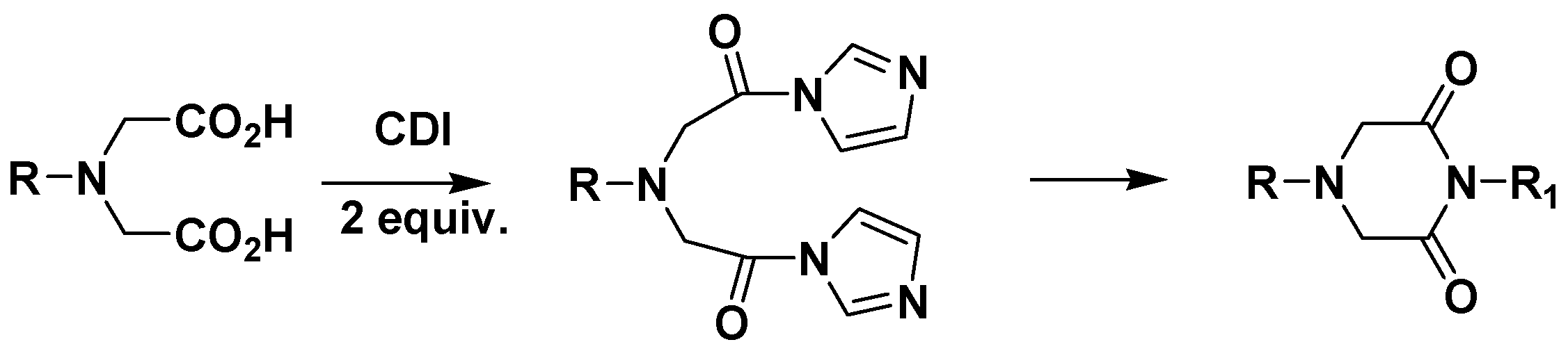
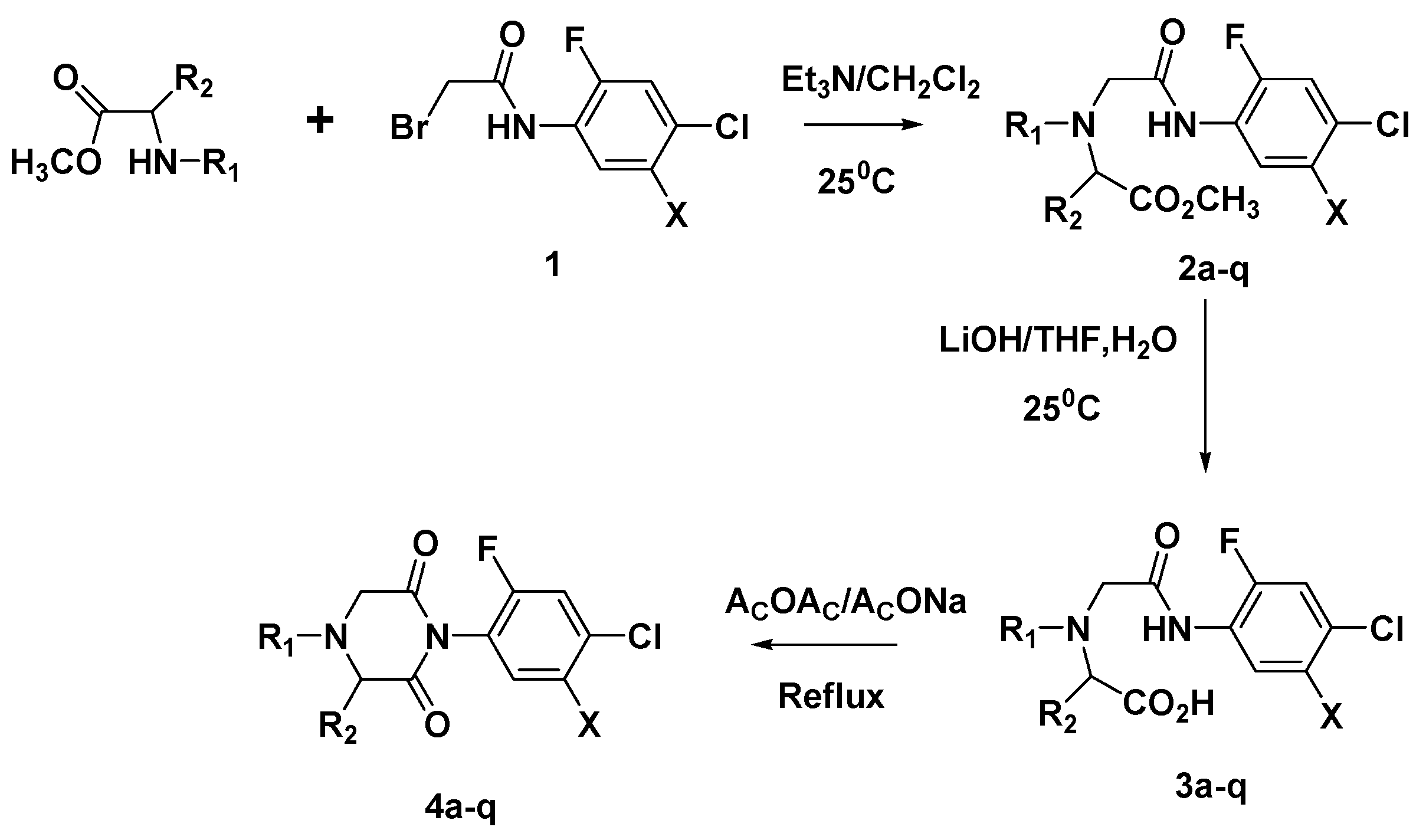
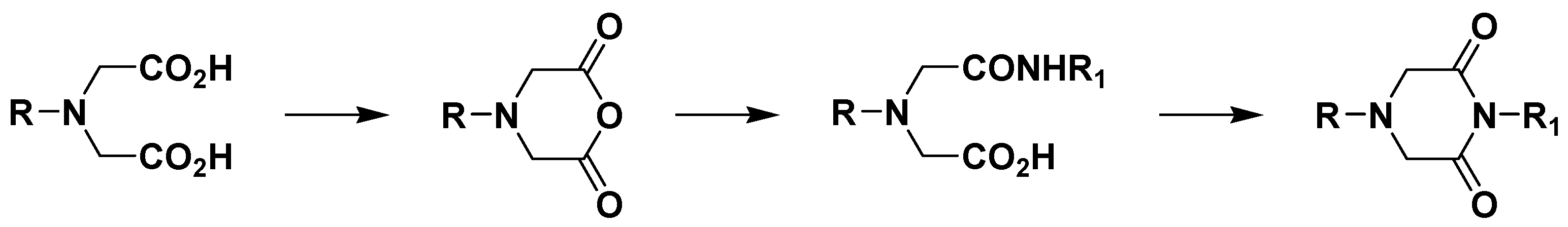General Method for the Preparation of 1-Phenyl-piperazine-2,6-diones 4a-q.
A solution of bromoacetyl bromide (11 mmol) in dichloromethane (10 mL) was added dropwise with stirring at room temperature to a solution of aniline (10 mmol) and triethylamine (11 mmol) in dichloromethane (20 mL). The reaction was monitored by TLC (ethyl acetate/hexane=1:1 to 1:5). The reaction came to completion usually in 1h and then ethyl acetate (200 mL) was added. The organic layer was washed with water (100 mL), saturated aqueous sodium bicarbonate (100 mL), brine (100 mL), dried over MgSO4 and concentrated under reduced pressure to afford the intermediate ester amide 1. The yields were from 92% to 98%. The purities ranged from 95% to 99%, as estimated by 1H-NMR.
To a solution of methyl N-substituted iminomonoacetate (6 mmol) and triethylamine (6 mmol) in dichloromethane (10 mL) the solution of 2-bromo-N-phenylacetamide (5 mmol) in dichloromethane (10 mL) was added dropwise with stirring at room temperature. The reaction was monitored by TLC (ethyl acetate/hexane=1:1 to 1:5). The reaction came to completion usually in 4h and then ethyl acetate (100 mL) was added. The organic layer was washed with water (60 mL), brine (60 mL), dried over MgSO4 and concentrated under reduced pressure to afford the intermediate ester 2. The yields were from 88% to 96%. The purity was from 90% to 99% as estimated by 1H-NMR.
To a mixture of the above ester 2 (4 mmol) in THF (10 mL) and water (10 mL) lithum hydroxide (6 mmol) was added with stirring at room temperature. The reaction was monitored by TLC (ethyl acetate/hexane=1:1 to 1:5). The reaction came to completion usually in 2h and then ethyl acetate (50 mL) and water (30 mL) were added. The organic layer was washed with water (20 mL). The aqueous layers were combined, acidified with 2N HCl to pH=3, and extracted with ethyl acetate (2 X 50 mL). The combined organic layer was washed with brine (60 mL), dried over MgSO4 and concentrated under reduced pressure to afford the intermediate acid 3. The yields were from 81% to 90%. The purity was from 95% to 99% as estimated by 1H-NMR.
The intermediate acid 3 (2 mmol) and catalytic amount of sodium acetate was added to acetic anhydride (10 mL) with stirring at room temperature. The reaction mixture was heated to reflux. The reaction was monitored by TLC (ethyl acetate/hexane=1:1 to 1:5). The reaction came to completion usually in 1h. After cooled down to room temperature, water (100 mL) and excess sodium carbonate was added to maintain the mixture basic (pH=7-9). The mixture was extracted with ethyl acetate (2 x 100 mL). The combined organic layer was washed with water (100 mL), brine (100 mL), dried over MgSO4 and concentrated under reduced pressure to afford the target compound 4. The yields ranged from 50% to 85%. The purity was from 85% to 95% as estimated by 1H-NMR.
2-(4-Chloro-2-fluorophenyl)-tetrahydro-2H-pyrido[1,2-a]pyrazine-1,3(4H,6H)-dione (4a): yield 80%; oil; 1H-NMR δ 1.238-1.409 (m, 1H), 1.608-1.778 (m, 4H), 2.192-2.306 (m, 2H), 2.963-3.017 (m, 2H), 3.346 (dd, J1 = 16.8 Hz, J2 = 12 Hz, 1H, COCH2N), 3.755 (dd, J1 = 16.8 Hz, J2 = 1.5 Hz, 1H, COCH2N), 7.083-7.254 (m, 3H); 1C-NMR δ 23.53 ; 25.15, 27.49, 55.10, 59.14, 64.02, 117.87, 120.88, 125.47, 131.63, 136.33, 158.05 (d, J = 253 Hz, CF), 169.18 (NCOCH2N), 171.46 (NCOCHN); MS m/z = 296 (M+, 5%), 268 (20%), 97 (100%).
2-(4,5-Dichloro-2-fluorophenyl)-tetrahydro-2H-pyrido[1,2-a]pyrazine-1,3(4H,6H)-dione (4b): yield 71%; oil; 1H-NMR δ 1.237-1.425 (m, 1H), 1.615-1.784 (m, 4H), 2.048-2.310 (m, 2H), 2.975-3.031 (m, 2H), 3.287 (dd, J1 = 16.8 Hz, J2 = 12.3 Hz, 1H, COCH2N), 3.433 (dd, J1 = 16.8 Hz, J2 = 3.6 Hz, 1H, COCH2N), 7.291-7.360 (m, 2H).
2-(4-Chloro-2-fluoro-5-methoxy phenyl)-tetrahydro-2H-pyrido[1,2-a]pyrazine-1,3(4H,6H)-dione (4c): yield 65%; semi-solid; 1H-NMR δ 1.308-1.479 (m, 1H), 1.529-1.892 (m, 4H), 2.212-2.401 (m, 2H), 2.974-3.125 (m, 2H), 3.355 (dd, J1 = 16.8 Hz, J2 = 1.2 Hz, 1H, COCH2N), 3.775 (dd, J1 = 16.8 Hz, J2 = 0.5 Hz, 1H, COCH2N), 6.750 (d, J=6Hz, 1H), 7.289 (d, 9Hz, 1H).
2-(4-Chloro-5-cyclopentyloxy-2-fluorophenyl)-tetrahydro-2H-pyrido[1,2-a]pyrazine-1,3(4H,6H)-dione (4d): yield 75%; oil; 1H-NMR δ 1.310-1.489 (m, 3H), 1.518-1.896 (m, 10H), 2.215-2.400 (m, 2H), 2.943-3.155 (m, 2H), 3.382 (dd, J1 = 16.8 Hz, J2 = 1 Hz, 1H, COCH2N), 3.795 (dd, J1 = 16.8 Hz, J2 = 0.3 Hz, 1H, COCH2N), 4.699 (m, 1H), 6.700 (d, J=6Hz, 1H), 7.254 (d, 9Hz, 1H).
2-(4-Chloro-5-cyclopentyloxy-2-fluorophenyl)-tetrahydropyrrolo[1,2-a]pyrazine-1,3(2H,4H)-dione (4e): yield 73%; oil; 1H-NMR δ 1.508-1.698 (m, 2H), 1.718-1.995 (m, 7H), 2.210-3.108 (m, 5H), 3.345-3.556 (m, 1H), 3.702 (dd, J1 = 17.4 Hz, J2 = 36.3 Hz, 1H, COCH2N), 4.000 (dd, J1 = 17.4 Hz, J2 = 0.9 Hz, 1H, COCH2N), 4.702 (m, 1H), 6.709 (d, J=6Hz, 1H), 7.249 (d, 9Hz, 1H).
1-(4-Chloro-5-ethoxycarbonylmethoxy-2-fluorophenyl)-4-methylpiperazine-2,6-dione (4f): yield 82%; m.p. 74-77°C; 1H-NMR δ 1.251 (t, J=7.2Hz, 3H), 2.493 (s, 3H), 3.514 (d, JAB=16.5Hz, 2H), 3.581 (d, JAB=16.5Hz, 2H), 4.244 (q, J=7.2Hz, 2H), 4.643 (s, 2H), 6.730 (d, J=6Hz, 1H), 7.296 (d, 9Hz, 1H).
1-(4-Chloro-5-propargyloxy-2-fluorophenyl)-4-methylpiperazine-2,6-dione (4g): yield 77%; semi-solid; 1H-NMR δ 2.504 (s, 3H), 2.567 (t, J=2.4Hz, 1H), 3.529 (d, JAB=16.5Hz, 2H), 3.621 (d, JAB=16.5Hz, 2H), 4.735 (d, J=2.4Hz, 1H), 6.900 (d, J=6.3Hz, 1H), 7.298 (d, J=9Hz, 1H)
1-(4-Chloro-5-cyclopentyloxy-2-fluorophenyl)-4-methylpiperazine-2,6-dione(4h): yield 55%; m.p. 111-113°C; 1H-NMR δ 1.520-1.662 (m, 2H), 1.790-1.920 (m, 6H), 2.497 (s, 3H), 3.519 (d, JAB=16.5Hz, 2H), 3.601 (d, JAB=16.5Hz, 2H), 4.665 (m, 1H), 6.690 (d, J=6.3Hz, 1H), 7.250 (d, J=9Hz, 1H).
4-tert-Butyl-1-(4-chloro-2-fluorophenyl)piperazine-2,6-dione (4i): yield 71%; m.p. 144-146°C; 1H-NMR δ 1.170 (s, 9H), 3.580 (d, JAB=16.5Hz, 2H), 3.750 (d, JAB=16.5Hz, 2H), 7.150-7.270 (m, 3H).
4-tert-Butyl-1-(4-chloro-5-methoxy-2-fluorophenyl)piperazine-2,6-dione (4j): yield 80%; semi-solid; 1H-NMR δ 1.174 (s, 9H), 3.525 (d, JAB=16.5Hz, 2H), 3.749 (d, JAB=16.5Hz, 2H), 3.866 (s, 3H), 6.730 (d, J=6.3Hz, 1H), 7.268 (d, J=9Hz, 1H).
4-tert-Butyl-1-(4-chloro-2-fluoro-5-propargyloxyphenyl)piperazine-2,6-dione (4k): yield 83%; semi-solid; 1H-NMR δ 1.174 (s, 9H), 2.598 (t, J=2.4Hz, 1H), 3.527 (d, JAB=16.5Hz, 2H), 3.750 (d, JAB=16.5Hz, 2H), 4.729 (d, J=2.4Hz, 1H), 6.910 (d, J=6.3Hz, 1H), 7.305 (d, J=9Hz, 1H).
4-tert-Butyl-1-(4-chloro-5-(cyclopentyloxy)-2-fluorophenyl)piperazine-2,6-dione (4l): yield 83%; m.p. 142-144°C; 1H-NMR δ 1.175 (s, 9H) 1.522-1.661 (m, 2H), 1.794-1.928 (m, 6H), 3.525 (d, JAB=16.5Hz, 2H), 3.752 (d, JAB=16.5Hz), 4.691 (m, 1H), 6.781 (d, J=6.3Hz, 1H), 7.247 (d, J=9Hz, 1H).
4-Benzyl-1-(4-chloro-2-fluorophenyl)piperazine-2,6-dione (4m): yield 85%; Oil; 1H-NMR δ 3.548 (d, JAB=16.5Hz, 2H), 3.660 (d, JAB=16.5Hz, 2H), 3.748 (s, 2H), 7.147-7.375 (m, 8H).
4-Benzyl-1-(4,5-dichloro-2-fluorophenyl)piperazine-2,6-dione (4n): yield 60%; semi-solid; 1H-NMR δ 3.584 (d, JAB=16.5Hz, 2H), 3.667 (d, JAB=16.5Hz, 2H), 3.769 (s, 2H), 7.361 (m, 7H).
4-Benzyl-1-(4-chloro-5-ethoxycarbonylmethoxy-2-fluorophenyl)piperazine-2,6-dione (4o): yield 83%; oil; 1H-NMR δ 1.260 (t, J=7.2Hz, 3H), 3.590 (d, JAB=16.5Hz, 2H), 3.655 (d, JAB=16.5Hz, 2H), 3.743 (s, 2H), 4.180 (q, J=7.2Hz, 2H), 4.644 (s, 2H), 6.720 (d, J=6.3Hz, 1H), 7.276-7.361 (m, 6H).
4-Benzyl-1-(4-chloro-2-fluoro-5-iso-propoxycarbonylphenyl)piperazine-2,6-dione (4p): yield 70%; semi-solid; 1H-NMR δ 1.370 (d, J=6.3Hz, 6H), 3.530 (d, JAB=16.5Hz, 2H), 3.651 (d, JAB=16.5Hz, 2H), 3.725 (s, 2H), 5.247 (sept, J=6.3Hz, 1H), 7.300-7.346 (m, 6H), 7.792 (d, J=6.3Hz, 1H).
4-Benzyl-1-(4-chloro-5-cyclopentyloxy-2-fluorophenyl)piperazine-2,6-dione (4q): yield 80%; m.p. 128-129°C; 1H-NMR δ 1.521-1.663 (m, 2H), 1.791-1.925 (m, 6H), 3.450 (d, JAB=16.5Hz, 2H), 3.651 (d, JAB=16.5Hz, 2H), 3.746 (s, 2H), 4.701 (m, 1H), 6.790 (d, J=6.3Hz, 1H), 7.245 (d, J=9Hz, 1H), 7.339-7.379 (m, 5H).