-3b, H-5b), 7.40 (d, J = 8.6 Hz, 1H, H-7’), 7.46 (d, J = 2.3 Hz, 1H, H-2’), 7.71 (dd, J = 8.6 Hz, 1.5 Hz, 1H, H-6’), 8.46 (s, 1H, H-4’), 11.28 (d, J = 1.8 Hz, 1H, H-1’). 13C NMR (125 MHz, DMSO-d6, 25°C): δ 31.1 (C-1’’’), 34.9 (C-2’’’), 35.6 (C-1’’), 38.7 (C-2’’), 110.6 (C-7’), 115.2 (C-3’), 118.4 (C-5), 120.2 (C-5’), 121.1 (C-3), 121.4 (C-6’), 121.8 (C-4’), 124.6 (C-2’), 124.7 (C-3a’), 125.2 (C-4b), 126.4 (C-4), 127.4 (C-3b, C-5b), 127.6 (C-2b, C-6b), 129.8 (C-6), 129.9 (C-1), 138.2 (C-7a’), 139.8 (C-1b), 150.0 (C-2), 167.7 (C-5a’).
3-[2-(2-Aminophenyl)-1-(phenethylthio)ethyl]-2,3-dihydro-1H,1'H-2,3'-biindole-5'-carboxylic acid (10). This compound was prepared according to Procedure A using indole, indole-5-carboxylic acid (equimolar quantities) and 2-phenylethanethiol. Yield 1%; HRMS: M+H+ (C33H32N3O2S) 534.2212; calculated 534.2215.
1H-NMR (500 MHz, DMSO-d6, 25°C): δ 2.23 (m, 2H, 2H-1’’’), 2.30, 2.37 (m, 2H, 2H-2’’’), 2.65 (m, 1H, Ha-1’’), 2.94 (m, 1H, Hb-1’’), 3.54 (m, 1H, H-2’’), 3.98 (m, 1H, H-3’), 5.24 (d, J = 7.6 Hz, 1H, H-2’), 6.64 (d, J = 7.5 Hz, 1H, H-7’), 6.71 (m, 1H, H-5’), 6.72 (m, 1H, H-3), 6.78 (XX´ part of AA´XX´system, 2H, H-2, H-6), 6.91(m, 1H, H-5), 6.93 (m, 1H, H-4), 7.05 (m, 1H, H-6), 7.06 (m, 1H, H-6’), 7.11 (AA´ part of AA´XX´system, 2H, H-3, H-5), 7.14 (m, 1H, H-4), 7.31 (d, J = 7.3 Hz, 1H, H-4’), 7.35 (m, 1H, H-2’), 7.45 (d, J = 8.6 Hz, 1H, H-7’), 7.74 (dd, J = 8.6 Hz, 1.5 Hz, 1H, H-6’), 8.26 (s, 1H, H-4’), 11.35 (m, 1H, H-1’), 12.36 (br, 1H, COOH); 13C-NMR (125 MHz, DMSO-d6, 25°C): δ 32.5 (C-1’’’), 34.4 (C-1’’), 34.5 (C-2’’’), 47.1 (C-2’’), 53.1 (C-3’), 57.9 (C-2’), 108.5 (C-7’), 110.6 (C-7’), 117.2 (C-5’), 117.5 (C-5), 117.8 (C-3’), 120.3 (C-5’), 121.6 (C-4’), 121.8 (C-6’), 123.7 (C-4’), 124.4 (C-2’), 124.5 (C-3a’), 125.2 (C-4), 126.9 (C-6), 126.9 (C-6’), 127.3 (C-3, 5), 127.5 (C-2, 6), 130.5 (C-4), 138.6 (C-7a’), 139.6 (C-1), 167.5 (C-COOH).
3-[2-(2-Aminophenyl)-1-(phenethylthio)ethyl]-1H,1'H-2,3'-biindole-5'-carboxylic acid (11). Compound 11 was synthesized from 10 (5 mg) when the latter was dissolved in DMSO-d6 (0.7 mL) and the solution was allowed to stand in a NMR tube at room temperature for one week.
1H-NMR (500 MHz, DMSO-d6, 25°C): δ 2.21, 2.30 (m, 4H, 2H-1’’’, 2H-2’’’), 3.39 (m, 1H, Ha-1’’), 3.45 (m, 1H, Hb-1’’), 4.64 (dd, J = 8.9 Hz, 5.9 Hz, 1H, H-2’’), 6.55 (XX´ part of AA´XX´system, 2H, H-2, H-6), 6.70 (d, J = 7.4 Hz, 1H, H-6), 6.80 (m, 1H, H-2’), 6.94 (m, 1H, H-4), 6.96 (AA´ part of AA´XX´system, 2H, H-3, H-5), 7.03 (m, 1H, H-5), 7.03 (m, 1H, H-4), 7.07 (m, 1H, H-5’), 7.13 (m, 1H, H-6’), 7.42 (d, J = 8.0 Hz, 1H, H-7’), 7.48 (d, J = 8.6 Hz, 1H, H-7’), 7.79 (dd, J = 8.6 Hz, 1.5 Hz, 1H, H-6’), 8.02 (d, J = 7.9 Hz, 1H, H-4’), 8.36 (s, 1H, H-4’), 11.20 (m, 1H, H-1’), 11.74 (m, 1H, H-1’), 12.50 (br, 1H, COOH); 13C-NMR (125 MHz, DMSO-d6, 25°C): δ 31.5 (C-1’’’), 35.1 (C-1’’, 2’’’), 39.9 (C-2’’), 107.9 (C-3’), 109.6 (C-3’), 110.7 (C-7’, 7’), 117.7 (C-5’), 119.4 (C-4’), 120.2 (C-6’), 121.4 (C-5’), 121.9 (C-4’), 122.3(C-6’), 125.0 (C-4), 125.2 (C-3a’), 125.3 (C-2’), 125.5 (C-3a’), 126.8 (C-4, 5), 127.2 (C-3, 5), 127.3 (C-2, 6), 129.6 (C-2), 129.7 (C-6), 130.6 (C-1, 2’), 136.5 (C-7a’), 137.9 (C-7a’), 139.4 (C-1), 167.5 (C-COOH).
Procedure C. Preparation of 3,3'-{1,1'-[ethane-1,2-diylbis(sulfanediyl)]bis[2-(2-acetamido-5-carboxyphenyl)ethane-1,1-diyl]}bis(1H-indole-5-carboxylic acid) (6b). Compound 6a (6.6 mg, 6.24 µmol) was dissolved in DMF (180 µL) and acetic anhydride (30 µL, 318 µmol) was added. The mixture was allowed to stand for 40 h, diluted with water to 1 mL volume, centrifuged and the clear solution in several portions applied onto an HPLC semipreparative column (10 x 250 mm, Vydac RP C18 90Å Pharmaceutical 201HS1010), eluent - 22 % MeCN in water + 0.1% TFA, flow 5 mL/min, detection at 280 nm. Eluate fractions containing pure putative 6b were pooled and freeze-dried. Yield ofoff-white powder was 3.4 mg (57%); HRMS: M+H+ (C42H39N4O10S2) 823.2129; calculated 823.2107. M-H- (C42H37N4O10S2) 821.1979; calculated 821.1951; Elemental analysis: found, %: N 6.1, C 54.3, H 4.6; calculated for 3C42H38N4O10S2·2CF3COOH·9H2O, %: N 5.88. C 54.62, H 4.72.
1H-NMR (500 MHz, DMSO-d6, 25°C): δ 1.88, 1.90 (2s, 6H, H-2c, H-2c), 2.3 – 2.5 (m, 4H, 2H-1’’’, 2H-1’’’), 3.25 - 3.40 (m, 4H, 2H-1’’, 2H-1’’), 4.42, 4.44 (2m, 2H, H-2’’, H-2’’), 7.19 (m, 2H, H-2’, H-2’), 7.33 (dd, J = 8.5 Hz, 2.7 Hz, 2H, H-7’, H-7’), 7.45 (d, J = 8.6 Hz, 2H, H-3, H-3), 7.64 (m, 2H, H-4, H-4), 7.66 (m, 2H, H-6, H-6), 7.68 (m, 2H, H-6’, H-6’), 8.38, 8.39 (2m, 2H, H-4’, H-4’), 9.35, 9.36 (2s, 2H, H-2a, H-2a), 11.14 (m, 2H, H-1’, H-1’), 12.5 (br, 2H, H-5a, H-5a); 13C NMR (125 MHz, DMSO-d6, 25°C): δ 22.4(C-2c, C-2c), 30.1 (C-1’’’, C-1’’’), 36.3 (C-1’’, C-1’’), 40.5 (C-2’’, C-2’’), 110.6 (C-7’, C-7’), 114.5 (C-3’, C-3’), 120.4 (C-5’, C-5’), 121.6 (C-4’, C-4’), 126.1 (C-5, C-5), 121.8 (C-6’, C-6’), 124.3 (C-3a’, C-3a’), 124.6 (C-2’, C-2’), 126.9 (C-4, C-4), 131.0 (C-6, C-6), 132.3 (C-1, C-1), 138.6 (C-7a’, C-7a’), 140.0 (C-2, C-2), 166.2 (C-5a, C-5a), 167.8 (C-5a’, C-5a’), 167.8 (C-2b, C-2b).
The following compounds were similarly prepared by Procedure C:
1-Acetyl-2,3-dihydro-1H,1'H-2,3'-biindole-5,5'-dicarboxylic acid (2b). From 2a. HRMS: M+H+ (C20H17N2O5) 365.1138; calculated 365.1137; M-H- (C20H15N2O5) 363.0961; calculated 363.0981; Elemental analysis: found, %: N 6.3, C 56.1, H 4.2; calculated for 2C20H16N2O5·CF3COOH·3H2O, %: N 6.25. C 56.25, H 4.38.
1H-NMR (400 MHz, DMSO-d6, 25°C): δ 2.07 (s, 3H, CH3), 3.01 (d, J = 16.0 Hz, 1H, H-3), 3.79 (dd, J = 16.0, 10.4 Hz, 1H, H-3), 6.04 (dd, J = 10.0, 2.0 Hz, 1H, H-2), 7.17 (d, J = 2.0 Hz, 1H, H-2’), 7.40 (d, J = 8.8 Hz, 1H, H-7), 7.69 (dd, J = 8.8, 1.6 Hz, 1H, H-6), 7.77 (d, J = 1.6 Hz, 1H, H-4), 7.88 (dd, J = 8.4, 2.0 Hz, 1H, H-6’), 7.98 (s, 1H, H-4’), 8.18 (m, 1H, H-7’), 11.34 (br s, 1H, H-1’).
3,3'-[2-(2-Acetamido-5-carboxyphenyl)ethane-1,1-diyl]bis(1H-indole-5-carboxylic acid) (3b). From 3a. HRMS: M+H+ (C29H24N3O7) 526.162, calculated 526.1614. M-H- (C29H22N3O7) 524.1467, calculated 524.1458; Elemental analysis: found, %: N 6.2, C 53.4, H 4.5; calculated for C29H23N3O4·CF3COOH·3H2O, %: N 6.06. C 53.68, H 4.36.
1H-NMR (400 MHz, DMSO-d6, 25°C): δ 1.88 (s, 3H, CH3), 3.54 (d, J = 7.6 Hz, 2H, 2H-1’’), 4.83 (dd, J = 7.6, 7.6 Hz, 1H, 1H-2’’), 7.27 (d, J = 2.4 Hz, 2H, H-1’, H-1’), 7.31 (d, J = 8.8 Hz, 2H, H-7’, H-7’), 7.48 (d, J = 8.4 Hz, 1H, H-3), 7.59 (dd, J = 8.8, 1.6 Hz, 2H, H-6’, H-6’), 7.61 (dd, J = 8.4, 2.0 Hz, 1H, H-4), 7.69 (d, J = 2.0 Hz, 1H, H-6), 8.11 (d, J = 1.6 Hz, 2H, H-4’, H-4’), 9.31 (s, 1H, H-2a), 11.13 (d, J = 2.4 Hz, 2H, H-1’, H-1’).
3-[2-(2-Acetamido-5-carboxyphenyl)-1-(2-mercaptoethylthio)ethyl]-1H-indole-5-carboxylic acid (5b). From 5a. HRMS: M+H+ (C22H23N2O5S2) 459.1037; calculated 459.1048; Elemental analysis: found, %: N 5.70, C 56.39, H 4.81; calculated for 8C22H22N2O5S2·CF3COOH, %: N 5.92. C 56.52, H 4.72.
1H-NMR (400 MHz, DMSO-d6, 25°C): δ 1.95 (s, 3H, CH3), 2.34(m, 1H, SH), 2.46 and 2.53 (2m, 4H, 2H-1’’’ and 2H-2’’’), 3.34 (m, 1H, 1H-1’’), 3.42 (dd, J = 14.4, 8.4 Hz, 1H, 1H-1’’), 4.49 (m, 1H, 1H-2’’), 7.31 (d, J = 2.4 Hz, 1H, H-2’), 7.36 (d, J = 8.8 Hz, 1H, H-7’), 7.45 (d, J = 8.4 Hz, 1H, H-3), 7.67 (m, 1H, H-4), 7.69 (dd, J = 8.4, 1.6 Hz, 1H, H-6’), 7.73 (d, J = 1.6 Hz, 1H, H-6), 8.41 (d, J = 1.6 Hz, 1H, H-4’), 9.44 (s, 1H, H-2a), 11.23 (d, J = 2.0 Hz, 1H, H-1’).
3-[2-(2-Acetamido-5-carboxyphenyl)-1-(phenethylthio)ethyl]-1H-indole-5-carboxylic acid (8a). From 7a. HRMS: M+H+ (C28H27N2O5S) 503.1636; calculated 503.1640; Elemental analysis: found, %: N 5.17, C 66.28, H 5.44; calculated for 3C28H26N2O5S·H2O, %: N 5.51. C 66.12, H 5.28.
1H-NMR (270 MHz, DMSO-d6, 25°C): δ 1.92 (s, 3H, CH3), 2.55 and 2.60 (2m, 4H, 2H-1’’’, 2H-2’’’), 3.30-3.49 (m, 2H, 2H-1’’), 4.48 (m, 1H, H-2’’), 7.03 (XX´ part of AA´XX´system, 2H, H-2b, H-6b), 7.08-7.22 (m, 1H, H-4b), 7.08-7.22 (AA´ part of AA´XX´system, 2H, H-3b, H-5b), 7.32 (d, J = 2.3 Hz, 1H, H-2’), 7.37 (d, J = 8.6 Hz, 1H, H-7’), 7.47 (d, J = 8.3 Hz, 1H, H-3), 7.69 (m, 2H, H-4, H-6’), 7.75 (d, J = 1.3 Hz, 1H, H-6), 8.43 (d, J = 1.3 Hz, 1H, H-4’), 9.46 (br s, 1H, H-2a), 11.25 (d, J = 2.3 Hz, 1H, H-1’).
3-[2-(2-Acetamido-4-carboxyphenyl)-1-(phenethylthio)ethyl]-1H-indole-6-carboxylic acid (8b). From 7b. HRMS: M+H+ (C28H27N2O5S) 503.1656; calculated 503.1640; Elemental analysis: found, %: N 5.30, C 65.24, H 5.17; calculated for 4C28H26N2O5S·3H2O, %: N 5.43. C 65.16, H 5.37.
1H-NMR (270 MHz, DMSO-d6, 25°C): δ 1.97 (s, 3H, CH3), 2.54 and 2.61 (2m, 4H, 2H-1’’’, 2H-2’’’), 3.31-3.51 (m, 2H, 2H-1’’), 4.50 (m, 1H, H-2’’), 7.04 (XX´ part of AA´XX´system, 2H, H-2b, H-6b), 7.09-7.24 (m, 1H, 4b), 7.09-7.24 (AA´ part of AA´XX´system, 2H, H-3b, H-5b), 7.09-7.24 (m, 1H, H-6), 7.42 (d, J = 2.6Hz, 1H, H-2’), 7.51 (dd, J = 7.9, 2.0 Hz, 1H, H-5), 7.57(dd, J = 8.3, 1.6 Hz, 1H, H-5’), 7.73 (d, J = 8.3 Hz, 1H, H-4’), 7.84 (d, J = 2.0 Hz, 1H, H-3), 7.96 (d, J = 1.6 Hz, 1H, H-7’), 9.48 (br s, 1H, H-2a), 11.27 (d, J = 2.3 Hz, 1H, H-1’).
3-{2-[2-Acetamido-5-(diethylcarbamoyl)phenyl]-1-(phenethylthio)ethyl}-N,N-diethyl-1H-indole-5-carboxamide (8c). From 7c. HRMS: M+H+ (C36H45N5O3S) 613.3205; calculated 613.3212; Elemental analysis: found, %: N 8.63, C 69.01, H 7.03; calculated for 6C36H44N5O3S·CF3COOH H2O, %: N 8.83. C 68.74, H 7.07.
1H-NMR (400 MHz, DMSO-d6, 25°C): δ 0.74 and 1.08 (2m, 12H, 4CH3), 1.99 (s, 3H, COCH3), 2.51-2.66 (m, 4H, 2H-1’’’, 2H-2’’’), 3.24-3.44 (m, 10H, 4CH2 NEt, 2H-1’’), 4.46 (dd, J = 8.8, 6.4 Hz, 1H, H-2’’), 6.89 (d, J = 2.0 Hz, 1H, H-6), 7.02-7.06 (XX´ part of AA´XX´system, 2H, H-2b, H-6b), 7.02-7.06 (m, 2H, H-4, H-6’), 7.09-7.13 (m, 1H, H-4b), 7.16-7.20 (AA´ part of AA´XX´system, 2H, H-3b, H-5b), 7.23 (d, J = 2.4 Hz, 1H, H-2’), 7.31 (d, J = 8.0 Hz, 1H, H-3), 7.32 (d, J = 8.4 Hz, 1H, H-7’), 7.68 (m, 1H, H-4’), 9.45 (br s, 1H, H-2a), 11.06 (d, J = 2.4 Hz, 1H, H-1’).