Leishmanicidal and Cholinesterase Inhibiting Activities of Phenolic Compounds from Allanblackia monticola and Symphonia globulifera
Abstract
:Introduction
Results and Discussion
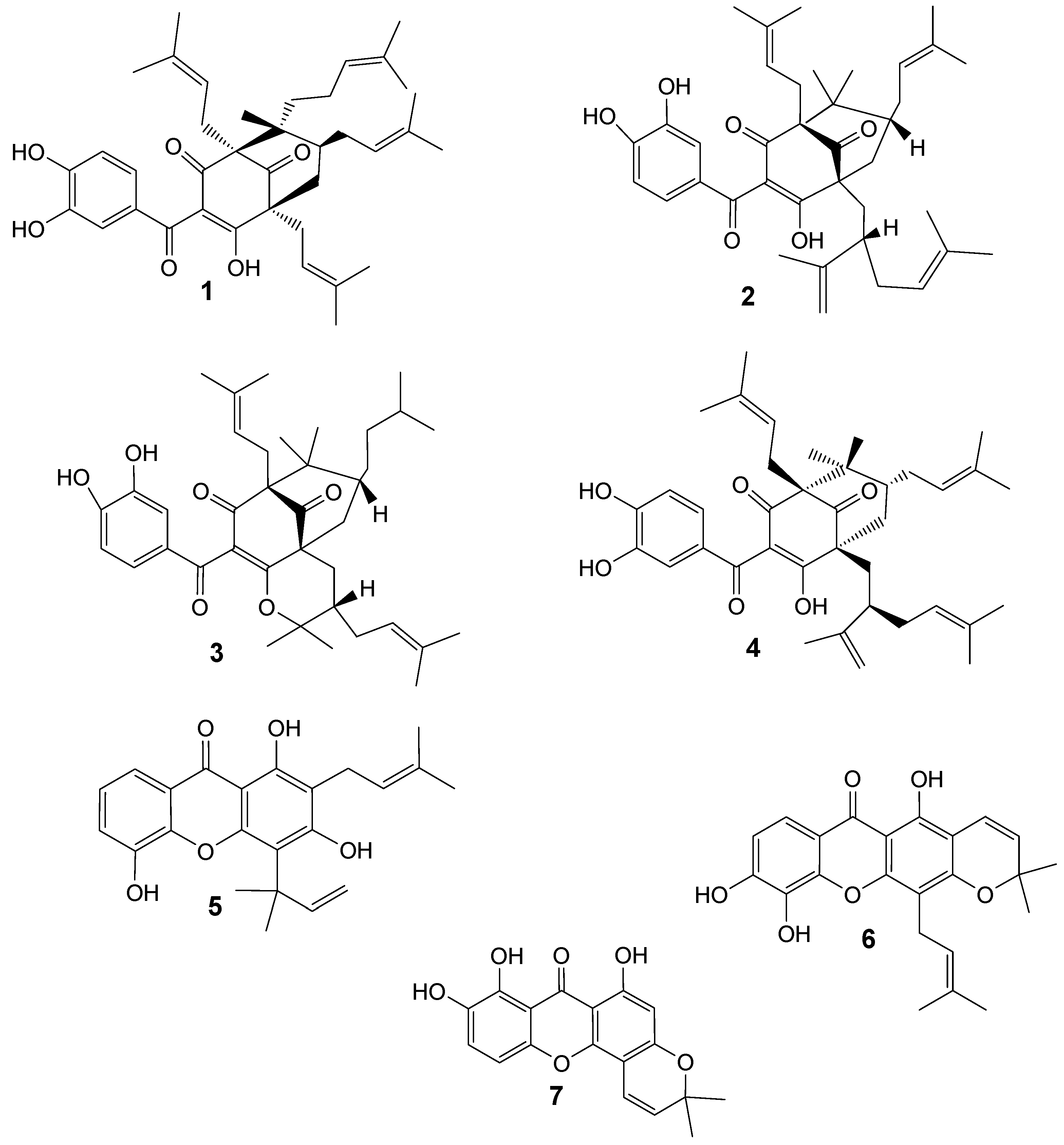
| Parasite growth inhibition (%) tested at: | ||
|---|---|---|
| 0.8 μg/mL | 4.8 μg/mL | |
| Guttiferone A (1) | 82.1 | 98.3 |
| Guttiferone F (4) | 58.2 | 98.2 |
| Allanxanthone A (5) | 13.3 | 57.4 |
| Xanthone V1 (6) | 99.6 | 71.9 |
| Globulixanthone C (7) | 98.7 | 49.7 |
| miltefosinea | 57.6 | 95.0 |
| L. donovani | L6 cells | ||
|---|---|---|---|
| Compounds | IC50 (µM) | IC50 (µM) | SIc |
| Guttiferone A (1) | 0.16 | 7.3 | 46 |
| Garcinol (2) | 0.82 | - | 2.7 |
| Camboginol (3) | 0.33 | - | 6.1 |
| Guttiferone F(4) | 0.20 | 5.4 | 27 |
| Xanthone V1 (6) | 1.40 | 18.0 | 13 |
| Standard | 0.47 | 0.048 | - |
| Compounds | AChE±SEMa | BChE±SEMa |
|---|---|---|
| Guttiferone A (1) | 0.88 ± 0.04 | 2.77 ± 0.02 |
| Garcinol (2) | 0.66 ± 0.02 | 7.39 ± 0.23 |
| Cambogin (3) | 1.13 ± 0.06 | 8.30 ± 0.01 |
| Guttiferone F (4) | 0.95 ± 0.01 | 3.50 ± 0.15 |
| Allanxanthone A | 95± 0.30 | 19.10± 0.90 |
| Galanthamineb | 0.5 ± 0.01 | 8.5 ± 0.01 |
Experimental
General
Plant material
Isolation and characterisation
In vitro leishmanicidal activity
Cytotoxicity
In vitro cholinesterase inhibition assay and determination of IC50
Acknowledgements
References
- Dondji, B.; Dereure, J.; Poste, B; Same-Ekobo, A.; Dedet, J. P. Leishmaniose viscérale au Cameroun. Enquête séro-épidémiologique dans la région de Kousseri, Nord-Cameroun. Bull. Soc. Path. Exo. 2001, 94, 418–420. [Google Scholar]
- Rama Rao, A. V.; Venkatswamy, G.; Pendse, A. D. Camboginol and cambogin. Tetrahedron Lett. 1980, 21, 1975–1978. [Google Scholar]
- Gustafson, K. R.; Blunt, J. W.; Munro, M. H. G.; Fuller, R. W.; Mc Kee, T. C.; Cardenilla, J. H. H.; Mc Mahon, J. B.; Boyd, M. R. The guttiferones, HIV-inhibitory benzophenones from Symphonia globulifera, Garcinia livingstonei, Garcinia ovalifolia and Clusia rosea. Tetrahedron 1992, 48, 10093–10102. [Google Scholar] [CrossRef]
- i>Fuller, R. W.; Blunt, J. W.; Boswell, J. L.; Cardellina, J. H.; Boyd, M. R. Guttiferone F, the first prenylated benzophenone from Allanblackia stuhlmannii. J. Nat. Prod. 1999, 62, 130–132. [Google Scholar] [CrossRef] [PubMed]
- Nkengfack, A. E.; Azebaze, G. A.; Vardamides, J. C.; Fomum, Z. T.; van Heerden, F. R. A prenylated xanthone from Allanblackia floribunda. Phytochemistry 2002, 60, 381–384. [Google Scholar] [PubMed]
- Nkengfack, A. E.; Mkounga, P.; Meyer, M.; Fomum, Z. T.; Bodo, B. Globulixanthones C, D and E: three prenylated xanthones with antimicrobial properties from the root bark of Symphonia globulifera. Phytochemistry 2002, 61, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Hay, A. E.; Hélesbeux, J. J.; Duval, O.; Labaïed, M.; Grellier, P.; Richomme, P. Antimalarial xanthones from Calophyllum caledonicum and Garcinia vieillardii. Life Sci. 2004, 75, 3077–3085. [Google Scholar] [CrossRef] [PubMed]
- Lenta, B. N; Ngouela, S.; Noungoue, D. T; Tsamo, E.; Connolly, J. D. Symphonin a prenylated xanthone with antimicrobial activity from Symphonia globulifera. Bull. Chem. Soc. Ethiop. 2004, 18, 175–180. [Google Scholar]
- Pedro, M.; Cerqueira, F.; Sousa, M. E.; Nascimento, M.S.; Pinto, M. Xanthones as inhibitors of growth of human cancer cell lines and their effects on the proliferation of human lymphocytes in vitro. Bioorg. Med. Chem. 2002, 10, 3725–3730. [Google Scholar] [CrossRef] [PubMed]
- Jiang, D. J; Dai, Z.; Li, Y. J. Pharmacological effects of xanthones as cardiovascular protective agents. Cardiovasc. Drug Rev. 2004, 22, 91–102. [Google Scholar]
- Dharmaratne, H. R.; Tan, G. T.; Marasinghe, G. P.; Pezzuto, J. M. Inhibition of HIV-1 reverse transcriptase and HIV-1 replication by Calophyllum coumarins and xanthones. Planta Med. 2002, 68, 86–87. [Google Scholar] [CrossRef] [PubMed]
- Dharmaratne, H. R; Wijesinghe, W. M; Thevanasem, V. Antimicrobial activity of xanthones from Calophyllum species, against methicillin-resistant Staphylococcus aureus (MRSA). J. Ethnopharmacol. 1999, 66, 339–342. [Google Scholar]
- Wang, L. W; Kang, J. J.; Chen, I. J.; Teng, C. M.; Lin, C. N. Antihypertensive and vasorelaxing activities of synthetic xanthone derivatives. Bioorg. Med. Chem. 2002, 10, 567–572. [Google Scholar]
- Kelly, J. X.; Winter, R.; Peyton, D. H.; Hinrichs, D. J.; Riscoe, M. Optimization of xanthones for antimalarial activity: the 3,6-bis-omega-diethylaminoalkoxyxanthone series. Antimicrob. Agents Chemother. 2002, 46, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Pinto, M. M. M.; Sousa, M. E.; Nascimento, M. S. J. Xanthones derivatives: New insights in biological activities. Current Med. Chem. 2005, 12, 2517–2538. [Google Scholar]
- Miura, T.; Ichiki, H.; Hashimoto, I.; Iwamoto, N.; Kato, M.; Kubo, M.; Ishihara, E.; Komatsu, Y.; Okada, M.; Ishida, T.; Tanigawa, K. Antidiabetic activity of a xanthone compound, mangiferin. Phytomedicine 2001, 8, 85–87. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J. X.; Ignatushchenko, M. V.; Bouwer, H. G.; Peyton, D. H.; Hinrichs, D. J; Winter, R. W.; Riscoe, M. Antileishmanial drug development: exploitation of parasite heme dependency. Mol. Biochem. Parasitol. 2003, 126, 43–49. [Google Scholar]
- Iinuma, M.; Tosa, H.; Tanaka, T.; Kanamaru, S.; Asai, F.; Kobayashi, Y.; Miyauchi, K.; Shimano, R. Antibacterial activity of some Garcinia benzophenone derivatives against methicillin-resistant Staphylococcus aureus. Biol. Pharm. Bull. 1996, 19, 311–314. [Google Scholar] [PubMed]
- Epifano, F.; Genovese, S.; Menghini, L.; Curini, M. Chemistry and pharmacology of oxyprenylated secondary metabolites. Phytochemistry 2007, 68, 939–953. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, F.; Ariga, T.; Yoshimura, Y.; Nakazawa, H. Antioxidative and Anti-Glycation Activity of Garcinol from Garcinia indica Fruit Rind. J. Agr. Food Chem. 2000, 48, 180–185. [Google Scholar]
- Yamaguchi, F.; Saito, M.; Ariga, T.; Yoshimura, Y.; Nakazawa, H. Free Radical Scavenging Activity and Antiulcer Activity of Garcinol from Garcinia indica Fruit Rind. J. Agr. Food Chem. 2000, 48, 2320–2325. [Google Scholar]
- Min-Hsiung, P.; Won-Ling, C.; Shoei-Yn, L.; Chi-Tang, H.; Jen-Kun, L. Induction of Apoptosis by Garcinol and Curcumin through Cytochrome c Release and Activation of Caspases in Human Leukemia HL-60 Cells. J. Agr. Food Chem. 2001, 49, 1464–1474. [Google Scholar]
- Tougu, V. Acetylcholinesterase: mechanism of catalysis and inhibition. Curr. Med. Chem. 2001, 1, 155–170. [Google Scholar]
- Ballard, C. G. Advances in the treatment of Alzheimer’s disease: benefits of dual cholinesterase inhibition. Eur. Neurol. 2002, 47, 64–70. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, M.; Glick, D.; Loewensten, Y.; Soreq, H. Engineering of human cholinesterases explains and predicts diverse consequences of administration of various drugs and poisons. Pharmacol Ther. 1995, 67, 283–322. [Google Scholar] [CrossRef] [PubMed]
- Khalid, A. Biochemical studies on new natural inhibitors of cholinesterase. Ph. D. Thesis, H.E.J. Research Institute of Chemistry, University of Karachi, Karachi, Pakistan, 2002. [Google Scholar]
- Lenta, B. N.; Vonthron-Sénécheau, C.; Soh, R. F.; Tantangmo, F.; Ngouela, S.; Kaiser, M.; Tsamo, E.; Anton, R.; Weniger, B. In vitro antiprotozoal activities and cytotoxicity of some selected Cameroonian medicinal plants. J. Ethnopharmacol. 2007, 111, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Abe, F.; Nagafuji, S.; Okabe, H.; Akahane, H.; Estrada-Muñiz, E.; Huerta-Reyes, M.; Reyes-Chilpa, R. Trypanocidal Constituents in Plants 3. Leaves of Garcinia intermedia and Heartwood of Calophyllum brasiliense. Biol. Pharm. Bull. 2004, 27, 141–143. [Google Scholar]
- Zhang, X. Cholinergic activity and amyloid precursor protein processing in aging and Alzheimer’s diseases. Curr. Drug Targets: CNS Neurol. Disord. 2004, 3, 137–152. [Google Scholar]
- Cunningham, I. New culture medium for maintenance of tsetse tissues and growth of trypanosomatids. J. Protozool. 1977, 24, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Brun, R.; Schönenberger, M. Cultivation and in vitro cloning or procyclic culture forms of Trypanosoma brucei in a semi-defined medium. Acta Trop. 1979, 36, 289–292. [Google Scholar] [PubMed]
- Räz, B.; Iten, M.; Grether, Y.; Kaminsky, R.; Brun, R. The Alamar Blue Assay to Determine drug Sensitivity of African Trypanosomes (T. b. rhodesiense and T. b. gambiense) in vitro. Acta Trop. 1997, 68, 139–147. [Google Scholar]
- Pagé, C.; Pagé, M.; Noel, C. A New Fluorimetric Assay for Cytotoxicity Measurements in vitro. Int. J. Oncol. 1993, 3, 473–476. [Google Scholar]
- Ahmed, S. A.; Gogal, R. M.; Walsh, J. E. A new rapid and simple non-radioactive assay to monitor and determine the proliferation of lymphocytes: an alternative to [3H]thymidine incorporation assay. J. Immun. Meth. 1994, 170, 211–224. [Google Scholar] [CrossRef]
- Ellman, G. L.; Courtney, K. D.; Andres, V.; Featherstone, R. M. A new and rapid colorimetric determination of actetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef] [PubMed]
- Sample Availability: Contact the authors.
© 2007 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Lenta, B.N.; Vonthron-Sénécheau, C.; Weniger, B.; Devkota, K.P.; Ngoupayo, J.; Kaiser, M.; Naz, Q.; Choudhary, M.I.; Tsamo, E.; Sewald, N. Leishmanicidal and Cholinesterase Inhibiting Activities of Phenolic Compounds from Allanblackia monticola and Symphonia globulifera. Molecules 2007, 12, 1548-1557. https://doi.org/10.3390/12081548
Lenta BN, Vonthron-Sénécheau C, Weniger B, Devkota KP, Ngoupayo J, Kaiser M, Naz Q, Choudhary MI, Tsamo E, Sewald N. Leishmanicidal and Cholinesterase Inhibiting Activities of Phenolic Compounds from Allanblackia monticola and Symphonia globulifera. Molecules. 2007; 12(8):1548-1557. https://doi.org/10.3390/12081548
Chicago/Turabian StyleLenta, Bruno Ndjakou, Catherine Vonthron-Sénécheau, Bernard Weniger, Krishna Prasad Devkota, Joseph Ngoupayo, Marcel Kaiser, Qamar Naz, Muhammad Iqbal Choudhary, Etienne Tsamo, and Norbert Sewald. 2007. "Leishmanicidal and Cholinesterase Inhibiting Activities of Phenolic Compounds from Allanblackia monticola and Symphonia globulifera" Molecules 12, no. 8: 1548-1557. https://doi.org/10.3390/12081548
APA StyleLenta, B. N., Vonthron-Sénécheau, C., Weniger, B., Devkota, K. P., Ngoupayo, J., Kaiser, M., Naz, Q., Choudhary, M. I., Tsamo, E., & Sewald, N. (2007). Leishmanicidal and Cholinesterase Inhibiting Activities of Phenolic Compounds from Allanblackia monticola and Symphonia globulifera. Molecules, 12(8), 1548-1557. https://doi.org/10.3390/12081548





