Antibacterial Activity of Some 3-(Arylideneamino)-2-phenylquinazoline-4(3H)-ones: Synthesis and Preliminary QSAR Studies
Abstract
:Introduction
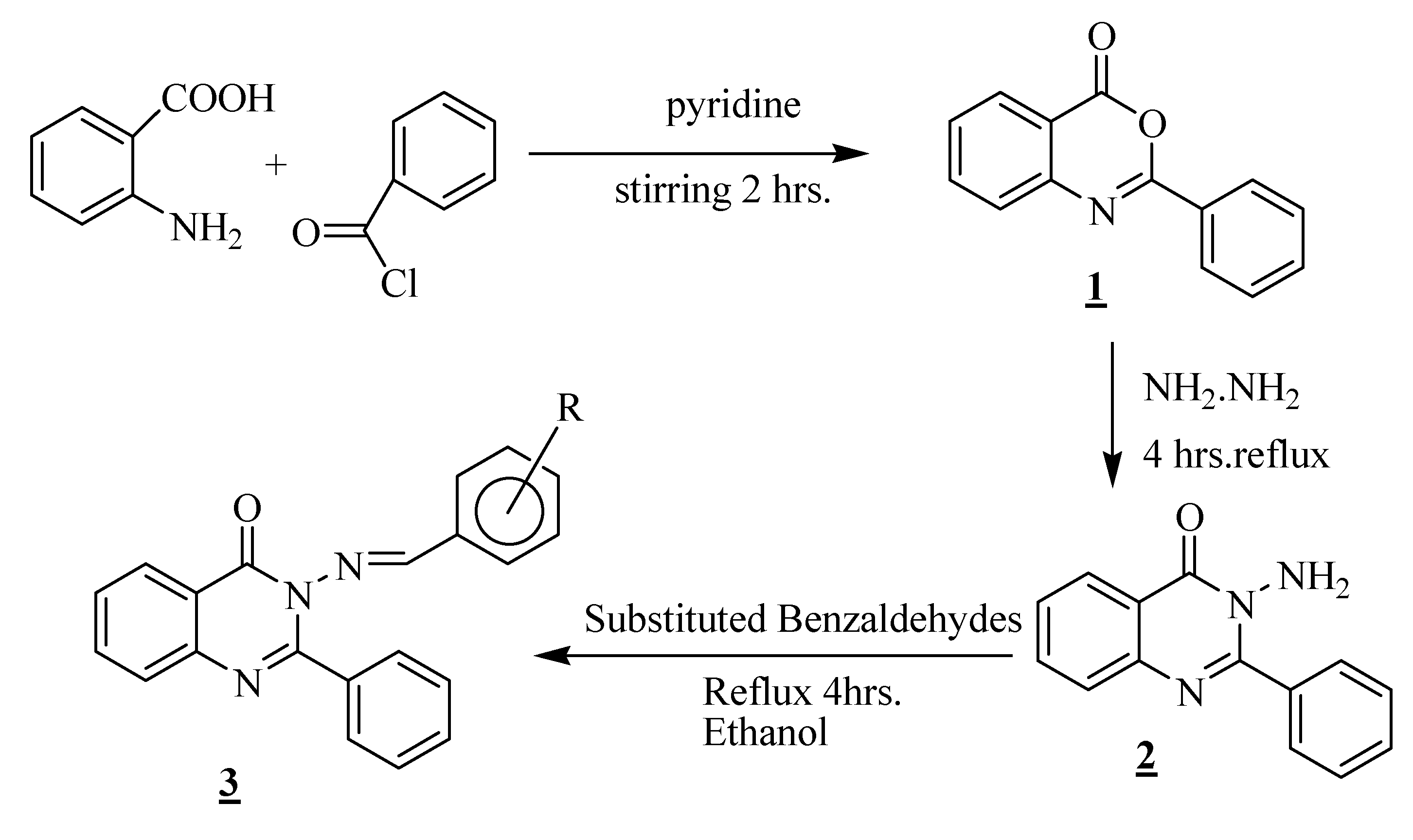
Results and Discussion
Synthesis of 3-(arylideneamino)-2-phenylquinazolin-4(3H)-ones 3a-3j
Interpretation of spectral data
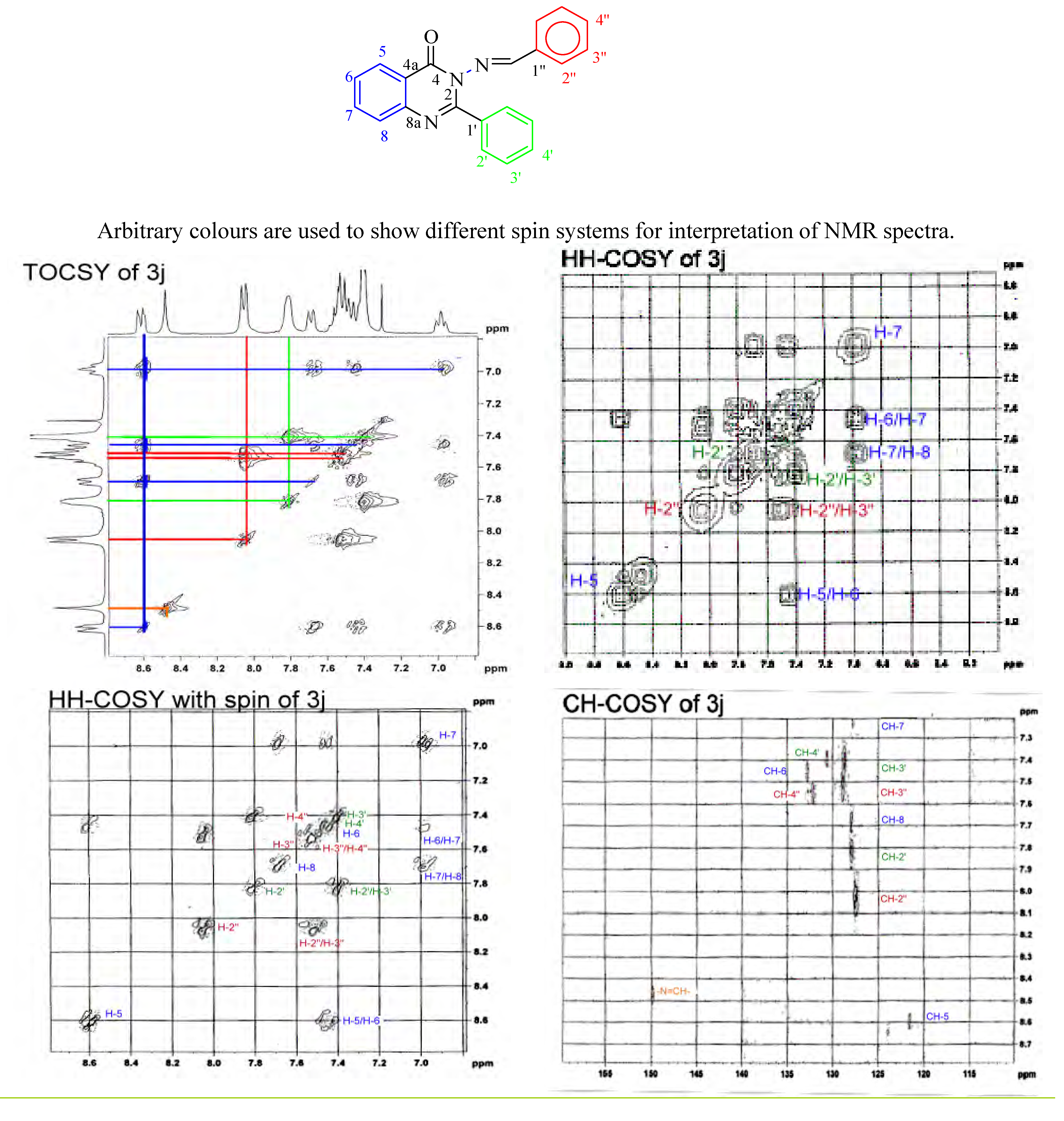
Antibacterial activity
| Compound | R | M.F | Gram-Positive | Gram-Negative | ||
|---|---|---|---|---|---|---|
| B. subtilis* | S. aureus 6571* | E. coli K12* | S. dysenteriae 6* | |||
| 1 | - | C14H9NO2 | - | - | - | - |
| 2 | - | C14H11N3O | 400 | - | 400 | - |
| 3a | 2΄΄-OH | C21H15N3O2 | 300 | 200 | 200 | 300 |
| 3b | 4΄΄-OCH3 | C22H17N3O2 | 100 | 200 | 200 | 300 |
| 3c | 4΄΄-F | C21H14N3OF | 200 | 100 | 200 | 200 |
| 3d | 4΄΄-N(CH3)2 | C23H20N4O | 100 | 100 | 100 | 200 |
| 3e | 4΄΄-Cl | C21H14N3OCl | 100 | 100 | 200 | 300 |
| 3f | 3΄΄-OCH3 | C22H17N3O2 | 100 | 200 | 400 | 300 |
| 3g | 4’-OH | C21H15N3O2 | 300 | 200 | 400 | 400 |
| 3h | 3΄΄-OCH3, 4΄΄-OH | C22H17N3O3 | 200 | 200 | 100 | 200 |
| 3i | 3΄΄-NO2 | C21H14N4O3 | 200 | 300 | 200 | 200 |
| 3j | H | C21H15N3O | 100 | 100 | 200 | 200 |
Preliminary QSAR study.
| Compounds | 3a | 3b | 3c | 3d | 3e | 3f | 3g | 3h | 3i | 3j |
|---|---|---|---|---|---|---|---|---|---|---|
| Log(P/C) for Gm –ve bacteria E. coli K12 | 7.9607 | 8.7209 | 8.6894 | 9.14782 | 9.4108 | 8.7204 | 7.9627 | 8.0490 | 3.7543* | 8.8084 |
| Log(P/C) for Gm +ve bacteria B. subtilis | 8.1369 | 8.4199 | 8.6894 | 9.1478 | 9.1098 | 8.1184 | 7.8378 | 8.3500 | 3.7543* | 8.5074 |
| MR | 2.85 | 7.87 | 0.92 | 15.55 | 6.03 | 7.87 | 2.85 | 10.72 | 7.37 | 1.03 |
| σm | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.12 | 0.00 | 0.12 | 0.71 | 0.00 |
| σp | 0.00 | -0.27 | 0.06 | -0.83 | 0.23 | 0.00 | -0.37 | -0.37 | 0.00 | 0.00 |
| Ova | 1.5561 | 1.5970 | 1.5630 | 1.6166 | 1.5746 | 1.5937 | 1.5642 | 1.6012 | 1.5945 | 1.5506 |
| L | 2.85 | 3.98 | 2.65 | 3.53 | 3.52 | 3.98 | 2.85 | 6.83 | 3.44 | 2.06 |
| B1 | 2.74 | 1.35 | 1.35 | 1.5 | 1.8 | 1.35 | 2.74 | 4.09 | 1.7 | 1.00 |
| B4 | 1.35 | 2.87 | 1.35 | 2.8 | 1.8 | 2.87 | 1.35 | 4.22 | 2.44 | 1.00 |
| L | B1 | B2 | B3 | B4 | MR | OVA | σm | σp | |
|---|---|---|---|---|---|---|---|---|---|
| B1 | 0.119 | 1.000 | 0.171 | 0.220 | 0.024 | 0.071 | 0.116 | 0.163 | -0.203 |
| B2 | 0.881 | 0.171 | 1.000 | 0.854 | 0.854 | 0.834 | 0.815 | 0.326 | -0.355 |
| B3 | 0.738 | 0.220 | 0.854 | 1.000 | 0.805 | 0.786 | 0.861 | 0.521 | -0.355 |
| B4 | 0.929 | 0.024 | 0.854 | 0.805 | 1.000 | 0.786 | 0.768 | 0.456 | -0.304 |
| MR | 0.767 | 0.071 | 0.834 | 0.786 | 0.786 | 1.000 | 0.841 | 0.318 | -0.544 |
| OVA | 0.705 | 0.116 | 0.815 | 0.861 | 0.768 | 0.841 | 1.000 | 0.342 | -0.484 |
| σm | 0.382 | 0.163 | 0.326 | 0.521 | 0.456 | 0.318 | 0.342 | 1.000 | -0.034 |
| σp | -0.297 | -0.203 | -0.355 | -0.355 | -0.304 | -0.544 | -0.484 | -0.034 | 1.000 |
| Equations | Eqn. Nos | t | sig | ||
|---|---|---|---|---|---|
| Gram +ve | Log(P/C) = 8.240 + 0.397mr + 0.319 σp + (-1.06) σm N = 10 F = 52.72( sig 0.000) r = 0.853 Rsqr = 0.968 SEE = 0.377 D/W = 1.11 | A | Intercept mr σp σm | 37.01 3.389 2.715 -12.37 | 0.000 0.015 0.035 0.000 |
| Gram -ve | Log(P/C) = 8.131 + 0.356mr + 0.230 σp + (-1.05) σm N = 10 F = 75.46( sig 0.000) r = 0.559 Rsqr = 0.974 SEE = 0.3053 D/W = 1.762 | B | Intercept mr σp σm | 45.09 3.620 2.333 -14.593 | 0.000 0.011 0.058 0.000 |
| Gram +ve | Log(P/C) = -34.998 + 0.386Ova + 0.286 σp + (-1.109) σm N = 10 F = 51.72( sig 0.000) r = 0.870 Rsqr = 0.963 SEE = 0.380 D/W = 0.852 | C | Intercept Ova σp σm | -2.668 3.339 -11.910 2.562 | 0.037 0.016 0.000 0.043 |
| Gram -ve | Log(P/C) = -26.253 + 0.319Ova + 0.183 σp + (-1.084) σm N = 10 F = 55.271( sig 0.000) r = 0.757 Rsqr = 0.965 SEE = 0.355 D/W = 1.818 | D | Intercept Ova σp σm | -2.146 2.850 1.686 -12.02 | 0.070 0.029 0.000 0.113 |
| Gram +ve | Log(P/C) = 8.456 + 0.599L + (-0.345)B1 + 0.062 σp + (-1.003) σm N = 10 F = 41( sig 0.000) r = 0.62 Rsqr = 0.97 SEE = 0.369 D/W = 1.044 | E | Intercept L B1 σp σm | 23.30 3.269 3.339 0.752 -12.6 | 0.000 0.022 0.021 0.486 0.150 |
| Gram -ve | Log(P/C) = 8.061 + 0.304L + (-0.209)B1 + 0.019σp + (-1.002) σm N = 10 F = 34.287( sig 0.001) r = 0.762 Rsqr = 0.965 SEE = 0.390 D/W = 1.727 | F | Intercept L B1 σp σm | 21.01 2.576 -1.844 0.214 -11.465 | 0.000 0.050 0.124 0.839 0.693 |
| Equation Nos. | A | C | E | ||||
| 3a | 7.9607 | 8.6350 | -0.67432 | 8.4214 | -0.46072 | 8.0947 | -0.13400 |
| 3b | 8.7209 | 8.8798 | -0.15895 | 9.1579 | -0.43709 | 9.3161 | -0.59528 |
| 3c | 8.6894 | 8.4678 | 0.221566 | 8.7047 | -1.53E-02 | 8.8419 | -0.15254 |
| 3d | 9.1478 | 9.0093 | 0.138470 | 8.8645 | 0.283317 | 8.8503 | 0.29751 |
| 3e | 9.4108 | 9.4590 | -4.8E-02 | 9.2819 | 0.128812 | 9.0131 | 0.39762 |
| 3f | 8.7204 | 8.4049 | 0.315474 | 8.5034 | 0.216909 | 8.5288 | 0.19152 |
| 3g | 7.9627 | 8.0178 | -5.5E-02 | 8.0919 | -0.12929 | 7.9749 | -1.2E-02 |
| 3h | 8.0490 | 8.1824 | -0.13346 | 8.1587 | -0.10971 | 8.0473 | 1.664E-03 |
| 3i | 3.7543 | 3.7850 | -3.07E-02 | 3.7724 | -1.81E-02 | 3.7869 | -3.26E-02 |
| 3j | 8.8084 | 8.3829 | 0.42540 | 8.2671 | 0.54127 | 8.7700 | 3.83E-02 |
| Equation Nos. | B | D | F | ||||
|---|---|---|---|---|---|---|---|
| Compound No. | Observed value of log(P/C) | Predicted value of log(P/C) | Residual | Predicted value of log(P/C) | Residual | Predicted value of log(P/C) | Residual |
| 3a | 8.1369 | 8.4728 | -0.3359 | 8.3139 | -0.1771 | 8.1600 | -2.31E-02 |
| 3b | 8.4199 | 8.7608 | -0.3410 | 8.9742 | -0.5543 | 9.0213 | -0.601454 |
| 3c | 8.6894 | 8.3112 | 0.3781 | 8.5231 | 0.1662 | 8.5692 | 0.120200 |
| 3d | 9.1478 | 9.0311 | 0.1166 | 8.8937 | 0.2541 | 8.7516 | 0.396140 |
| 3e | 9.1098 | 9.1205 | -1.067E-02 | 8.9361 | 0.1736 | 8.7483 | 0.361502 |
| 3f | 8.1184 | 8.1903 | -7.194E-02 | 8.2388 | -0.1205 | 8.2062 | -8.78E-02 |
| 3g | 7.8378 | 8.0436 | -0.2058 | 8.1529 | -0.3151 | 8.1240 | -0.286304 |
| 3h | 8.3501 | 8.1024 | 0.2476 | 8.0655 | 0.2844 | 8.2693 | 8.077E-02 |
| 3i | 3.7543 | 3.7840 | -2.968E-02 | 3.7820 | -2.77E-02 | 3.7531 | 1.197E-03 |
| 3j | 8.5075 | 8.2548 | 0.2526 | 8.1911 | 0.31631 | 8.4684 | 3.898E-02 |
Conclusions
Experimental
General
General method for the preparation of 3a-j
Antimicrobial activity
QSAR calculations
Acknowledgments
References and Notes
- Spirkova, K.; Stankovsky, S.; Mrvova, A.; Cipak, L. Synthesis and Biological Activity of Some 2-substituted Quinazolin-4-ones. Chem. Pap. 1999, 53, 272–275. [Google Scholar]
- Alagarsamy, V.; Giridhar, R.; Yadav, H. R.; Revathi, R.; Rukmani, K.; De Clercq, E. Anti HIV, antibacterial and antifungal activities of some novel1,4-disubstituted-1,2,4-triazolo[4,3a] quinazolin-5(4h)-ones. Indian J. Pharm. Sci. 2006, 68, 532–535. [Google Scholar] [CrossRef]
- Srivastava, V. K.; Singh, A.; Gucati, A.; Shankar, K. Antiparkinsonian agent from qunazonyl thioazolidinones and azetidinones. Indian J. Chem. 1987, 26, 652–656. [Google Scholar]
- Gupta, D. P.; Ahmed, S.; Kumar, A.; Shankar, K. Newer Quinazolinone Derivatives as Anthelmintic Agents. Indian J. Chem. 1988, 27, 1060–1062. [Google Scholar]
- Jatav, V.; Mishra, P.; Kaswa, S.; Stabes, J. P. Synthesis and CNS depressant activity of some novel 3-[5-substituted 1,3,4-thiadiazole-2-yl]-2-styryl quinazoline-4(3H)-ones. Eur. J. Med. Chem. 2007. ?. [Google Scholar]
- Joshi, V.; Chaurasia, R. P. Synthesis of Some New 4-Quinazolinone-2-carboxylhydrazides and their Tosyl Derivatives Having Potential Biological Activity. Indian J. Chem. 1987, 26, 602–604. [Google Scholar]
- Yale, H. J.; Kalkstein, M. Substituted 2,3-dihydro-4(1H)-quinazolinones, a new class of inhibitors of cell multiplication. J. Med. Chem. 1967, 10, 334–336. [Google Scholar] [CrossRef]
- Neil, G. L.; Li, L. H.; Buskirk, H. H.; Moxley, T. E. Antitumor effects of the antispermatogenicagent, 2,3-dihydro-2-(1-naphthyl)-4(1H)-quinazolinone (NSC-145669). Cancer Chemother. Rep. 1972, 56, 163–173. [Google Scholar]
- Jiang, J. B; Hesson, D. P.; Dusak, B. A.; Dexter, D. L.; Kang, G. J.; Harmel, E. Synthesis and Biological Evaluation of 2-Styrylquinazolin-4(3H)-ones, a New Class of Antimitotic Anticancer Agents which Inhibit Tubulin Polymerization. J. Med. Chem. 1990, 33, 1721–1728. [Google Scholar] [CrossRef]
- Lin, C. M.; Kang, G. J.; Roach, M. C.; Jiang, J. B.; Hesson, D. P.; Luduena, R. F.; Hamel, E. Investigation of the mechanism of the interaction of tubulin with derivatives of 2-styryl-quinazolin-4(3H)-one. Mol. Pharmacol. 1991, 40, 827–832. [Google Scholar]
- Pandey, V. K.; Tusi, S.; Tusi, Z.; Raghubir, R.; Dixit, M.; Joshi, M. N. Heterocyclic Compounds Thiazolyl quinazolones as Potential Antiviral and Antihypertensive Agents. Indian J. Chem. 2004, 43, 180–184. [Google Scholar]
- Kamal, A.; Devaiah, V.; Sankaraiah, N.; Reddy, K. L. A Polymer-assisted Solution-Phase Strategy for the Synthesis of Fused [2,1-b] quinazolones and the Preparation of Optically Active Vasicinone. Synlett 2006, 2609–2612. [Google Scholar]
- Chohan, Z. H.; Supuran, C.T.; Scozzafava, A; Farroq, M.A. Antibacterial Schiff bases of oxalyl-hydrazine/diamide incorporating pyrrolyl and salicylyl moieties and their zinc(II) complexes. J. Enzym. Inhib. Med, Chem. 2002, 17, 1–7. [Google Scholar] [CrossRef]
- Patel, J. A.; Mistry, B. D.; Desai, K. R. Synthesis and Antibacterial Activity of Newer quinazolones. Molecules 2006, 3, 97–102. [Google Scholar]
- Alagarsamy, V.; Salomon, V. R.; Vanikavitha, G.; Paluchamy, V.; Chandran, M. R.; Sajin, A. A.; Thangathiruppathy, A.; Amuthalakshmi, S.; Revathi, R. Synthesis, Analgesic, Anti-inflamatory and Antibacterial Activities of Some Novel 2-phenyl-3-substituted Quinazolin-4(3H)ones. Biol. Pharm. Bull. 2002, 25, 1432–1435. [Google Scholar]
- Mukherjee, S.; Chakraborty, R. Incidence of class-1 integrons in multiple antibiotic-resistant Gram-negative copiotrophic bacteria from the River Torsa in India. Res. Microbiol. 2006, 157, 220–226. [Google Scholar] [CrossRef]
- Karleson, M.; Lobanov, V. S.; Katrizky, A. R. Quantum Chemical Descriptors in QSAR/QSPR studies. Chem. Rev. 1996, 96, 1027–1043. [Google Scholar] [CrossRef]
- Zhang, Li; Xiafiu, Y.; Cia, L.-H.; Hu, Y.-J.; Yin, J.; Hu, P.-Z. Inhibitory Study of Some Novel Schiff base derivatives on Staphylococcus aureus by microcalorimetry. Thermochim. Acta 2006, 440, 51–56. [Google Scholar] [CrossRef]
- Hantzsh, C.; Lee, A. Electronic Parameters: Substituent Constants for Correlation Analysis in Chemistry and Biology; Jonh Wiley & Sons: New York, 1979; Chapter 1; pp. 1–7. [Google Scholar]
- Verloop, A.; Hoogenstraaten, W.; Tipker, J. Development and application of New Steric Substituent Parameters in Drug Design: Drug Design, Vol-VII; Ariens, E. J., Ed.; Academic Press: New York, 1976; Chapter 4; pp. 165–207. [Google Scholar]
- Sample Availability: Samples of compounds 1, 2 and 3a-j are available from the authors.
© 2007 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Nanda, A.K.; Ganguli, S.; Chakraborty, R. Antibacterial Activity of Some 3-(Arylideneamino)-2-phenylquinazoline-4(3H)-ones: Synthesis and Preliminary QSAR Studies. Molecules 2007, 12, 2413-2426. https://doi.org/10.3390/12102413
Nanda AK, Ganguli S, Chakraborty R. Antibacterial Activity of Some 3-(Arylideneamino)-2-phenylquinazoline-4(3H)-ones: Synthesis and Preliminary QSAR Studies. Molecules. 2007; 12(10):2413-2426. https://doi.org/10.3390/12102413
Chicago/Turabian StyleNanda, Ashis Kumar, Subarna Ganguli, and Ranadhir Chakraborty. 2007. "Antibacterial Activity of Some 3-(Arylideneamino)-2-phenylquinazoline-4(3H)-ones: Synthesis and Preliminary QSAR Studies" Molecules 12, no. 10: 2413-2426. https://doi.org/10.3390/12102413
APA StyleNanda, A. K., Ganguli, S., & Chakraborty, R. (2007). Antibacterial Activity of Some 3-(Arylideneamino)-2-phenylquinazoline-4(3H)-ones: Synthesis and Preliminary QSAR Studies. Molecules, 12(10), 2413-2426. https://doi.org/10.3390/12102413




