Human β-Defensin 2 and Its Postulated Role in Modulation of the Immune Response
Abstract
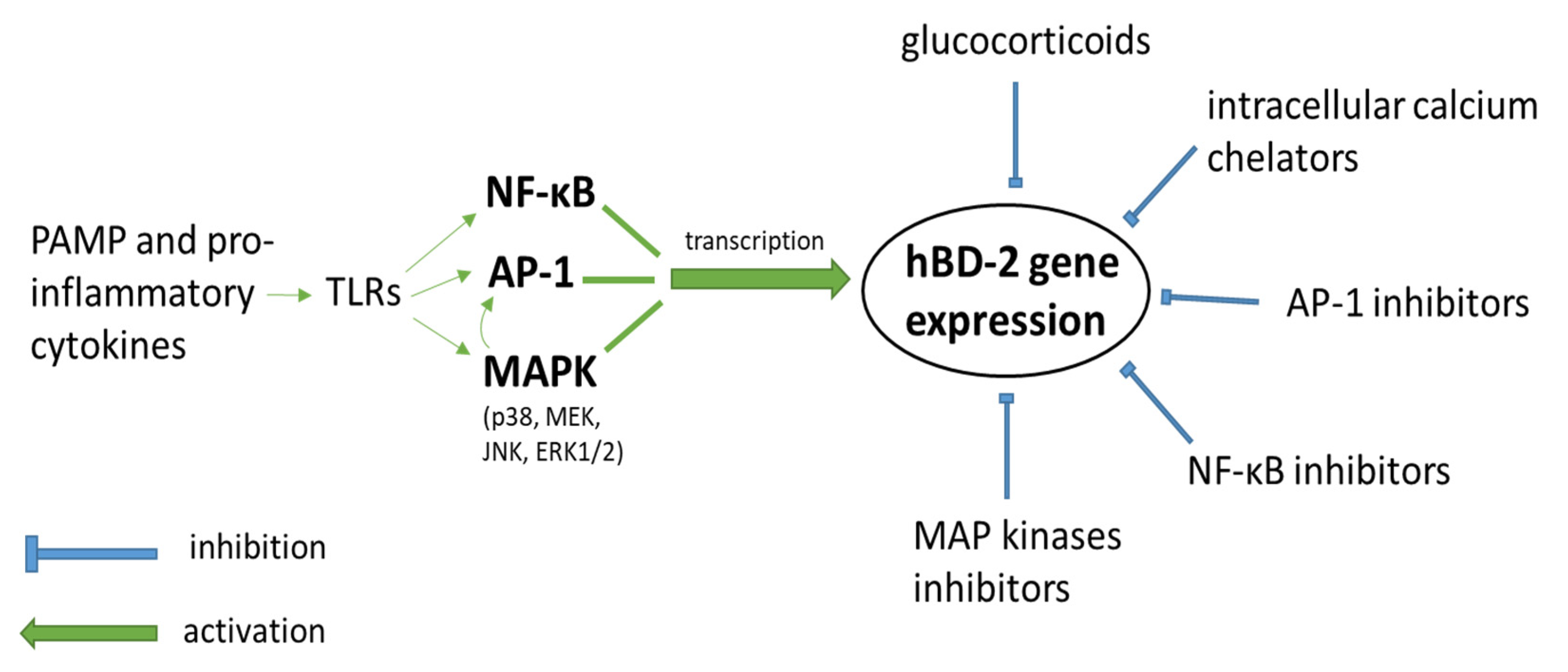
1. Introduction
2. hBD-2 and Oral Cavity Epithelium
3. hBD-2 and Gastric Mucosa
4. hBD-2 and Intestinal Epithelium
5. hBD-2 and Cells from Central Nervous System
6. hBD-2 and the Epithelium Lining the Respiratory System
7. hBD-2 in Skin and Wounds
8. hBD-2 in Pregnancy and Infancy
9. hBD-2 and Genitourinary Tract
10. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Deptuła, J.; Tokarz-Deptuła, B.; Deptuła, W. Defensins in humans and animals. Postepy Hig. Med. Dosw. 2019, 73, 152–158. [Google Scholar] [CrossRef]
- Xu, D.; Lu, W. Defensins: A Double-Edged Sword in Host Immunity. Front. Immunol. 2020, 11, 764. [Google Scholar] [CrossRef] [PubMed]
- Weinberg, A.; Jin, G.; Sieg, S.; McCormick, T.S. The Yin and Yang of human Beta-defensins in health and disease. Front. Immunol. 2012, 3, 294. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.; Chertov, O.; Bykovskaia, S.N.; Chen, Q.; Buffo, M.J.; Shogan, J.; Anderson, M.; Schröder, J.M.; Wang, J.M.; Howard, O.M.; et al. Beta-defensins: Linking innate and adaptive immunity through dendritic and T cell CCR6. Science 1999, 286, 525–528. [Google Scholar] [CrossRef] [PubMed]
- Harder, J.; Bartels, J.; Christophers, E.; Schröder, J.M. A peptide antibiotic from human skin. Nature 1997, 387, 861. [Google Scholar] [CrossRef] [PubMed]
- Schröder, J.M.; Harder, J. Human beta-defensin-2. Int. J. Biochem. Cell Biol. 1999, 31, 645–651. [Google Scholar] [CrossRef]
- De Paula, V.S.; Valente, A.P. A Dynamic Overview of Antimicrobial Peptides and Their Complexes. Molecules 2018, 23, 2040. [Google Scholar] [CrossRef]
- Taylor, K.; Barran, P.E.; Dorin, J.R. Structure-activity relationships in beta-defensin peptides. Biopolymers 2008, 90, 1–7. [Google Scholar] [CrossRef]
- Huang, L.C.; Redfern, R.L.; Narayanan, S.; Reins, R.Y.; McDermott, A.M. In vitro activity of human beta-defensin 2 against Pseudomonas aeruginosa in the presence of tear fluid. Antimicrob. Agents Chemother. 2007, 51, 3853–3860. [Google Scholar] [CrossRef]
- Tomita, T.; Nagase, T.; Ohga, E.; Yamaguchi, Y.; Yoshizumi, M.; Ouchi, Y. Molecular mechanisms underlying human beta-defensin-2 gene expression in a human airway cell line (LC2/ad). Respirology 2002, 7, 305–310. [Google Scholar] [CrossRef]
- Steubesand, N.; Kiehne, K.; Brunke, G.; Pahl, R.; Reiss, K.; Herzig, K.H.; Schubert, S.; Schreiber, S.; Fölsch, U.R.; Rosenstiel, P.; et al. The expression of the beta-defensins hBD-2 and hBD-3 is differentially regulated by NF-kappaB and MAPK/AP-1 pathways in an in vitro model of Candida esophagitis. BMC Immunol. 2009, 10, 36. [Google Scholar] [CrossRef] [PubMed]
- Wehkamp, K.; Schwichtenberg, L.; Schröder, J.M.; Harder, J. Pseudomonas aeruginosa- and IL-1beta-mediated induction of human beta-defensin-2 in keratinocytes is controlled by NF-kappaB and AP-1. J. Investig. Dermatol. 2006, 126, 121–127. [Google Scholar] [CrossRef]
- Yoon, Y.M.; Lee, J.Y.; Yoo, D.; Sim, Y.S.; Kim, Y.J.; Oh, Y.K.; Kang, J.S.; Kim, S.; Kim, J.S.; Kim, J.M. Bacteroides fragilis enterotoxin induces human beta-defensin-2 expression in intestinal epithelial cells via a mitogen-activated protein kinase/I kappaB kinase/NF-kappaB-dependent pathway. Infect. Immun. 2010, 78, 2024–2033. [Google Scholar] [CrossRef]
- Hertz, C.J.; Wu, Q.; Porter, E.M.; Zhang, Y.J.; Weismüller, K.H.; Godowski, P.J.; Ganz, T.; Randell, S.H.; Modlin, R.L. Activation of Toll-like receptor 2 on human tracheobronchial epithelial cells induces the antimicrobial peptide human beta defensin-2. J. Immunol. 2003, 171, 6820–6826. [Google Scholar] [CrossRef]
- Vora, P.; Youdim, A.; Thomas, L.S.; Fukata, M.; Tesfay, S.Y.; Lukasek, K.; Michelsen, K.S.; Wada, A.; Hirayama, T.; Arditi, M.; et al. Beta-defensin-2 expression is regulated by TLR signaling in intestinal epithelial cells. J. Immunol. 2004, 173, 5398–5405. [Google Scholar] [CrossRef] [PubMed]
- Seo, E.S.; Blaum, B.S.; Vargues, T.; De Cecco, M.; Deakin, J.A.; Lyon, M.; Barran, P.E.; Campopiano, D.J.; Uhrín, D. Interaction of human β-defensin 2 (HBD2) with glycosaminoglycans. Biochemistry 2010, 49, 10486–10495. [Google Scholar] [CrossRef]
- Bhat, S.; Song, Y.H.; Lawyer, C.; Milner, S.M. Modulation of the complement system by human beta-defensin 2. J. Burns Wounds 2007, 5, e10. [Google Scholar] [PubMed]
- Mathews, M.; Jia, H.P.; Guthmiller, J.M.; Losh, G.; Graham, S.; Johnson, G.K.; Tack, B.F.; McCray, P.B., Jr. Production of beta-defensin antimicrobial peptides by the oral mucosa and salivary glands. Infect. Immun. 1999, 67, 2740–2745. [Google Scholar] [CrossRef]
- Gürsoy, U.K.; Salli, K.; Söderling, E.; Gürsoy, M.; Hirvonen, J.; Ouwehand, A.C. Regulation of hBD-2, hBD-3, hCAP18/LL37, and Proinflammatory Cytokine Secretion by Human Milk Oligosaccharides in an Organotypic Oral Mucosal Model. Pathogens 2021, 10, 739. [Google Scholar] [CrossRef]
- Kozubska, A.; Grzegorczyk, J.; Konieczka, M.; Szczepańska, J. Analysis of the level of non-specific and specific immunity parameters in saliva of children with osteogenesis imperfecta and study of relationships between selected proteins, disease symptoms and sociodemographic factors. New. Med. 2020, 24, 67–75. [Google Scholar]
- Dale, B.A.; Fredericks, L.P. Antimicrobial peptides in the oral environment: Expression and function in health and disease. Curr. Issues Mol. Biol. 2005, 7, 119–133. [Google Scholar]
- Joly, S.; Organ, C.C.; Johnson, G.K.; McCray, P.B., Jr.; Guthmiller, J.M. Correlation between beta-defensin expression and induction profiles in gingival keratinocytes. Mol. Immunol. 2005, 42, 1073–1084. [Google Scholar] [CrossRef]
- Mahanonda, R.; Sa-Ard-Iam, N.; Eksomtramate, M.; Rerkyen, P.; Phairat, B.; Schaecher, K.E.; Fukuda, M.M.; Pichyangkul, S. Cigarette smoke extract modulates human beta-defensin-2 and interleukin-8 expression in human gingival epithelial cells. J. Periodontal Res. 2009, 44, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Khurshid, Z.; Naseem, M.; Sheikh, Z.; Najeeb, S.; Shahab, S.; Zafar, M.S. Oral antimicrobial peptides: Types and role in the oral cavity. Saudi Pharm. J. 2016, 24, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Krisanaprakornkit, S.; Kimball, J.R.; Weinberg, A.; Darveau, R.P.; Bainbridge, B.W.; Dale, B.A. Inducible expression of human beta-defensin 2 by Fusobacterium nucleatum in oral epithelial cells: Multiple signaling pathways and role of commensal bacteria in innate immunity and the epithelial barrier. Infect. Immun. 2000, 68, 2907–2915. [Google Scholar] [CrossRef]
- Krisanaprakornkit, S.; Kimball, J.R.; Dale, B.A. Regulation of human beta-defensin-2 in gingival epithelial cells: The involvement of mitogen-activated protein kinase pathways, but not the NF-kappaB transcription factor family. J. Immunol. 2002, 168, 316–324. [Google Scholar] [CrossRef]
- Ouhara, K.; Komatsuzawa, H.; Shiba, H.; Uchida, Y.; Kawai, T.; Sayama, K.; Hashimoto, K.; Taubman, M.A.; Kurihara, H.; Sugai, M. Actinobacillus actinomycetemcomitans outer membrane protein 100 triggers innate immunity and production of beta-defensin and the 18-kilodalton cationic antimicrobial protein through the fibronectin-integrin pathway in human gingival epithelial cells. Infect. Immun. 2006, 74, 5211–5220. [Google Scholar] [CrossRef]
- Krisanaprakornkit, S.; Jotikasthira, D.; Dale, B.A. Intracellular calcium in signaling human beta-defensin-2 expression in oral epithelial cells. J. Dent. Res. 2003, 82, 877–882. [Google Scholar] [CrossRef] [PubMed]
- Wassing, G.M.; Ilehag, N.; Frey, J.; Jonsson, A.B. Modulation of Human Beta-Defensin 2 Expression by Pathogenic Neisseria meningitidis and Commensal Lactobacilli. Antimicrob. Agents Chemother. 2021, 65, e02002–e02020. [Google Scholar] [CrossRef] [PubMed]
- Wassing, G.M.; Lidberg, K.; Sigurlásdóttir, S.; Frey, J.; Schroeder, K.; Ilehag, N.; Lindås, A.C.; Jonas, K.; Jonsson, A.B. DNA Blocks the Lethal Effect of Human Beta-Defensin 2 Against Neisseria meningitidis. Front. Microbiol. 2021, 12, 697232. [Google Scholar] [CrossRef]
- Casaroto, A.R.; da Silva, R.A.; Salmeron, S.; Rezende, M.; Dionísio, T.J.; Santos, C.; Pinke, K.H.; Klingbeil, M.; Salomão, P.A.; Lopes, M.; et al. Candida albicans-Cell Interactions Activate Innate Immune Defense in Human Palate Epithelial Primary Cells via Nitric Oxide (NO) and β-Defensin 2 (hBD-2). Cells 2019, 8, 707. [Google Scholar] [CrossRef] [PubMed]
- Salem, A.; Almahmoudi, R.; Hagström, J.; Stark, H.; Nordström, D.; Salo, T.; Eklund, K.K. Human β-Defensin 2 Expression in Oral Epithelium: Potential Therapeutic Targets in Oral Lichen Planus. Int. J. Mol. Sci. 2019, 20, 1780. [Google Scholar] [CrossRef]
- Tang, E.; Khan, I.; Andreana, S.; Arany, P.R. Laser-activated transforming growth factor-β1 induces human β-defensin 2: Implications for laser therapies for periodontitis and peri-implantitis. J. Periodontal Res. 2017, 52, 360–367. [Google Scholar] [CrossRef]
- Ruggiero, P. Helicobacter pylori and inflammation. Curr. Pharm. Des. 2010, 16, 4225–4236. [Google Scholar] [CrossRef]
- Lamb, A.; Chen, L.F. Role of the Helicobacter pylori-induced inflammatory response in the development of gastric cancer. J. Cell. Biochem. 2013, 114, 491–497. [Google Scholar] [CrossRef]
- Testerman, T.L.; Morris, J. Beyond the stomach: An updated view of Helicobacter pylori pathogenesis, diagnosis, and treatment. World J. Gastroenterol. 2014, 20, 12781–12808. [Google Scholar] [CrossRef] [PubMed]
- Cadamuro, A.C.; Rossi, A.F.; Maniezzo, N.M.; Silva, A.E. Helicobacter pylori infection: Host immune response, implications on gene expression and microRNAs. World J. Gastroenterol. 2014, 20, 1424–1437. [Google Scholar] [CrossRef]
- Kalisperati, P.; Spanou, E.; Pateras, I.S.; Korkolopoulou, P.; Varvarigou, A.; Karavokyros, I.; Gorgoulis, V.G.; Vlachoyiannopoulos, P.; Sougioultzis, S. Inflammation, DNA Damage, Helicobacter pylori and Gastric Tumorigenesis. Front. Genet. 2017, 8, 20. [Google Scholar] [CrossRef] [PubMed]
- Cunliffe, R.N.; Mahida, Y.R. Expression and regulation of antimicrobial peptides in the gastrointestinal tract. J. Leukoc. Biol. 2004, 75, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Jäger, S.; Stange, E.F.; Wehkamp, J. Antimicrobial peptides in gastrointestinal inflammation. J. Inflamm. Res. 2010, 2010, 910283. [Google Scholar] [CrossRef] [PubMed]
- Bajaj-Elliott, M.; Fedeli, P.; Smith, G.V.; Domizio, P.; Maher, L.; Ali, R.S.; Quinn, A.G.; Farthing, M.J. Modulation of host antimicrobial peptide (β-defensins 1 and 2) expression during gastritis. Gut 2002, 51, 356–361. [Google Scholar] [CrossRef]
- Hamanaka, Y.; Nakashima, M.; Wada, A.; Ito, M.; Kurazono, H.; Hojo, H.; Nakahara, Y.; Kohno, S.; Hirayama, T.; Sekine, I. Expression of human beta-defensin 2 (hBD-2) in Helicobacter pylori induced gastritis: Antibacterial effect of hBD-2 against Helicobacter pylori. Gut 2001, 49, 481–487. [Google Scholar] [CrossRef]
- Isomoto, H.; Mukae, H.; Ishimoto, H.; Nishi, Y.; Wen, C.Y.; Wada, A.; Ohnita, K.; Hirayama, T.; Nakazato, M.; Kohno, S. High concentrations of human β-defensin 2 in gastric juice of patients with Helicobacter pylori infection. World J. Gastroenterol. 2005, 11, 4782–4787. [Google Scholar] [CrossRef] [PubMed]
- Bauer, B.; Wex, T.; Kuester, D.; Meyer, T.; Malfertheiner, P. Differential expression of human beta defensin 2 and 3 in gastric mucosa of Helicobacter pylori-infected individuals. Helicobacter 2013, 18, 6–12. [Google Scholar] [CrossRef]
- Nuding, S.; Gersemann, M.; Hosaka, Y.; Konietzny, S.; Schaefer, C.; Beisner, J.; Schroeder, B.O.; Ostaff, M.J.; Saigenji, K.; Ott, G.; et al. Gastric antimicrobial peptides fail to eradicate Helicobacter pylori infection due to selective induction and resistance. PLoS ONE 2013, 8, e73867. [Google Scholar] [CrossRef]
- Karkhah, A.; Ebrahimpour, S.; Rostamtabar, M.; Koppolu, V.; Darvish, S.; Vasigala, V.K.R.; Validi, M.; Nouri, H.R. Helicobacter pylori evasion strategies of the host innate and adaptive immune responses to survive and develop gastrointestinal diseases. Microbiol. Res. 2019, 218, 49–57. [Google Scholar] [CrossRef]
- Cullen, T.W.; Giles, D.K.; Wolf, L.N.; Ecobichon, C.; Boneca, I.G.; Trent, M.S. Helicobacter pylori versus the host: Remodeling of the bacterial outer membrane is required for survival in the gastric mucosa. PLoS Pathog. 2011, 7, e1002454. [Google Scholar] [CrossRef]
- Ohara, T.; Morishita, T.; Suzuki, H.; Masaoka, T.; Nishizawa, T.; Hibi, T. Investigation of the possibility of human-beta defensin 2 (hBD2) as a molecular marker of gastric mucosal inflammation. Hepato-gastroenterology 2005, 52, 1320–1324. [Google Scholar] [PubMed]
- Wehkamp, J.; Fellermann, K.; Herrlinger, K.; Bevins, C.L.; Stange, E.F. Mechanisms of Disease: Defensins in gastrointestinal diseases. Nat. Rev. Gastroenterol. Hepatol. 2005, 2, 406–415. [Google Scholar] [CrossRef]
- Kim, J.M. Antimicrobial proteins in intestine and inflammatory bowel diseases. Intest. Res. 2014, 12, 20–33. [Google Scholar] [CrossRef] [PubMed]
- Cobo, E.R.; Chadee, K. Antimicrobial Human β-Defensins in the Colon and Their Role in Infectious and Non-Infectious Diseases. Pathogens 2013, 2, 177–192. [Google Scholar] [CrossRef]
- Fusco, A.; Savio, V.; Donniacuo, M.; Perfetto, B.; Donnarumma, G. Antimicrobial Peptides Human Beta-Defensin-2 and -3 Protect the Gut During Candida albicans Infections Enhancing the Intestinal Barrier Integrity: In Vitro Study. Front. Cell. Infect. Microbiol. 2021, 11, 666900. [Google Scholar] [CrossRef] [PubMed]
- O’Neil, D.A.; Porter, E.M.; Elewaut, D.; Anderson, G.M.; Eckmann, L.; Ganz, T.; Kagnoff, M.F. Expression and regulation of the human beta-defensins hBD-1 and hBD-2 in intestinal epithelium. J. Immunol. 1999, 163, 6718–6724. [Google Scholar] [PubMed]
- Konno, Y.; Ashida, T.; Inaba, Y.; Ito, T.; Tanabe, H.; Maemoto, A.; Ayabe, T.; Mizukami, Y.; Fujiya, M.; Kohgo, Y. Isoleucine, an Essential Amino Acid, Induces the Expression of Human β Defensin 2 through the Activation of the G-Protein Coupled Receptor-ERK Pathway in the Intestinal Epithelia. Food Sci. Nutr. 2012, 3, 548–555. [Google Scholar] [CrossRef]
- Fusco, A.; Savio, V.; Cammarota, M.; Alfano, A.; Schiraldi, C.; Donnarumma, G. Beta-Defensin-2 and Beta-Defensin-3 Reduce Intestinal Damage Caused by Salmonella typhimurium Modulating the Expression of Cytokines and Enhancing the Probiotic Activity of Enterococcus faecium. J. Immunol. Res. 2017, 2017, 6976935. [Google Scholar] [CrossRef] [PubMed]
- Huang, F.C.; Lu, Y.T.; Liao, Y.H. Beneficial effect of probiotics on Pseudomonas aeruginosa-infected intestinal epithelial cells through inflammatory IL-8 and antimicrobial peptide human beta-defensin-2 modulation. Innate Immun. 2020, 26, 592–600. [Google Scholar] [CrossRef]
- Huang, F.C. Differential regulation of interleukin-8 and human beta-defensin 2 in Pseudomonas aeruginosa-infected intestinal epithelial cells. BMC Microbiol. 2014, 14, 275. [Google Scholar] [CrossRef] [PubMed]
- Zaalouk, T.K.; Bajaj-Elliott, M.; George, J.T.; McDonald, V. Differential regulation of beta-defensin gene expression during Cryptosporidium parvum infection. Infect. Immun. 2004, 72, 2772–2779. [Google Scholar] [CrossRef]
- Carryn, S.; Schaefer, D.A.; Imboden, M.; Homan, E.J.; Bremel, R.D.; Riggs, M.W. Phospholipases and cationic peptides inhibit Cryptosporidium parvum sporozoite infectivity by parasiticidal and non-parasiticidal mechanisms. J. Parasitol. 2012, 98, 199–204. [Google Scholar] [CrossRef]
- Ayala-Sumuano, J.T.; Téllez-López, V.M.; Domínguez-Robles, M.D.C.; Shibayama-Salas, M.; Meza, I. Toll-like Receptor Signaling Activation by Entamoeba histolytica Induces Beta Defensin 2 in Human Colonic Epithelial Cells: Its Possible Role as an Element of the Innate Immune Response. PLoS Negl. Trop. Dis. 2013, 7, e2083. [Google Scholar] [CrossRef]
- Ramasundara, M.; Leach, S.T.; Lemberg, D.A.; Day, A.S. Defensins and inflammation: The role of defensins in inflammatory bowel disease. J. Gastroenterol. Hepatol. 2009, 24, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Wehkamp, J.; Fellermann, K.; Herrlinger, K.R.; Baxmann, S.; Schmidt, K.; Schwind, B.; Duchrow, M.; Wohlschläger, C.; Feller, A.C.; Stange, E.F. Human beta-defensin 2 but not beta-defensin 1 is expressed preferentially in colonic mucosa of inflammatory bowel disease. Eur. J. Gastroenterol. Hepatol. 2002, 14, 745–752. [Google Scholar] [CrossRef]
- Wehkamp, J.; Harder, J.; Weichenthal, M.; Mueller, O.; Herrlinger, K.R.; Fellermann, K.; Schroeder, J.M.; Stange, E.F. Inducible and constitutive beta-defensins are differentially expressed in Crohn’s disease and ulcerative colitis. Inflamm. Bowel Dis. 2003, 9, 215–223. [Google Scholar] [CrossRef]
- Aldhous, M.C.; Noble, C.L.; Satsangi, J. Dysregulation of human beta-defensin-2 protein in inflammatory bowel disease. PLoS ONE 2009, 4, e6285. [Google Scholar] [CrossRef] [PubMed]
- Rahman, A.; Fahlgren, A.; Sitohy, B.; Baranov, V.; Zirakzadeh, A.; Hammarström, S.; Danielsson, A.; Hammarström, M.L. Beta-defensin production by human colonic plasma cells: A new look at plasma cells in ulcerative colitis. Inflamm. Bowel Dis. 2007, 13, 847–855. [Google Scholar] [CrossRef] [PubMed]
- Meisch, J.P.; Nishimura, M.; Vogel, R.M.; Sung, H.C.; Bednarchik, B.A.; Ghosh, S.K.; Fu, P.; McCormick, T.; Weinberg, A.; Levine, A.D. Human β-defensin 3 peptide is increased and redistributed in Crohn’s ileitis. Inflamm. Bowel Dis. 2013, 19, 942–953. [Google Scholar] [CrossRef]
- Langhorst, J.; Junge, A.; Rueffer, A.; Wehkamp, J.; Foell, D.; Michalsen, A.; Musial, F.; Dobos, G.J. Elevated human beta-defensin-2 levels indicate an activation of the innate immune system in patients with irritable bowel syndrome. Am. J. Gastroenterol. 2009, 104, 404–410. [Google Scholar] [CrossRef]
- Koeninger, L.; Armbruster, N.S.; Brinch, K.S.; Kjaerulf, S.; Andersen, B.; Langnau, C.; Autenrieth, S.E.; Schneidawind, D.; Stange, E.F.; Malek, N.P.; et al. Human β-Defensin 2 Mediated Immune Modulation as Treatment for Experimental Colitis. Front. Immunol. 2020, 11, 93. [Google Scholar] [CrossRef]
- Mailänder-Sánchez, D.; Kjaerulf, S.; Sidelmann Brinch, K.; Andersen, B.; Stange, E.F.; Malek, N.; Nordkild, P.; Wehkamp, J. DOP083 Recombinant subcutaneous human beta-Defensin 2 (hBD2) ameliorates experimental colitis in different in vivo models. J. Crohn’s Colitis 2017, 11, S75–S76. [Google Scholar] [CrossRef][Green Version]
- Koeninger, L.; Armbruster, N.S.; Hu, Z.; Jensen, B.; Stange, E.F.; Nordkild, P.; Malek, N.P.; Zender, L.; Wehkamp, J. P851 Oral delivery of Human β-defensin 2 is reversibly increasing microbiome diversity and is effective in the treatment of experimental colitis. J. Crohn’s Colitis 2018, 12, S547. [Google Scholar] [CrossRef]
- Tamboli, C.P.; Neut, C.; Desreumaux, P.; Colombel, J.F. Dysbiosis in inflammatory bowel disease. Gut 2004, 53, 1–4. [Google Scholar] [CrossRef]
- Mirsepasi-Lauridsen, H.C.; Vrankx, K.; Engberg, J.; Friis-Møller, A.; Brynskov, J.; Nordgaard-Lassen, I.; Petersen, A.M.; Krogfelt, K.A. Disease-Specific Enteric Microbiome Dysbiosis in Inflammatory Bowel Disease. Front. Med. 2018, 5, 304. [Google Scholar] [CrossRef]
- Nishida, A.; Inoue, R.; Inatomi, O.; Bamba, S.; Naito, Y.; Andoh, A. Gut microbiota in the pathogenesis of inflammatory bowel disease. Clin. J. Gastroenterol. 2018, 11, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Borysowski, J.; Międzybrodzki, R.; Przybylski, M.; Owczarek, B.; Weber-Dąbrowska, B.; Górski, A. The effects of T4 and A5/80 phages on the expression of immunologically important genes in differentiated Caco-2 cells. Postepy Hig. Med. Dosw. 2020, 74, 371–376. [Google Scholar] [CrossRef]
- Aarli, J.A. The immune system and the nervous system. J. Neurol. 1983, 229, 137–154. [Google Scholar] [CrossRef]
- Steinman, L. Elaborate interactions between the immune and nervous systems. Nat. Immunol. 2004, 5, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Kioussis, D.; Pachnis, V. Immune and nervous systems: More than just a superficial similarity? Immunity 2009, 31, 705–710. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, K.; Nakajima, K. Role of the Immune System in the Development of the Central Nervous System. Front. Neurosci. 2019, 13, 916. [Google Scholar] [CrossRef] [PubMed]
- Hao, H.N.; Zhao, J.; Lotoczky, G.; Grever, W.E.; Lyman, W.D. Induction of human beta-defensin-2 expression in human astrocytes by lipopolysaccharide and cytokines. J. Neurochem. 2001, 77, 1027–1035. [Google Scholar] [CrossRef]
- Tiszlavicz, Z.; Endrész, V.; Németh, B.; Megyeri, K.; Orosz, L.; Seprényi, G.; Mándi, Y. Inducible expression of human β-defensin 2 by Chlamydophila pneumoniae in brain capillary endothelial cells. Innate Immun. 2011, 17, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Williams, W.M.; Castellani, R.J.; Weinberg, A.; Perry, G.; Smith, M.A. Do β-defensins and other antimicrobial peptides play a role in neuroimmune function and neurodegeneration? Sci. World J. 2012, 2012, 905785. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bals, R.; Wang, X.; Wu, Z.; Freeman, T.; Bafna, V.; Zasloff, M.; Wilson, J.M. Human beta-defensin 2 is a salt-sensitive peptide antibiotic expressed in human lung. J. Clin. Investig. 1998, 102, 874–880. [Google Scholar] [CrossRef] [PubMed]
- Olvera, D.P.R.; Gutiérrez, C.C. Multifunctional Activity of the β-Defensin-2 during Respiratory Infections. In Immune Response Activation and Immunomodulation; Tyagi, R.K., Bisen, P.S., Eds.; IntechOpen: London, UK, 2018; Available online: https://www.intechopen.com/chapters/63967 (accessed on 14 September 2021).
- Borchers, N.S.; Santos-Valente, E.; Toncheva, A.A.; Wehkamp, J.; Franke, A.; Gaertner, V.D.; Nordkild, P.; Genuneit, J.; Jensen, B.; Kabesch, M. Human β-Defensin 2 Mutations Are Associated With Asthma and Atopy in Children and Its Application Prevents Atopic Asthma in a Mouse Model. Front. Immunol. 2021, 12, 636061. [Google Scholar] [CrossRef]
- Ora, J.; Calzetta, L.; Matera, M.G.; Cazzola, M.; Rogliani, P. Advances with glucocorticoids in the treatment of asthma: State of the art. Expert Opin. Pharmacother. 2020, 21, 2305–2316. [Google Scholar] [CrossRef]
- Witthöft, T.; Pilz, C.S.; Fellermann, K.; Nitschke, M.; Stange, E.F.; Ludwig, D. Enhanced Human β-Defensin-2 (hBD-2) Expression by Corticosteroids Is Independent of NF-κB in Colonic Epithelial Cells. Dig. Dis. Sci. 2005, 50, 1252–1259. [Google Scholar] [CrossRef] [PubMed]
- Yanagi, S.; Ashitani, J.; Imai, K.; Kyoraku, Y.; Sano, A.; Matsumoto, N.; Nakazato, M. Significance of human beta-defensins in the epithelial lining fluid of patients with chronic lower respiratory tract infections. Clin. Microbiol. Infect. 2007, 13, 63–69. [Google Scholar] [CrossRef]
- Liu, S.; He, L.R.; Wang, W.; Wang, G.H.; He, Z.Y. Prognostic value of plasma human β-defensin 2 level on short-term clinical outcomes in patients with community-acquired pneumonia: A preliminary study. Respir. Care 2013, 58, 655–661. [Google Scholar] [CrossRef]
- MacRedmond, R.; Greene, C.; Taggart, C.C.; McElvaney, N.; O’Neill, S. Respiratory epithelial cells require Toll-like receptor 4 for induction of Human β-defensin 2 by Lipopolysaccharide. Respir. Res. 2005, 6, 116. [Google Scholar] [CrossRef]
- Alekseeva, L.; Huet, D.; Féménia, F.; Mouyna, I.; Abdelouahab, M.; Cagna, A.; Guerrier, D.; Tichanné-Seltzer, V.; Baeza-Squiban, A.; Chermette, R.; et al. Inducible expression of beta defensins by human respiratory epithelial cells exposed to Aspergillus fumigatus organisms. BMC Microbiol. 2009, 9, 33. [Google Scholar] [CrossRef] [PubMed]
- Hielpos, M.S.; Ferrero, M.C.; Fernández, A.G.; Bonetto, J.; Giambartolomei, G.H.; Fossati, C.A.; Baldi, P.C. CCL20 and Beta-Defensin 2 Production by Human Lung Epithelial Cells and Macrophages in Response to Brucella abortus Infection. PLoS ONE 2015, 10, e0140408. [Google Scholar] [CrossRef]
- Chen, L.; Sun, B.B.; Wang, T.; Wang, X.; Li, J.Q.; Wang, H.X.; Zhang, S.F.; Liu, D.S.; Liu, L.; Xu, D.; et al. Cigarette smoke enhances β-defensin 2 expression in rat airways via nuclear factor-κB activation. Eur. Respir. J. 2010, 36, 638–645. [Google Scholar] [CrossRef]
- Park, M.S.; Kim, J.I.; Lee, I.; Park, S.; Bae, J.Y.; Park, M.S. Towards the Application of Human Defensins as Antivirals. Biomol. Ther. (Seoul) 2018, 26, 242–254. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Ghosh, S.K.; Basavarajappa, S.C.; Muller-Greven, J.; Penfield, J.; Brewer, A.; Ramakrishnan, P.; Buck, M.; Weinberg, A. Molecular dynamics simulations and functional studies reveal that hBD-2 binds SARS-CoV-2 spike RBD and blocks viral entry into ACE2 expressing cells. bioRxiv 2021. [Google Scholar] [CrossRef]
- Xu, C.; Wang, A.; Marin, M.; Honnen, W.; Ramasamy, S.; Porter, E.; Subbian, S.; Pinter, A.; Melikyan, G.B.; Lu, W.; et al. Human Defensins Inhibit SARS-CoV-2 Infection by Blocking Viral Entry. Viruses 2021, 13, 1246. [Google Scholar] [CrossRef]
- Solanki, S.S.; Singh, P.; Kashyap, P.; Sansi, M.S.; Ali, S.A. Promising role of defensins peptides as therapeutics to combat against viral infection. Microb. Pathog. 2021, 155, 104930. [Google Scholar] [CrossRef] [PubMed]
- Al-Bayatee, N.T.; Ad’hiah, A.H. Human beta-defensins 2 and 4 are dysregulated in patients with coronavirus disease 19. Microb. Pathog. 2021, 160, 105205. [Google Scholar] [CrossRef]
- Clausen, M.L.; Jungersted, J.M.; Andersen, P.S.; Slotved, H.C.; Krogfelt, K.A.; Agner, T. Human β-defensin-2 as a marker for disease severity and skin barrier properties in atopic dermatitis. Br. J. Dermatol. 2013, 169, 587–593. [Google Scholar] [CrossRef]
- Clausen, M.L.; Slotved, H.C.; Krogfelt, K.A.; Agner, T. Measurements of AMPs in stratum corneum of atopic dermatitis and healthy skin–tape stripping technique. Sci. Rep. 2018, 8, 1666. [Google Scholar] [CrossRef]
- Goo, J.; Ji, J.H.; Jeon, H.; Kim, M.J.; Jeon, S.Y.; Cho, M.Y.; Lee, S.H.; Choi, E.H. Expression of antimicrobial peptides such as LL-37 and hBD-2 in nonlesional skin of atopic individuals. Pediatr. Dermatol. 2010, 27, 341–348. [Google Scholar] [CrossRef] [PubMed]
- Alexander, H.; Paller, A.S.; Traidl-Hoffmann, C.; Beck, L.A.; De Benedetto, A.; Dhar, S.; Girolomoni, G.; Irvine, A.D.; Spuls, P.; Su, J.; et al. The role of bacterial skin infections in atopic dermatitis: Expert statement and review from the International Eczema Council Skin Infection Group. Br. J. Dermatol. 2020, 182, 1331–1342. [Google Scholar] [CrossRef]
- Kim, J.; Krueger, J.G. The immunopathogenesis of psoriasis. Dermatol. Clin. 2015, 33, 13–23. [Google Scholar] [CrossRef]
- Sticherling, M. Psoriasis and autoimmunity. Autoimmun. Rev. 2016, 15, 1167–1170. [Google Scholar] [CrossRef] [PubMed]
- Niyonsaba, F.; Ushio, H.; Nagaoka, I.; Okumura, K.; Ogawa, H. The human beta-defensins (-1, -2, -3, -4) and cathelicidin LL-37 induce IL-18 secretion through p38 and ERK MAPK activation in primary human keratinocytes. J. Immunol. 2005, 175, 1776–1784. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Cho, D.H.; Park, H.J. IL-18 and Cutaneous Inflammatory Diseases. Int. J. Mol. Sci. 2015, 16, 29357–29369. [Google Scholar] [CrossRef] [PubMed]
- Chieosilapatham, P.; Ogawa, H.; Niyonsaba, F. Current insights into the role of human β-defensins in atopic dermatitis. Clin. Exp. Immunol. 2017, 190, 155–166. [Google Scholar] [CrossRef]
- Hollox, E.J.; Huffmeier, U.; Zeeuwen, P.L.; Palla, R.; Lascorz, J.; Rodijk-Olthuis, D.; van de Kerkhof, P.C.; Traupe, H.; de Jongh, G.; den Heijer, M.; et al. Psoriasis is associated with increased beta-defensin genomic copy number. Nat. Genet. 2008, 40, 23–25. [Google Scholar] [CrossRef] [PubMed]
- Jansen, P.A.; Rodijk-Olthuis, D.; Hollox, E.J.; Kamsteeg, M.; Tjabringa, G.S.; de Jongh, G.J.; van Vlijmen-Willems, I.M.; Bergboer, J.G.; van Rossum, M.M.; de Jong, E.M.; et al. Beta-defensin-2 protein is a serum biomarker for disease activity in psoriasis and reaches biologically relevant concentrations in lesional skin. PLoS ONE 2009, 4, e4725. [Google Scholar] [CrossRef] [PubMed]
- Jin, T.; Sun, Z.; Chen, X.; Wang, Y.; Li, R.; Ji, S.; Zhao, Y. Serum Human Beta-Defensin-2 Is a Possible Biomarker for Monitoring Response to JAK Inhibitor in Psoriasis Patients. Dermatology 2017, 233, 164–169. [Google Scholar] [CrossRef]
- Hu, S.C.; Yu, H.S.; Yen, F.L.; Lin, C.L.; Chen, G.S.; Lan, C.C. Neutrophil extracellular trap formation is increased in psoriasis and induces human β-defensin-2 production in epidermal keratinocytes. Sci. Rep. 2016, 6, 31119. [Google Scholar] [CrossRef]
- Mi, B.; Liu, J.; Liu, Y.; Hu, L.; Liu, Y.; Panayi, A.C.; Zhou, W.; Liu, G. The Designer Antimicrobial Peptide A-hBD-2 Facilitates Skin Wound Healing by Stimulating Keratinocyte Migration and Proliferation. Cell. Physiol. Biochem. 2018, 51, 647–663. [Google Scholar] [CrossRef]
- Gonzalez-Curiel, I.; Trujillo, V.; Montoya-Rosales, A.; Rincon, K.; Rivas-Calderon, B.; deHaro-Acosta, J.; Marin-Luevano, P.; Lozano-Lopez, D.; Enciso-Moreno, J.A.; Rivas-Santiago, B. 1,25-dihydroxyvitamin D3 induces LL-37 and HBD-2 production in keratinocytes from diabetic foot ulcers promoting wound healing: An in vitro model. PLoS ONE 2014, 9, e111355. [Google Scholar] [CrossRef]
- Kotani, H.; Matsubara, K.; Koshizuka, T.; Nishiyama, K.; Kaneko, H.; Tasaka, M.; Sugiyama, T.; Suzutani, T. Human β-defensin-2 as a biochemical indicator of vaginal environment in pregnant women. Hypertens. Res. Pregnancy 2018, 6, 68–72. [Google Scholar] [CrossRef]
- Soto, E.; Espinoza, J.; Nien, J.K.; Kusanovic, J.P.; Erez, O.; Richani, K.; Santolaya-Forgas, J.; Romero, R. Human beta-defensin-2: A natural antimicrobial peptide present in amniotic fluid participates in the host response to microbial invasion of the amniotic cavity. J. Matern. Fetal Neonatal Med. 2007, 20, 15–22. [Google Scholar] [CrossRef]
- Brunner, A.; Medvecz, M.; Makra, N.; Sárdy, M.; Komka, K.; Gugolya, M.; Szabó, D.; Gajdács, M.; Ostorházi, E. Human beta defensin levels and vaginal microbiome composition in post-menopausal women diagnosed with lichen sclerosus. Sci. Rep. 2021, 11, 15999. [Google Scholar] [CrossRef] [PubMed]
- Madhusudhan, N.; Pausan, M.R.; Halwachs, B.; Durdević, M.; Windisch, M.; Kehrmann, J.; Patra, V.; Wolf, P.; Boukamp, P.; Moissl-Eichinger, C.; et al. Molecular Profiling of Keratinocyte Skin Tumors Links Staphylococcus aureus Overabundance and Increased Human β-Defensin-2 Expression to Growth Promotion of Squamous Cell Carcinoma. Cancers 2020, 12, 541. [Google Scholar] [CrossRef]
- Amabebe, E.; Anumba, D. The Vaginal Microenvironment: The Physiologic Role of Lactobacilli. Front. Med. 2018, 5, 181. [Google Scholar] [CrossRef]
- James, C.P.; Bajaj-Elliott, M.; Abujaber, R.; Forya, F.; Klein, N.; David, A.L.; Hollox, E.J.; Peebles, D.M. Human beta defensin (HBD) gene copy number affects HBD2 protein levels: Impact on cervical bactericidal immunity in pregnancy. Eur. J. Hum. Genet. 2018, 26, 434–439. [Google Scholar] [CrossRef]
- Garcia-Lopez, G.; Flores-Espinosa, P.; Zaga-Clavellina, V. Tissue-specific human beta-defensins (HBD)1, HBD2, and HBD3 secretion from human extra-placental membranes stimulated with Escherichia coli. Reprod. Biol. Endocrinol. 2010, 8, 146. [Google Scholar] [CrossRef]
- Zaga-Clavellina, V.; Ruiz, M.; Flores-Espinosa, P.; Vega-Sanchez, R.; Flores-Pliego, A.; Estrada-Gutierrez, G.; Sosa-Gonzalez, I.; Morales-Méndez, I.; Osorio-Caballero, M. Tissue-specific human beta-defensins (HBD)-1, HBD-2 and HBD-3 secretion profile from human amniochorionic membranes stimulated with Candida albicans in a two-compartment tissue culture system. Reprod. Biol. Endocrinol. 2012, 10, 70. [Google Scholar] [CrossRef] [PubMed]
- Kotani, H.; Koshizuka, T.; Matsubara, K.; Nishiyama, K.; Sugiyama, T.; Suzutani, T. Relationship Between Human β-Defensin 2 and the Vaginal Environment. Jpn. J. Infect. Dis. 2020, 73, 214–220. [Google Scholar] [CrossRef]
- Feng, Y.; Pan, X.; Huang, N.; Feng, Y.; Wu, Q.; Wang, B. The human beta-defensins expression in female genital tract and pregnancy-related tissues. Sichuan Da Xue Xue Bao Yi Xue Ban 2003, 34, 217–219. [Google Scholar]
- Hanson, L.A. Breastfeeding provides passive and likely long-lasting active immunity. Ann. Allergy Asthma Immunol. 1999, 81, 523–533, quiz 533-4, 537. Erratum in: Ann. Allergy Asthma Immunol. 1999, 82, 478. [Google Scholar] [CrossRef]
- Cacho, N.T.; Lawrence, R.M. Innate Immunity and Breast Milk. Front. Immunol. 2017, 8, 584. [Google Scholar] [CrossRef] [PubMed]
- Palmeira, P.; Carneiro-Sampaio, M. Immunology of breast milk. Rev. Assoc. Med. Bras. 1992, 62, 584–593. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Cao, R.; Li, J.; Wu, J.; Wu, S.; Chen, T. Identification of sociodemographic and clinical factors associated with the levels of human β-defensin-1 and human β-defensin-2 in the human milk of Han Chinese. Br. J. Nutr. 2014, 111, 867–874. [Google Scholar] [CrossRef]
- Savilahti, E.M.; Kukkonen, A.K.; Kuitunen, M.; Savilahti, E. Soluble CD14, α-and β-defensins in breast milk: Association with the emergence of allergy in a high-risk population. Innate Immun. 2015, 21, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Baricelli, J.; Rocafull, M.A.; Vázquez, D.; Bastidas, B.; Báez-Ramirez, E.; Thomas, L.E. β-defensin-2 in breast milk displays a broad antimicrobial activity against pathogenic bacteria. J. Pediatr. 2015, 91, 36–43. [Google Scholar] [CrossRef]
- Trend, S.; Strunk, T.; Hibbert, J.; Kok, C.H.; Zhang, G.; Doherty, D.A.; Richmond, P.; Burgner, D.; Simmer, K.; Davidson, D.J.; et al. Antimicrobial protein and Peptide concentrations and activity in human breast milk consumed by preterm infants at risk of late-onset neonatal sepsis. PLoS ONE 2015, 10, e0117038. [Google Scholar] [CrossRef] [PubMed]
- Olbrich, P.; Pavón, A.; Rosso, M.L.; Molinos, A.; de Felipe, B.; Sanchez, B.; Praena-Fernández, J.M.; Jimenez, F.; Obando, I.; Neth, O. Association of human beta-defensin-2 serum levels and sepsis in preterm neonates. Pediatr. Crit. Care Med. 2013, 14, 796–800. [Google Scholar] [CrossRef]
- Schaller-Bals, S.; Schulze, A.; Bals, R. Increased levels of antimicrobial peptides in tracheal aspirates of newborn infants during infection. Am. J. Respir. Crit. Care Med. 2002, 165, 992–995. [Google Scholar] [CrossRef] [PubMed]
- Spencer, J.D.; Schwaderer, A.L.; Becknell, B.; Watson, J.; Hains, D.S. The innate immune response during urinary tract infection and pyelonephritis. Pediatr. Nephrol. 2014, 29, 1139–1149. [Google Scholar] [CrossRef] [PubMed]
- Caterino, J.M.; Hains, D.S.; Camargo, C.A.; Quraishi, S.A.; Saxena, V.; Schwaderer, A.L. A Prospective, Observational Pilot Study of the Use of Urinary Antimicrobial Peptides in Diagnosing Emergency Department Patients with Positive Urine Cultures. Acad. Emerg. Med. 2015, 22, 1226–1230. [Google Scholar] [CrossRef] [PubMed]
- Lehmann, J.; Retz, M.; Harder, J.; Krams, M.; Kellner, U.; Hartmann, J.; Hohgräwe, K.; Raffenberg, U.; Gerber, M.; Loch, T.; et al. Expression of human beta-defensins 1 and 2 in kidneys with chronic bacterial infection. BMC Infect. Dis. 2002, 2, 20. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Jung, J.R.; Kim, H.J.; Lee, S.Y.; Chang, I.H.; Lee, T.J.; Kim, W.; Myung, S.C. Expression of human β-defensin-2 in the prostate. BJU Int. 2011, 107, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Ko, K.; Kim, H.J.; Lee, J.; Myung, S.C. Zinc induces LPS-mediated upregulation of HBD-2 via ERK1/2 and p38MAPK signaling pathways in human prostate epithelial cells. Anim. Cells Syst. 2016, 20, 317–324. [Google Scholar] [CrossRef]
- Liu, A.Y.; Destoumieux, D.; Wong, A.V.; Park, C.H.; Valore, E.V.; Liu, L.; Ganz, T. Human beta-defensin-2 production in keratinocytes is regulated by interleukin-1, bacteria, and the state of differentiation. J. Investig. Dermatol. 2002, 118, 275–281. [Google Scholar] [CrossRef]
| Conditions Where Beneficial Effect of hBD-2 Is Suggested and Proposed as a Therapeutic Agent | |
|---|---|
| fungal intestinal infection [52] | upregulation of intestinal epithelium integrity |
| experimental colitis [68,69,70] | suppression of inflammation |
| premature delivery [113] | protective effect |
| atopic asthma [84] | protective effect |
| house dust mite allergy [84] | protective effect |
| smoking-induced cell damage [23,92] | protection of the respiratory tract against oxidative stress and infections |
| wound healing [33,111,112] | increasing the migration and proliferation of cells |
| oral lichen planus [32] | enhancement of the level of hBD-2 reduced by the disease |
| virus diseases [94,95] | possible inhibition of virus entry into the host cells |
| Conditions Where Elevated Levels of hBD-2 were Noted and Proposed to Be a Marker |
|---|
| gingival inflammation [18,22,23,24,25] |
| infection with H. pylori [41,42,43,44,45,46] |
| ulcerative colitis [62,63,64] |
| Crohn’s disease [66] |
| irritable bowel syndrome [67] |
| intra-amniotic infections [114] |
| lichen sclerosus [115] |
| smoking [92] |
| atopic dermatitis [98,99] |
| psoriasis [107,108] |
| Tissue/Cells | Stimulus | mRNA hBD-2 | hBD-2 Protein |
|---|---|---|---|
| oral keratinocytes/oral epithelial cells | TNF-α IL-1β IFN-γ LPS IL-2, IL-6 C. albicans | ↑ * [18,22,23,25,31]/0 ** [29] | ↑ * [18,22,23,25,31] |
| human milk oligosaccharides | - | ↑ [19] | |
| cigarette smoke | ↓ [23] | ↓ [23] | |
| laser therapy | ↑ [33] | ↑ [33] | |
| gastric epithelial cell | H. pylori | ↑ [41,42,44,45] | ↑ [43] |
| colon epithelial cells | IL-1α IL-22 bacteria enterotoxins parasites | ↑ [13,53,60,62] | ↑ [13,53,60,62] |
| probiotic bacteria | ↑ [56] | ↑ [56] | |
| T4 bacteriophage | ↑ [74] | - | |
| genitourinary tract | fungi bacteria LPS IL-1β TNF-α | ↑ [118,134,135] | ↑ [113,118,133,135] |
| placental tissue | fungi bacteria | - | ↑ [119,120] |
| astrocytes | TNF-α IL-1β LPS | ↑ [79] | ↑ [79] |
| brain capillary | Chlamydophila pneumoniae | ↑ [80] | ↑ [80] |
| lung epithelium | LPS bacterial (e.g., Pseudomonas) infection fungal infection IL-1β | ↑ [89,90] | ↑ [87,89,90] |
| glucocorticoids (dexamethasone) | ↓ [10]/↑ [86] | - | |
| B. abortus infections | 0 ** [91] | 0 ** [91] | |
| skin keratinocytes | bacteria IL-1α IL-1β | ↑ [116] | ↑ [137] |
| D3 vitamin | ↑ [112] | ↑ [112] | |
| NET | ↑ [110] | ↑ [110] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cieślik, M.; Bagińska, N.; Górski, A.; Jończyk-Matysiak, E. Human β-Defensin 2 and Its Postulated Role in Modulation of the Immune Response. Cells 2021, 10, 2991. https://doi.org/10.3390/cells10112991
Cieślik M, Bagińska N, Górski A, Jończyk-Matysiak E. Human β-Defensin 2 and Its Postulated Role in Modulation of the Immune Response. Cells. 2021; 10(11):2991. https://doi.org/10.3390/cells10112991
Chicago/Turabian StyleCieślik, Martyna, Natalia Bagińska, Andrzej Górski, and Ewa Jończyk-Matysiak. 2021. "Human β-Defensin 2 and Its Postulated Role in Modulation of the Immune Response" Cells 10, no. 11: 2991. https://doi.org/10.3390/cells10112991
APA StyleCieślik, M., Bagińska, N., Górski, A., & Jończyk-Matysiak, E. (2021). Human β-Defensin 2 and Its Postulated Role in Modulation of the Immune Response. Cells, 10(11), 2991. https://doi.org/10.3390/cells10112991






