Manure Source and Cropping System Affect Nutrient Uptake by Cactus (Nopalea cochenillifera Salm Dyck)
Abstract
1. Introduction
2. Materials and Methods
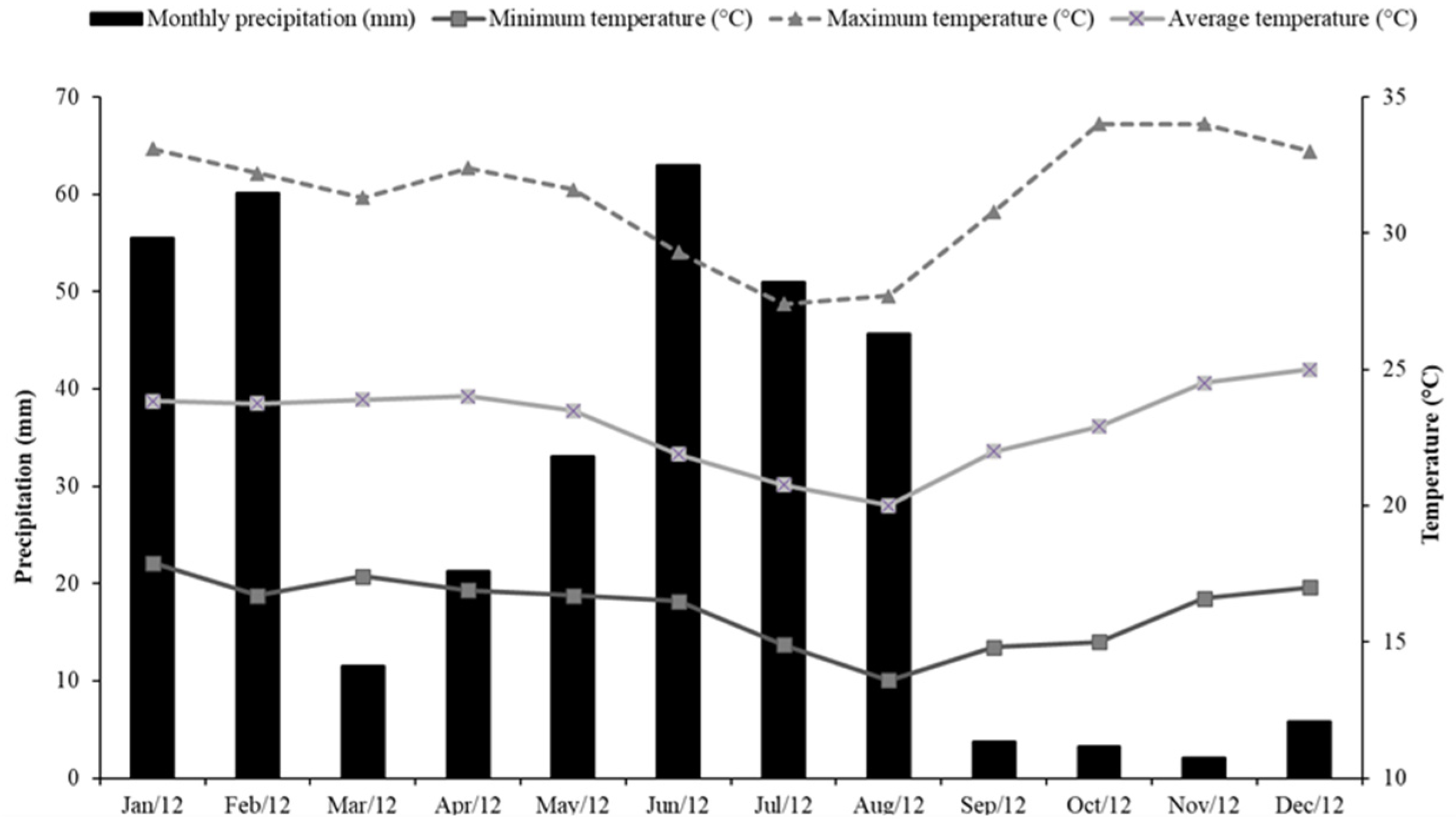
2.1. Site Description
2.2. Experimental Design
2.3. Sample Preparation and Analysis
2.4. Statistical Analysis
3. Results
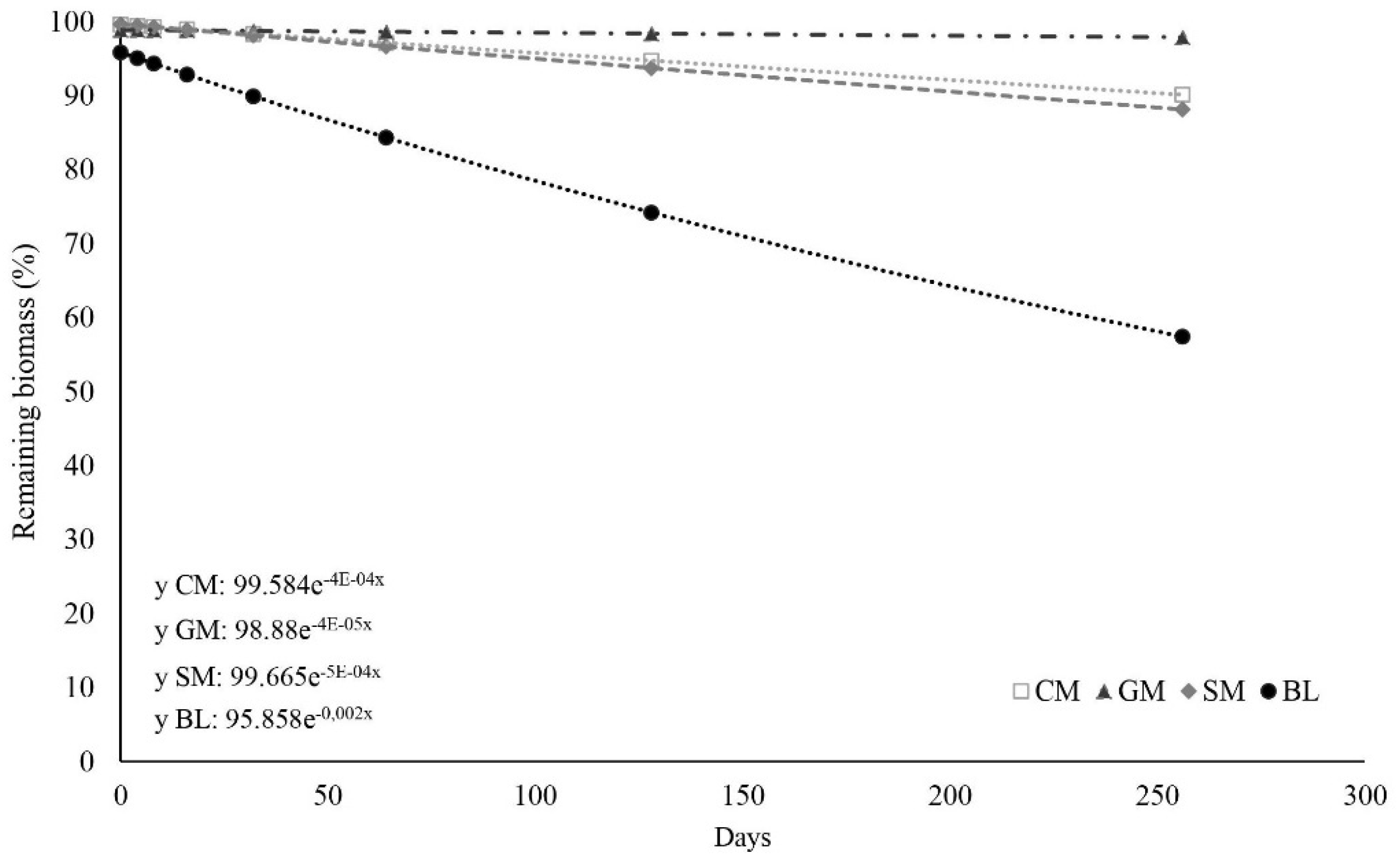
3.1. Manure Decomposition
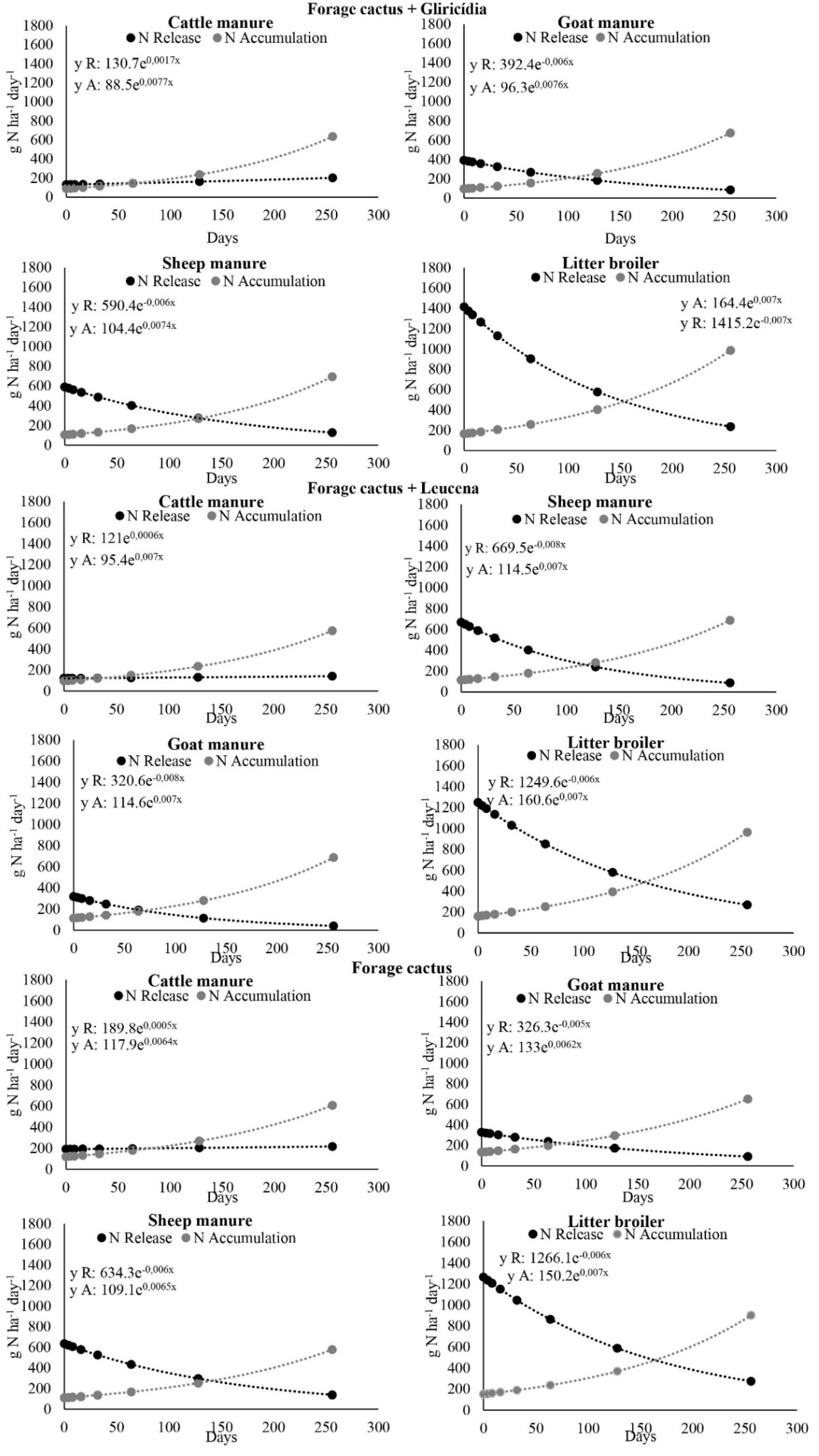
3.2. Nitrogen Accumulation in Aboveground Cactus Biomass and Release from Manure
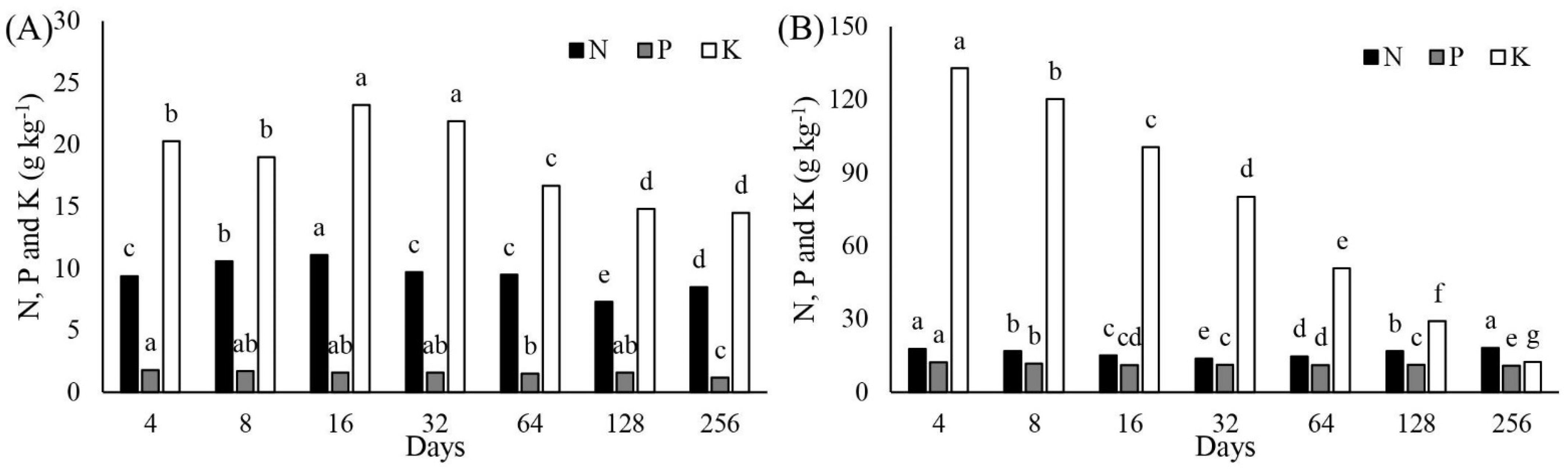
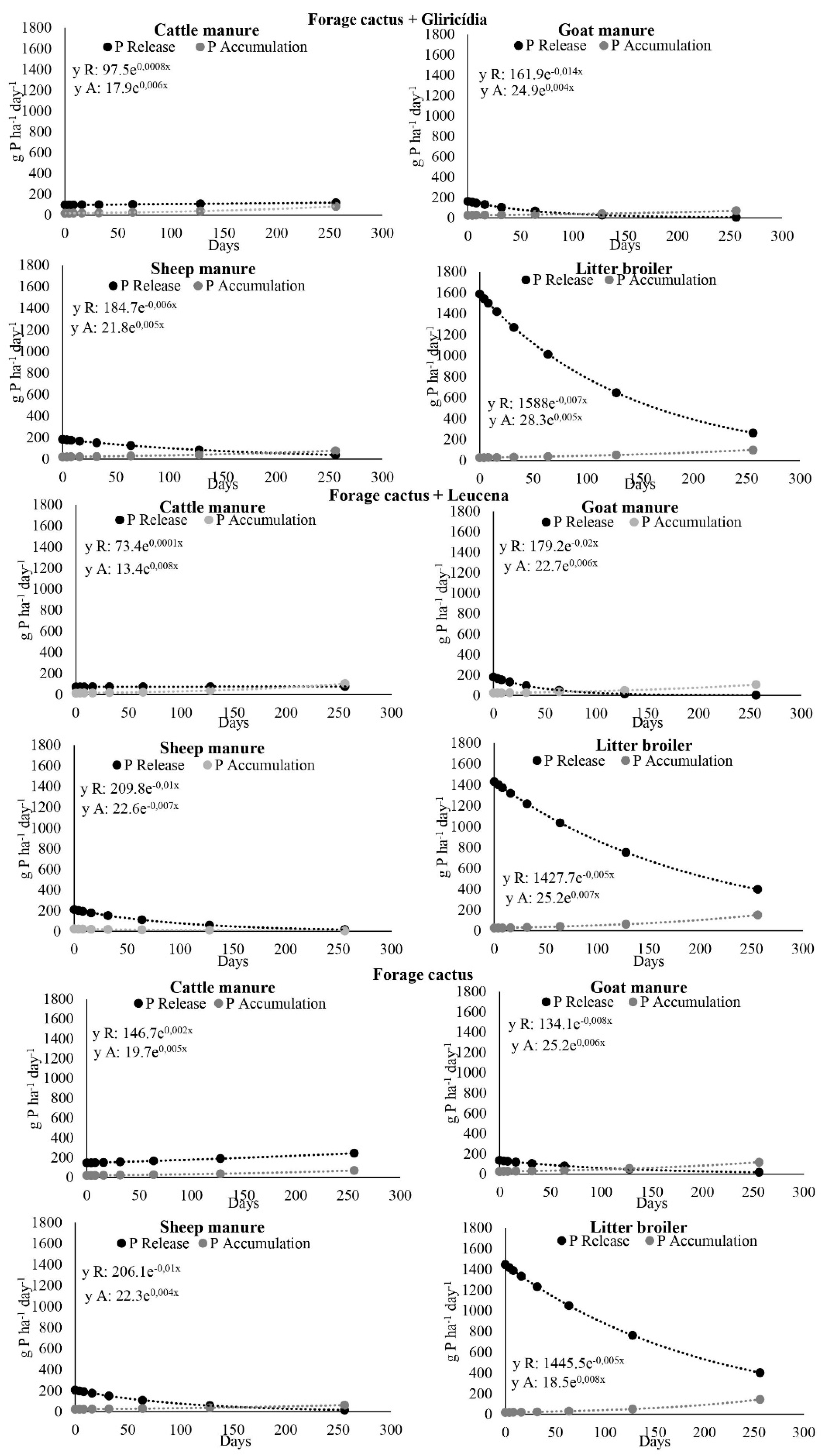
3.3. Phosphorus Accumulation in Cactus Aboveground Biomass and Release from Manure
3.4. Potassium Accumulation Rate in Cactus Aboveground Biomass and K Release from Manure
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Miranda, K.R.D.; Dubeux Júnior, J.C.B.; Mello, A.C.L.; Silva, M.C.; Santos, M.V.F.; Santos, D.C. Forage Production and Mineral Composition of Cactus Intercropped with Legumes and Fertilized with Different Sources of Manure. Ciência Rural. 2019, 49, e20180324. [Google Scholar] [CrossRef]
- Castro, I.N.; Santos, S.R.; Kondo, M.K.; Maia, V.; Santos, C.C.R. Agronomic Performance and Water Use Efficiency of Irrigated Cactus Pear Cultivars. Rev. Caatinga 2020, 33, 529–540. [Google Scholar] [CrossRef]
- Souza, T.C.; Santos, M.V.F.; Dubeux Júnior, J.C.B.; Lira, M.A.; Santos, D.C.; Cunha, M.V.; Silva, R.R. Productivity and Nutrient Concentration in Spineless Cactus Under Different Fertilizations and Plant Densities. Rev. Bras. De Ciências Agrárias 2017, 12, 555–560. [Google Scholar] [CrossRef]
- Vasconcelos, A.G.V.; Lira, M.A.; Cavalcanti, V.L.B.; Santos, M.V.F.; Willadino, L. Selection of Prickly-Pear Clones Resistant to Carmine Cochineal Dactylopius sp. Rev. Bras. De Zootec. 2009, 38, 827–831. [Google Scholar] [CrossRef]
- Edvan, R.L.; Mota, R.R.M.; Silva, T.P.S.; Nascimento, R.R.; Sousa, S.V.; Silva, A.L.; Araújo, J.S. Resilience of Cactus Pear Genotypes in A Tropical Semi-Arid Region Subject to Climatic Cultivation Restriction. Sci. Rep. 2020, 10, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Mayer, J.A.; Cushman, J.C. Nutritional and Mineral Content of Prickly Pear Cactus: A Highly Water-Use Efficient Forage, Fodder and Food Species. J. Agron. Crop. Sci. 2019, 205, 625–634. [Google Scholar] [CrossRef]
- Schumacher, M.V.; Szymczak, D.A.; Trüby, P.; Londero, E.K.; Marafiga, J. Litter and Nutrient Input in Seasonal Forest in The Central Region of Rio Grande do Sul state. Ciência Florest. 2018, 28, 532–541. [Google Scholar] [CrossRef]
- Oli, P.S.; Mandal, T.N.; Adhikari, U. Effect of Leaf Litter Treatment on Soil Microbial Biomass. Open J. Soil Sci. 2018, 8, 175–185. [Google Scholar] [CrossRef]
- Santos, F.M.S.C.; Lima Junior, D.M.; Cardoso, D.B.; Maciel, M.V.; Carvalho, F.F.R. Replacement of Tifton 85 Hay with Maniçoba Hay in The Spineless Cactus Diet of Sheep. Rev. Caatinga 2021, 1, 219–227. [Google Scholar] [CrossRef]
- Silva, L.L.H.; Oliveira, E.; Calegari, L.; Pimenta, M.C.; Dantas, M.K.L. Características Dendrométricas, Físicas e Químicas da Myracrodruon Urundeuva e da Leucaena Leucocephala. Floresta e Ambiente 2017, 24, 1–8. [Google Scholar] [CrossRef][Green Version]
- Silva, J.A.D.; Donato, S.L.; Donato, P.E.; Souza, E.D.S.; Padilha Júnior, M.C.; Junior, S. Extraction/Export of Nutrients in Opuntia ficus-indica Under Different Spacings and Chemical Fertilizers. Rev. Bras. De Eng. Agrícola E Ambient. 2016, 20, 236–242. [Google Scholar] [CrossRef]
- Silva, N.G.M.; Santos, M.V.F.; Dubeux Júnior, J.C.B.; Cunha, M.V.; Lira, M.A.; Ferraz, I. Effects of Planting Density and Organic Fertilization Doses on Productive Efficiency of Cactus Pear. Rev. Caatinga 2016, 29, 976–983. [Google Scholar] [CrossRef]
- Zhu, Y.; Merbold, L.; Leitner, S.; Pelster, D.E.; Okoma, S.A.; Ngetich, F.; Butterbach-Bahl, K. The Effects of Climate on Decomposition of Cattle, Sheep and Goat Manure in Kenyan Tropical Pastures. Plant Soil 2020, 451, 325–343. [Google Scholar] [CrossRef]
- Food and Agriculture Organization (FAO). World Reference Base for Soil Resources: International Soil Classification System for Naming Soils and Creating Legends for Soil Maps. World Soil Resour. Rep. 2014, 106, 192. [Google Scholar]
- Almeida, B.G.; Donagemma, G.K.; Ruiz, H.A.; Braida, J.A.; Viana, J.H.M.; Reichert, J.M.M.; Passos, R.R. Standardization of Methods for Granulometric Analyses in Brazil. Embrapa Solos-Comun. Técnico 2012, 66, 11. [Google Scholar]
- Empresa Brasileira de Pesquisa Agropecuária (EMBRAPA). Manual de métodos de análise de solo, 2nd ed.; Embrapa Solos: Rio de Janeiro, Brazil, 2011; 230p. [Google Scholar]
- Dubeux Júnior, J.C.B.; Sollenberger, L.E.; Interrante, S.M.; Vendramini, J.M.B.; Stewart Jr, R.L. Litter Decomposition and Mineralization in Bahiagrass Pastures Managed at Different Intensities. Crop. Sci. 2006, 46, 1305–1310. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemists. Official Methods of Analysis, 18th ed.; AOAC: Arlington, TX, USA, 2005. [Google Scholar]
- Silva, F.C. Manual de Análises Químicas de Solos, Plantas e Fertilizantes, 2nd ed.; Embrapa Solos: Rio de Janeiro, Brazil, 1999; pp. 171–223. [Google Scholar]
- Wagner, G.H.; Wolf, D.C. Carbon Transformation and Soil Organic Matter Formations. In Principles and Applications of Soil Microbiology; Sylvia, D.M., Hartel, P.G., Fuhrmann, J.J., Zubere, D.A., Eds.; Prentice Hall: Englewood Cliffs, NJ, USA, 1999; pp. 218–258. [Google Scholar]
- SAS Institute Inc. SAS/STAT® 14.1 User’s Guide; SAS Institute Inc.: Cary, NC, USA, 2015. [Google Scholar]
- Yan, X.; Wei, Z.; Hong, Q.; Lu, Z.; Wu, J. Phosphorus Fractions and Sorption Characteristics in a Subtropical Paddy Soil as Influenced by Fertilizer Sources. Geoderma 2017, 295, 80–85. [Google Scholar] [CrossRef]
- Fei, C.; Zhang, S.R.; Wei, W.L.; Liang, B.; Ding, X.D. Straw and Optimized Nitrogen Fertilizer Decreases Phosphorus Leaching Risks in a Long-Term Greenhouse Soil. J. Soils Sediments 2019, 20, 1199–1207. [Google Scholar] [CrossRef]
- Nobile, C.; Bravin, M.N.; Becquer, T.; Paillat, J. Phosphorus Sorption and Availability in an Andosol After a Decade of Organic or Mineral Fertilizer Applications: Importance of pH and Organic Carbon Modifications in Soil as Compared to Phosphorus Accumulation. Chemosphere 2020, 239, 124709. [Google Scholar] [CrossRef]
- Horwath, W.R. The Role of the Soil Microbial Biomass in Cycling Nutrients. Microb. Biomass: A Paradig. Shif Terr. Biogeochem. 2017, 41–66. [Google Scholar] [CrossRef]
- Slade, E.M.; Riutta, T. Interacting Effects of Leaf Litter Species and Macrofauna on Decomposition in Different Litter Environments. Basic Appl. Ecol. 2012, 13, 423–431. [Google Scholar] [CrossRef]
- Cardoso, E.J.B.N.; Andreote, F.D. Soil Microbiology, 2nd ed.; ESALQ: Piracicaba, Brazil, 2016. [Google Scholar]
- Silva, V.B.; Silva, A.P.; Dias, B.O.; Araujo, J.L.; Santos, D.; Franco, R.P. Decomposition and Mineralization of N, P and K of Cattle Manure and Poultry Litter Isolated or Mixed. Rev. Bras. De Ciência Do Solo 2014, 38, 1537–1546. [Google Scholar] [CrossRef]
- Nguyen, X.P.; Jho, H.; Jeong, Y.J.; Jo, Y.T.; Islam, M.N.; Park, J.H. Characterization of Composted Chicken Manures Discharged from Farms in South Korea. Environ. Eng. Res. 2015, 20, 254–259. [Google Scholar] [CrossRef]
- Barros, F.M.D.R.; Fracetto, G.G.M.; Fracetto, F.J.C.; Mendes Júnior, J.P.; Araújo, V.L.V.P.; Lira, M.A., Jr. Silvopastoral Systems Drive The Nitrogen Cycling Bacterial Community in Soil. Ciência E Agrotecnologia 2018, 42, 281–290. [Google Scholar] [CrossRef]
- Apolinário, V.X.O.; Dubeux, J.C.B., Jr.; Lira, M.A.; Ferreira, R.L.C.; Mello, A.C.L.; Santos, M.V.F.; Sampaio, E.V.S.B.; Muir, J.P. Tree Legumes Provide Marketable Wood and add Nitrogen in Warm-Climate Silvopasture Systems. Agron. J. 2015, 107, 1915–1921. [Google Scholar] [CrossRef]
- Sarabia-Salgado, L.; Solorio-Sánchez, F.; Ramírez-Avilés, L.; Alves, B.J.R.; Ku-Vera, J.; Aguilar-Pérez, C.; Boddey, R.M. Increase in Milk Yield from Cows Through Improvement of Forage Production using The N2-fixing Legume Leucaena leucocephala in a Silvopastoral System. Animals 2020, 10, 734. [Google Scholar] [CrossRef]
- Donato, P.E.; Donato, S.L.; Silva, J.A.; Pires, A.J.; Junior, S. Extraction/Exportation of Macronutrients by Cladodes of ‘Gigante’cactus Pear under Different Spacings and Organic Fertilization. Rev. Bras. De Eng. Agrícola E Ambient. 2017, 21, 238–243. [Google Scholar] [CrossRef]
- Oladipupo, A.J.; Alade, A.A.; Adewuyi, S.; Ajiboye, G.A.; Blessing, O.T. Soil Phosphorus Fractions, Reaction, and Conductivity in Some Southwestern Nigerian Soils as Affected by Animal Manure Mixtures. Commun. Soil Sci. Plant Anal. 2020, 51, 2616–2632. [Google Scholar] [CrossRef]
- Donato, P.E.; Donato, S.L.; Silva, J.A.; Pires, A.J.; Rosa, R.C.; Aquino, A.A. Nutrition and Yield of ‘Gigante’ Cactus Pear Cultivated with Different Spacings and Organic Fertilizer. Rev. Bras. De Eng. Agrícola E Ambient. 2016, 20, 1083–1088. [Google Scholar] [CrossRef][Green Version]
- Lédo, A.A.; Donato, S.L.; Aspiazu, I.; Silva, J.A.D.; Donato, P.E.; Carvalho, A.J.D. Nutrient Concentration and Nutritional Efficiency in ‘Gigante’ Cactus Pear Submitted to Different Spacings and Fertilizations. Rev. Bras. De Eng. Agrícola E Ambient. 2020, 24, 154–161. [Google Scholar] [CrossRef]
- Somda, Z.C.; Powell, J.M. Seasonal Decomposition of Sheep Manure and Forage Leaves in Soil. Commun. Soil Sci. Plant Anal. 1998, 29, 2961–2979. [Google Scholar] [CrossRef]


| Attributes | Soil Layer (m) | ||
|---|---|---|---|
| 0–0.10 | 0–0.20 | 0.20–0.40 | |
| Sand (g kg−1) 1 | 746 | 748 | - |
| Silt (g kg−1) 1 | 146 | 136 | - |
| Clay (g kg−1) 1 | 108 | 116 | - |
| Textural class | Sandy | ||
| pH (H2O) 2 | 4.8 | 4.7 | 4.7 |
| P (mg/dm3) 2 | 23.5 | 15.5 | 6.2 |
| Ca (cmolc/dm3) 2 | 1.93 | 1.76 | 1.61 |
| Mg (cmolc/dm3) 2 | 0.39 | 0.45 | 0.34 |
| Na (cmolc/dm3) 2 | 0.8 | 0.7 | 0.9 |
| K (cmolc/dm3) 2 | 0.2 | 0.1 | 0.1 |
| Al (cmolc/dm3) 2 | 0.27 | 0.28 | 0.31 |
| Al + H (cmolc/dm3) 2 | 2.38 | 2.32 | 2.29 |
| TOC (g.kg−1) | 9.59 | 8.75 | 8.91 |
| SOM (g.kg−1) | 16.5 | 15.1 | 15.7 |
| Manure | N | P | K | C | C/N | C/P | C/K | OM |
|---|---|---|---|---|---|---|---|---|
| g kg−1 | g kg−1 | |||||||
| Cattle | 11 | 6 | 142 | 232 | 21 | 39 | 2 | 400 |
| Goat | 16 | 5 | 107 | 290 | 18 | 58 | 3 | 500 |
| Sheep | 20 | 5 | 157 | 348 | 17 | 70 | 2 | 600 |
| Broiler litter | 35 | 31 | 173 | 522 | 15 | 17 | 3 | 900 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saraiva, F.M.; Dubeux, J.C.B., Jr.; Cunha, M.V.d.; Menezes, R.S.C.; Santos, M.V.F.d.; Camelo, D.; Ferraz, I. Manure Source and Cropping System Affect Nutrient Uptake by Cactus (Nopalea cochenillifera Salm Dyck). Agronomy 2021, 11, 1512. https://doi.org/10.3390/agronomy11081512
Saraiva FM, Dubeux JCB Jr., Cunha MVd, Menezes RSC, Santos MVFd, Camelo D, Ferraz I. Manure Source and Cropping System Affect Nutrient Uptake by Cactus (Nopalea cochenillifera Salm Dyck). Agronomy. 2021; 11(8):1512. https://doi.org/10.3390/agronomy11081512
Chicago/Turabian StyleSaraiva, Felipe Martins, José Carlos Batista Dubeux, Jr., Márcio Vieira da Cunha, Rômulo Simões Cezar Menezes, Mércia Virginia Ferreira dos Santos, Dayanne Camelo, and Ivan Ferraz. 2021. "Manure Source and Cropping System Affect Nutrient Uptake by Cactus (Nopalea cochenillifera Salm Dyck)" Agronomy 11, no. 8: 1512. https://doi.org/10.3390/agronomy11081512
APA StyleSaraiva, F. M., Dubeux, J. C. B., Jr., Cunha, M. V. d., Menezes, R. S. C., Santos, M. V. F. d., Camelo, D., & Ferraz, I. (2021). Manure Source and Cropping System Affect Nutrient Uptake by Cactus (Nopalea cochenillifera Salm Dyck). Agronomy, 11(8), 1512. https://doi.org/10.3390/agronomy11081512






