Bis(trimethylsilyl)chromate Catalyzed Oxidations of Alcohols to Aldehydes and Ketones with Periodic Acid
Abstract
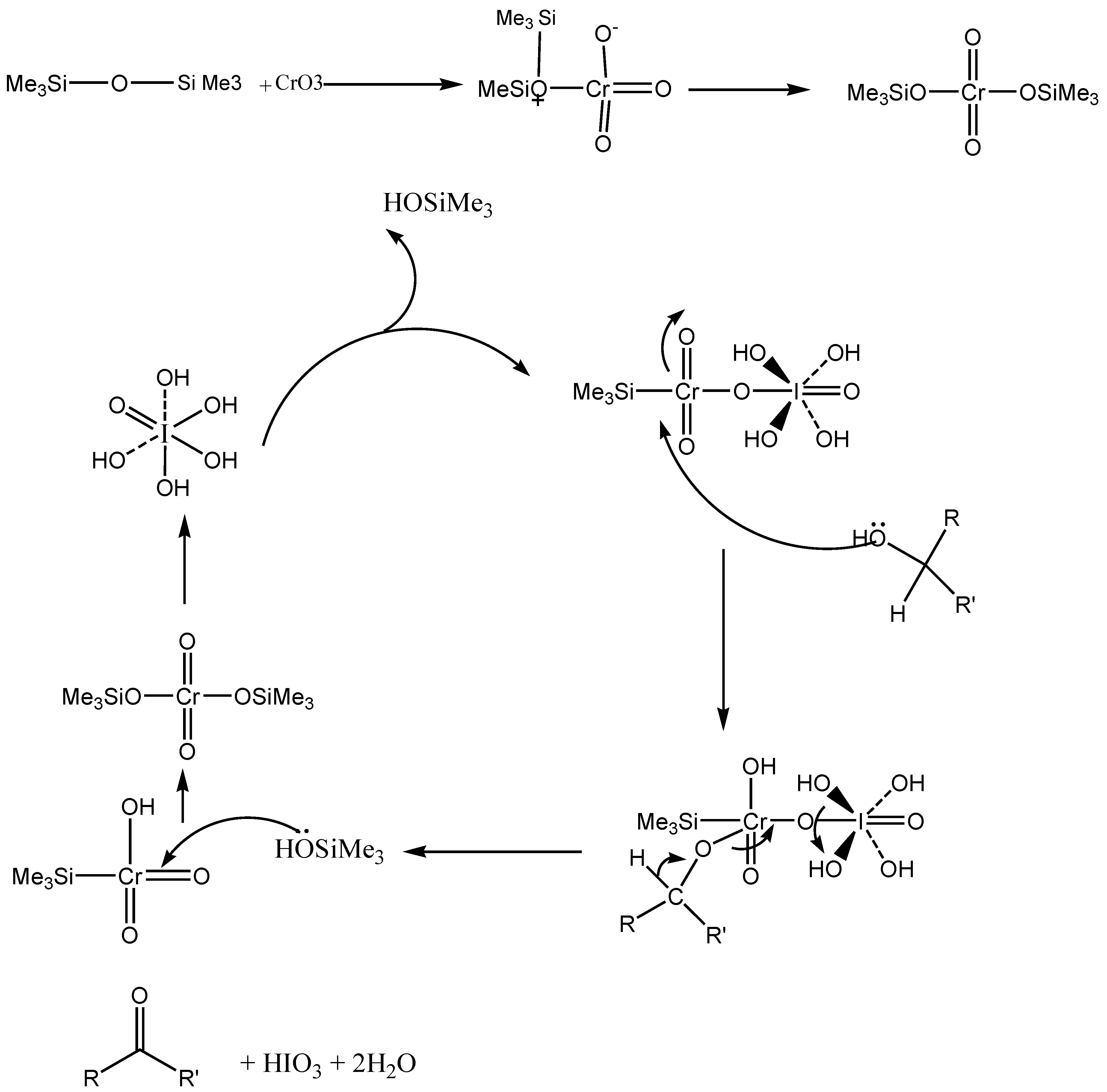
:Introduction
Results and Discussion
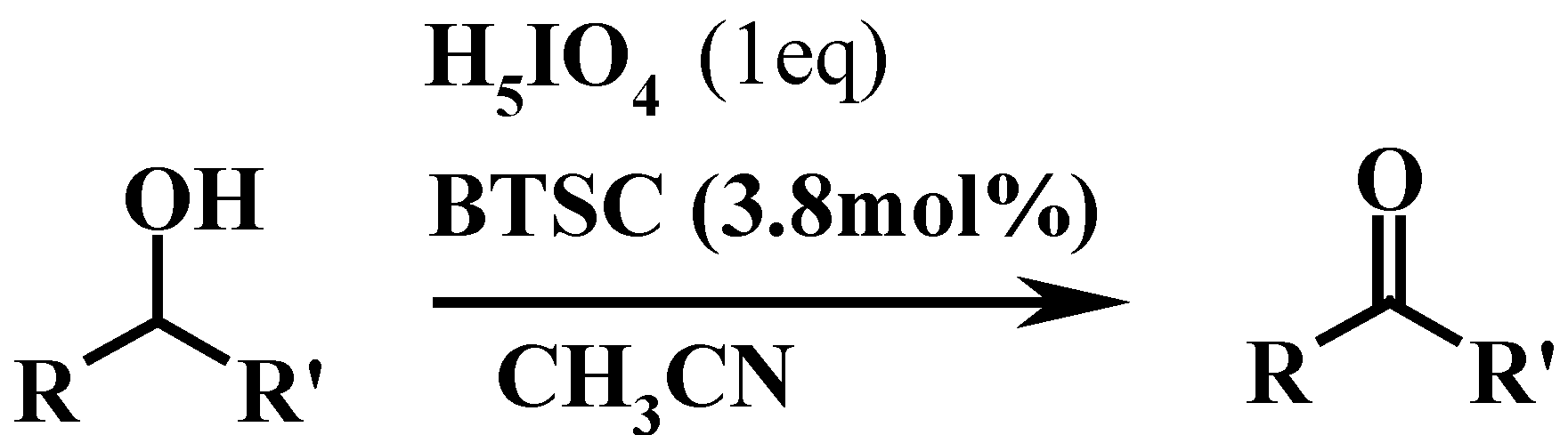
| Entry | Compound | Product | Solvent | Temp(°C) | Time(h) | Yieldb (%) | m.p or b.p.(˚C) | |
|---|---|---|---|---|---|---|---|---|
| Found | Lit. | |||||||
| 1 | 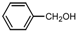 | 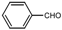 | CH3CN | 40-50 | 1 | 96 | 179 c | 181 |
| 2 | 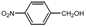 | 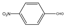 | CH3CN | 80 | 3 | 80 | 106 | 104 |
| 3 | 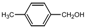 |  | THF | 60 | 3 | 91 | 204-5 | 202 |
| 4 | 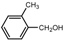 | 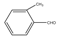 | CH3CN | 50-60 | 3 | 82 | 200 c | 200-3 |
| 5 | 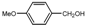 | 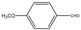 | THF | 50-60 | 2 | 96 | 248 c | 248 |
| 6 | 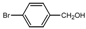 | 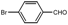 | THF | 50 | 3 | 87 | 67 | 65 |
| 7 |  |  | THF | 60 | 3 ½ | 79 | 196-7 c | 194 |
| 8 | 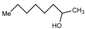 |  | CH3CN | 80 | 4 ½ | 83 | 172-3 | 170 |
| 9 | 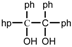 | 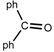 | THF | 60 | 2 | 86 | 48 | 47 |
| 10 |  |  | CH3CN | 80 | 4 | 81 | 131 | 129 |
| 11 |  | 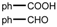 | CH3CN | 80 | 2 1/2 | 82 | 121 | 118 |
Conclusions
Experimental
General
Oxidation of benzyl alcohol: Typical procedure
Acknowledgments
References and Notes
- Hudlicky, M. Oxidation in Organic Chemistry; American Chemical Society: Washington, DC, 1990. [Google Scholar]
- Varma, R. S.; Saini, R. K. Wet alumina supported chromium (VI) oxide: Selective oxidation of alcohols in solventless system. Tetrahedron Lett. 1998, 39, 1481–1482. [Google Scholar] [CrossRef]
- Velusamy, S.; Srenvasan, A.; Punniyamurthy, T. Copper (II) catalyzed selective oxidation of primary alcohols to aldehydes with atmospheric oxygen. Tetrahedron Lett. 2006, 47, 923–926. [Google Scholar] Herrerias, C. I.; Zhang, T. Y.; Li, Ch. Catalytic oxidations of alcohols to carbonyl compounds by oxygen under solvent-free and transition-metal-free conditions. Tetrahedron Lett. 2006, 47, 13–17. [Google Scholar] Mardani, H. R.; Golchobian, H. Effective oxidation of benzylic and aliphatic alcohols with hydrogen peroxide catalyzed by a manganese(III) Schiff-base complex under solvent-free conditions. Tetrahedron Lett. 2006, 47, 2349–2352. [Google Scholar] Farhadi, S.; Afshari, M.; Maleki, M.; Babazadeh, Z. Photocatalytic oxidation of primary and secondary benzylic alcohols to carbonyl compounds catalyzed by H3PW12O40/SiO2 under an O2 atmosphere. Tetrahedron Lett. 2005, 46, 8483–8486. [Google Scholar] Hunsen, M. Pyridinium chlorochromate catalyzed oxidation of alcohols to aldehydes and ketones with periodic acid. Tetrahedron Lett. 2005, 46, 1651–1653. [Google Scholar]
- Heravi, M. M.; Behbahani, F. K.; Bamoharram, F. F. H-14[NaP5W30O110]: A heteropoly acid catalyzed acetylation of alcohols and phenols in acetic anhydride. J. Mol Catal. A. Chem. 2006, 253, 16–19. [Google Scholar] Heravi, M. M.; Bakhtiari, Kh.; Bamoharram, F. F. An efficient and chemoselective synthesis of acylals from aromatic aldehydes and their regeneration, catalyzed by 12-molybdo-phosphoric acid. Catal. Commun. 2006, 7, 499–501. [Google Scholar] Heravi, M. M.; Bakhtiari, Kh.; Bamoharram, F. F. 12-Molybdophosphoric acid: A recyclable catalyst for the synthesis of Biginelli-type 3,4-dihydropyrimidine-2(1H)-ones. Catal. Commun. 2006, 7, 373–376. [Google Scholar] Bamoharram, F. F.; Heravi, M. M.; Roshani, M.; Tawakoli, N. A catalytic method for synthesis of δ-butyrolactone, є-caprolactone and 2-cumaranone in the presence of Preyssler's anion, [NaP5W30O110] (14-), as a green and reusable catalyst. J. Mol. Catal. A. Chem. 2006, 252, 90–95. [Google Scholar] Heravi, M. M.; Motamedi; Siefi, N.; Bamoharram, F. F. Catalytic synthesis of 6-aryl-1H-pyrazolo[3,4-d]pyrimidin-4[5H]-ones by heteropolyacid: H-14[NaP5W30O110] and H3PW12O40. J. Mol. Catal. A. 2006, 249, 1–3. [Google Scholar] Bamoharram, F. F.; Heravi, M. M.; Roshani, M.; Jahangiri, M.; Gharib, A. Preyssler catalyst, [NaP5W30O110](14-): A green, efficient and reusable catalyst for esterification of salicylic acid with aliphatic and benzylic alcohols. Applied Cat. A. 2006, 302, 42–47. [Google Scholar] Oskooei, H. A.; Heravi, M. M.; Bakhtiari, Kh; Zadsirjan, V. H-14[NaP5W30O110] as an efficient catalyst for the one-pot synthesis of alpha-amino nitriles. Synlett 2006, 11, 1768–1770. [Google Scholar] Heravi, M. M.; Derikvand, F.; Haghigi, M. “On water”: Rapid Knoevenagel condensation using sodium pyruvate. Lett. Org. Chem. 2006, 3, 297–299. [Google Scholar] Heravi, M. M.; Derikvand, F.; Oskooie, H. A.; Hekmatshoar, R. Zeofen: a user friendly agent for oxidation of the Hantzsch 1,4-dihydro-pyridines. J. Chem. Res. 2006, 3, 168–169. [Google Scholar] Fotouhi, L.; Nematolahi, D.; Heravi, M. M.; Tamari, E. An efficient electrochemical method for a unique synthesis of new derivatives of 7H-thiazolo[3,2-b]-1,2,4-triazin-7-one. Tetrahedron Lett 2006, 47, 1713–1716. [Google Scholar]
- Heravi, M. M.; Derikvand, F.; Ghassemzadeh, M. Tetrameric DABCO-bromine complex: A novel reagent for regeneration of carbonyl compounds from aldoximes and ketoximes. Synth Commun. 2006, 36, 581–585. [Google Scholar] Heravi, M. M.; Derikvand, F.; Ghassemzadeh, M.; Neumuller, B. Synthesis, characterization and structure of a tetrameric DABCO–bromine complex: a novel oxidizing agent for oxidation of alcohols to carbonyl compounds. Tetrahedron Lett. 2005, 46, 6243–6245. [Google Scholar] Heravi, M. M.; Mirza Aghajan, Z. Microwave-assisted oxidation of alcohols using wet alumina supported ammonium chlorochromate in solventless system. Naturforsch. 1999, 54, 815–817. [Google Scholar] Heravi, M. M.; Ajami, D.; Tabar hydra, K.; Ghassemzadeh, M. Remarkable fast microwave-assisted zeolite HZSM-5 catalyzed oxidation of alcohols with chromium trioxide under solvent-free conditions. J. Chem. Res. 1999, 5, 334–335. [Google Scholar] Heravi, M. M.; Ajami, D.; Aghapoor, K.; Ghassemzadeh, M. ‘Zeofen’, a user-friendly oxidizing reagent. Chem. Commun. 1999, 89, 833–834. [Google Scholar] Heravi, M. M.; Ajami, D.; Ghassemzadeh, M. Wet alumina supported chromium (VI) oxide: A mild, efficient and inexpensive reagent for oxidative deprotection of trimethylsilyl and tetrahydropyranyl ethers in a solventless system. Synthesis 1999, 3, 393–394. [Google Scholar]
- Heravi, M. M.; Derikvand, F.; Oskooei, H. A.; Hekmatshoar, R. Silica gel-supported bis(trimethylsilyl) chromate: Oxidation of 1,4-dihydropyridines to pyridines. Synth. Commun. 2006, 36, 77–82. [Google Scholar] Heravi, M. M.; Ajami, D. Oxidative deprotection of tetrahydropyranyl ethers to carbonyl compounds with montmorillonite K-10 supported bis(trimethylsilyl) chromate under non-aqueou conditions. J. Chem. Res. 1998, 718–719. [Google Scholar] Heravi, M. M.; Ajami, D.; Tabar-Heydar, K.; Mojtahedi, M. M. Direct oxidative deprotection using montmorillonite supported bis-(trimethylsilyl)chromate. J. Chem. Res. 1998, 9, 620–621. [Google Scholar] Heravi, M. M.; Ajami, D.; Tajbakhsh, M.; Ghassemzadeh, M. Clay supported bis-(trimethylsilyl)-chromate: An efficient reagent for oxidative deoximation. Monatsh. Chem. 2000, 131, 1109–1113. [Google Scholar] Heravi, M. M.; Ajami, D. Clay supported bis(trimethylsilyl)chromate: Oxidative deprotection of tetrahydropyranyl ethers under solvent-free conditions using microwaves. Monatsch. Chem. 1999, 130, 709–712. [Google Scholar] Khaleghi, Sh.; Heravi, M. M.; Derikvand, F. Bis(trimethylsilyl) chromate: An efficient reagent for cleavage of phenylhydrazones. Phosphorus Sulfur Silicon 2006, 181, 227–231. [Google Scholar]
- Zhao, M.; Li, J.; Song, Z.; Desmond, R.; Taschaen, D. M.; Grabowski, E. J. J.; Reider, P. A novel chromium trioxide catalyzed oxidation of primary alcohols to the carboxylic acids. Tetrahedron Lett. 1998, 39, 5323–5326. [Google Scholar]
- Heravi, M. M.; Beheshtiha, Y. S.; Ghasemzadeh, M.; Hekmat Shoar, R.; Sarmadi, N. Solid state deoximation with clay supported ammonium chlorochromate: Regeneration of carbonyl compounds using microwaves in a solventless system. Monatsh. Chem. 2000, 31, 187–190. [Google Scholar] Heravi, M. M.; Rajabzadeh, G.; Bamoharram, F. F.; Seifi, N. An eco-friendly catalytic route for synthesis of 4-amino-pyrazolo[3,4-d]pyrimidine derivatives by Keggin heteropolyacids under classical heating and microwave irradiation. J. Mol. Catal. A: Chem. 2006, 256, 238–241. [Google Scholar]
- Kim, S. S.; Nehru, K.; Kim, S. S.; Kim, D. W.; Jung, H. C. A Mild and Highly Efficient Oxidation of Sulfides to Sulfoxides with Periodic Acid Catalyzed by FeCl3. Synthesis 2002, 17, 2484–2486. [Google Scholar]
- Kim, S. S.; Nehru, K. A Mild and Efficient Oxidation of Alcohols to Carbonyl Compounds by Periodic Acid Catalyzed by 2,2,6,6-Tetramethylpiperidinyl-1-oxyl. Synlett 2002, 4, 616–617. [Google Scholar]
- Sample availability: Samples of the compounds presented in this paper are available from the authors.
© 2007 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Asadolah, K.; Heravi, M.M.; Hekmatshoar, R.; Majedi, S. Bis(trimethylsilyl)chromate Catalyzed Oxidations of Alcohols to Aldehydes and Ketones with Periodic Acid. Molecules 2007, 12, 958-964. https://doi.org/10.3390/12050958
Asadolah K, Heravi MM, Hekmatshoar R, Majedi S. Bis(trimethylsilyl)chromate Catalyzed Oxidations of Alcohols to Aldehydes and Ketones with Periodic Acid. Molecules. 2007; 12(5):958-964. https://doi.org/10.3390/12050958
Chicago/Turabian StyleAsadolah, Karim, Majid M Heravi, Rahim Hekmatshoar, and Soma Majedi. 2007. "Bis(trimethylsilyl)chromate Catalyzed Oxidations of Alcohols to Aldehydes and Ketones with Periodic Acid" Molecules 12, no. 5: 958-964. https://doi.org/10.3390/12050958
APA StyleAsadolah, K., Heravi, M. M., Hekmatshoar, R., & Majedi, S. (2007). Bis(trimethylsilyl)chromate Catalyzed Oxidations of Alcohols to Aldehydes and Ketones with Periodic Acid. Molecules, 12(5), 958-964. https://doi.org/10.3390/12050958




