Synthesis and Spectroscopic Studies of New Schiff Bases
Abstract
:Introduction
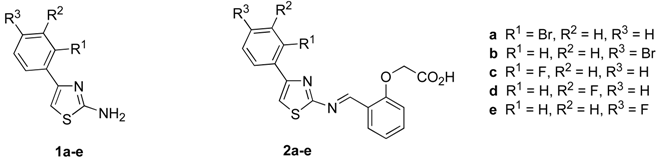
Results and Discussion
Experimental
General
Synthesis of 2-amino-4-(2’-bromophenyl)-thiazole (1a)
2-amino-4-(4’-bromophenyl)thiazole (1b)
2-amino-4-(2’-fluorophenyl)thiazole (1c)
2-amino-4-(3’-fluorophenyl)thiazole (1d)
2-amino-4-(4’-fluorophenyl)thiazole (1e)
Preparation of (2-{[4-(2-bromophenyl)thiazol-2-ylimino]methyl}phenoxy)acetic acid (2a):
Preparation of (2-{[4-(4-bromophenyl)-thiazole-2-ylimino]methyl}phenoxy)acetic acid (2b)
Preparation of (2-{[4-(2’-fluorophenyl)-thiazole-2-ylimino]methyl}phenoxy)acetic acid (2c)
Preparation of (2-{[4-(3’-fluorophenyl)thiazole-2-ylimino]methyl}phenoxy)acetic acid (2d)
Preparation of (2-{[4-(4’-fluorophenyl)-thiazole-2-ylimino]methyl}phenoxy)acetic acid (2e)
Acknowledgements
References
- Karia, F. D.; Parsania, P.H. Synthesis, biological and thermal properties of Schiff bases of bisphenol-C. Asian J. Chem. 1999, 11, 991–995. [Google Scholar]
- More, P. G.; Bhalvankar, R. B.; Pattar, S. C. Synthesis and biological activities of Schiff bases of aminothiazoles. J. Indian Chem. Soc. 2001, 78, 474–475. [Google Scholar]
- El-Masry, A. H.; Fahmy, H. H.; Abdelwahed, S. H. A. Synthesis and antimicrobial activity of some new benzimidazole derivatives. Molecules 2000, 5, 1429–1438. [Google Scholar]
- Baseer, M. A.; Jadhav, V. D.; Phule, R. M.; Archana, Y. V.; Vibhute, Y. B. Synthesis and antimicrobial activity of some new Schiff bases. Orient. J. Chem. 2000, 16, 553–556. [Google Scholar]
- Pandeya, S. N.; Sriram, D.; Nath, G.; De Clercq, E. Synthesis and antimicrobial activity of Schiff and Mannich bases of isatin and its derivatives with pyrimidine. IL Farmaco 1999, 54, 624–628. [Google Scholar]
- Singh, W. M.; Dash, B. C. Synthesis of some new Schiff bases containing thiazole and oxazole nuclei and their fungicidal activity. Pesticides 1988, 22, 33–37. [Google Scholar]
- Hodnett, E. M.; Dunn, W. J. Structure-antitumour activity correlation of some Schiff bases. J. Med. Chem. 1970, 13, 768–770. [Google Scholar]
- Desai, S. B.; Desai, P. B.; Desai, K.R. Synthesis of some Schiff bases, thiazolidones, and azetidinones derived from 2,6-diaminobenzo[1,2-d:4,5-d’]bisthiazole and their anticancer activities. Heterocycl. Commun. 2001, 7, 83–90. [Google Scholar]
- Pathak, P.; Jolly, V. S.; Sharma, K.P. Synthesis and biological activities of some new substituted arylazo Schiff bases. Oriental. J. Chem. 2000, 16, 161–162. [Google Scholar]
- Samadhiya, S.; Halve, A. Synthetic utility of Schiff bases as potential herbicidal agents. Orient. J. Chem. 2001, 17, 119–122. [Google Scholar]
- Aydogan, F.; Öcal, N.; Turgut, Z.; Yolacan, C. Transformations of aldimines derived from pyrrole-2-carboxaldehyde. Synthesis of thiazolidino-fused compounds. Bull. Korean Chem. Soc. 2001, 22, 476–480. [Google Scholar]
- Taggi, A. E.; Hafez, A. M.; Wack, H.; Young, B.; Ferraris, D.; Lectka, T. The development of the first catalysed reaction of ketenes and imines: catalytic asymmetric synthesis of β-lactams. J. Am. Chem. Soc. 2002, 124, 6626–6635. [Google Scholar]
- King, L.C.; Hlavacek, R.J. Reaction of ketones with iodine and thiourea. J. Am. Chem. Soc. 1950, 72, 3722. [Google Scholar]
- Zav’Yalov, S.I.; Dorofeeva, O.V.; Rumyantseva, E.E.; Kulikova, L.B.; Ezhova, G.I.; Kravchenko, N.E.; Zavozin, A.G. Synthesis of 2-aminothiazole derivatives. Pharm. Chem. J. 2001, 35, 96–98. [Google Scholar]
- Sample availability: Contact the authors
© 2006 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Siddiqui, H.L.; Iqbal, A.; Ahmad, S.; Weaver, W. Synthesis and Spectroscopic Studies of New Schiff Bases. Molecules 2006, 11, 206-211. https://doi.org/10.3390/11020206
Siddiqui HL, Iqbal A, Ahmad S, Weaver W. Synthesis and Spectroscopic Studies of New Schiff Bases. Molecules. 2006; 11(2):206-211. https://doi.org/10.3390/11020206
Chicago/Turabian StyleSiddiqui, Hamid Latif, Amjid Iqbal, Saeed Ahmad, and W. Weaver. 2006. "Synthesis and Spectroscopic Studies of New Schiff Bases" Molecules 11, no. 2: 206-211. https://doi.org/10.3390/11020206
APA StyleSiddiqui, H. L., Iqbal, A., Ahmad, S., & Weaver, W. (2006). Synthesis and Spectroscopic Studies of New Schiff Bases. Molecules, 11(2), 206-211. https://doi.org/10.3390/11020206



