A General Synthesis of Tris-Indole Derivatives as Potential Iron Chelators
Abstract
:Introduction
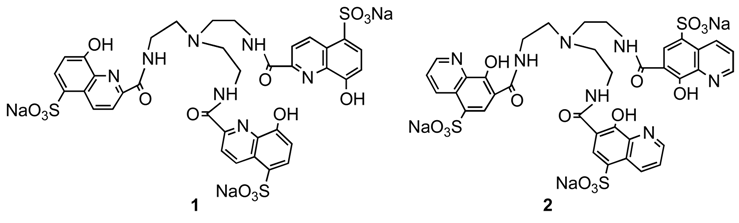
Results and Discussion
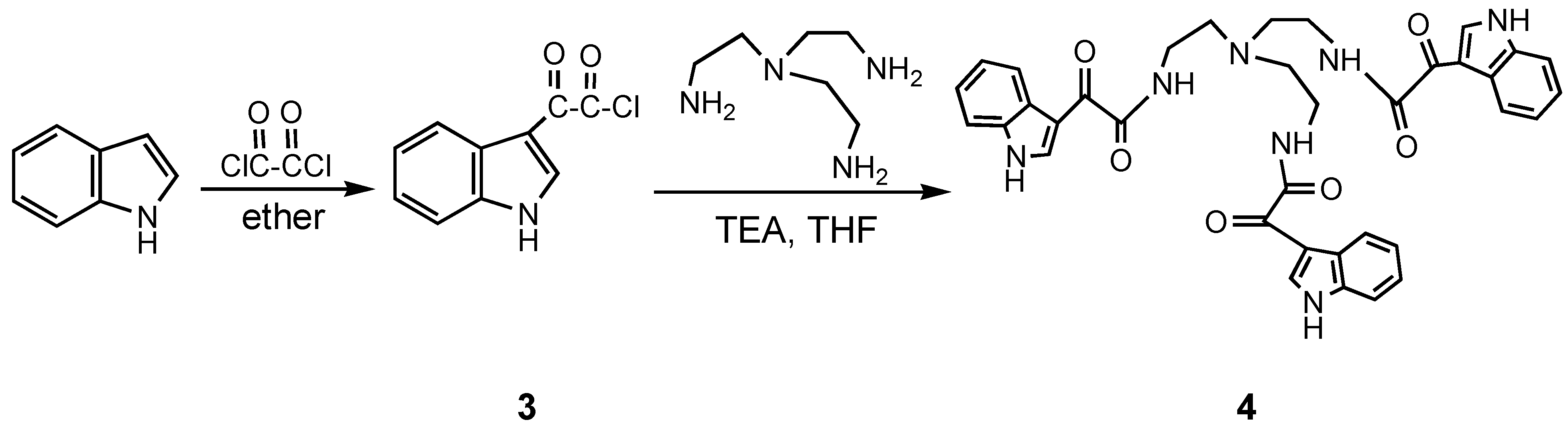
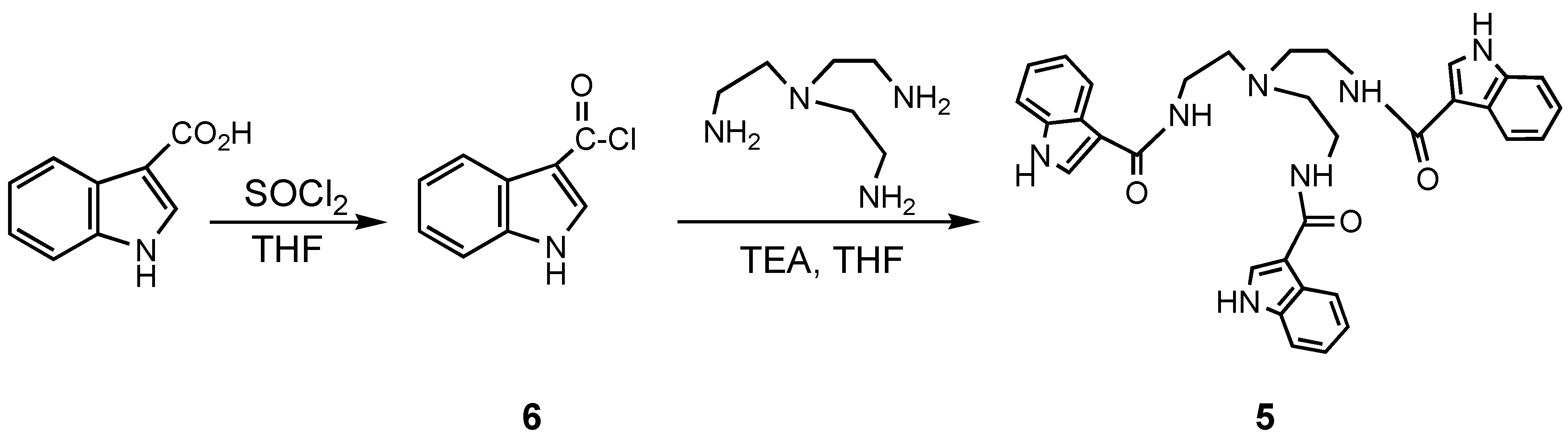
Conclusions
Experimental
General
References
- Shrader, W. D.; Celebuski, J.; Kline, S. J.; Johnson, D. Tetrahedron Lett. 1988, 29, 1351.
- Baret, P.; Béguin, C. G.; Boukhalfa, H.; Caris, C.; Laulhére, J.-P.; Pierre, J.-L.; Serratrice, G. J. Am. Chem. Soc. 1995, 117, 9760.
- a) Dobbin, P.S.; Hider, R. C. Chem. Br. 1990, 565.; b) Rakba, N.; Aouad, F.; Henry, C.; Caris, C.; Morel, I.; Baret, P.; Pierre, J.-L.; Brissot, R.; Ward, R. J.; Lescoat, G.; Chichton, R. R. Biochem. Pharmacol. 1998, 55, 1797..
- a) Sundberg, R. J. The Chemistry of Indoles; Academic: New York, 1996. [Google Scholar]; b) Sakagami, M.; Muratake, H.; Natsume, M. Chem. Pharm. Bull. 1994, 42, 1393.; c) Fukuyama, T.; Chen, X. J. Am. Chem. Soc. 1994, 116, 3125..
- Moore, R. E.; Cheuk, C.; Yang, X. Q.; Patterson, G. M. L.; Bonjouklian, R.; Smita, T. A.; Mynderse, J.; Foster, R. S.; Jones, N. D.; Skiartzendruber, J. K.; Deeter, J. B. J. Org. Chem. 1987, 52, 1036.
- a) Yang, C.; Patel, H. H.; Ku, Y.; Shah, R.; Sawick, D. Synth. Commun. 1997, 27, 2125.; b) Bergman, J.; Venemalm, L. Tetrahedron 1990, 46, 6061.; c) Ketcha, D. M.; Gribble, G. W. J. Org. Chem. 1985, 50, 5451.; d) Eyley, S. C.; Giles, R. G.; Heaney, H. Tetrahedron Lett. 1985, 26, 4649.; e) Keasling, H. H.; Willette, R. E.; Szmuszkovicz, J. J. Med. Chem. 1964, 7, 94..
- Carpenter, R. A.; Farley, A. R.; Cox, J. R.; Dobson, A. J.; Whitlock, C. R. J. Undergrad. Chem. Res. 2004, 1, 11.
- Sample availability: Not available.
© 2005 by MDPI (http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Sears, R.; Carpenter, R.; Whitlock, C. A General Synthesis of Tris-Indole Derivatives as Potential Iron Chelators. Molecules 2005, 10, 488-491. https://doi.org/10.3390/10020488
Sears R, Carpenter R, Whitlock C. A General Synthesis of Tris-Indole Derivatives as Potential Iron Chelators. Molecules. 2005; 10(2):488-491. https://doi.org/10.3390/10020488
Chicago/Turabian StyleSears, R., R. Carpenter, and C. Whitlock. 2005. "A General Synthesis of Tris-Indole Derivatives as Potential Iron Chelators" Molecules 10, no. 2: 488-491. https://doi.org/10.3390/10020488
APA StyleSears, R., Carpenter, R., & Whitlock, C. (2005). A General Synthesis of Tris-Indole Derivatives as Potential Iron Chelators. Molecules, 10(2), 488-491. https://doi.org/10.3390/10020488



