Anthocyanin Characterization of Pilot Plant Water Extracts of Delonix regia Flowers
Abstract
:Introduction
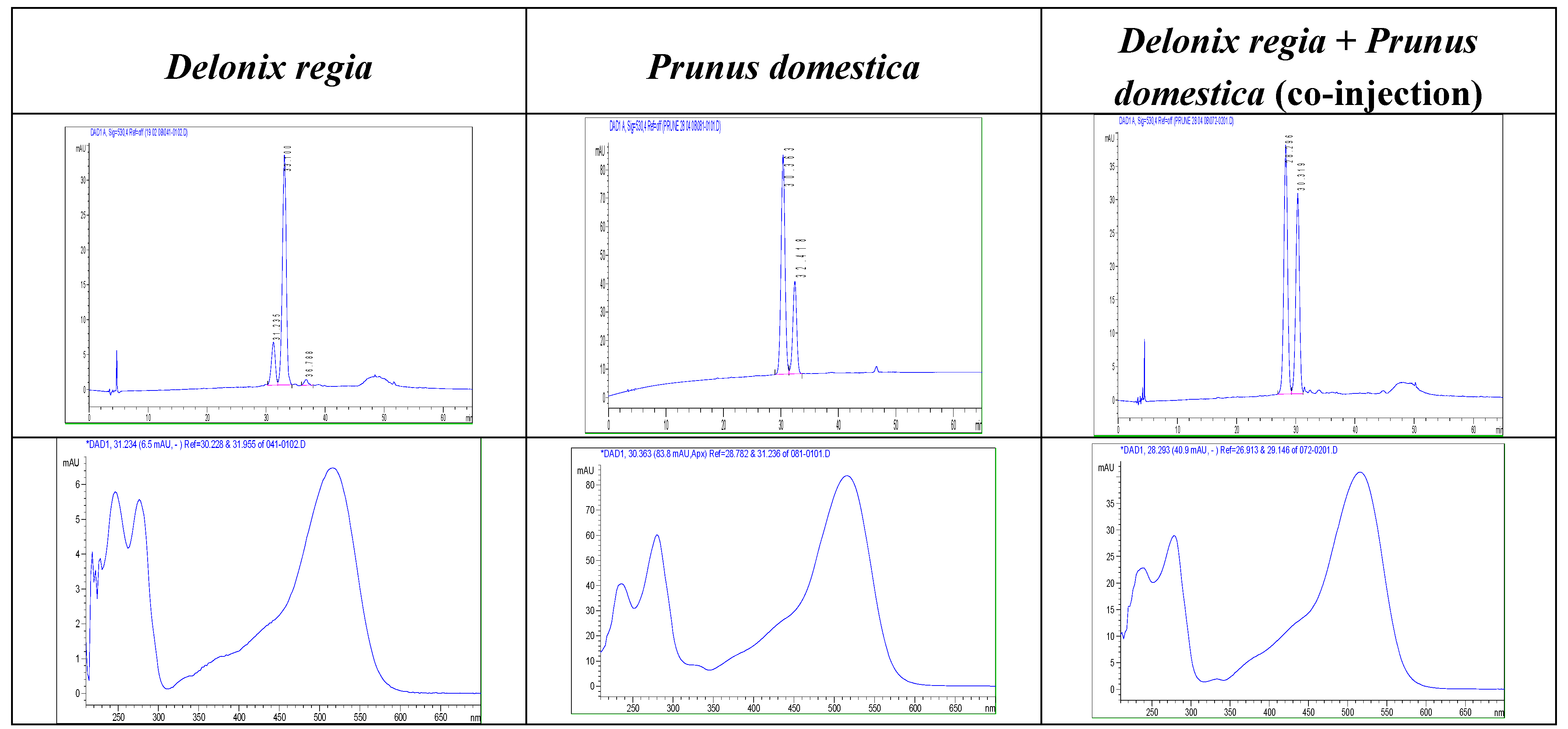
Results and Discussion

| HPLC peak number | RT (min) | λmax (nm) | [M+H] + m/z | [M-X] + m/z | Identified anthocyanin | Ref. number |
|---|---|---|---|---|---|---|
| 1 | 16.5 | 516 | 449 | 287 [M-162] | cyanidin 3-O-glucoside | [4] |
| 2 | 19.1 | 516 | 595 | 449 [M-146] (a) 287 [M-162] (b) | cyanidin 3-O-rutinoside | - |
| 3 | 23.1 | 506 | 579 | 433 [M-146] (a) 271 [M-162] (b) | pelargonidin 3-O-rutinoside | - |
| Identified Anthocyanins | HPCL-DAD UV-vis spectra | LC-MS-ESI fragmentations | |
|---|---|---|---|
| cyanidin 3-O-glucoside |  |  | MS1 |
| cyanidin 3-O-rutinoside |  |  | MS2 |
| pelargonidin 3-O-rutinoside |  |  | MS2 |
Experimental
Biological Material
Extraction of anthocyanins
| Anthocyanins | Molecular structures | mg/L a |
|---|---|---|
| cyanidin 3-O-glucoside |  | 3.4±0.2 |
| cyanidin 3-O-rutinoside |  | 10.7±0.6 |
| pelargonidin 3-O-rutinoside |  | 0.9±0.1 |
HPLC-DAD analyses.
HPLC-MS analyses.
Acknowledgements
References
- Joy, P.P.; Mathew, J.S.; Skaria, B.P. Medicinal plants. Trop. Horticult. 2001, 2, 449–632. [Google Scholar]
- Jyothi, M.V.; Mandayan, S.N.; Kotamballi, N.C.; Bhagyalakshmi, N. Antioxidative efficacies of floral petal extracts of Delonix regia Raffin. Int. J. Biomed. Pharmaceut. Sci. 2007, 1, 73–82. [Google Scholar]
- Sammour, R.H.; El-Shanshoury; Abd El Raheem, R. Antimicrobial activity of legume seed proteins. Bot. Bull. Acad. Sin. 1992, 33, 185–90. [Google Scholar]
- Aqil, F.; Ahmad, I. Broad-spectrum antibacterial and antifungal properties of certain traditionally used Indian medicinal plants. World J. Microbiol. Biotechnol. 2003, 19, 653–657. [Google Scholar] [CrossRef]
- Gupta, R.K.; Chandra, S. Chemical investigation of Delonix regia flowers. Indian J. Pharm. 1971, 33, 75. [Google Scholar]
- Banerjee, A.; De, B. Anthocyanins in some flowers of West Bengal. J. Med. Aromat. Plant Sci. 2001, 23, 600–604. [Google Scholar]
- Soltan, M.E.; Sirry, S.M. Usefulness of some plant flowers as natural acid-base indicators. J. Chin. Chem. Soc. 2002, 49, 63–68. [Google Scholar]
- Saleh, N. A. M.; Ishak, M. S. Anthocyanins of some leguminosae flowers and their effect on colour variation. Phytochemistry 1976, 15, 835–836. [Google Scholar] [CrossRef]
- Meng, L.; Lozano, Y.; Bombarda, I.; Gaydou, E.M.; Li, B. Anthocyanin and flavonoid production from Perilla frutescens: pilot plant scale processing including cross-flow microfiltration and reverse osmosis. J. Agric. Food Chem. 2006, 54, 4297–4303. [Google Scholar] [CrossRef]
- Santos-Buelga, C.; Garcia-Viguera, C.; Tomas-Barberan, F. A. On-line identification of flavonoids by HPLC coupled to diode array detection. In Methods in polyphenols analysis; The Royal Society: Cambridge, 2003; pp. 92–124. [Google Scholar]
- Blando, F.; Gerardi, C.; Nicoletti, I. Sour cherry (Prunus cerasus L.) anthocyanins as ingredients for functional foods. J. Biomed. Biotechnol. 2004, 5, 253–258. [Google Scholar] [CrossRef]
- Los, J.; Wilska-Jeszka, J.; Pawlak, M. Polyphenolic compounds of plums (Prunus domestica). Pol. J. Food Nutrit. Sci. 2000, 9/50, 35–38. [Google Scholar]
- Will, F.; Dietrich, H. Optimised processing technique for colour and cloud stable plum juices and stability of bioactive substances. Eur. Food Res. Technol. 2006, 223, 419–425. [Google Scholar] [CrossRef]
- Hong, V.; Wrolstad, R.E. Use of HPLC separation photodiode array detection for characterization of anthocyanins. J. Agric. Food Chem. 1990, 38, 708–715. [Google Scholar] [CrossRef]
- Pawlowska, A.M.; Oleszek, W.; Braca, A. Quali-quantitative analyses of flavonoids of Morus nigra L. and Morus alba L. (Moraceae) fruits. J. Agric. Food Chem. 2008. accepted for publication, Feb 29th. [Google Scholar]
- Longo, L.; Vasapollo, G. Determination of anthocyanins in Ruscus aculeatus L. Berries. J. Agric. Food Chem. 2005, 53, 475–479. [Google Scholar] [CrossRef]
- Sample Availability: Samples of pilot plant water-extracts of Delonix regia flowers are available from the authors.
© 2008 by the authors. Licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Adje, F.; Lozano, Y.F.; Meudec, E.; Lozano, P.; Adima, A.; N’zi, G.A.; Gaydou, E.M. Anthocyanin Characterization of Pilot Plant Water Extracts of Delonix regia Flowers. Molecules 2008, 13, 1238-1245. https://doi.org/10.3390/molecules13061238
Adje F, Lozano YF, Meudec E, Lozano P, Adima A, N’zi GA, Gaydou EM. Anthocyanin Characterization of Pilot Plant Water Extracts of Delonix regia Flowers. Molecules. 2008; 13(6):1238-1245. https://doi.org/10.3390/molecules13061238
Chicago/Turabian StyleAdje, Felix, Yves F. Lozano, Emmanuelle Meudec, Paul Lozano, Augustin Adima, Georges Agbo N’zi, and Emile M. Gaydou. 2008. "Anthocyanin Characterization of Pilot Plant Water Extracts of Delonix regia Flowers" Molecules 13, no. 6: 1238-1245. https://doi.org/10.3390/molecules13061238
APA StyleAdje, F., Lozano, Y. F., Meudec, E., Lozano, P., Adima, A., N’zi, G. A., & Gaydou, E. M. (2008). Anthocyanin Characterization of Pilot Plant Water Extracts of Delonix regia Flowers. Molecules, 13(6), 1238-1245. https://doi.org/10.3390/molecules13061238




