Abstract
In a bioassay-guided search for acetylcholinesterase (AChE) inhibitors from 180 medicinal plants, an ethyl acetate extract of whole plants of Agrimonia pilosa ledeb yielded tiliroside (1), 3-methoxy quercetin (2), quercitrin (3) and quercetin (4). We report herein for the first time that all four flavonol compounds showed significant inhibitory effects on AChE, particularly quercetin (4), which showed twice the activity of dehydroevodiamine (DHED).
Introduction
Alzheimer’s disease is a chronic neurodegenerative disorder which is characterised by a loss of cognitive ability, severe behavioral abnormalities and ultimately death. Alzheimer’s disease is the most common cause of dementia among the elderly and there are currently 2.5 to 4.0 million estimated Alzheimer’s disease patients in the United States and some 17 to 25 million worldwide [1,2]. One treatment strategy to enhance cholinergic functions is the use of acetylcholinesterase (AChE, EC3.1.1.7) inhibitors to increase the amount of acetylcholine present in the synapses between cholinergic neurons. In this respect, use of a selective inhibitor for acetylcholinesterase has attracted particular attention for treatment of the Alzheimer-type dementia [3]. Some AChE inhibitors like physostigmine or tacrine are known to have limitations such as short half life or side effects like hepatotoxicity. Alkylpyridium polymers, dehydroevodiamine (DHED) and carbamate type AChE inhibitors have been reported, but because of bioavailability problems and possible side effects, there is still great interest in finding better AChE inhibitors [4].
In our continued search for biologically active compounds from traditional medicines, we wanted to identify AChE-inhibiting small molecules from herbal medicinal plants. The stems and the leaves of Agrimonia pilosa ledeb have shown diverse pharmacological properties such as anti-tumor, analgesic, antibacterial, anti-inflammatory [5], hypoglycemic and vasoconstrictor activities. The plant is used in the treatment of abdominal pain, sore throat, headaches, bloody and mucoid dysentery and heat stroke. Over the past two decades several research groups have reported the isolation of two dozen various compounds from this plant [6,7,8]. This paper describes the isolation and characterization of flavonoid compounds and from Agrimonia pilosa ledeb the first reported acetylcholinesterase (AChE) inhibitory activity of components of this sp.
Results and Discussion
During screening for AChE inhibitors from 180 medicinal plants, we found that the ethyl acetate extract of the whole plants of Agrimonia pilosa ledeb showed significant AChE inhibition. To isolate the AChE inhibitory constituents, bioactivity-guided fractionation and subsequent chromatography of the ethyl acetate extract on silica gel, Sephadex LH-20 and MCI-gel columns (summarized in Figure 1) led to the isolation of four flavonoids as active constituents: tiliroside (1) [9,10,11,12,13], 3-methoxyquercetin (2) [14], quercitrin (3) [9,14,15] and quercetin (4) [8,14], respectively. The structures of these compounds (Figure 2) were fully elucidated on the basis of physicochemical and spectral data (in particular 1H-NMR, 13C-NMR, DEPT, HMBC, TOCSY and NOESY) and by comparison with published data [8,10,14].
For example, compound 1 showed UV absorption bands at 266 and 310 nm with a shoulder at 278 nm. The IR spectrum presented bands at 3262 (OH), 1654 (C=O) and 1606 cm-1 (C=C). The 13C-NMR and DEPT spectra showed 30 signals, six of which could be assigned to a β-glucopyranosyl moiety. Fifteen other signals were similar to those of kaempferol. The remaining seven signals are compatible with a p-coumaroyl moiety. In the 1H-NMR spectrum, the main difference is due to the B-ring of the flavonoid skeleton: the spectrum showed two signals, both integrating for two protons, with J=7.7 Hz at δ 7.86 (2H, H2'/H6') and at δ 6.71 (2H, H3'/H5'), clearly indicating a kaempferol derivative. The singlets at δ 6.02 and δ 6.18 were assigned to H6, H8, respectively. Two other doublets at δ 6.71 (J=7.3) and δ 7.20 (J=7.2 Hz) were attributed to H3'''/H5''' and H2'''/ H6''' of the p-coumaroyl moiety, respectively. The two doublets (J=13.2) at d 5.99 and 7.31 were assigned to Hγ and Hß of a p-coumaroyl moiety with trans stereochemistry, respectively. The signal at δ 5.13 (J=7.0 Hz) was assigned to a D-glucose moiety with ß-configuration.
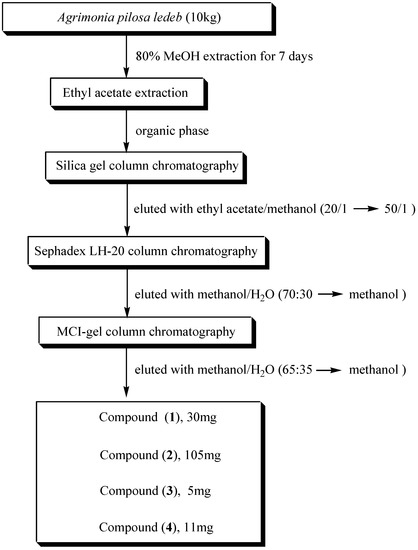
Figure 1.
Isolation of acetylcholinesterase inhibitors from Agrimonia pilosa ledeb.
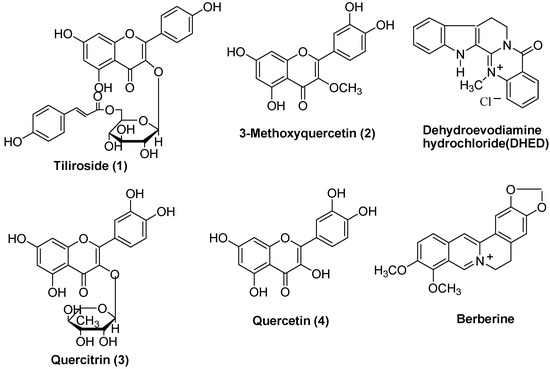
Figure 2.
The flavonoids 1-4 isolated from Agrimonia pilosa ledeb and structures of DHED and berberine.
The 13C-NMR spectrum presented two intense signals at δ 130.9 and at δ 114.7, assigned to H2'/H6' and H3'/H5', respectively. The signals at δ 121.4 and at δ 160.2 are typical of an unsubstituted B-ring of a kaempferol-like structure. The signal at δ 63.6 (CH2) shows that the p-coumaroyl linkage was at C-6 of the glucose unit. In the HREI-MS, the molecular ion peak of was observed at 594 a.m.u. After distortionless enhancement by polarization transfer (DEPT), the molecular formula of compound 1 was determined to be C30H26O13. On the basis of HREI-MS and NMR data, compound (1) was thus determined to be tiliroside (kaempferol-3-o-β-D-6’’(p-coumaroyl)glucopyranoside). The 1H-NMR and 13C-NMR, 2D-NMR, and IR data of compound 1 are shown below (Figure 3, Figure 4, Figure 5, Figure 6 and Figure 7).
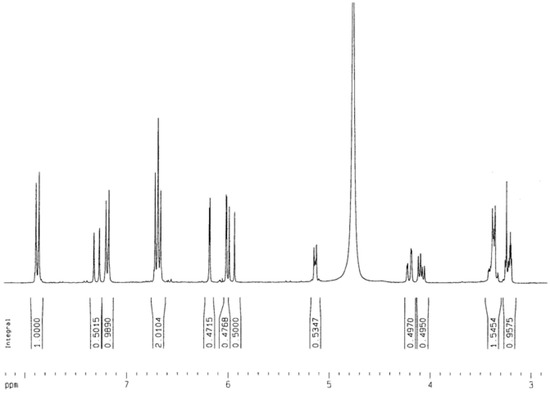
Figure 3.
1H-NMR spectrum of compound 1 (CD3OD, 250MHz).
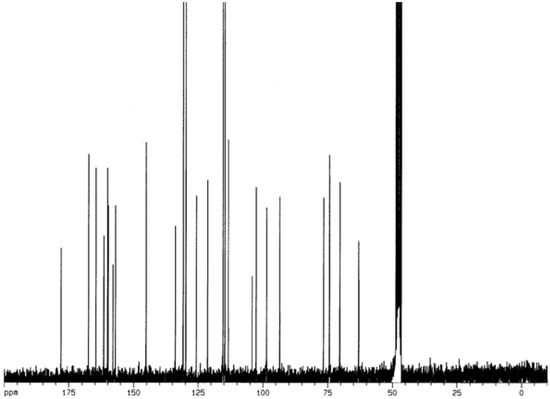
Figure 4.
13C-NMR spectrum of compound 1 (CD3OD, 250MHz).
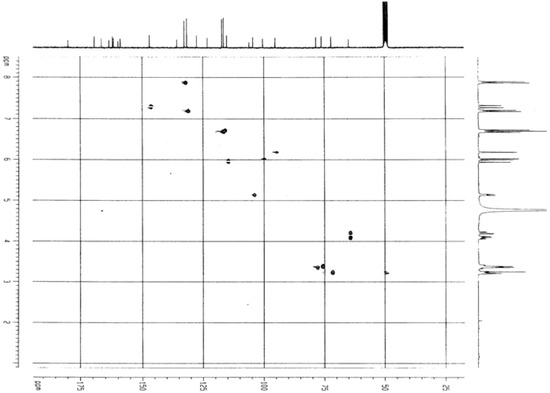
Figure 5.
HMBC spectrum of compound 1 (CD3OD, 250MHz).
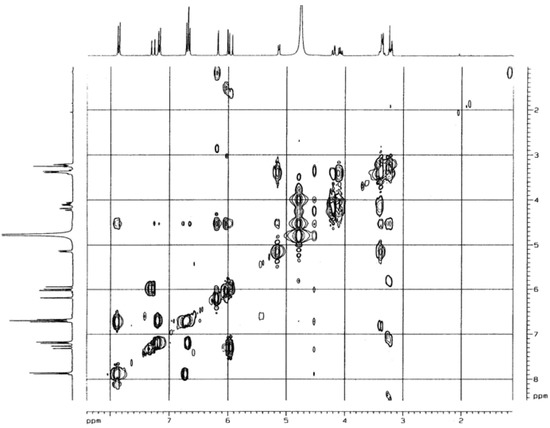
Figure 6.
2D-COSY spectrum of compound 1 (CD3OD, 250MHz).
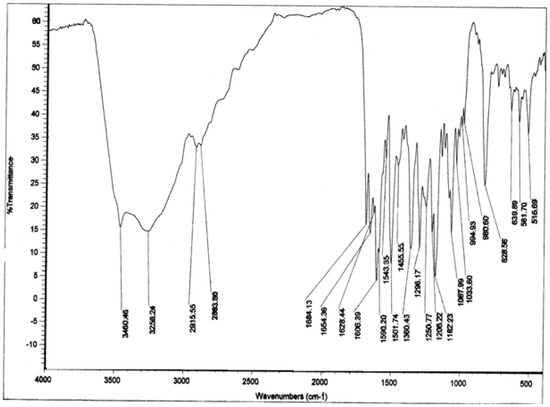
Figure 7.
IR spectrum of compound 1.
Biological activity
These compounds inhibited AChE activity in a dose dependent manner and the IC50 (50% AChE inhibitory effect) values of 1-4 were determined to be 23.5, 37.9, 66.9 and 19.8 μM, respectively, while the IC50 value of a positive control, dehydroevodiamine (DHED) hydrochloride, was 37.8 μM (Table 1).

Table 1.
The inhibitory activities of compounds 1-4 on AChE.
| Compound | +IC50 (μM) |
| Tiliroside (1) | 23.5 |
| 3-Methoxy quercetin (2) | 37.9 |
| Quercitrin (3) | 66.9 |
| Quercetin (4) | 19.8 |
| Dehydroevodiamine(DHED) | 37.8 |
+ The values indicate 50% AChE inhibitory effect and are the means of triplicate data.
These flavonoids are less effective than the clinically useful tacrine (IC50 = 0.1 μM) and berberine (IC50 = 0.7 μM). However, tiliroside 1 and quercetin 4 showed higher AChE ihhibitory activity (IC50) than dehydroevodiamine hydrochloride [16].
Conclusions
In summary, tiliroside, quercetin, quercitrin and 3-methoxyquercetin were isolated from the ethyl acetate extracts of the whole plants of Agrimonia pilosa ledeb, with tiliroside (1) and 3-methoxy-quercetin (2) being isolated for the first time from this plant sp. The present study also confirms that these compounds have significant anticholinesterase activity. Particularly quercetin (4) was twice as active against AChE than DHED, which showed more in vivo antiamnesic activty than the clinically useful tacrine [16] and thus, quercetin or its derivatives might have the therapeutic potential for the treatment in Alzheimer’s disease.
Experimental
General
Chemical grade reagents were obtained from Sigma-Aldrich (Milwaukee, Wisconsin). The reference inhibitor compound, tacrine, was purchased from Acros Organics (Geel, Belgium). Electric eel acetylcholinesterase, acetylthiocholine iodide, and 5-5′-thiobis-2-nitrobenzoic acid (DTNB) were purchased from Sigma (St. Louis, Mo.). Buffers and other chemicals were of analytical grade. To purify the active compounds, silica gel (particle size: 0.040-0.063 mm, Merck Co., Darmstadt, Germany), ODS RP-18 (YMC-GEL ODS-A, 120 A, S-150 um, YMC, Kyoto, Japan), Sephadex LH-20 (Amersham Pharmacia Biotech Co., Uppsala, Sweden), and thin layer chromatography precoated silica gel (20 PLC plates 20 × 20, silica gel 60 F254, 0.5 mm thickness, Merck Co., Darnstadt, Germany) or Gel (CHP20P, 75-150 µ, Mitsubishi Chemical Co., Tokyo, Japan) were used. The high purity solvents used for HPLC were from Fisher Scientific and Burdick & Jackson (Allied Signal, Muskegon, USA), and other solvents for the isolation were obtained from Duck-San Co. (Ansan, Korea).
Plant material
The dried whole plants of Agrimonia pilosa ledeb were purchased from the local market in Seoul, Korea and botanically identified. A voucher specimen is deposited in the Bioorganic & Medicinal Chemistry Laboratory, Yonsei University.
Extraction, Screening and Isolation
The dried leaves of Agrimonia pilosa ledeb (10 kg) were cut into small pieces and extracted three times with 80% MeOH at room temperature for 7 days. The MeOH extract were filtered and concentrated under reduced pressure. The liquid residue was suspended in water and then partitioned successively in turn with hexane, methylene chloride, ethyl acetate and n-butyl alcohol. The concentration of each extract was adjusted to 1 mg/mL. Extracted sample (50 µL) was added to AChE reaction mixture (1.5 mL; final concentration of each extract = 30 µg/mL). The samples possessing high inhibitory activity towards AChE (details below) were selected for further isolation of active compounds as AChE inhibitors. The active ethyl acetate extract (40g, % yield 4x10-3% from the natural medicine) was fractionated into twelve fractions on silica gel 60 (70-230 mesh, Merck, stepwise elution with 50:1 EtOAc-MeOH). The active fraction 5-3 was concentrated and then rechromatographed on silica gel column using EtOAc with increasing amounts of MeOH (40:1). The tiliroside eluate was purified by Sephadex LH-20 column chromatography with pure MeOH, followed by an MCI-gel column eluted with 70% aqueous MeOH to give compound 1 (30 mg, 7x10-4 % yield). Fraction 6-5 was eluted stepwise with EtOAc-MeOH (20-1), concentrated and then rechromatographed on silica gel column using EtOAc with increasing amounts of MeOH (10:1). The quercetin eluate was purified by Sephadex LH-20 column chromatography with MeOH, followed by an MCI-gel column with 65% aqueous MeOH to give compound 2 (105㎎, % yield mg, 2.6x10-3). Fraction 5-5 was eluted stepwise with EtOAc-MeOH (50:1), concentrated and then chromatographed again on a silica gel column using EtOAc with increasing amounts of MeOH (40:1) as eluent. The quercitrin eluate was purified by Sephadex LH-20 column chromatography with MeOH, followed by an MCI-gel column with 70% aqueous MeOH to give compound 3 (5 mg, 1x10-4 % yield). Finally, Fraction 6-7 was eluted stepwise with EtOAc-MeOH (30-1), concentrated and then chromatographed on a silica gel column using EtOAc with increasing amounts of MeOH (20:1). The 3-methoxy-quercetin eluate was purified by Sephadex LH-20 column chromatography with MeOH, followed by an MCI-gel column with 70% aqueous MeOH to give compound 4 (11 mg, 2x10-4 % yield).
Compound 1: Pale yellow solid; mp 269-271 °C; MS m/z 594 (M+); 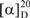 = -62.5 °(C. 0.5 MeOH), C30H26O13; 1H-NMR (CD3OD) δ: 3.21 (2H, m, C3″,4″), 3.37 (1H, m, C2″), 3.38 (1H, m, C5″), 4.19 (2H, m, C6″), 5.13 (1H, m, C1″), 5.99 (1H, d, Cγ), 6.02 (1H, d, C6), 6.18 (1H, d, C8), 6.71 (4H, t, C3′,5′,3″′,5″′), 7.20 ( 2H, d, C2″’,6″′), 7.31 (1H, Cß ), 7.86 (2H, d, C2′,6′); 13C NMR: 63.06 (C6″), 70.42 (C4″), 74.44 (C3″), 74.50 (C2″), 76.71 (C5″), 93.57 (C8), 98.70 (C6), 102.72 (C1″), 104.28 (C10), 113.44 (Cß), 114.74 (C3′, 5′), 115.48 (C3″′, 5″′), 121.40 (C1’), 125.78 (C1″′), 129.89 (C2″′, 6″″), 130.93 (C2′, 6′), 133.93 (C3), 145.26 (Cγ), 157.07 (C9), 158.01 (C2), 159.87 (C4″′), 160.21 (C4′), 161.63 (C5), 164.65 (C7), 167.53 (Cα), 178.09 (C4); IR υmax 3262 (OH), 1654 (C=O), 1606 (C=C) cm-1.
= -62.5 °(C. 0.5 MeOH), C30H26O13; 1H-NMR (CD3OD) δ: 3.21 (2H, m, C3″,4″), 3.37 (1H, m, C2″), 3.38 (1H, m, C5″), 4.19 (2H, m, C6″), 5.13 (1H, m, C1″), 5.99 (1H, d, Cγ), 6.02 (1H, d, C6), 6.18 (1H, d, C8), 6.71 (4H, t, C3′,5′,3″′,5″′), 7.20 ( 2H, d, C2″’,6″′), 7.31 (1H, Cß ), 7.86 (2H, d, C2′,6′); 13C NMR: 63.06 (C6″), 70.42 (C4″), 74.44 (C3″), 74.50 (C2″), 76.71 (C5″), 93.57 (C8), 98.70 (C6), 102.72 (C1″), 104.28 (C10), 113.44 (Cß), 114.74 (C3′, 5′), 115.48 (C3″′, 5″′), 121.40 (C1’), 125.78 (C1″′), 129.89 (C2″′, 6″″), 130.93 (C2′, 6′), 133.93 (C3), 145.26 (Cγ), 157.07 (C9), 158.01 (C2), 159.87 (C4″′), 160.21 (C4′), 161.63 (C5), 164.65 (C7), 167.53 (Cα), 178.09 (C4); IR υmax 3262 (OH), 1654 (C=O), 1606 (C=C) cm-1.
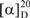 = -62.5 °(C. 0.5 MeOH), C30H26O13; 1H-NMR (CD3OD) δ: 3.21 (2H, m, C3″,4″), 3.37 (1H, m, C2″), 3.38 (1H, m, C5″), 4.19 (2H, m, C6″), 5.13 (1H, m, C1″), 5.99 (1H, d, Cγ), 6.02 (1H, d, C6), 6.18 (1H, d, C8), 6.71 (4H, t, C3′,5′,3″′,5″′), 7.20 ( 2H, d, C2″’,6″′), 7.31 (1H, Cß ), 7.86 (2H, d, C2′,6′); 13C NMR: 63.06 (C6″), 70.42 (C4″), 74.44 (C3″), 74.50 (C2″), 76.71 (C5″), 93.57 (C8), 98.70 (C6), 102.72 (C1″), 104.28 (C10), 113.44 (Cß), 114.74 (C3′, 5′), 115.48 (C3″′, 5″′), 121.40 (C1’), 125.78 (C1″′), 129.89 (C2″′, 6″″), 130.93 (C2′, 6′), 133.93 (C3), 145.26 (Cγ), 157.07 (C9), 158.01 (C2), 159.87 (C4″′), 160.21 (C4′), 161.63 (C5), 164.65 (C7), 167.53 (Cα), 178.09 (C4); IR υmax 3262 (OH), 1654 (C=O), 1606 (C=C) cm-1.
= -62.5 °(C. 0.5 MeOH), C30H26O13; 1H-NMR (CD3OD) δ: 3.21 (2H, m, C3″,4″), 3.37 (1H, m, C2″), 3.38 (1H, m, C5″), 4.19 (2H, m, C6″), 5.13 (1H, m, C1″), 5.99 (1H, d, Cγ), 6.02 (1H, d, C6), 6.18 (1H, d, C8), 6.71 (4H, t, C3′,5′,3″′,5″′), 7.20 ( 2H, d, C2″’,6″′), 7.31 (1H, Cß ), 7.86 (2H, d, C2′,6′); 13C NMR: 63.06 (C6″), 70.42 (C4″), 74.44 (C3″), 74.50 (C2″), 76.71 (C5″), 93.57 (C8), 98.70 (C6), 102.72 (C1″), 104.28 (C10), 113.44 (Cß), 114.74 (C3′, 5′), 115.48 (C3″′, 5″′), 121.40 (C1’), 125.78 (C1″′), 129.89 (C2″′, 6″″), 130.93 (C2′, 6′), 133.93 (C3), 145.26 (Cγ), 157.07 (C9), 158.01 (C2), 159.87 (C4″′), 160.21 (C4′), 161.63 (C5), 164.65 (C7), 167.53 (Cα), 178.09 (C4); IR υmax 3262 (OH), 1654 (C=O), 1606 (C=C) cm-1.Compound 2: White powder; formula: C15H10O7; MS: Negative ES-MS, m/z (rel. int.): 301 ([M+-H] 100); 1H-NMR (CD3OD) δ: 7.77 (1H, d, 2.1, H2′), 7.67 (1H, dd, J=2.1 and 8.5 Hz, H6′), 6.92 (1H, d, J =8.5 Hz, H5′), 6.43 (1H, d, J=1.9 Hz, H8), 6.22 (1H, d, J=1.9 Hz, H6); 13C-NMR δ: 175.7 (C4), 163.9 (C7), 160.7 (C5), 156.2 (C9), 146.9 (C4′), 147.6 (C2), 145.0 (C3′), 135.6 (C3), 122.0 (C1′), 120.0 (C6′), 115.6 (C5′), 115.3 (C2′), 103.0 (C10), 98.2 (C6), 93.4 (C8).
Compound 3: Formula: C21H20O11; MS: Negative ES-MS, m/z (rel. int.): 447 ([M+-H] 100); 1H-NMR: (CD3OD) δ: 7.38 (d, H-2′, J(2′-6′)=2.1 Hz), 7.35 (dd, H-6′, J(6′-5′)=8.3 Hz), 6.95 (d, H-5′), 6.41 (d, H-8, J(8-6)=2.2 Hz), 6.24 (d, H-6), 5.40 (d, H-1″, J(1″-2″)=1.7 Hz), 4.26 (dd, H-2″, J(2″-3″)= 3.4 Hz), 3.79 (dd, H-3″, J(3″-4″)=9.3 Hz), 3.46 (dq, H-5″, J(4″-5″)=9.6 Hz, J(5″ 6″)=6.0 Hz), 3.37 (dd, H-4″), 0.98 (d, H-6″).
Compound 4: White powder; formula: C15H10O7; MS: Negative ES-MS, m/z (rel. int.): 301 ([M+-H] 100); 1H-NMR (CD3OD) δ: 7.77 (1H, d, J=2.1 Hz, H2′), 7.67 (1H, dd, J=2.1 and 8.5 Hz, H6′), 6.92 (1H, d, J=8.5 Hz, H5′), 6.43 (1H, d, J=1.9, H8), 6.22 (1H, d, J=1.9 Hz, H6), 3.64 (3H, s, OCH3); 13C-NMR δ: 175.7 (C4), 163.9 (C7), 160.7 (C5), 156.2 (C9), 146.9 (C4′), 147.6 (C2), 145.0 (C3′), 135.6 (C3), 122.0 (C1′), 120.0 (C6′), 115.6 (C5′), 115.3 (C2′), 103.0 (C10), 98.2 (C6), 93.4 (C8), 56.1 (C3).
Determination of inhibitory activity against acetylcholinesterase
Acetylcholinesterase inhibition was determined spectrophotometically using acetylthiocholine as substrate by modifying the Ellman method [17]. The reaction was carried in 100 mM sodium phosphate buffer (pH 8.0) at 25 °C. In a typical assay, buffer (140 µL), enzyme preparation (20 mL) and test compound solution (20 µL) were mixed and incubated for 30 min. DTNB (10 µL) was added and the reaction was then started by adding acetylthiocholine (10 µL). The hydrolysis of acetylthiocholine was determined by monitoring the formation of yellow 5-thio-2-nitrobenzoate anion as a result of the reaction of 5,5’-dithiobis-2-nitrobenzoic acid with thiocholine released by the enzymatic hydrolysis of acetylthiocholine at a wavelength of 410 nm on microtitre plate spectrophotometer.
Acknowledgments
This study was supported by a research grant from the Ministry of Health and Welfare (Project No. B040002), the Republic of Korea. M. P. acknowledges the fellowship of the BK 21 program from the Ministry of Education and Human Resources Development.
References
- Parnetti, L.; Senin, U.; Mecocci, P. Cognitive enhancement therapy for Alzheimer’s disease. The way forward. Drugs 1997, 53, 752–768. [Google Scholar]
- Brinton, R.D.; Yamazaki, R.S. Advances and Challenges in the Prevention and Treatment of Alzeimer’s Disease. Pharmaceut. Res. 1998, 15, 386–398. [Google Scholar] [CrossRef]
- Kim, W.G.; Cho, K.M.; Lee, C.K.; Yoo, I.D. Terreulactone A, a novel meroterpenoid with anti-acetylcholinesterase activity from Aspergillus terreus. Tetrahedron Lett. 2002, 43, 3197–3198. [Google Scholar] [CrossRef]
- Schneider, L.S. J. Clin. New therapeutic approaches to Alzeimer’s Disease. Psychiatry 1996, 57, 30–36. [Google Scholar]
- Sala, A.; Recio, M.C.; Schinella, G.R.; Manez, S.; Giner, R.M.; Cerda-Nicolas, M.; Rios, J-L. Assessment of the anti-inflammatory activity and free radical scavenger activity of tiliroside. Eur. J. Pharmacol. 2003, 461, 53–61. [Google Scholar]
- a) Pei, Y.H.; Li, X.; Zhu, T.R. Studies on the chemical constituents from the root-sprouts of Agrimonia pilosa Ledeb. Yao Hsueh Hsueh Pao 1989, 24, 431–437. [Google Scholar] b) Pei, Y.H.; Li, X.; Zhu, T.R.; Wu, L.J. Studies on the structurs of a new flavanonol glucoside of the root-sprouts of Agrimonia pilosa ledeb. Yao Xue Xue Bao 1990, 25, 267–270. [Google Scholar]
- Isao, K.; Naosuke, B.; Yumiko, O.; Nobusuke, K. Triterpenoids from Agrimonia pilosa. Phytochemistry 1988, 27, 297–299. [Google Scholar] [CrossRef]
- Su., G.; Su, S.; Zhu, T. Studies on bacteriostatic components from Agrimonia pilosa Ledeb. Shenyang Yaoxueyuan Xuebao 1984, 1, 44–50. [Google Scholar]
- Powell, R.G.; Smith, C.R.; Bajaj, R.; Chang, C.J.; McLaughlin, J. Tiliroside from the seeds of Eremocarpus setigerus. J. Nat. Prod. 1986, 49, 1174–1175. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.Y.; Oh, S.R.; Lee, I.S.; Ahn, K.S.; Lee, J.J.; Lee, H.K. Anti-complement activity of tiliroside from the flower buds of Magnolia fargesii. Biol. Pharm. Bull. 1998, 21, 1077–1078. [Google Scholar] [PubMed]
- Liu, H.; Orjala, J.; Sticher, O.; Rali, T. Acylated flavonol glycosides from the leaves of Stenocleane palustris. J. Nat. Prod. 1999, 62, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Dimas, K.; Demetzos, C.; Mitaku, S.; Marselos, M.; Tzavaras, T.; Kokkinopoulos, D. Cytotoxic activity of Kaempferol glycosides against human leukaemic cell lines in vitro. Pharmacol Res. 2000, 41, 85–88. [Google Scholar]
- Matsuda, H.; Ninomiya, K.; Shimoda, H.; Yoshikawa, M. Hepatoprotective principles from the flowers of Tila argentea (Linden): Structure requirements of tiliroside and mechanisms of action. Bioorg. Med. Chem. 2002, 10, 707–712. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.F.; Strack, D.; Baumert, A.; Subramaniam, R.; Goh, N.K.; Chia, T.F.; Tan, S.N.; Chia, L. S. Antioxidant flavonoids from leaves of Polygonum hydropiper L. Phytochemistry 2003, 62, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Buckova, A.; Eisenreichova, E.; Homola, V.; Leifertova, I.; Licha, K.; Natherova, L. Acta Facult. Pharm. Univ. Comenianae. 1972, 22, 47–73.
- Park, C.H.; Kim, S.H.; Choi, W.; Lee, Y.J.; Kim, J.S.; Kang, S.S.; Suh, Y.H. Novel Anticholinesterase and antiamnestic activites of dehydroevodiamine, a constituent of Evodia rutaecarpa. Planta Med. 1996, 62, 405–409. [Google Scholar] [CrossRef] [PubMed]
- Ellman, G.L.; Courtney, K.D.; Andres, V.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Milligram quantities of compounds 1 and 2 are available from the authors.
© 2007 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.