Synthesis of Novel Benzimidazolyl-substituted Acrylonitriles and Amidino-substituted Benzimidazo[1,2-a]Quinolines
Abstract
:Introduction
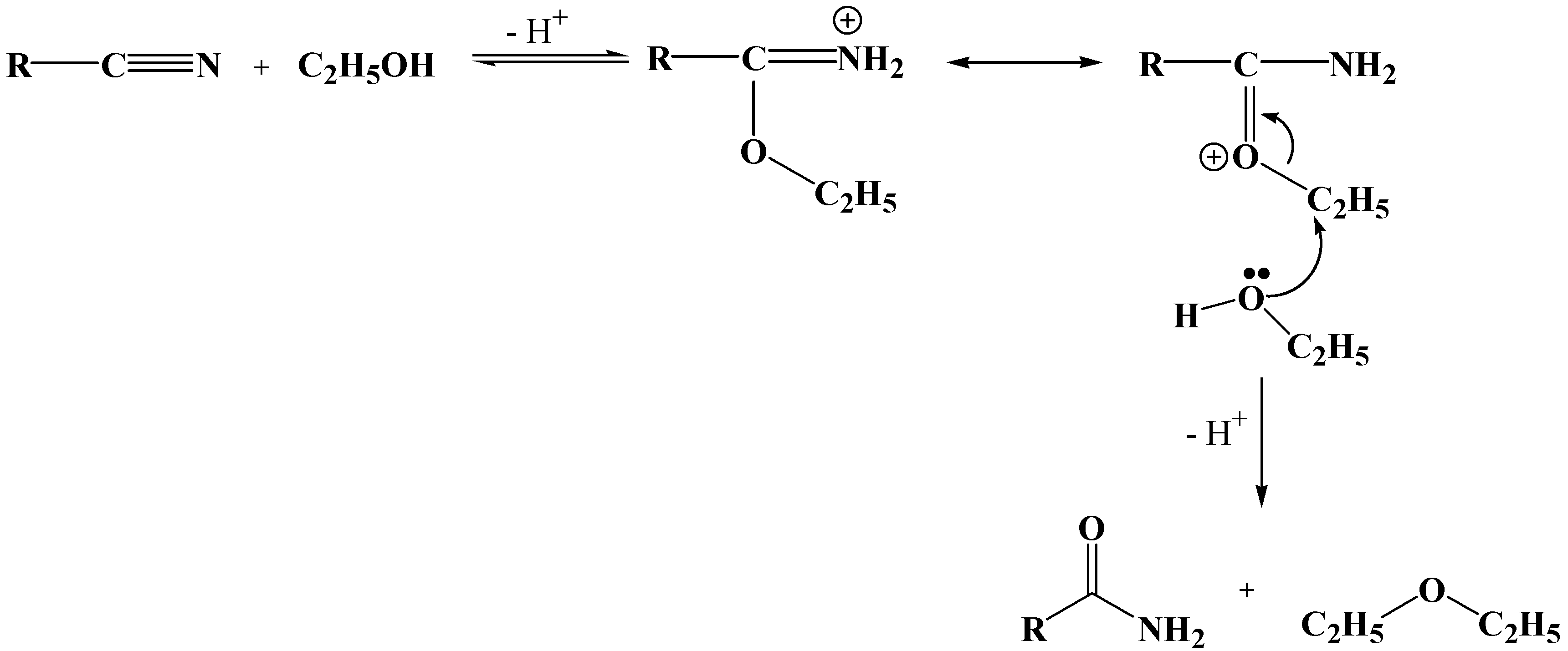
Results and Discussion
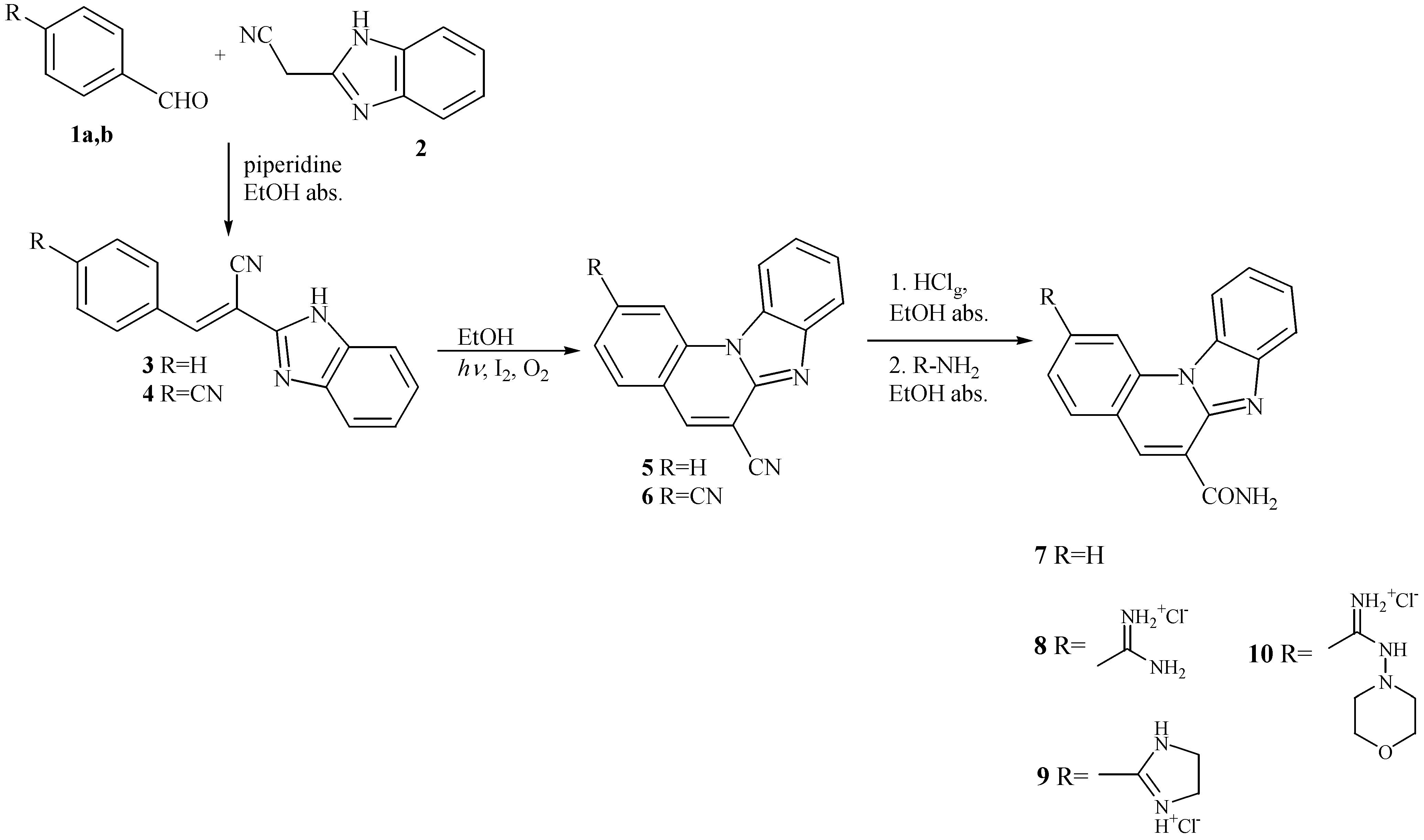
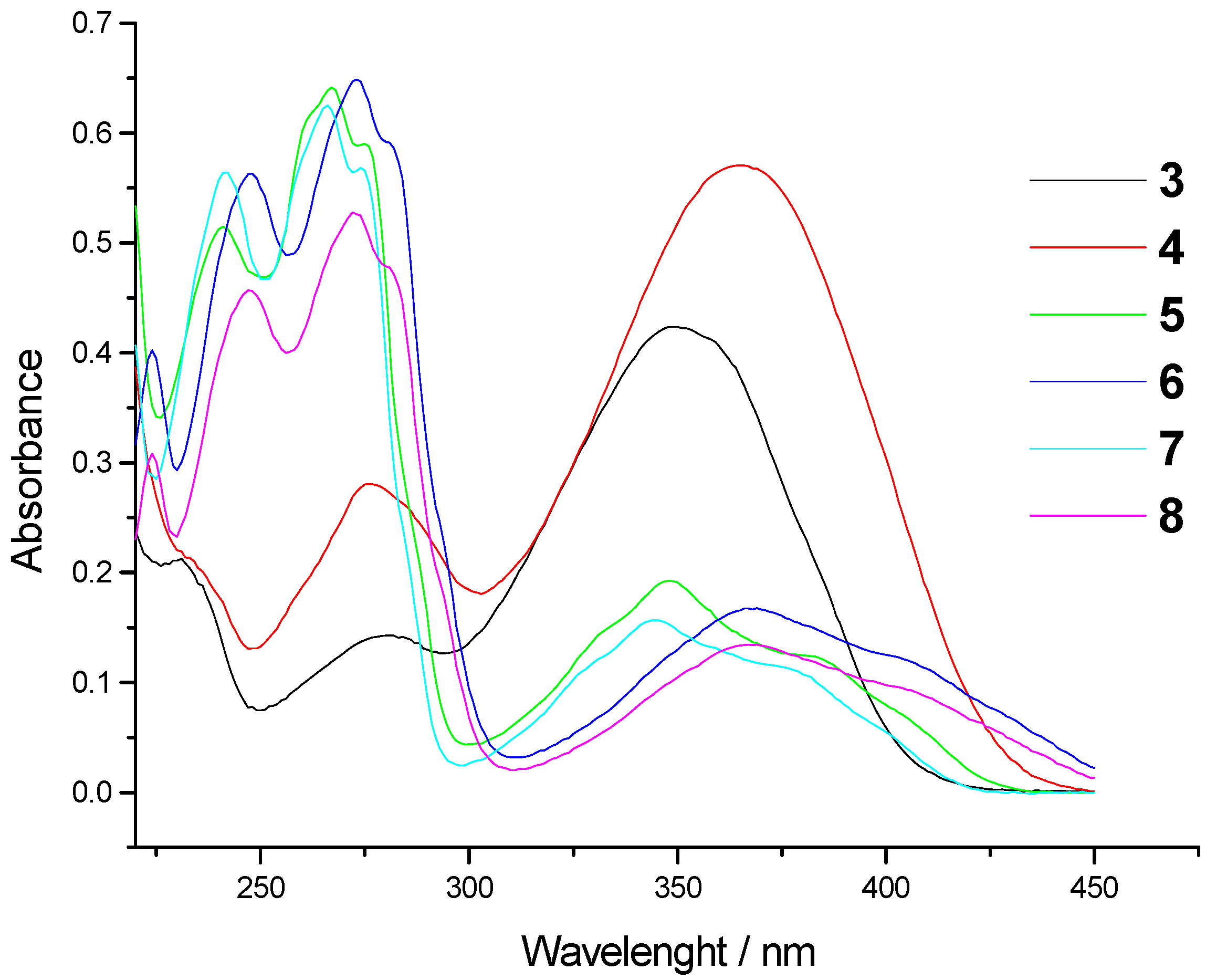
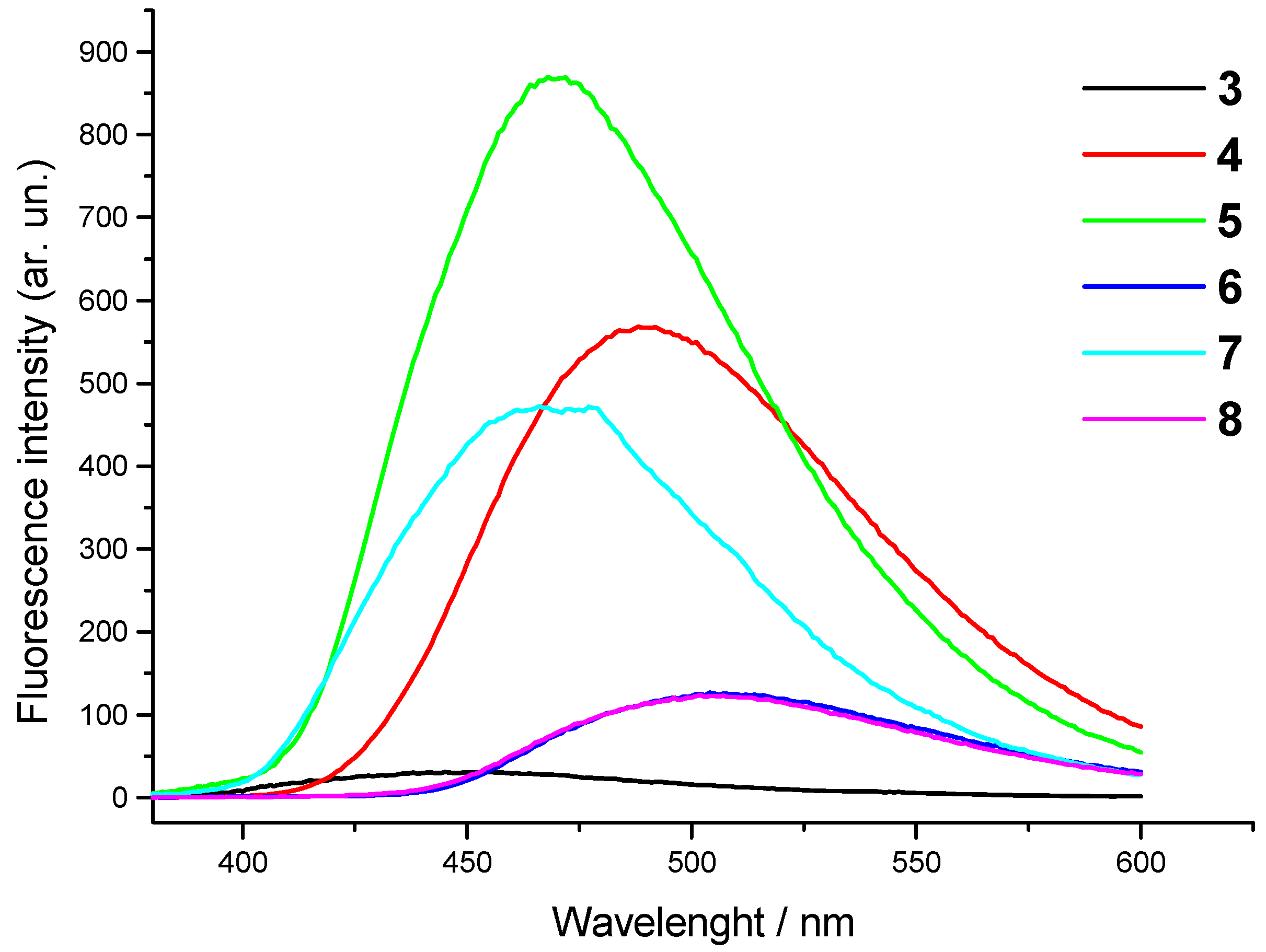
| Comp. | λmax/nm | ε x 103 (dm3mol-1 cm-1) | λemiss/nm | λexc/nm | Int (arb. un.) |
|---|---|---|---|---|---|
| 3 | 355 280 | 21.7 8.4 | 445 | 352 280 | 28.6 |
| 4 | 371 278 | 17.6 12.1 | 490 | 367 274 | 570.0 |
| 5 | 387 351 270 | 6.4 9.9 35.5 | 469 | 350 268 245 | 864.5 |
| 6 | 370 276 | 7.9 32.5 | 507 | 370 274 247 | 126.3 |
| 7 | 375 344 267 | 7.8 4.6 28.9 | 467 | 345 268 240 | 469.5 |
| 8 | 374 276 | 8.1 35.2 | 507 | 368 272 249 | 121.1 |
Conclusions
Experimental
General
General method for preparation of 3 and 4.
2-(1H-benzimidazol-2-yl)-3-acrylonitrile (3)
2-(1H-benzimidazol-2-yl)-3-(4-cyanophenyl)-acrylonitrile (4)
General method for preparation of 5 and 6
Benzimidazo[1,2-a]quinoline-6-carbonitrile (5)
Benzimidazo[1,2-a]quinoline-2,6-dicarbonitrile (6)
General method for preparation of benzimidazo[1,2-a]quinoline-6-carboxamide 7 and 2-amidino-benzimidazo[1,2-a]quinoline-6-carboxamides 8-10
Benzimidazo[1,2-a]quinoline-6-carboxamide (7)
2-Carboxamidine-benzimidazo[1,2-a]quinoline-6-carboxamide hydrochloride (8)
2-(1H-imidazol-2-yl)-benzimidazo[1,2-a]quinoline-6-carboxamide Hydrochloride (9)
N-Morpholin-4-yl-benzimidazo[1,2-a]quinoline-6-carboxamide hydrochloride (10)
Acknowledgements
References
- Lobato, J.; Canizares, P.; Rodrigo, M. A.; Linares, J. J.; Manjavacas, G. Synthesis and characterisation of poly[2,2-(m-phenylene)-5,5-bibenzimidazole] as polymer electrolyte membrane for high temperature PEMFCs. J. Membrane Sci. 2006, 280, 351–362. [Google Scholar]; Rodembusch, F. S.; Paulus Leusin, F.; Bordignon, L. B.; Gallas, M. R.; Stefani, V. New fluorescent monomers and polymers displaying an intramolecular proton-transfer mechanism in the electronically excited state (ESIPT): Part II. Synthesis, spectroscopic characterization and solvatochromism of new benzazolylvinylene derivatives. J. Photochem. Photobiol. A: Chem. 2005, 173, 81–92. [Google Scholar].
- Costela, A.; García-Moreno, I.; Mallavia, R.; Amat-Guerri, F.; Barroso, J.; Sastre, R. Proton-transfer lasers based on solid copolymers of modified 2-(2′-hydroxyphenyl)benzimidazoles with methacrylate monomers. Opt. Commun. 1998, 152, 89–95. [Google Scholar]; Zhou, L. L.; Sun, H.; Zhang, X. H.; Wu, S. K. An effective fluorescent chemosensor for the detection of copper(II). Spectrochim. Acta A: Mol. Biomol. Spectrosc. 2005, 61, 61–65. [Google Scholar].
- Mavrova, A. T.; Anichina, K. K; Vuchev, D. I.; Tsenov, J. A.; Kondeva, M. S.; Micheva, M. K. Synthesis and antitrichinellosis activity of some 2-substituted-[1,3]thiazolo[3,2-a]benzimidazol-3(2H)-ones. Bioorg. Med. Chem. 2005, 13, 5550–5559. [Google Scholar] [CrossRef]
- He, Y.; Yang, J.; Baogen, W.; Risen, L.; Swayze, E. E. Synthesis and biological evaluations of novel benzimidazoles potential antibacterial agents. Bioorg. Med. Chem. Lett. 2004, 14, 1217–1220. [Google Scholar] [CrossRef]
- Göker, H.; Ozden, S.; Yıldız, S.; Boykin, D. W. Synthesis and Potent Antibacterial Activity against MRSA of Some Novel 1,2-Disubstituted-1H-Benzimidazole-N-alkylated-5-carboxamidines. Eur. J. Med. Chem. 2005, 40, 1062–1069. [Google Scholar]
- Göker, H.; Kus, C.; Boykin, D. W.; Yildiz, S.; Altanlar, N. Synthesis of some new 2-substituted-phenyl-1H-benzimidazole-5-carbonitriles and their potent activity against candida species. Bioorg. Med. Chem. 2002, 10, 2589–2596. [Google Scholar] [CrossRef]
- Demirayak, S.; Abu Mohsen, U.; Cagri Karaburun, A. Synthesis and anticancer and anti-HIV testing of some pyrazino[1,2-a]benzimidazole derivatives. Eur. J. Med. Chem. 2002, 37, 255–260. [Google Scholar] [CrossRef]
- Raymond, A. Ng.; Guan, J.; Alford, V. C.; Lanter, J. C.; Allan, G. F.; Sbriscia, T.; Linton, O.; Lundeen, L. S.; Sui, Z. Synthesis of potent and tissue-selective androgen receptor modulators (SARMs): 2-(2,2,2)-Trifluoroethyl-benzimidazole scaffold. Bioorg. Med. Chem. Lett. 2007, 17, 1784–1787. [Google Scholar]
- Kumar, D.; Jacob, M. R.; Reynolds, M. B.; Kerwin, S. M. Synthesis and evaluation of anticancer benzoxazoles and benzimidazoles related to UK-1. Bioorg. Med. Chem. 2002, 10, 3997–4004. [Google Scholar]
- Ismail, M. A.; Brun, R.; Wenzler, T.; Tanious, F. A.; Wilson, W. D.; Boykin, D. W. Dicationic biphenyl benzimidazole derivatives as antiprotozoal agents. Bioorg. Med. Chem. 2004, 12, 5405–5413. [Google Scholar]
- Cox, O.; Jackson, H.; Vargas, V. A.; Bàez, A.; Colón, J. I.; Gonzalez, B. C.; De León, M. Synthesis and Biological Activity of Benzothiazolo- and Benzoxazolo[3,2-a]quinolinium Salts. J. Med. Chem. 1982, 25, 1378–1381. [Google Scholar] [CrossRef]
- Vivas-Mejía, P.; Cox, O.; Gonzalez, F. A. Inhibition of human topoisomerase II by anti-neoplastic benzazolo[3,2-α]quinolinium chlorides. Mol. Cell. Biochem. 1998, 178, 203–212. [Google Scholar]
- Brana, M. F.; Castellano, J. M.; Keilhauer, G.; Machuca, A.; Martín, Y.; Redondo, C.; Schlick, E.; Walker, N. Benzimidazo[1,2-c]quinazolines: a new class of antitumor compounds. Anti-Cancer Drug Des. 1994, 9, 527–538. [Google Scholar]
- Chaires, J. B. Drug-DNA interactions. Curr. Opin. Struct. Biol. 1998, 8, 314–320. [Google Scholar]
- (a)Demeunynck, M.; Bailly, C.; Wilson, W. D. In DNA and RNA Binders; Wiley-VCH: Weinheim, 2002. [Google Scholar]; Bailly, C. Topoisomerase I Poisons and Suppressors as Anticancer Drugs. Curr. Med. Chem. 2000, 7, 39–58. [Google Scholar].
- Hirano, K.; Oderaotoshi, Y.; Minataka, S.; Komatsu, M. Unique Fluorescent Properties of 1-aryl-3,4-diphenylpyrido[1,2-a]benzimidazoles. Chem. Lett. 2001, 1262–1263. [Google Scholar]; Vivas-Mejía, P.; Rodríguez-Cabán, J. L.; Díaz-Velázquez, M.; Hernández-Pérez, M. G.; Cox, O.; Gonzalez, F. A. DNA binding-independent anti-proliferative action of benzazolo[3,2-α]quinolinium DNA intercalators. Mol. Cell. Biochem. 1997, 177, 69–77. [Google Scholar].
- Fairley, T. A.; Tidwell, R. R.; Donkor, I.; Naiman, N. A.; Ohemeng, K. A.; Lombardy, R. J.; Bentley, J. A.; Cory, M. Structure, DNA minor-Groove Binding and Base-Pair Specificity of Alkyl-Linked and Aryl-Linked Bis(amidinobenzimidazoles) and Bis(amidinoindoles). J. Med. Chem. 1993, 36, 1746–1753. [Google Scholar]
- Jarak, I.; Kralj, M.; Šuman, L.; Pavlović, G.; Dogan, J.; Piantanida, I.; Žinić, M.; Pavelić, K.; Karminski-Zamola, G. Novel Cyano- and N-isopropylamidino-Substituted Derivatives of Benzo[b]thiophene-2-carboxanilides and Benzo[b]thieno[2, 3-c]quinolones: Synthesis, Photochemical Synthesis, Crystal Structure Determination and Antitumor Evaluation. Part 2. J. Med. Chem. 2005, 48, 2346–2360. [Google Scholar] [CrossRef]
- Starčević, K.; Karminski-Zamola, G.; Piantanida, I.; Žinić, M.; Šuman, L.; Kralj, M. Photoinduced Switch of a DNA/RNA Inactive Molecule into a Classical Intercalator. J. Am. Chem. Soc. 2005, 127, 1074–1075. [Google Scholar] [CrossRef]
- Buechi, J.; Zwicky, H.; Aebi, A. Synthese einiger 1,2-Benzimidazol-Derivate. Arch. Pharm. 1960, 8, 759–766. [Google Scholar]
- Saczewski, F.; Reszka, P.; Gdaniec, M.; Grunert, R.; Bednarski, P. J. Synthesis, X-ray Crystal Structures, Stabilities, and in Vitro Cytotoxic Activities of New Heteroarylacrylonitriles. J. Med. Chem. 2004, 47, 3438–3449. [Google Scholar]
- Ganushchak, N. I.; Lesyuk, A. I.; Fedorovich, I. S.; Obushak, N. D.; Andrushenko, V. N. 5-Aryl-2-furaldehydes in the synthesis of 2-substituted 1,3-benzazoles. Russ. J. Org. Chem. 2003, 39, 1295–1300. [Google Scholar] [CrossRef]
- Cooper, G.; Irwin, W. J. Photocyclisation of 1-Styrylimidazoles. A Novel Route to N-Bridgehead Compounds. J. Chem. Soc. Perkin I. 1975, 75–80. [Google Scholar]
- Guenther, D. Benzimidazo[1,2-a]Quinolines. US Patent 4,124,589, 1978. [Google Scholar]; Guenther, D. Benzimidazo[1,2-a]Quinolines. US Patent 4,219,651, 1980. [Google Scholar]; Hoechst, AG. Benzimidazo-(1,2-a)-quinoleines et leur preparation. FR 2364214, 1978. [Chem. Abstr. 1978, 89, 7595]. [Google Scholar].
- Boyd, G. V. Recent Advances in the Synthesis of Amidines. In The Chemistry of Amidines and Imidates; Patai, S., Ed.; John Wiley & Sons: New York, 1991; Vol. 2, p. 339. [Google Scholar]
- Hranjec, M.; Grdiša, M.; Pavelić, K.; Boykin, D. W.; Karminski-Zamola, G. Synthesis and antitumor evaluation of some new substituted amidino-benzimidazolyl-furyl-phenyl-acrylates and naphtho[2,1-b]furan-carboxylates. Farmaco 2003, 58, 1319–1324. [Google Scholar]
- Ozden, S.; Atabey, D.; Yildiz, S.; Göker, H. Synthesis and potent antimicrobial activity of some novel methyl or ethyl 1H-benzimidazole-5-carboxylates derivatives carrying amide or amidine groups. Bioorg. Med. Chem. 2005, 13, 1587–1597. [Google Scholar] [CrossRef]
- Sample Availability: Contact the authors.
© 2007 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Hranjec, M.; Karminski-Zamola, G. Synthesis of Novel Benzimidazolyl-substituted Acrylonitriles and Amidino-substituted Benzimidazo[1,2-a]Quinolines. Molecules 2007, 12, 1817-1828. https://doi.org/10.3390/12081817
Hranjec M, Karminski-Zamola G. Synthesis of Novel Benzimidazolyl-substituted Acrylonitriles and Amidino-substituted Benzimidazo[1,2-a]Quinolines. Molecules. 2007; 12(8):1817-1828. https://doi.org/10.3390/12081817
Chicago/Turabian StyleHranjec, Marijana, and Grace Karminski-Zamola. 2007. "Synthesis of Novel Benzimidazolyl-substituted Acrylonitriles and Amidino-substituted Benzimidazo[1,2-a]Quinolines" Molecules 12, no. 8: 1817-1828. https://doi.org/10.3390/12081817
APA StyleHranjec, M., & Karminski-Zamola, G. (2007). Synthesis of Novel Benzimidazolyl-substituted Acrylonitriles and Amidino-substituted Benzimidazo[1,2-a]Quinolines. Molecules, 12(8), 1817-1828. https://doi.org/10.3390/12081817




