Preparation of Benzalkonium Salts Differing in the Length of a Side Alkyl Chain
Abstract
:Introduction
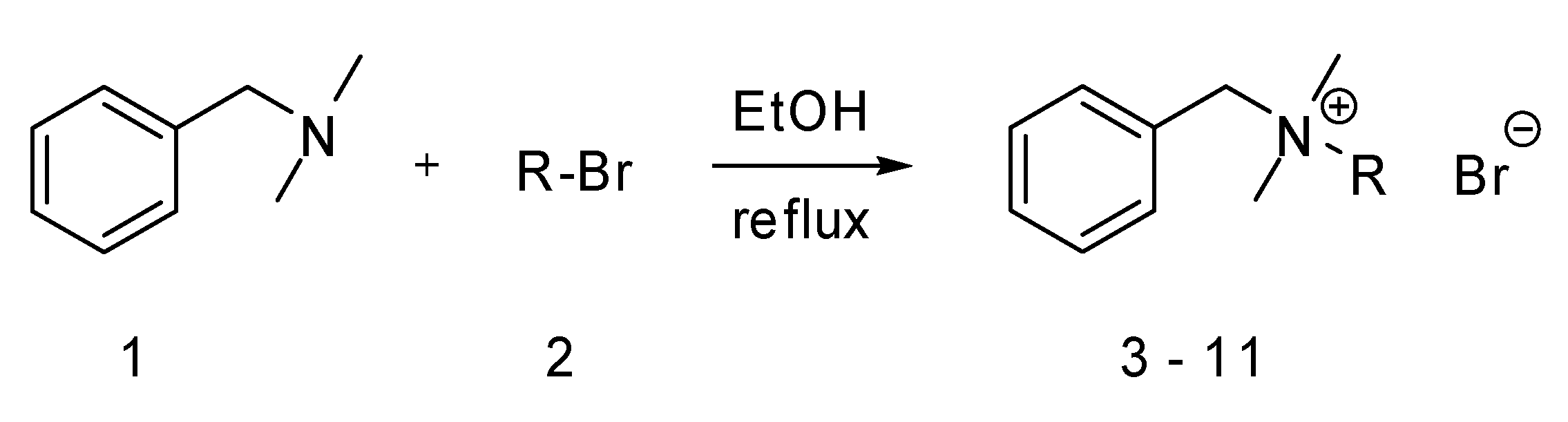
Results and Discussion
| Compound | Yield (%) | m.p. (°C) | TLC Rf | HPLC Rt |
|---|---|---|---|---|
| 3 | 94 | 120.5-121.5 | 0.083 | 4.76 |
| 4 | 44 | 150.0-152.0 | 0.260 | 5.84 |
| 5 | 54 | 122.0-123.5 | 0.420 | 7.13 |
| 6 | 61 | 53.0-56.0 | 0.512 | 8.47 |
| 7 | 61 | 34.0-37.5 | 0.558 | 9.92 |
| 8 | 80 | 37.0-40.0 | 0.592 | 11.55 |
| 9 | 71 | 43.5-47.5 | 0.612 | 13.43 |
| 10 | 58 | 50.0-53.0 | 0.634 | 15.60 |
| 11 | 71 | 81.0-83.0 | 0.651 | 18.15 |
| 12 | 64 | 85.0-88.0 | 0.664 | 21.25 |
Conclusions
Experimental
General
Synthesis
Acknowledgments
References and Notes
- Grillitsch, B.; Gans, O.; Kreuzinger, N.; Scharf, S.; Uhl, M.; Fuerhacker, M. Environmental risk assessment for quaternary ammonium compounds: a case study from Austria. Wat. Sci. Technol. 2006, 54, 111–118. [Google Scholar]
- Daoud, N.N.; Dickinson, N.A.; Gilbert, P. Determination of benzalkonium chloride by chemical ionization mass spectroscopy. Microbios 1983, 37, 73–85. [Google Scholar]
- Debreceni, G.; Meggyesi, R.; Mestyan, G. Efficacy of spray disinfection with a 2-propanol and benzalkonium chloride containing solution before epidural catheter insertion--a prospective, randomized, clinical trial. Br. J. Anaesth. 2007, 98, 131–135. [Google Scholar] [CrossRef]
- Chung, S.H.; Lee, S.K.; Cristol, S.M.; Lee, E.S.; Lee, D.W.; Seo, K.Y.; Kim, E.K. Impact of short-term exposure of commercial eyedrops preserved with benzalkonium chloride on precorneal mucin. Mol. Vis. 2006, 12, 415–421. [Google Scholar]
- Brill, F.; Goroncy-Bermes, P.; Sand, W. Influence of growth media on the sensitivity of Staphylococcus aureus and Pseudomonas aeruginosa to cationic biocides. Int. J. Hyg. Environ. Health. 2006, 209, 89–95. [Google Scholar] [CrossRef]
- Rabenau, H.F.; Kampf, G.; Cinatl, J.; Doerr, H.W. Efficacy of various disinfectants against SARS coronavirus. J. Hosp. Infect. 2005, 61, 107–111. [Google Scholar] [CrossRef]
- Labranche, L.P.; Dumont, S.N.; Levesque, S.; Carrier, A. Rapid determination of total benzalkonium chloride content in ophthalmic formulation. J. Pharm. Biomed. Anal. 2007, 43, 989–993. [Google Scholar]
- Cabal, J.; Kassa, J.; Severa, J. A comparison of the decontamination efficacy of foam-making blends based on cationic and nonionic tensides against organophosphorus compounds determined in vitro and in vivo. Hum. Experim. Toxicol. 2003, 22, 507–514. [Google Scholar]
- Rudakova, A.W.; Popova, L.P. Study of reaction capability of lygnoceryl alcohol (In Russian). Z. Obs. Chim. 1966, 36, 242–244. [Google Scholar]
- Novaki, L.P.; El Soud, O.A. Solvatochromism in aqueous micellar solutions: effects of the molecular structures of solvatochromic probes and cationic surfactants. Phys. Chem. Chem. Phys. 1999, 1, 1957–1964. [Google Scholar] [CrossRef]
- Stepanenko, B.N.; Ulámána, T.S.; Zelenkova, V.V. Synthesis of some qauternary salts of ammonium compounds (In Russian). Chim. Farm. Z. 1974, 8, 21–24. [Google Scholar]
- Echols, L.; Maier, V.P.; Poling, S.M.; Sterling, P.R. New bioregulators of gibberellin biosynthesis in Gibberella fujikuroi. Phytochemistry 1981, 20, 433–438. [Google Scholar]
- Kuca, K.; Kivala, M.; Dohnal, V. A general method for the quaternization of N,N-dimethyl benzylamines with long chain n-alkylbromides. J. Appl. Biomed.. 2004, 2, 195–198. [Google Scholar]
- Prince, S.J.; McLaury, H.J.; Allen, L.V.; McLaury, P. Analysis of benzalkonium chloride and its homologs: HPLC versus HPCE. J. Pharm. Biomed. Anal. 1999, 19, 877–882. [Google Scholar]
- Elrod Jr., L.; Golich, T.G.; Morley, J.A. Determination of benzalkonium chloride in eye care products by high-performance liquid chromatography and solid-phase extraction or on-line column switching. J. Chromatogr. 1992, 625, 362–367. [Google Scholar] [CrossRef]
- Ambrus, G.; Takahashi, L.T.; Marty, P.A. Direct determination of benzalkonium chloride in ophthalmic systems by reversed-phase high-performance liquid chromatography. J. Pharm. Sci. 1987, 76, 174–176. [Google Scholar] [CrossRef]
- Kuca, K.; Dohnal, V.; Bielavska, M.; Cabal, J. Determination of benzalkonium bromide homologues in desinfection products using high-performance liquid chromatography. Anal. Lett. 2005, 38, 673–682. [Google Scholar]
- European Pharmacopoeia 5 – Directorate for the Quality of Medicines of the Council of Europe; Council of Europe: Aubin (France), 2005; Vol. 1, pp. 1068–1070.
- Sample Availability: Samples of all prepared compounds are available from the authors.
© 2007 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Kuca, K.; Marek, J.; Stodulka, P.; Musilek, K.; Hanusova, P.; Hrabinova, M.; Jun, D. Preparation of Benzalkonium Salts Differing in the Length of a Side Alkyl Chain. Molecules 2007, 12, 2341-2347. https://doi.org/10.3390/12102341
Kuca K, Marek J, Stodulka P, Musilek K, Hanusova P, Hrabinova M, Jun D. Preparation of Benzalkonium Salts Differing in the Length of a Side Alkyl Chain. Molecules. 2007; 12(10):2341-2347. https://doi.org/10.3390/12102341
Chicago/Turabian StyleKuca, Kamil, Jan Marek, Petr Stodulka, Kamil Musilek, Petra Hanusova, Martina Hrabinova, and Daniel Jun. 2007. "Preparation of Benzalkonium Salts Differing in the Length of a Side Alkyl Chain" Molecules 12, no. 10: 2341-2347. https://doi.org/10.3390/12102341
APA StyleKuca, K., Marek, J., Stodulka, P., Musilek, K., Hanusova, P., Hrabinova, M., & Jun, D. (2007). Preparation of Benzalkonium Salts Differing in the Length of a Side Alkyl Chain. Molecules, 12(10), 2341-2347. https://doi.org/10.3390/12102341




