Identification of Minor Secondary Metabolites from the Latex of Croton lechleri (Muell-Arg) and Evaluation of Their Antioxidant Activity
Abstract
:Introduction
Results and Discussion
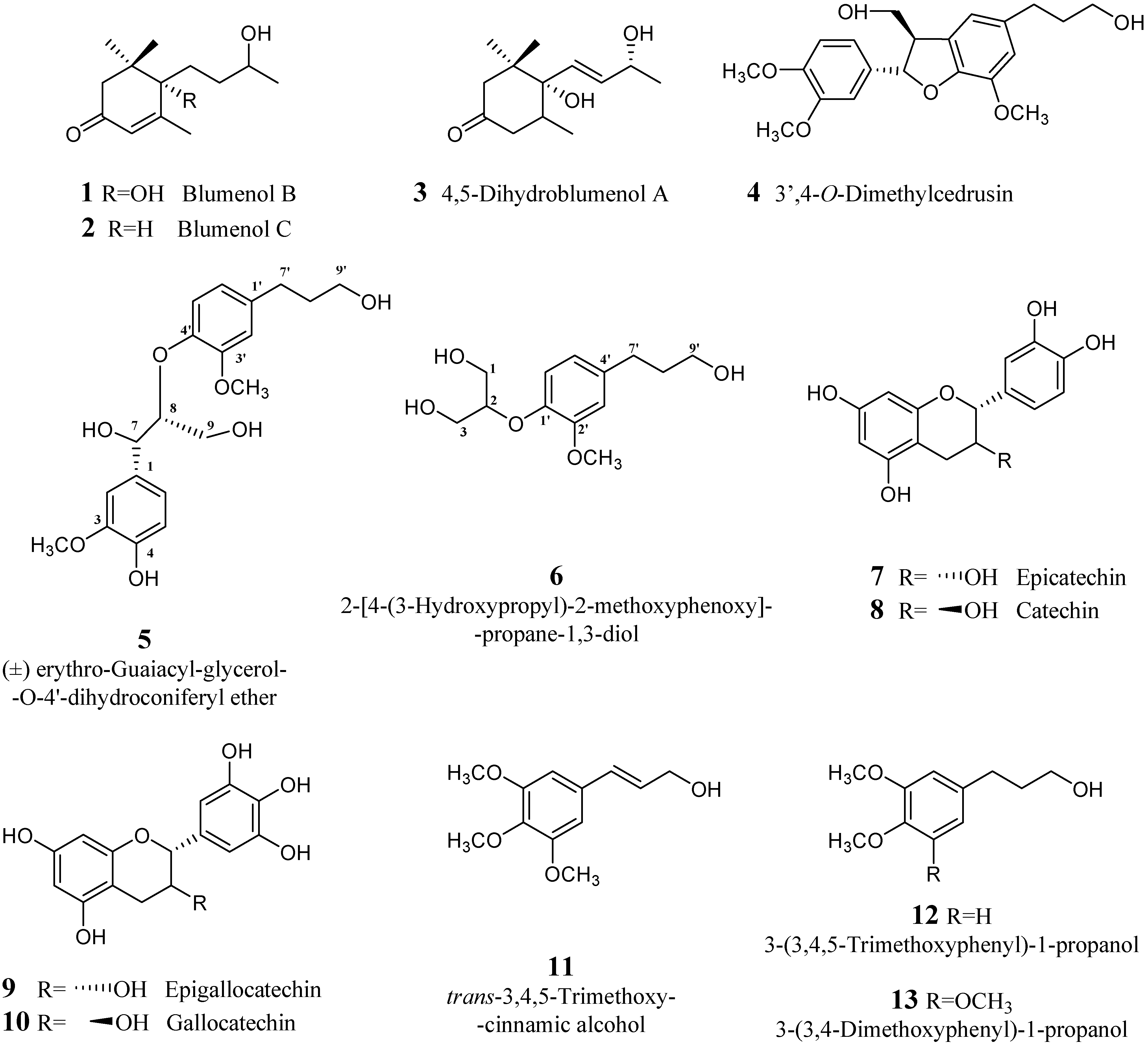
| 5 | 6 | ||||
|---|---|---|---|---|---|
| Position | δH | δC | Position | δH | δC |
| 1 | - | 132.7 | 1 | 3.75 d (J= 5.1 Hz) | 61.7 |
| 2 | 7.02 br s (J= 1.6 Hz) | 118.6 | 2 | 4.15 q | 83.0 |
| 3 | - | 147.7 | 3 | 3.75 d (J= 5.1 Hz) | 61.7 |
| 4 | - | 146.2 | 1’ | - | 146.5 |
| 5 | 6.77 d (J= 8.1 Hz) | 115.6 | 2’ | - | 151.6 |
| 6 | 6.87 dd (J= 8.1, 1.6 Hz) | 119.5 | 3’ | 6.86 d (J=1.5 Hz) | 113.8 |
| 7 | 4.88 d (J= 4.7 Hz) | 73.2 | 4’ | - | 138.1 |
| 8 | 4.20 ddd (J= 4.7, 3.8, 3.0 Hz) | 87.0 | 5’ | 6.74 dd (J= 8.1, 1.5 Hz) | 121.6 |
| 9 | 3.72 dd (J= 12.1, 3.0 Hz) 3.47 dd (J= 12.1, 3.8 Hz) | 60.7 | 6’ | 6.99 d (J= 8.1 Hz) | 119.2 |
| 1’ | - | 137.2 | 7’ | 2.62 t (J= 8.1 Hz) | 32.4 |
| 2’ | 6.87 d (J= 1.7 Hz) | 112.8 | 8’ | 1.82 m | 35.3 |
| 3’ | - | 150.5 | 9’ | 3.56 t (J= 6.6 Hz) | 61.9 |
| 4’ | - | 146.4 | -OCH3 | 3.84 s | 56.1 |
| 5’ | 6.98 d (J= 8.1 Hz) | 110.5 | |||
| 6’ | 6.73 dd (J= 8.1, 1.7 Hz) | 120.9 | |||
| 7’ | 2.63 t (J= 8.0 Hz) | 31.8 | |||
| 8’ | 1.82 m | 34.6 | |||
| 9’ | 3.55 t (J= 6.4 Hz) | 61.0 | |||
| -OCH3 in 3 | 3.83 s | 55.1 | |||
| -OCH3 in 3’ | 3.85 s | 55.3 | |||
Antioxidant activity.
| Sample | Phenols (mg/g extract) | Antioxidant capacity (mEq uric acid) | DPPH (IC50) | |
|---|---|---|---|---|
| 1 μM | 10 μM | μM | ||
| Ascorbic acid | n.a | 0.36±0.02 | 0.33±0.02 | 9.65 μM |
| Trolox® | n.a | 0.85±0.07 | 1.77±0.18 | 12.9 μM |
| Quercetin | n.a | 0.75±0.06 | 2.17±0.17 | 4.37 μM |
| n-Hexane extract | 4.84 | / | / | / |
| CHCl3 extract | 41.87 | 0.09±0.01 | 0.15±0.01 | 0.09 μM |
| n-BuOH extract | 306.01 | 0.10±0.01 | 0.65±0.05 | 0.875 μM |
| H2O residue extract | 34.37 | 0.05±0.01 | 0.12±0.01 | 12.4 μM |
| Fraction 1 | 212.90 | 0.01±0.01 | 0.83±0.07 | 13.1 μM |
| Fraction 2 | 314.55 | 0.02±0.01 | 1.81±0.14 | 6.10 μM |
| Fraction 3 | 546.47 | 0.01±0.01 | 1.11±0.08 | 10.9 μM |
| Fraction 4 | 330.51 | 0.03±0.01 | 0.66±0.05 | 1.41 μM |
| Gallocatechin | n.a | 0.16±0.03 | 1.35±0.10 | 10.0 μM |
| Epigallocatechin | n.a | 0.39±0.05 | 2.05±0.11 | 0.561 μM |
| Epicatechin | n.a | 0.09±0.01 | 0.36±0.03 | 19.3 μM |
Conclusions
Experimental
General
Plant Material
Extraction and Isolation
Isolation and identification of alkaloids
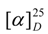 = 0° (MeOH, c 0.09); EI-MS m/z 378 [M]+ (C20H26O7); 1H- and 13C-NMR see Table 1.
= 0° (MeOH, c 0.09); EI-MS m/z 378 [M]+ (C20H26O7); 1H- and 13C-NMR see Table 1.Determination of phenolic content
Total antioxidant capacity
DPPH scavenging test
Acknowledgements
References
- Milanowsky, D.J.; Winter, R.E.; Elvin-Lewis, M.P.; Lewis, W.H. Geographic distribution of the alkaloid chemotypes of Croton lechleri. J. Nat. Prod. 2002, 65, 814–819. [Google Scholar] [CrossRef]
- Desmarchelier, C.; Witting Schaus, F.; Coussio, J.; Cicca, G. Effects of Sangre de Drago from Croton lechleri Muell.-Arg. on the production of active oxygen radicals. J. Ethnopharmacol. 1997, 58, 103–108. [Google Scholar] [CrossRef]
- Risco, E.; Ghia, F.; Vila, R.; Iglesias, J.; Alvarez, E.; Canigueral, S. Immunomodulatory activity and chemical characterisation of sangre de drago (dragon’s blood) from Croton lechleri. Planta Med. 2003, 69, 785–794. [Google Scholar] [CrossRef]
- Ubillas, R.; Jolad, S.D.; Bruening, R.C.; Kernan, M.R.; King, S.R.; Sesin, D.F.; Barrett, M.; Stoddart, C.A.; Flaster, T. SP-303, an antiviral oligomeric proanthocyanidin from the latex of Croton lechleri (Sangre de Drago). Phytomedicine 1994, 1, 77–106. [Google Scholar] [CrossRef]
- Pieters, L.; de Bruyne, T.; Claeys, M.; Vlietinck, A.; Calomme, M.; Vanden Berghe, D. Isolation of a dihydrobenzofuran lignan from South American Dragon's Blood (Croton spp.) as an inhibitor of cell proliferation. J. Nat. Prod. 1993, 56, 899–906. [Google Scholar] [CrossRef]
- Carlson, T.J.S.; King, S.R. Sangre de drago (Croton lechleri Mell-Arg) a phytomedicine for the treatment of diarrhoea. Health Notes. Rev. Compl. Integr. Med. 2000, 7, 315–320. [Google Scholar]
- Di Cesare, D.; Du Pont, H.L.; Mathewson, J.J.; Ashley, D.; Martinez-Sandoval, F.; Pennington, J.E.; Porter, S.B. A double blind, randomized placebo-controlled study of SP-303 (Provir) in the symptomatic treatment of acute diarrhoea among travellers to Jamaica and Mexio. Am. J. Gastroent. 2002, 97, 2585–2588. [Google Scholar] [CrossRef]
- Holodniy, M.; Koch, J.; Mistal, M.; Schmidt, J.M.; Khandwala, A.; Pennington, J.E.; Porter, S.B. A double blind, randomized placebo-controlled phase II study to assess the safety and efficacy of orally administered SP-303 for symptomatic treatment of diarrhoea in patients with AIS. Am. J. Gastroent. 1999, 94, 3267–3273. [Google Scholar] [CrossRef]
- Fisher, H.; Machen, T.E.; Widdicombe, J.H.; Carlson, T.J.S.; King, S.R.; Chow, J.W.S.; Illek, B. A novel extract SB-300 from the steam bark latex of Croton lechleri inhibits CFTR-mediated chloride secretion in human colonic epithelial cells. 2004. J Ethnopharmacol 2004, 351–357. [Google Scholar]
- Jones, K. Review of Sangre de drago (Croton lechleri) a south American tree sap in the treatment of diarrhea, inflammation, insect bites, viral infections and wounds: traditional uses to clinical researh. J. Altern. Complement. Med. 2003, 9, 877–896. [Google Scholar] [CrossRef]
- Vaisberg, A.J.; Milla, M.; del Carmen Planas, M.; Cordova, J.L.; Rosas de Agusti, E.; Ferreyra, R.; del Carmen Mustiga, M.; Carlin, L.; Hammond, G.B. Taspine is the cicatrizant principle in Sangre de Grado extracted from Croton lechleri. Planta Med. 1989, 55, 140–143. [Google Scholar]
- Phillipson, J.D. Review. A matter of some sensitivity. Phytochemistry 1995, 38, 1319–1343. [Google Scholar] [CrossRef]
- Hartwell, J.L. Plants used against cancer : a survey. Lloydia 1969, 32, 158–176. [Google Scholar]
- Bettolo, R.M.; Scarpati, M.L. Alkaloids of Croton draconoides. Phytochemistry 1979, 18, 520. [Google Scholar] [CrossRef]
- Chen, Z.P.; Cai, Y.; Phillipson, J.D. Studies on the anti-tumour, anti-bacterial, and wound-healing properties of Dragon's Blood. Planta Med. 1994, 60, 541–5. [Google Scholar]
- Lopes e Lopes, M.I.; Saffi, J.; Echeverrigaray, S.; Pegas Henriques, J.A.; Salvador, M. Mutagenic and antioxidant activities of Croton lechleri sap in biological systems. J. Ethopharmacol. 2004, 95, 437–445. [Google Scholar] [CrossRef]
- Cai, Y.; Evans, F.J.; Roberts, M.F.; Phillipson, J.D.; Zenk, M.H.; Gleba, Y.Y. Biological and chemical investigation of Dragon's Blood from Croton species of South America. Part 1. Polyphenolic compounds from Croton lechleri. Phytochemistry 1991, 30, 2033–2040. [Google Scholar] [CrossRef]
- Cai, Y.; Chen, Z.P.; Phillipson, J.D. Biological and chemical investigation of Dragon's Blood from Croton species of South America. Part 3. Clerodane diterpenoids from Croton lechleri. Phytochemistry 1993, 34, 265–268. [Google Scholar] [CrossRef]
- Cai, Y.; Chen, Z.P.; Phillipson, J.D. Biological and chemical investigation of Dragon's Blood from Croton species of South America. Part 2. Diterpenes from Croton lechleri. Phytochemistry 1993, 32, 755–760. [Google Scholar]
- Galbraith, M.N.; Horn, D.H.S. Structures of the natural products blumenols A, B, and C. J. Chem. Soc. Chem. Comm. 1972, 3, 113–114. [Google Scholar] [CrossRef]
- De Marino, S.; Borbone, N.; Zollo, F.; Ianaro, A.; Di Meglio, P.; Iorizzi, M. Megastigmane and phenolic components from Laurus nobilis L. leaves and their inhibitory effects on nitric oxide production. J. Agric. Food Chem. 2004, 52, 7525–7531. [Google Scholar]
- Sinkkonen, J.; Karonen, M.; Liimatainen, J.; Pihlaja, K. Lignans from the bark extract of Pinus sylvestris L. Magn. Res. Chem. 2006, 44, 633–636. [Google Scholar] [CrossRef]
- Baltenweck-Guyot, R.; Trendel, J.M.; Albrecht, P.; Schaeffer, A. Glycosides and phenylpropanoid glycerol in Vitis vinifera cv. Gewurztraminer wine. J. Agric. Food Chem. 2000, 48, 6178–6182. [Google Scholar]
- Achenbach, H.; Hemrich, H. Constituents of tropical medicinal plants. Part 40. Alkaloids, flavonoids and phenylpropanoids of the West African plant Oxymitra velutina. Phytochemistry 1991, 30, 1957–1962. [Google Scholar] [CrossRef]
- Arnoldi, A.; Merlini, L. Asymmetric synthesis of 3-Methyl-2-phenyl-1,4-benzodioxanes. Absolute configuration of the neolignans eusiderin and eusiderin C and D. J. Chem. Soc. Perkin Trans. I 1985, 2555–2557. [Google Scholar] [CrossRef]
- Fang, J.; Lee, C.; Cheng, Y. Lignans from leaves of Juniperus chinensis. Phytochemistry 1992, 31, 3659–3661. [Google Scholar] [CrossRef]
- Kupchan, S.M.; Britton, R.W.; Ziegler, M.F.; Sigel, C.W. Bruceantin, a new potent antileukemic simaroubolide from Brucea antidysenterica. J. Org. Chem. 1973, 38, 178–179. [Google Scholar] [CrossRef]
- Visioli, F.; Vinceri, F.F.; Galli, C. ‘Waste water’ from olive oil production are rich in natural antioxidants. Experientia 1995, 51, 32–34. [Google Scholar]
- Visioli, F.; Caruso, D.; Plasmati, E.; Patelli, R.; Mulinacci, N.; Romani, A.; Galli, G.; Galli, C. Hydroxytyrosol, as a component of olive mill waste water, is dose-dependently absorbed and increases the antioxidant capacity of rat plasma. Free Radical Res. 2001, 34, 301–305. [Google Scholar] [CrossRef]
- Visioli, F.; Galli, C. The effect of minor constituents of olive oil on cardiovascular disease: new findings. Nutrition Rev. 1998, 56, 142–147. [Google Scholar]
- Sample Availability: Contact the authors
© 2008 by the authors. Licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
De Marino, S.; Gala, F.; Zollo, F.; Vitalini, S.; Fico, G.; Visioli, F.; Iorizzi, M. Identification of Minor Secondary Metabolites from the Latex of Croton lechleri (Muell-Arg) and Evaluation of Their Antioxidant Activity. Molecules 2008, 13, 1219-1229. https://doi.org/10.3390/molecules13061219
De Marino S, Gala F, Zollo F, Vitalini S, Fico G, Visioli F, Iorizzi M. Identification of Minor Secondary Metabolites from the Latex of Croton lechleri (Muell-Arg) and Evaluation of Their Antioxidant Activity. Molecules. 2008; 13(6):1219-1229. https://doi.org/10.3390/molecules13061219
Chicago/Turabian StyleDe Marino, Simona, Fulvio Gala, Franco Zollo, Sara Vitalini, Gelsomina Fico, Francesco Visioli, and Maria Iorizzi. 2008. "Identification of Minor Secondary Metabolites from the Latex of Croton lechleri (Muell-Arg) and Evaluation of Their Antioxidant Activity" Molecules 13, no. 6: 1219-1229. https://doi.org/10.3390/molecules13061219
APA StyleDe Marino, S., Gala, F., Zollo, F., Vitalini, S., Fico, G., Visioli, F., & Iorizzi, M. (2008). Identification of Minor Secondary Metabolites from the Latex of Croton lechleri (Muell-Arg) and Evaluation of Their Antioxidant Activity. Molecules, 13(6), 1219-1229. https://doi.org/10.3390/molecules13061219








