Results and Discussion
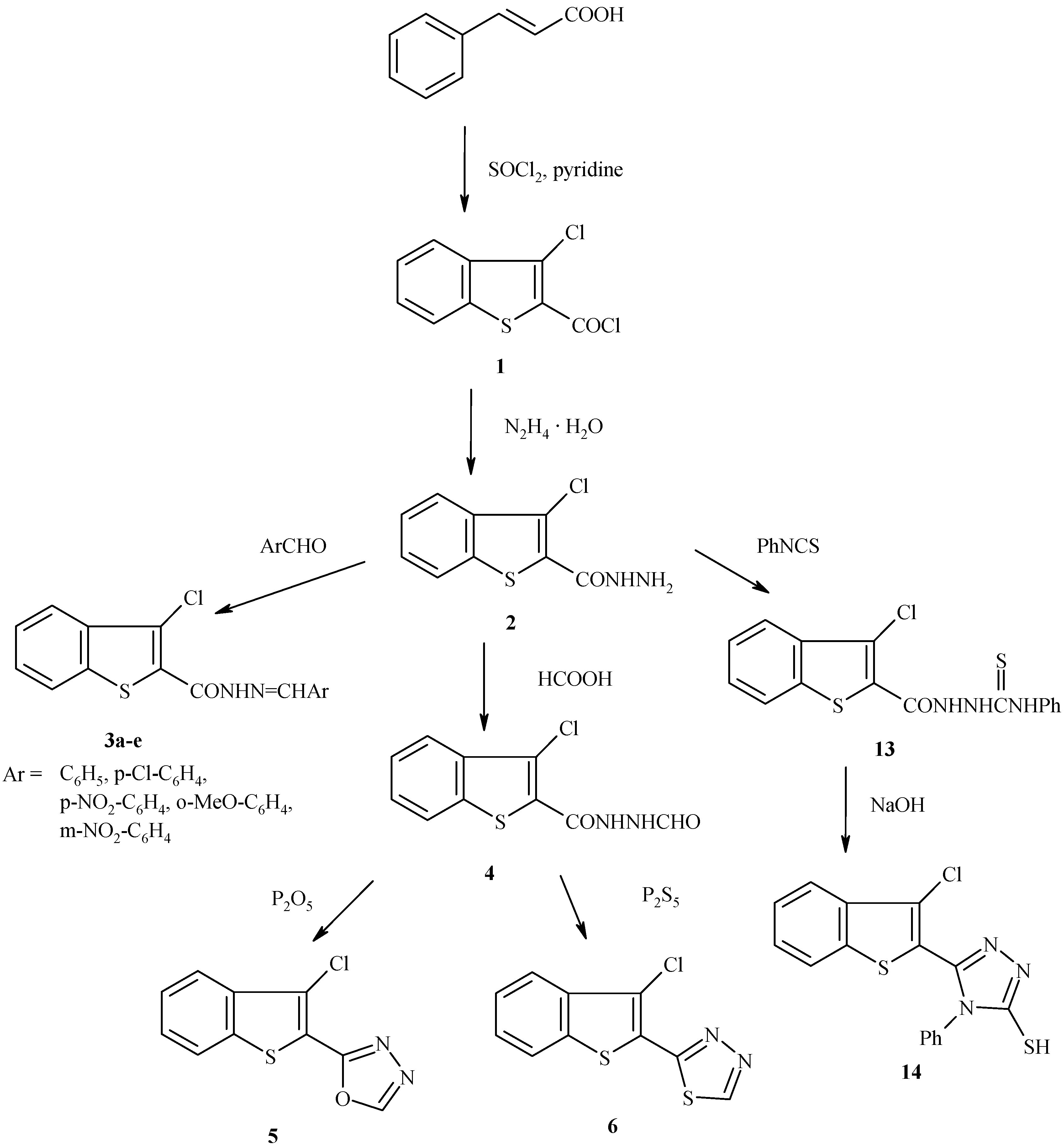
The new oxadiazole and thiadiazole derivatives were prepared following the reaction sequences depicted in
Scheme 1. The starting material for the synthesis of the targeted compounds is 3-chloro-2- chlorocarbonyl benzo[b]thiophene (
1) which was prepared by the reaction of cinnamic acid with thionyl chloride in DMF and dry pyridine according to the reported method [
10]. Reaction between
1 and hydrazine hydrate afforded the acid hydrazide
2 in good yield. The IR spectra showed the N-H stretching absorption near 3140 cm
-1 and the C=O stretching one at 1640 cm
-1. Condensation of the acid hydrazide
2 with aryl aldehydes in absolute ethanol gave the Schiff bases
3a-e.
The formation of these Schiff bases was indicated by the presence in their IR spectra of the azomethine (CH=N) stretching band at 1600 cm
-1, combined with the disappearance of the NH
2 stretching band. Further, 3-chloro-2-(N-formyl acid hydrazide)benzo[b]thiophene (
4) was obtained in 70% yield when the acid hydrazide
2 was allowed to react with formic acid[
11,
12]. The structure of compound
4 was confirmed by the presence of two amidic carbonyl stretching bands at 1720 cm
-1 and 1660 cm
-1 (CO-NH-NH-CHO).
Refluxing compound
4 with phosphorous pentoxide in xylene for one hour afforded 2-(3-chloro-1- benzothien-2-yl)-1,3,4- oxadiazole (
5), which displayed two bands at 1245 cm
-1 and 1080 cm
-1 for the C-O-C asymmetric and symmetric stretching, respectively [
12,
13], in addition to the band at 1620 cm
-1 for the C=N stretch. Moreover, 2-(3-chloro-1-benzothien-2-yl)-1,3,4-thiadiazole (
6) was obtained in 62-68% yield from the reaction of
4 with phosphorous pentasulphide under reflux in xylene solution. The absorption band at 1610 cm
-1 in the IR spectrum of
6 is due to C=N stetching. On the other hand the reaction of
1 with powdered thiosemicarbazide in dry benzene afforded the acylthiosemicarbazide
7, which upon ring closure with NaOH gave 5-(3-chloro-1-benzothien-2-yl)-4H-1,2,4-triazole-3-thiol (
8) [
14] which exists in a tautomeric thiol – thione equilibrium, as indicated by the C=S stretching band at 1190 cm
-1 and S-H stretch at 2600 cm
-1 [
15]. Alkylation of
8 gave compound
9, while treatment with secondary amines resulted in compound
10 by nucleophilic displacement of the SH group. Further, reaction of
8 with propargyl chloride led to the introduction of alkynyl functionality, thus affording compound
11, as indicated by the strong band at 2100 cm
-1 for the C≡C stretch and 3300 cm
-1 for the ≡C-H stretching vibration, respectively. Treatment of
11 with secondary cyclic amines, namely morpholine, piperidine or piperazine, under Mannich reaction conditions resulted into the Mannich bases
12a-c, which was indicated by the disapperance of the ≡C-H vibration band.
In another triazole preparation, condensation of acid hydrazide 2 with phenyl isothiocyanate afforded the corresponding thiosemicarbazide 13 in moderate yield. Ring closure of arylthiosemicarbazides in an alkaline medium is a well known method for the synthesis of 1,2,4-triazoles and thus 5–(3-chloro-1-benzothien-2-yl)-4-phenyl-4H-1,2,4-triazole-3-thiol (14) was obtained from the corresponding 13 by this method. The IR spectra of 1,4-substituted thiosemicarbazide derivative 13 display a C=O stretching band at 1680 cm-1 and C=S stretching band at 1240 cm-1. It is worth mentioning that compounds 8 and 14 exist as thiol – thione tautomers, as indicated by their IR spectra which showed a band due to SH and another band to N-C=S .
Experimental
General
Melting points were determined in open capillary tubes on a Gallenkamp mething point apparatus and are uncorrected. The IR spectra (KBr discs) were recorded with a Pye–Unicam SP-300 spectrometer. UV spectra were recorded on Hitachi–2000 spectrophotometer using absolute ethanol as solvent. 1H-NMR on a Hitachi–Perkin–Elmer 60 MHz NMR spectrometer in DMSO–d6 solutions and with tetramethylsilane as an internal standard. Elemental analysis were done on a 1106 Carlo – Erba instrument.
Preparation of 3-chloro-2-chloromethyl benzo[b]thiophene (1)
This compound was prepared according to the method reported in the literature [
10]; mp 112- 114°C (lit. mp 110-112°C [
10])
Preparation of 3- chloro benzo[b] thiophene-2-carboxylic acid hydrazide (2)
To a solution of compound 1 (0.001 mole) in dry chloroform (5 mL), hydrazine hydrate (0.0018 mole) was added dropwise. The mixture was refluxed for one hour. The solvent was removed in vacuo and the solid product was collected and crystallized from ethanol, mp 164-166°C; 73% yield; IR (υ, cm-1): 3280, 3140 (N-H), 1650(C =O), 1570 (C=C), 1070 (=C-Cl), 680 (C-S-C); UV (λmax): 297 nm.
Preparation of compounds 3a-e.
A mixture of 2 (0.01 mole) and the corresponding aryl halide (0.01 mole) in ethanol (25 mL) was refluxed for 3 hours and cooled. The precipitate was filtered and crystallized from ethanol. (3b) Yield 76%; mp 212-214; IR v (cm-1): 3180 (N-H), 3020 (C-H ar ), 1650 (C=O), 1600(C=N); UV: 295 nm; (3c) Yield 82%; mp 240-242; IR v (cm-1): 3200 (N-H), 3050 (C-H ar), 1640 (C=O), 1610 (C=N); UV: 289 nm.
Preparation of compound 4.
A solution of 2 (0.01 mole) in formic acid (20 mL) was refluxed for 30 minutes. The solvent was evaporated and the residue was crystallized from methanol; 73% yield; mp. 188-190°C; IR (υ, cm-1): 3200 (N-H), 1660, 1720 (C=O); UV (λmax): 306 nm; Calculated for C10H7N2O2SCl (%); C, 57.30; H, 3.09; N, 11.79; found (%): C, 56.95; H, 3.11; N, 11.65.
General Procedure for preparation of 2-(3-chloro-1-benzothien-2-yl)-1,3,4-oxadiazole (5) and 2-(3- chloro-1-benzothien-2-yl)-1,3,4-thiadiazole (6).
To a solution of 4 (0.01 mole) in xylene (150 mL), phosphorous pentoxide or phosphorous pentasulphide (0.01 mole) was added. The mixture was refluxed for one hour. The solvent was evaporated, water (10 mL) was added and the mixture was extracted with chloroform. The solvent was evaporated and the residue was recrystallized from benzene or ethylacetate. Compound 5: mp. 100- 102 °C; yield 53%; IR (υ, cm-1): 1620 (C=N), 1530 (C=C), 1245, 1080 (C-O-C); UV (λmax) 287 nm; 1H-NMR δ: 6.2 (s, 1H, oxadiazole), 7.0-7.5 (m, 4H, Ar-H); Compound 6: mp. 124-126°C; yield 63%; IR (υ, cm-1) 1610 (C=N), 1550 (C=C); UV (λmax) 294 nm; 1H-NMR δ: 6.6 (s, 1H, thiadiazole), 7.7-8 (m, 4H, Ar-H); Calcd. for C10H5N2S2Cl (%): C, 47.62; H,1.98; N,11.11; found %: C, 47.29; H,2.07; N, 10.98.
Preparation of 3- chloro benzo[b]thiophene -2- carboxylic thiosemicarbazide (7).
To a solution of compound 1 (0.01 mole) in dry benzene (50 mL) was added thiosemicarbazide (0.01 mole). The mixture was refluxed for 2 hours and after cooling the precipitate was filtered and crystallized from ethanol – water; mp 200-202°C; yield 78%; IR (υ, cm-1): 3170 (N-H); 1660 (C=O); 1220 (C=S); 1070 (C-Cl); 690 (C-S-C); UV (λmax): 298 nm; 1H-NMR δ (ppm): 10.4 (s, 1H, CONH); 9.7 (s, 1H, CSNH); 8-7.5 (m, 4H, Ar-H); 3.6 (s, 2H, CSNH2); Calcd. for C10H8N3OS2 Cl (%): C, 42.10; H, 2.80; N, 14.73; found (%): C,41.88; H,3.01; N,14.55.
Preparation of 5-(3-chloro-1-benzothien-2-yl)-4H-1,2,4-triazole-3-thiol (8).
A stirred mixture of compound 7 and aqueous sodim hydroxide (4%, 10 mL) was refluxed for 3 hours. The mixture was acidified with dil. HCl and the precipitate was collected and crystallized from ethanol; yield 54%; mp. 231(dec)°C; IR υ (cm-1): 3130 (N-H), 3045 (C-Har), 2600 (SH), 1650 (C=N), 1590 (C=C); 1190 (C=S); UV (λmax): 299.5 nm; Calcd. for C10H6N3S2Cl (%): C, 44.94; H, 2.28; N, 15.73; found (%): C, 44.59; H, 2.34; N, 15.48.
Preparation of compounds 9a-c.
To the stirred solution of compound 8 (0.001 mole) and sodium acetate (0.0024 mole) in ethanol (20 mL), alkyl/aryl halide (0.001 mole) was added. The mixture was refluxed for 3 hours. After cooling, the solution was poured onto crushed ice (30 g). The precipitate was filtered and recrystallized from acetone – water.
5-(3-chloro-1-benzothien-2-yl)-4H-1,2,4-triazole-3 methyl thiol (9a): Yield 52%; mp. 150-152°C; IR (υ, cm-1): 3330 (N-H), 3070 (C-Har), 1600 (C=N), 1550 (C=C), 1440 (S-CH3), 750 (C-S); UV (λmax): 252 nm; 1H-NMR (δ) ppm: 7.5-8.5 (m, 4H, Ar-H), 3.2 (s, 3H, SCH3).
5-(3-chloro-1-benzothien-2-yl)-4H-1,2,4-triazole-3-(4-nitrobenzyl) thiol (9b): Yield 40%; mp. 163- 165°C; IR (υ, cm-1): 3260 (N-H), 3070 (C-Har), 1600 (C=N), 1510 (C=C), 1510, 1340 (C–NO2); UV (λmax): 381 nm; Calcd. for C17H11N4O2S2Cl (%): C, 52.57; H, 2.83; N, 10.82; found (%): C, 52.34; H, 2.95; N, 10.66.
5-(3-chloro-1-benzothien-2-yl)-4H-1,2,4-triazole-3-(2,4-dinitrophenyl) thiol (9 c): Yield 67%; mp 161-163°C; IR (υ, cm-1): 3120 (N-H), 3050 (C-Har), 1620 (C=N), 1530 (C=C), 1540, 1350 (C-NO2), 760 (C-S); UV (λmax): 382 nm.
Preparation of compounds 10a-c.
To a stirred solution of compound 8 (0.003 mole) in dry dioxane (10 mL) was added a solution of the appropriate secondary amine (0.003 mole) in dry dioxane (5 mL). The mixture was refluxed for four hours. After cooling, the precipitate was filtered and crystallized from methanol.
5-(3-chloro-1-benzothien-2-yl)-4H-1,2,4-triazole-3-N-piperidine (10a): Yield 60%; mp 160-162°C; IR (υ, cm-1): 3340 (N-H), 3030 (C-Har ), 2860, 2970 (C-Hal), 1640 (C=N), 1575 (C=C); UV (λmax): 300 nm; 1H-NMR (δ) ppm: 2.6-2.8 (m, piperidine ring protons), 6.5-6.8 (m, 4H, Ar-H). Calcd. For C15H15N4SCl (%): C, 56.60; H, 4.71; N, 17.61; found (%): C, 56.33; H, 5.00; N, 17.44
General procedure for preparation of 5-(3-chloro-1-benzothien -2-yl)-4H-1,2,4-triazole-3-(prop-2- ynyl) thiol (11)
To a stirred solution of compound 8 (0.01 mole) and triethylamine (0.01 mole) in ethanol (25 mL), was added propargyl chloride (0.01 mole) dropwise. The mixture was refluxed for two hours in a water bath. Excess ethanol was removed in vacuo. The product was collected and crystallized from ethanol – water; Yield 75%; mp 108-110°C; IR (υ, cm-1): 3300 (≡ C-H), 3180 (N-H), 3060 (C-Har), 2970 (C-Hal), 1650 (C=N), 1590 (C=C), 2100 (C≡C); UV (λmax): 298nm.
General procedure for preparation of compounds 12a-c.
To a stirring solution of compound 11 (0.005 mole) in dioxane (25 mL) was added cuprous chloride (0.0025 g) and the mixture was heated for a few minutes, then paraformaldehyde (0.005 mole) and the appropriate secondary amine (0.005 mole) were added. The mixture was heated at 90°C for two hours. After cooling, the mixture was filtered and the poured onto ice water (100 mL). The residue was extracted with chloroform (3 x 25 mL) and purified on a column of silica gel using chloroform – petroleum spirit as eluent. The product is oily. Yield of 12b 68% (from piperidine); IR (υ, cm-1): 3400 (N-H), 3050(C-Har), 2920, 2880 (C-Hal ), 1645 (C=N), 1560 (C=C), 1180 (C-N); UV (λmax): 305 nm.
General procedure for preparation of compound 14.
A mixture of the acid hydrazide 2 (0.01 mole) and phenyl isothiocyanate (0.01 mole) in dry C6H6 was refluxed for 6 hours. The solid material obtained on cooling was filtered off and recrystallized from methanol to give compound 13. IR (υ, cm-1): 1680 (C=O), 1240 (C=S). A stirring mixture of compound 13 (1 mmole) and sodium hydroxide (40 mg, 1 mmole) was refluxed for 4 hours. After cooling, the solution was acidified with hydrochloric acid and the precipitate was filtered. The solid 5-(3-chloro-1-benzothien -2-yl-4-phenyl-4H-1,2,4-triazole-3-thiol (14) was recrystallized from ethyl acetate. Yield: 73%; mp 240-242°C; IR (υ, cm-1): 3320 (N-H thione), 3060 (C-Har), 1635 (C=N), 1530 (C=C), 1310 (C=S thione); UV (λmax): 313 nm; 1H-NMR (δ) ppm: 3.8 (s, 1H, N-H thione), 8.3 – 8.9 (m, 9H, Ar-H); Calcd. for C16H10N3S2Cl (%): C, 55.97; H, 2.91; N, 12.44; found (%): C, 55.59; H, 3.08; N, 12.23.