Antimicrobial and Antioxidative Activities of Bioactive Constituents from Hydnophytum formicarum Jack.
Abstract
:Introduction
Results and Discussion
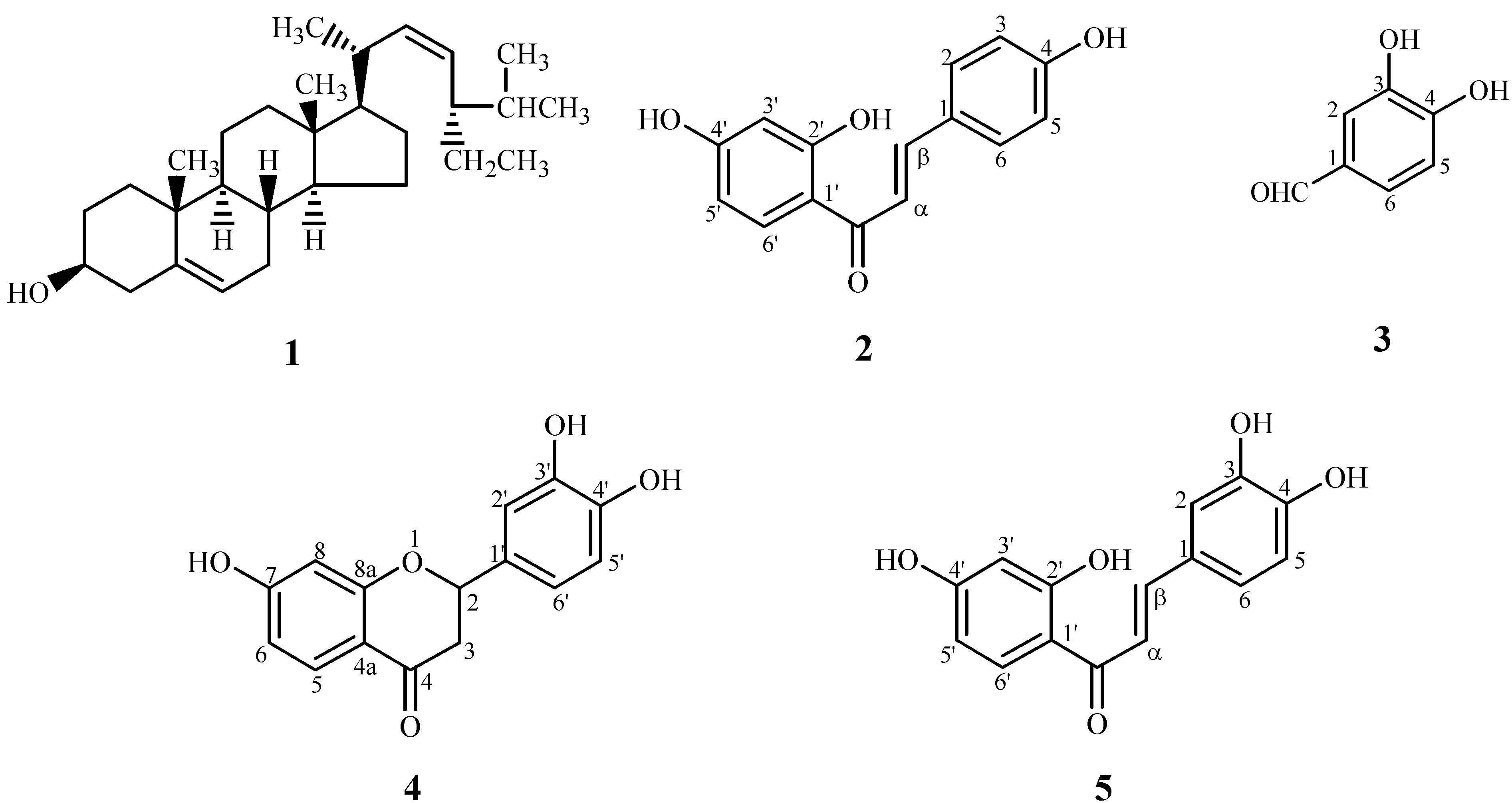
Isolation and structure elucidation
Biological activities
Antimicrobial activity
| Compounda,b | Microorganism | Gram | MIC c (μg/mL) |
|---|---|---|---|
| Crude hexane extract | C. diphtheriae NCTC 10356 | + | 256 |
| Crude dichloromethane extract | C. diphtheriae NCTC 10356 | + | 256 |
| Crude ethyl acetate extract | C. diphtheriae NCTC 10356 | + | 256 |
| A. xylosoxidan ATCC 2706 | - | 256 | |
| S. aureus ATCC 25923 | + | 256 | |
| M. lutens ATCC 10240 | + | 256 | |
| S. dysenteriae | - | 256 | |
| S. pyogenes II | + | 256 | |
| A. hydrophila | - | 256 | |
| B. cereus | + | 256 | |
| S. putrefaciens ATCC 8671 | - | 128 | |
| Crude methanol extract | C. diphtheriae NCTC 10356 | + | 256 |
| S. pyogenes II | + | 256 | |
| B. cereus | + | 256 | |
| Compound 3 | P. shigelloides | - | ≤ 60 |
| Ampicillin | P. shigelloides | - | 10 |
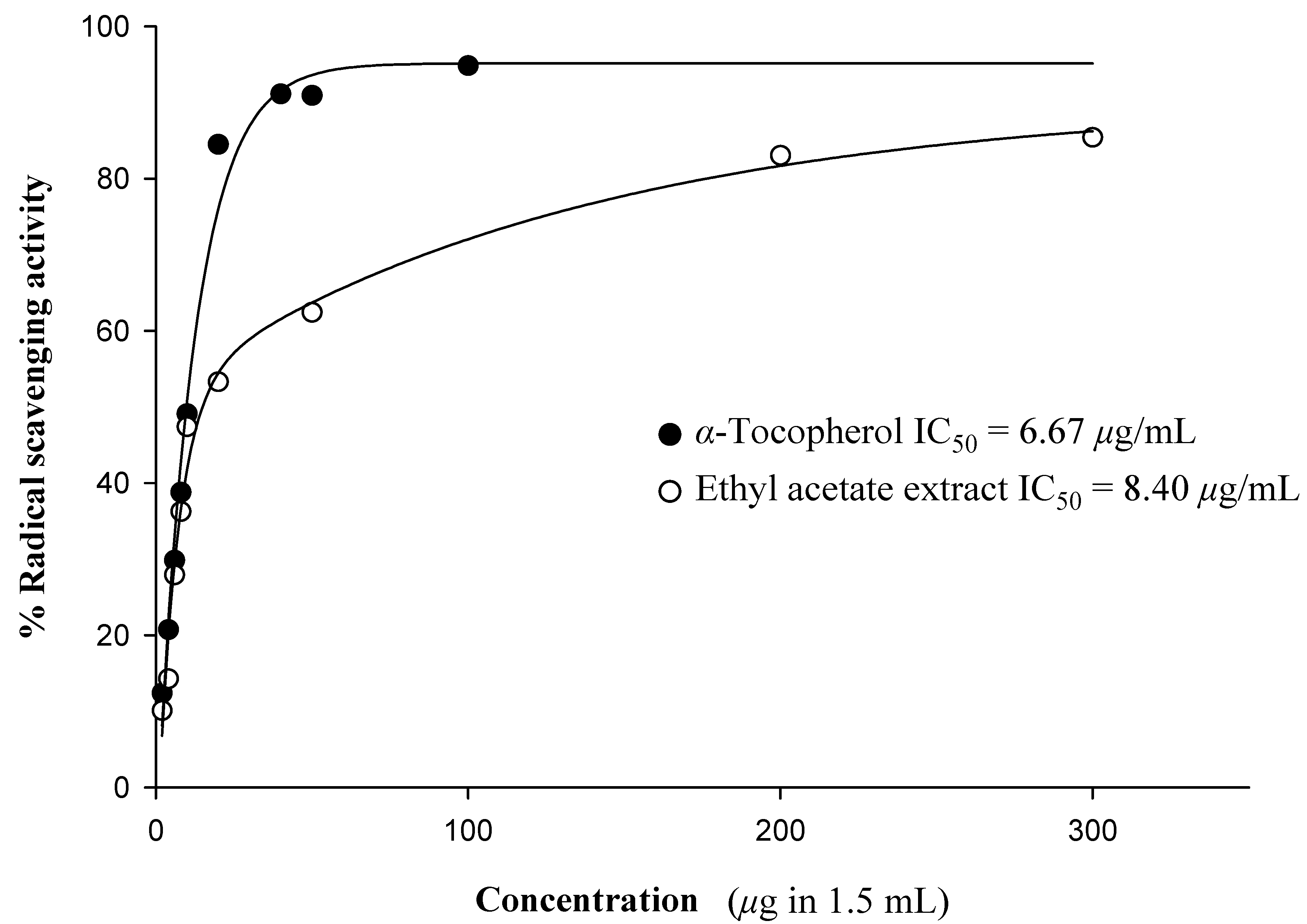
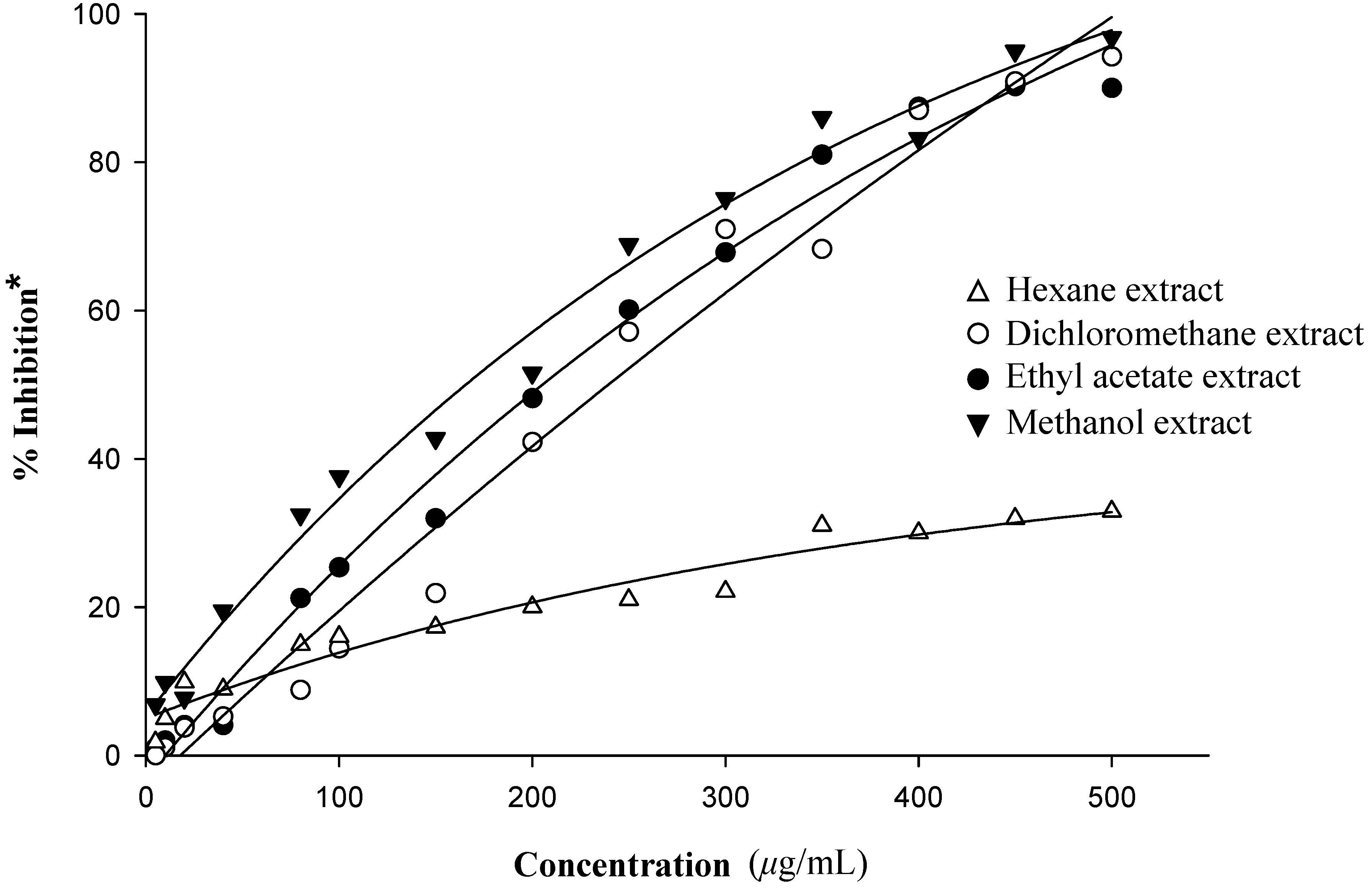
Antioxidant activity
| Compound | Radical scavenging activitya (%) (333.33 µg/mL) | Superoxide dismutase activityb (%) (300 µg/mL) |
|---|---|---|
| Crude hexane extract | 28.60 | 25.86 |
| Crude dichloromethane extract | 55.70 | 62.24 |
| Crude ethyl acetate extract | 83.31 | 67.91 |
| Crude methanol extract | 56.80 | 74.19 |
Cytotoxic activity
| Compound | ED50 (μg/mL)* | |
|---|---|---|
| HuCCA-1 cell line | KB cell line | |
| 3 | >10 | >10 |
Conclusions
Experimental
General
Plant material
Cell cultures
Extraction and Isolation
Extraction
Isolation
 -87° (c 0.03, MeOH)(-); lit. [17],–18.7°± 0.8°; c 0.5, MeOH [18]); 1H-NMR (CD3OD): δ 7.69 (1H, d, J = 8.7 Hz, H-5), 6.87 (1H, d, J = 1.8 Hz, H-2′), 6.77 (1H, d, J =8.2 Hz, H-5′), 6.74 (1H, dd, J = 8.2, 1.8 Hz, H-6′), 6.44 (1H, dd, J = 8.7, 2.2 Hz, H-6), 6.32 (1H, d, J = 2.2 Hz, H-8), 5.22 (1H, dd, J = 13.2, 2.8 Hz, H-2), 2.94 (1H, dd, J = 13.2, 17.0 Hz, H-3a), 2.65 (1H, dd, J = 17.0, 2.8 Hz, H-3b); 13C-NMR (CD3OD): δ 192.07 (CO), 164.86 (C-7), 163.77 (C-8a), 144.96 (C-3′), 144.59 (C-4′), 130.31 (C-1′), 128.74 (CH-5), 118.15 (CH-6′), 114.98 (CH-5′), 113.64 (C-4a), 113.21 (CH-2′), 110.65 (CH-6), 102.77 (CH-8), 79.45 (CH-2), 43.62 (CH2-3); EIMS m/z (% relative intensity): 272 (M+, 100), 271 (55), 255 (24), 163 (20), 150 (87), 137 (52), 136 (24) (lit. [17]: 272 (M+, 65)).
-87° (c 0.03, MeOH)(-); lit. [17],–18.7°± 0.8°; c 0.5, MeOH [18]); 1H-NMR (CD3OD): δ 7.69 (1H, d, J = 8.7 Hz, H-5), 6.87 (1H, d, J = 1.8 Hz, H-2′), 6.77 (1H, d, J =8.2 Hz, H-5′), 6.74 (1H, dd, J = 8.2, 1.8 Hz, H-6′), 6.44 (1H, dd, J = 8.7, 2.2 Hz, H-6), 6.32 (1H, d, J = 2.2 Hz, H-8), 5.22 (1H, dd, J = 13.2, 2.8 Hz, H-2), 2.94 (1H, dd, J = 13.2, 17.0 Hz, H-3a), 2.65 (1H, dd, J = 17.0, 2.8 Hz, H-3b); 13C-NMR (CD3OD): δ 192.07 (CO), 164.86 (C-7), 163.77 (C-8a), 144.96 (C-3′), 144.59 (C-4′), 130.31 (C-1′), 128.74 (CH-5), 118.15 (CH-6′), 114.98 (CH-5′), 113.64 (C-4a), 113.21 (CH-2′), 110.65 (CH-6), 102.77 (CH-8), 79.45 (CH-2), 43.62 (CH2-3); EIMS m/z (% relative intensity): 272 (M+, 100), 271 (55), 255 (24), 163 (20), 150 (87), 137 (52), 136 (24) (lit. [17]: 272 (M+, 65)).Biological evaluation: Antimicrobial assay [19]
| Gram-negative bacteria | Clinical specimens |
| Escherichia coli ATCC 25922 | Gram-negative bacteria |
| Klebsiella pneumoniae ATCC 700603 | Shigella dysenteriae |
| Serratia marcescens ATCC 8100 | Salmonella enteritidis type C |
| Salmonella typhimurium ATCC 13311 | Morganella morganii |
| Salmonella choleraesuis ATCC 10708 | Aeromonas hydrophila |
| Shewanella putrefaciens ATCC 8671 | Citrobacter freundii |
| Achromobacter xylosoxidans ATCC 2706 | Plesiomonas shigelloides |
| Pseudomonas aeruginosa ATCC 15442 | Gram-positive bacteria |
| Pseudomonas stutzeri ATCC 17587 | Streptococcus pyogenes II |
| Gram-positive bacteria | Bacillus cereus |
| Staphylococcus aureus ATCC 29213 | Listeria monocytogenes |
| Staphylococcus aureus ATCC 25923 | |
| Enterococcus faecalis ATCC 29212 | |
| Enterococcus faecalis ATCC 33186 | |
| Micrococcus lutens ATCC 10240 | |
| Bacillus subtilis ATCC 6633 | |
| Corynebacterium diphtheriae NCTC 10356 | |
| Yeasts | |
| Saccharomyces cereviseae ATCC 2601 | |
| Candida albicans ATCC 90028 |
Antioxidative assay [20, 21]
Cytotoxic assay [22]
Acknowledgements
References
- Morse, C. Ecology & Evolutionary Biology Conservatory. Available online: http://florawww.eeb.uconn.edu/acc_num/198500652.html (Online 2000, 30 March).
- Huxley, C. R.; Jebb, M. H. P. The tuberous epiphytes of the rubiaceae 1: A new subtribe-the hydnophytinae. Blumea 1991, 36, 1–20. [Google Scholar]
- Smitinund, T. Thai plant names (botanical names-vernacular names); Funny Publishing: Bangkok, 1980; pp. 183–184. [Google Scholar]
- Prommee, P. Thai traditional medicine; Mahachulalongkon Publishing: Bangkok, 1988; p. 51. [Google Scholar]
- Beckstrom-Sternberg, S. M.; Duke, J. A.; Wain, K. K. The Ethnobotany Database. Available online: http://probe.nal.usda.gov:8300/cgi-bin/webace?db=ethnobotdb& class =Taxon&object= Hydnophytum+formicarium (Online 1994, July).
- Nguyen, M. T.; Awale, S.; Tezuka, Y.; Tran, Q. L.; Watanabe, H.; Kadota, S. Xanthine oxidase inhibitory activity of Vietnamese medicinal plants. Biol. Pharm. Bull. 2004, 27, 1414–1421. [Google Scholar] [CrossRef]
- Ueda, J. Y.; Tezuka, Y.; Banskota, A. H.; Tran, Q. L.; Tran, Q. K.; Hariyama, Y.; Saiki, I.; Kadota, S. Antiproliferative activity of Vietnamese medicinal plants. Biol. Pharm. Bull. 2002, 25, 753–760. [Google Scholar] [CrossRef]
- Huxley, C. Symbiosis between ants and epiphytes. Biol. Rev. 1980, 55, 321–340. [Google Scholar] [CrossRef]
- Pouchert, C. J.; Bekke, J. The Aldrich library of 13C and 1H FTNMR spectra; Aldrich Chemical Company: Wisconsin, 1993; p. 569. [Google Scholar]
- Devon, T. K.; Scott, A. I. Handbook of naturally occurring compounds: acetogenins, shikimates and carbohydrates; Academic Press: New York, 1975; Vol. 1, pp. 10–167. [Google Scholar]
- Saitoh, T.; Noguchi, H.; Shibata, S. A new isoflavone and the corresponding isoflavanone of licorice root. Chem. Pharm. Bull. 1978, 26, 144–147. [Google Scholar] [CrossRef]
- Yenesew, A.; Midiwo, J. O.; Waterman, P. G. Rotenoids, isoflavones and chalcones from the stem bark of Mellettia usaramensis subspecies usaramensis. Phytochemistry 1998, 47, 295–300. [Google Scholar] [CrossRef]
- Mabry, T. J.; Markham, K. R.; Thomas, M. B. The systematic identification of flavonoids; Springer-Verlag: New York, 1970; pp. 165–166. [Google Scholar]
- Kuroyanagi, M.; Fukuoka, M.; Yoshihira, K; Natori, S. Chemical and toxicology studies on bracken fern, Pteridium aquilinum var. latiusculum. III. Further characterization of pterosins and pterosides, sesquiterpenes and the glucosides having 1-indanone skeleton, from the rhizomes. Chem. Pharm. Bull. 1979, 27, 592–601. [Google Scholar] [CrossRef]
- Hata, K.; Baba, K; Kozawa, M. Chemical studies on the heartwood of Cassia garrettiana CRAIB. II. nonanthraquinonic constituents. Chem. Pharm. Bull. 1979, 27, 984–989. [Google Scholar] [CrossRef]
- Santavy′, F.; Walterova′, D.; Hruban, L. Ultraviolet spectra, methoxy and methylenedioxy groups of some aromatic compounds and alkaloids. IV; The effect of hydroxyl, methoxyl and methylenedioxy groups on the ultraviolet spectra of aromatic compounds conjugated with an oxo-group or with and α, β-double bond in the side chain. Coll.t Czech. Chem. Commun. 1970, 37, 1825–1850. [Google Scholar]
- Van der Westhuizen, J. H.; Ferreira, D.; Roux, D. G. Photochemical deoxygenation of an α-ketol: The dihydroflavonol-flavanone conversion. J. Chem. Soc. Perkin Trans I. 1980, 1003–1006. [Google Scholar] [CrossRef]
- Roux, D. G.; Paulus, E. Condensed tannins. 10. Isolation of (-)-butin and butein from wattle heartwoods. Biochem. J. 1961, 80, 62–63. [Google Scholar]
- Baron, E. J.; Peterson, L. R.; Finegold, S. M. Methods for testing antimicrobial effectiveness. In Bailey and Scott’s diagnostic microbiology; Mosby-Year Book: St. Louis, MI, 1994; pp. 168–193. [Google Scholar]
- Yen, G. C.; Hsieh, G. C. Antioxidant effects on dopamine and related compounds. Biosci. Biotechnol. Biochem. 1997, 61, 1646–1649. [Google Scholar] [CrossRef]
- Piacham, T.; Isarankura-Na-Ayudhya, C.; Nantasenamat, C.; Yainoy, S.; Ye, L.; Prachayasittikul, V. Metalloantibiotic Mn(II)-bacitracin complex mimicking manganese superoxide dismutase. Biochem. Biophys. Res. Comm. 2006, 341, 925–930. [Google Scholar] [CrossRef]
- Tengchaisri, T.; Chawengkirttikul, R.; Rachaphaew, N.; Reutrakul, V.; Sangsuwan, R.; Sirisinha, S. Antitumor activity of triptolide against cholangiocarcinoma growth in vitro and in hamsters. Cancer Lett. 1998, 133, 169–175. [Google Scholar] [CrossRef]
- Bate-Smith, E. C.; Swain, T. The isolation of 2,4,4'-trihydroxychalcone from yellow varieties of Dahlia variabilis. J. Chem. Soc. 1953, 2185–2187. [Google Scholar]
- Vaya, J.; Belinky, P. A.; Aviram, M. Antioxidant constituents from licorice roots: isolation, structure elucidation and antioxidative capacity toward LDL oxidation. Free Radic. Biol. Med. 1997, 23, 302–313. [Google Scholar] [CrossRef]
- Kong, L. D.; Zhang, Y.; Pan, X.; Tan, R. X.; Cheng, C. H. Inhibition of xanthine oxidase by liquiritigenin and isoliquiritigenin isolated from Sinofranchetia chinensis. Cell. Mol. Life Sci. 2000, 57, 500–505. [Google Scholar] [CrossRef]
- Zheng, Z.-P.; Cheng, K.-W.; Chao, J.; Wu, J.; Wang, M. Tyrosinase inhibitors from paper mulberry (Broussonetia papyrifera). Food Chem. 2008, 106, 529–535. [Google Scholar] [CrossRef]
- Hayashi, H.; Hiraoka, N.; Ikeshiro, Y.; Yamamoto, H. Organ specific localization of flavonoids in Glycyrrhiza glabra L. Plant Sci. 1996, 116, 233–238. [Google Scholar] [CrossRef]
- Wegener, J. W.; Nawrath, H. Cardiac effects of isoliquiritigenin. Eur. J. Pharmacol. 1997, 326, 37–44. [Google Scholar] [CrossRef]
- Wegener, J. W.; Nawrath, H. Differential effects of isoliquiritigenin and YC-1 in rat aortic smooth muscle. Eur. J. Pharmacol. 1997, 323, 89–91. [Google Scholar] [CrossRef]
- Tawata, M.; Aida, K.; Noguchi, T.; Ozaki, Y.; Kume, S.; Sasaki, H.; Chin, M.; Onaya, T. Anti-platelet action of isoliquiritigenin, an aldose reductase inhibitor in licorice. Eur. J. Pharmacol. 1992, 212, 87–92. [Google Scholar] [CrossRef]
- Yu, S. M.; Kuo, S. C. Vasorelaxant effect of isoliquiritigenin, a novel soluble guanylate cyclase activator, in rat aorta. Br. J. Pharmacol. 1995, 114, 1587–1594. [Google Scholar] [CrossRef]
- Haraguchi, H.; Ishikawa, H.; Mizutani, K.; Tamura, Y.; Kinoshita, T. Antioxidative and superoxide scavenging activities of retrochalcones in Glycyrrhiza inflata. Bioorg. Med. Chem. 1998, 6, 339–347. [Google Scholar] [CrossRef]
- Kobayashi, S.; Miyamoto, T.; Kimura, I.; Kimura, M. Inhibitory effect of isoliquiritigenin, a compound in licorice root, on angiogenesis in vivo and tube formation in vitro. Biol. Pharm. Bull. 1995, 18, 1382–1386. [Google Scholar] [CrossRef]
- Hsu, Y.-L.; Kuo, P.-L.; Lin, C.-C. Isoliquiritigenin induces apoptosis and cell cycle arrest through p53-dependent pathway in Hep G2 cells. Life Sci. 2005, 77, 279–292. [Google Scholar] [CrossRef]
- Kuma, S.; Sharma, A.; Madan, B.; Singhal, V.; Ghosh, B. Isoliquiritigenin inhibits IkB kinase activity and ROS generation to block TNF-α induced expression of cell adhesion molecules on human endothelial cells. Biochem. Pharmacol. 2007, 73, 1602–1612. [Google Scholar] [CrossRef]
- Takahashi, T.; Takasuka, N.; Iigo, M.; Baba, M.; Nishino, H.; Tsuda, H.; Okuyama, T. Isoliquiritigenin, a flavonoid from licorice, reduces prostaglandin E2 and nitric oxide, causes apoptosis, and suppresses aberrant crypt foci development. Cancer Sci. 2004, 95, 448–453. [Google Scholar] [CrossRef]
- Jung, J. I.; Chung, E.; Seon, M. R.; Shin, H.-K.; Kim, E. J.; Lim, S. S.; Chung, W.-Y.; Park, K.-K.; Park, J. H. Y. Isoliquiritigenin (ISL) inhihits ErbB3 signaling in prostate cancer cells. BioFactors 2006, 28, 159–168. [Google Scholar] [CrossRef]
- Figueiredo, A. R.; Campos, F.; de Freitas, V.; Hogg, T.; Couto, J. A. Effect of phenolic aldehydes and flavonoids on growth and inactivation of Oenococcus oeni and Lactobacillus hilgardii. Food Microbiol. 2008, 25, 105–112. [Google Scholar] [CrossRef]
- Martin, T. S.; Kikuzaki, H.; Hisamoto, M.; Nakatani, N. Constituents of Amomum tsao-ko and their radical scavenging and antioxidant activities. J. Am. Oil Chem. Soc. 2000, 77, 667–673. [Google Scholar] [CrossRef]
- Lee, T. Y.; Chang, H. H.; Wang, G. J.; Chiu, J. H.; Yang, Y. Y.; Lin, H. C. Water-soluble extract of Salvia miltiorrhiza ameliorates carbon tetrachloride-mediated hepatic apoptosis in rats. J. Pharm. Pharmacol. 2006, 58, 659–665. [Google Scholar] [CrossRef]
- Lu, Y.; Yeap, F. L. Salvianolic acid L, a potent phenolic antioxidant from Salvia officinalis. Tetrahedron Lett. 2001, 42, 8223–8225. [Google Scholar] [CrossRef]
- Lee, S.; Shim, S. H.; Kim, J. S.; Shin, K. H.; Kang, S. S. Aldose reductase inhibitors from the fruiting bodies of Ganoderma applanatum. Biol. Pharm. Bull. 2005, 28, 1103–1105. [Google Scholar] [CrossRef]
- Shibata, M.-A.; Hirose, M.; Yamada, M.; Tatematsu, M.; Uwagawa, S.; Ito, N. Epithelial cell proliferation in rat forestomach and glandular stomach mucosa induced by catechol and analogous dihydroxybenzenes. Carcinogenesis 1990, 11, 997–1000. [Google Scholar] [CrossRef]
- Kim, Y. S.; Kim, N. H.; Lee, S. W.; Lee, Y. M.; Jang, D. S.; Kim, J. S. Effect of protocatechualdehyde on receptor for advanced glycation end products and TGF-β1 expression in human lens epithelial cells cultured under diabetic conditions an on lens opacity in streptozotocin-diabetic rats. Eur. J. Pharmacol. 2007, 569, 171–179. [Google Scholar]
- Lai, P. K.; Oh-Hara, T.; Tamura, Y.; Kawazoe, Y.; Konno, K.; Sakagami, H.; Tanaka, A.; Nonoyama, M. Polymeric phenylpropenoids are the active components in the pine cone extract that inhibit the replication of type-1 human immunodeficiency virus in vitro. J. Gen. Appl. Microbiol. 1992, 38, 303–312. [Google Scholar] [CrossRef]
- Bhargava, S. K. Estrogenic and postcoital anticonceptive activity in rats of butin isolated from Butea monosperma seed. J. Ethnopharmacol. 1986, 18, 95–101. [Google Scholar] [CrossRef]
- Lee, M.-H.; Lin, Y.-P.; Hsu, F.-L.; Zhan, G.-R.; Yen, K.-Y. Bioactive constituents of Spatholobus suberectus in regulating tyrosinase-related proteins and mRNA in HEMn cells. Phytochemistry 2006, 67, 1262–1270. [Google Scholar] [CrossRef]
- Perkin F. R. S., A. G.; Hummel, J. J. CXLVII.-The colouring principle of the flowers of the Butea Frondosa. J. Chem. Soc. Trans. 1904, 85, 1459–1472. [Google Scholar]
- Shimokoriyama, M.; Geissman, T. A. Anthochlor pigments. XIV. The pigments of Viguiera multiflora (Nutt.) and Baeria chrysostoma (F. and M.). J. Org. Chem. 1960, 25, 1956–1959. [Google Scholar] [CrossRef]
- Geissman, T. A. Anthochlor pigments. III. The pigments of Cosmos sulphureus. J. Chem. Soc. 1942, 64, 1704–1707. [Google Scholar] [CrossRef]
- Geissman, T. A; Harborne, J. B.; Seikel, M. K. Anthochlor pigments. XI. The Constituents of Coreopsis maritima. Reinvestigation of Coreopsis gigantea. J. Am. Chem. Soc. 1956, 78, 825–829. [Google Scholar] [CrossRef]
- Cheng, Z.-J.; Kuo, S.-C.; Chan, S.-C.; Ko, F.-N.; Teng, C.-M. Antioxidant properties of butein isolated from Dalbergia odorifera. Biochim. Biophys. Acta. 1998, 1392, 291–299. [Google Scholar] [CrossRef]
- Chan, S. C.; Chang, Y. S.; Wang, J. P.; Chen, S. C.; Kuo, S. C. Three new flavonoids and antiallergic, anti-inflammatory constituents from the heartwood of Dalbergia odorifera. Planta Med. 1998, 64, 153–158. [Google Scholar] [CrossRef]
- Sogawa, S.; Nihro, Y.; Ueda, H.; Miki, T.; Matsumoto, H.; Satoh, T. Protective effects of hydroxychalcones on free radical-induced cell damage. Biol. Pharm. Bull. 1994, 17, 251–256. [Google Scholar] [CrossRef]
- Lee, J. C.; Lim, K. T.; Jang, Y. S. Identification of Rhus verniciflua stokes compounds that exhibit free radical scavenging and anti-apoptotic properties. Biochim. Biophys. Acta. 2002, 1570, 181–191. [Google Scholar] [CrossRef]
- Chen, W.-J.; Song, J.-R.; Guo, P.; Wen, Z.-Y. Butein, a more effective antioxidant than α-tocopherol. J. Mol. Struct. THEOCHEM 2006, 763, 161–164. [Google Scholar] [CrossRef]
- Lee, S. H.; Seo, G. S.; Sohn, D. H. Inhibition of lipopolysaccharide-induced expression of inducible nitric oxide synthase by butein in RAW264.7 cells. Biochem. Biophys. Res. Comm. 2004, 323, 125–132. [Google Scholar] [CrossRef]
- Lee, S. H.; Seo, G. S.; Jin, X. Y.; Ko, G.; Sohn, D. H. Butein blocks tumor necrosis factor α-induced interleukin 8 and matrix metalloproteinase 7 production by inhibiting p38 kinase and osteopontin mediated signaling events in HT-29 cells. Life Sci. 2007, 81, 1535–1543. [Google Scholar] [CrossRef]
- Wang, Y.; Chan, F. L.; Chen, S.; Leung, L. K. The plant polyphenol butein inhibits testosterone-induced proliferation in breast cancer cells expressing aromatase. Life Sci. 2005, 77, 39–51. [Google Scholar] [CrossRef]
- Yu, S. M.; Cheng, Z. J.; Kuo, S. C. Endothelium-dependent relaxation of rat aorta by butein, a novel cyclic AMP-specific phosphodiesterase inhibitor. Eur. J. Pharmacol. 1995, 280, 69–77. [Google Scholar] [CrossRef]
- Kang, D. G.; Kim, Y. C.; Sohn, E. J.; Lee, Y. M.; Lee, A. S.; Yin, M. H.; Lee, H. S. Hypotensive effect of butein via the inhibition of angiotensin converting enzyme. Biol. Pharm. Bull. 2003, 26, 1345–1347. [Google Scholar] [CrossRef]
- Hayashi, K.; Nagamatsu, T.; Honda, S.; Sukuki, Y. Butein (3,4,2′,4′-tetrahydroxychalcone) ameliorates experimental anti-glomerular basement membrane antibody-associated glomerulonephritis. Eur J. Pharmacol. 1996, 316, 297–306. [Google Scholar]
- Kim, N. Y.; Pae, H. O.; Oh, G. S.; Kang, T. H.; Kim, Y. C.; Rhew, H. Y.; Chung, H. T. Butein, a plant polyphenol, induces apoptosis concomitant with increased caspase-3 activity, decreased Bcl-2 expression and increased Bax expression in HL-60 cells. Pharmacol. Toxicol. 2001, 88, 261–266. [Google Scholar]
- Iwashita, K.; Kobori, M.; Yamaki, K.; Tsushida, T. Flavonoids inhibit cell growth and induce apoptosis in B16 melanoma 4A5 cells. Biosci. Biotechnol. Biochem. 2000, 64, 1813–1820. [Google Scholar] [CrossRef]
- Hung, C.-Y.; Yen, G.-C. Extraction and identification of antioxidative components of Hsian-tsao (Mesona procumbens Hemsl.). Lebensm.-Wiss. u.-Technol. 2001, 34, 306–311. [Google Scholar] [CrossRef]
- Ramadan, M. F.; Zayed, R.; El-Shamy, H. Screening of bioactive lipids and radical scavenging potential of some solanaceae plants. Food Chem. 2007, 103, 885–890. [Google Scholar] [CrossRef]
- Batta, A. K.; Xu, G.; Honda, A.; Miyazaki, T.; Salen, G. Stigmasterol reduces plasma cholesterol levels and inhibits hepatic synthesis and intestinal absorption in the rat. Metabolism 2006, 55, 292–299. [Google Scholar] [CrossRef]
- Sample Availability: Contact the authors.
© 2008 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Prachayasittikul, S.; Buraparuangsang, P.; Worachartcheewan, A.; Isarankura-Na-Ayudhya, C.; Ruchirawat, S.; Prachayasittikul, V. Antimicrobial and Antioxidative Activities of Bioactive Constituents from Hydnophytum formicarum Jack. Molecules 2008, 13, 904-921. https://doi.org/10.3390/molecules13040904
Prachayasittikul S, Buraparuangsang P, Worachartcheewan A, Isarankura-Na-Ayudhya C, Ruchirawat S, Prachayasittikul V. Antimicrobial and Antioxidative Activities of Bioactive Constituents from Hydnophytum formicarum Jack. Molecules. 2008; 13(4):904-921. https://doi.org/10.3390/molecules13040904
Chicago/Turabian StylePrachayasittikul, Supaluk, Prasit Buraparuangsang, Apilak Worachartcheewan, Chartchalerm Isarankura-Na-Ayudhya, Somsak Ruchirawat, and Virapong Prachayasittikul. 2008. "Antimicrobial and Antioxidative Activities of Bioactive Constituents from Hydnophytum formicarum Jack." Molecules 13, no. 4: 904-921. https://doi.org/10.3390/molecules13040904
APA StylePrachayasittikul, S., Buraparuangsang, P., Worachartcheewan, A., Isarankura-Na-Ayudhya, C., Ruchirawat, S., & Prachayasittikul, V. (2008). Antimicrobial and Antioxidative Activities of Bioactive Constituents from Hydnophytum formicarum Jack. Molecules, 13(4), 904-921. https://doi.org/10.3390/molecules13040904




