Techniques and Algorithms for Hepatic Vessel Skeletonization in Medical Images: A Survey
Abstract
:1. Introduction
1.1. Liver Diseases and Vasculature
1.2. Growth of Hepatic Vessels
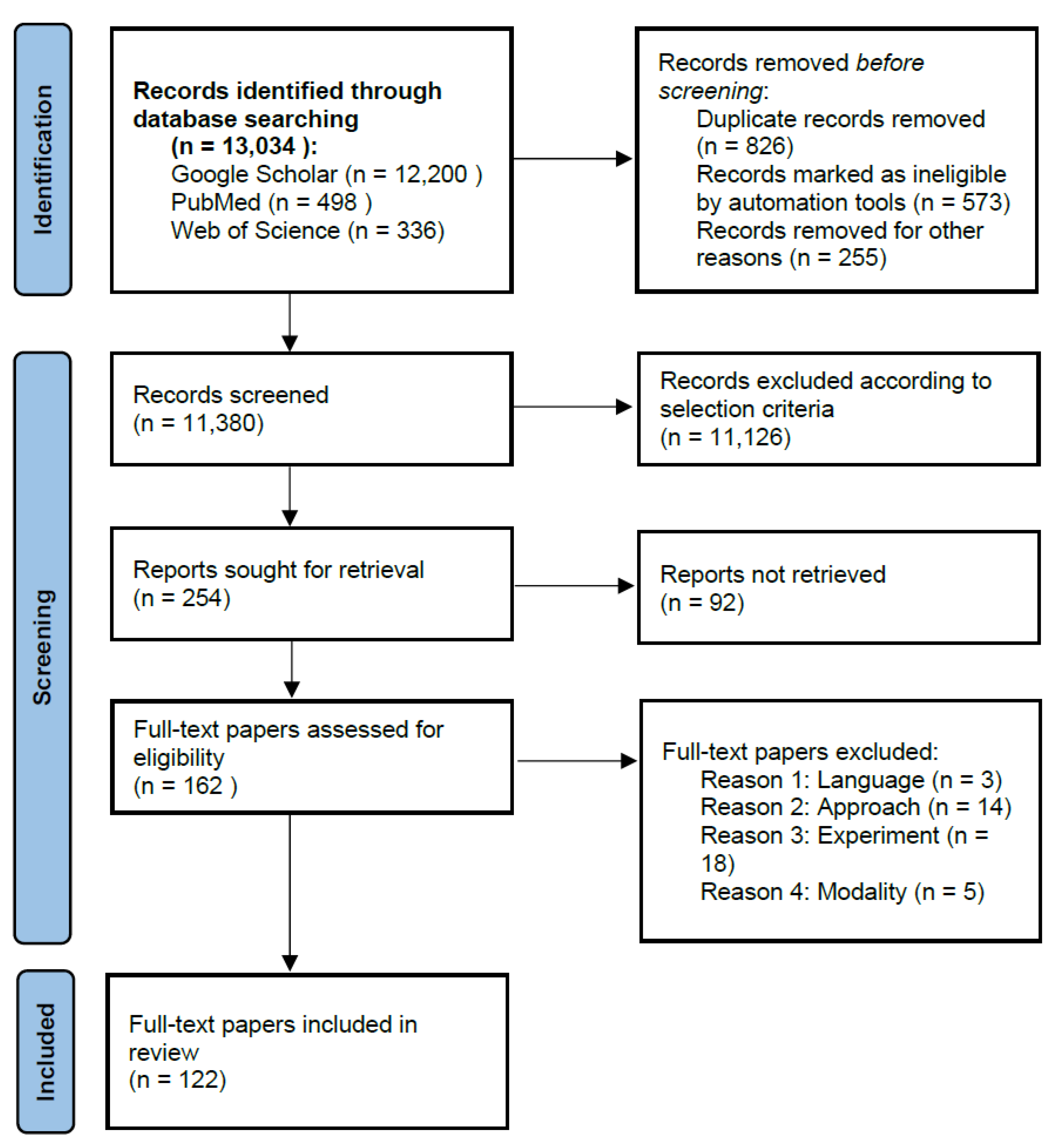
- To our knowledge, this is the first systematic review specifically on the skeletonization of hepatic vessel, which fills in gaps in the literature.
- With a survey from more than 120 papers, we provide comprehensive introductions, analyses and detailed statistics on recent and classical publications from different perspectives (such as methods, image modalities and evaluation criteria) in the related Sections, Tables and Figures.
- According to our survey and statistics, we reasonably put forward challenges and future trends in the Discussion and Conclusion.
2. Image Modality for Hepatic Vessels
3. Evaluation Criteria
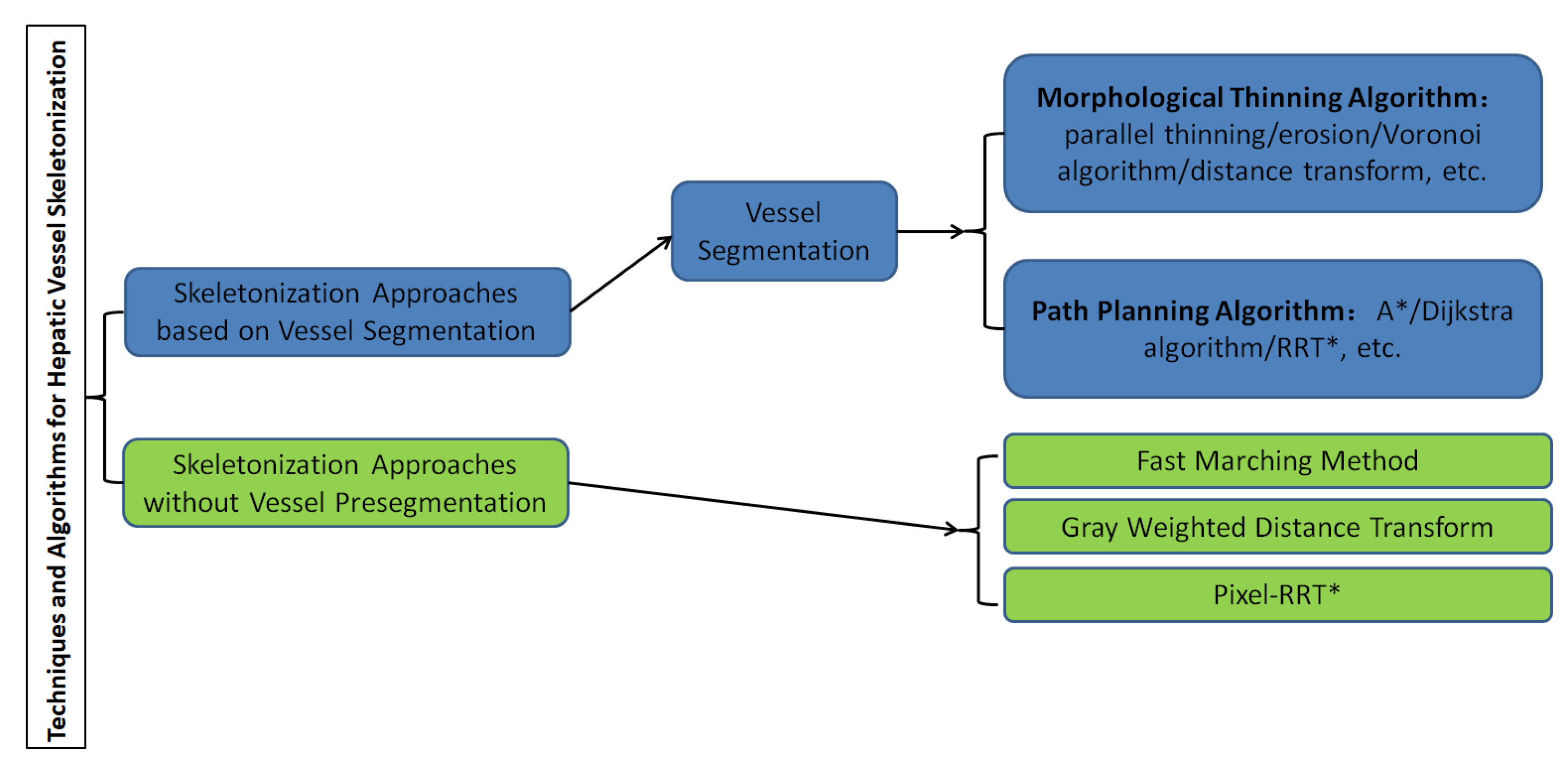
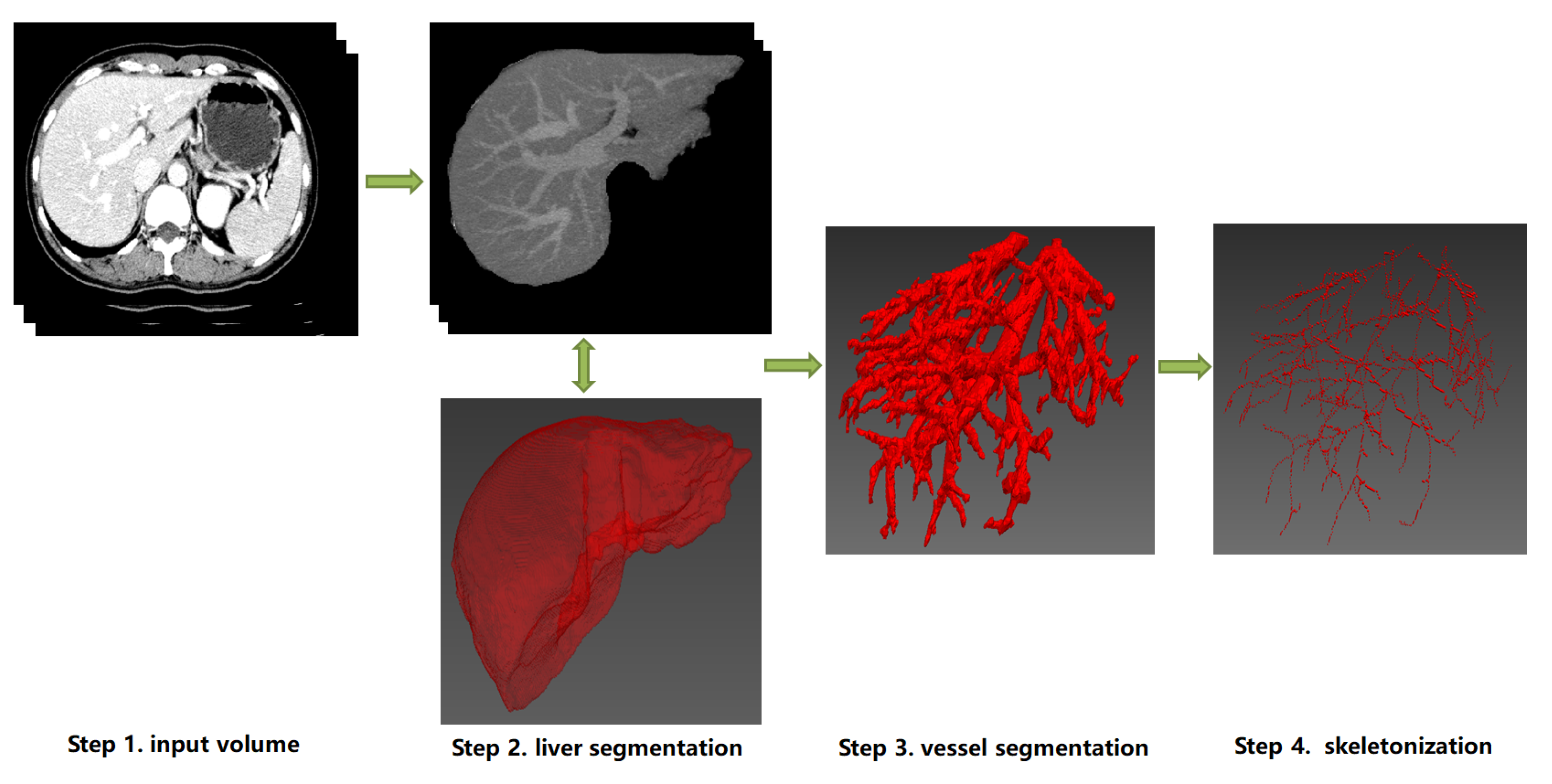
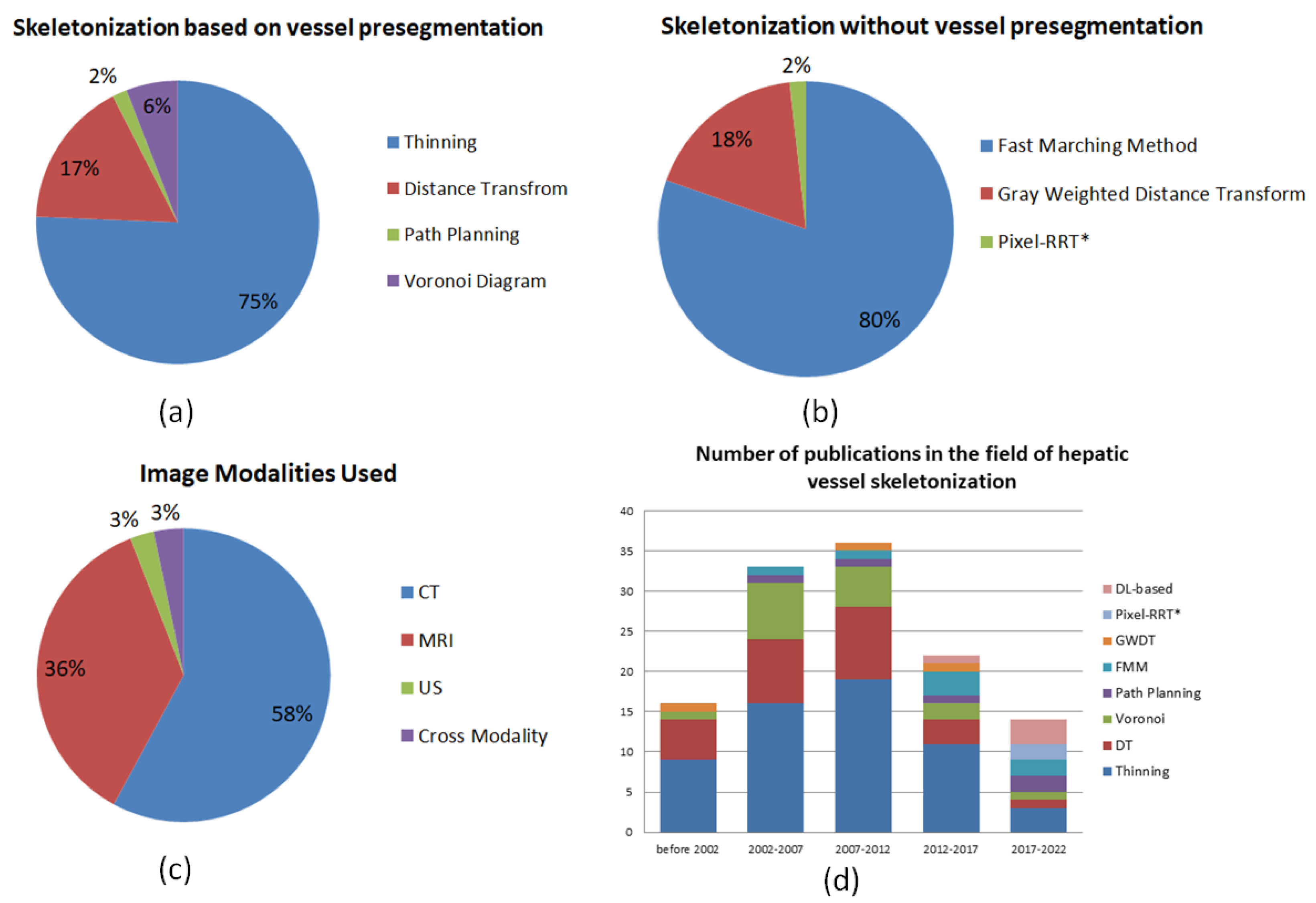
4. Skeletonization Approaches Based on Vessel Segmentation
4.1. Hepatic Vessel Segmentation
4.2. Morphological Thinning Algorithm
4.3. Path Planning Algorithm
| Algorithm 1: Dijkstra Framework. |
 |
5. Skeletonization Approaches without Vessel Pre-Segmentation
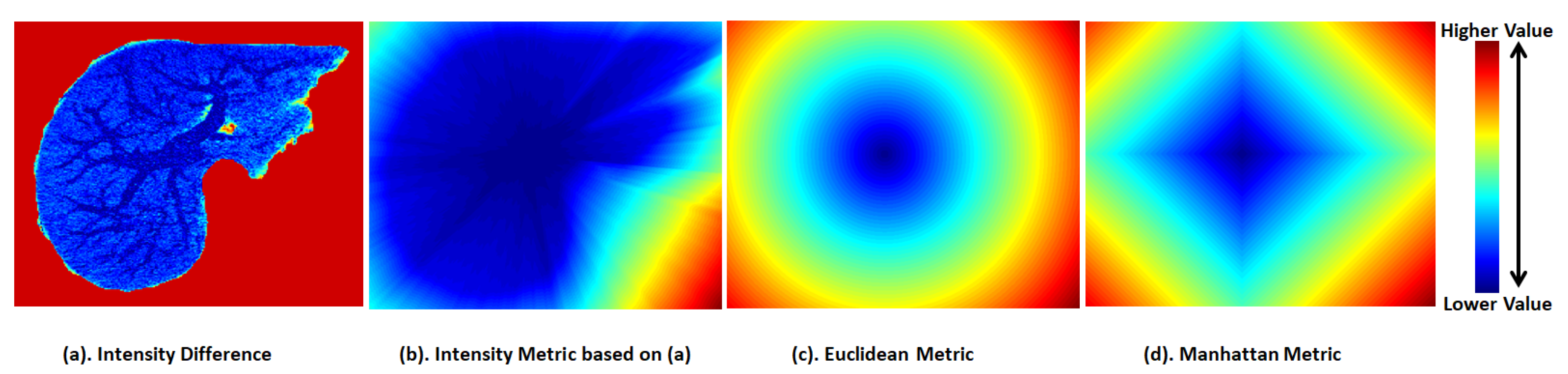
5.1. Fast Marching Method
5.2. Gray Weighted Distance Transform
5.3. Pixel-RRT*
6. Discussion and Conclusions
6.1. Challenges and Opportunities
6.2. Future Trends
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| G | Graph |
| V | Vertex of Graph |
| E | Edge of Graph |
| The i-th Edge of Graph | |
| Weight of the i-th Edge | |
| s | Starting point(initial vertex) |
| Initial distance of starting point | |
| S | The set of vertices with known shortest path |
| Q | The set of vertices not in S |
| ∅ | Empty set |
| u | Vertex of shortest path in Q |
| The set of neighbor vertices of u | |
| v | The element of |
| 3D | three dimensional |
| CT | Computed Tomography |
| MR | Magnetic Resonance |
| MRI | Magnetic Resonance Imaging |
| US | ultrasound |
| OCT | Optical Coherence Tomography |
| PET | Positron Emission computed Tomography |
| DICOM | Digital Imaging and Communications in Medicine |
| ROI | Region of Interest |
| ACM | active contour model |
| CNN | Convolutional Neural Networks |
| RRT* | Rapidly exploring Random Tree Star |
| DT | Distance Transform |
| DL | deep learning |
| FMM | Fast Marching Method |
| GWDT | Gray Weighted Distance Transform |
| ITK | The Insight Toolkit |
| VTK | The Visualization Toolkit |
| MITK | The Medical Imaging Interaction Toolkit |
| TP | true positives |
| FP | false positives |
| TN | true negatives |
| FN | false negatives |
| TPR | true positive rate |
| FPR | false positive rate |
| FNR | false negative rate |
| RMSE | root mean standard error |
| HD | Hausdorff distance |
| SLIVER07 | the Segmentation of the Liver Competition 2007 |
| LiTS | Liver Tumor Segmentation |
| CHAOS | Combined (CT-MR) Healthy Abdominal Organ Segmentation |
| 3D-IRCADb-01 | 3D Image Reconstruction for Comparison of Algorithm Database |
| TCGA-LIHC | The Cancer Genome Atlas Liver Hepatocellular Carcinoma |
| MSD | Medical Segmentation Decathlon |
| ISBI | The IEEE International Symposium on Biomedical Imaging |
| MICCAI | International Conference on Medical Image Computing and Computer |
| Assisted Intervention |
References
- Saha, P.K.; Borgefors, G.; di Baja, G.S. A survey on skeletonization algorithms and their applications. Pattern Recognit. Lett. 2016, 76, 3–12. [Google Scholar] [CrossRef]
- Sherman, T.F. On connecting large vessels to small. The meaning of Murray’s law. J. Gen. Physiol. 1981, 78, 431–453. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mitra, V.; Metcalf, J. Metabolic functions of the liver. Anaesth. Intensive Care Med. 2009, 10, 334–335. [Google Scholar] [CrossRef]
- Ozougwu, J.C. Physiology of the liver. Int. J. Res. Pharm. Biosci. 2017, 4, 13–24. [Google Scholar]
- Williams, R. Global challenges in liver disease. Hepatology 2006, 44, 521–526. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Kisseleva, T.; Brenner, D.A. Aging and liver disease. Curr. Opin. Gastroenterol. 2015, 31, 184. [Google Scholar] [CrossRef] [Green Version]
- Lieber, C.S. Prevention and treatment of liver fibrosis based on pathogenesis. Alcohol. Clin. Exp. Res. 1999, 23, 944–949. [Google Scholar] [CrossRef] [PubMed]
- Targher, G.; Bellis, A.; Fornengo, P.; Ciaravella, F.; Pichiri, I.; Perin, P.C.; Trimarco, B.; Marchesini, G. Prevention and treatment of nonalcoholic fatty liver disease. Dig. Liver Dis. 2010, 42, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Schiff, L.; Schiff, E.R. (Eds.) Diseases of the Liver; Lippincott Philadelphia: Philadelphia, PA, USA, 1993; Volume 2. [Google Scholar]
- Coulon, S.; Heindryckx, F.; Geerts, A.; Van Steenkiste, C.; Colle, I.; Van Vlierberghe, H. Angiogenesis in chronic liver disease and its complications. Liver Int. 2011, 31, 146–162. [Google Scholar] [CrossRef] [PubMed]
- Fernández, M.; Semela, D.; Bruix, J.; Colle, I.; Pinzani, M.; Bosch, J. Angiogenesis in liver disease. J. Hepatol. 2009, 50, 604–620. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Jiang, T.; Lu, F.; Wang, K.; Kong, D. Semiautomatic radiofrequency ablation planning based on constrained clustering process for hepatic tumors. IEEE Trans. Biomed. Eng. 2017, 65, 645–657. [Google Scholar]
- Ng, K.K.; Poon, R.T. Radiofrequency ablation for malignant liver tumor. Surg. Oncol. 2005, 14, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Lu, F.; Wu, F.; Jiang, T.; Xie, L.; Kong, D. An analytical solution for temperature distributions in hepatic radiofrequency ablation incorporating the heat-sink effect of large vessels. Phys. Med. Biol. 2018, 63, 235026. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Zhang, J.; Kong, D.; Lou, Q.; Lu, F. Fast calculation of 3D radiofrequency ablation zone based on a closed-form solution of heat conduction equation fitted by ex vivo measurements. Phys. Med. Biol. 2021, 66, 055022. [Google Scholar] [CrossRef] [PubMed]
- Lebre, M.A.; Vacavant, A.; Grand-Brochier, M.; Merveille, O.; Chabrot, P.; Abergel, A.; Magnin, B. Automatic 3-d skeleton-based segmentation of liver vessels from mri and ct for couinaud representation. In Proceedings of the 2018 25th IEEE International Conference on Image Processing (ICIP), Athens, Greece, 7–10 October 2018; pp. 3523–3527. [Google Scholar]
- Couinaud, C. Le Foie: Études Anatomiques et Chirurgicales; Masson: Issy-les-Moulineaux, France, 1957. [Google Scholar]
- Yan, Z.; Chen, F.; Kong, D. Liver venous tree separation via twin-line RANSAC and Murray’s law. IEEE Trans. Med. Imaging 2017, 36, 1887–1900. [Google Scholar] [CrossRef] [PubMed]
- Selle, D.; Preim, B.; Schenk, A.; Peitgen, H.O. Analysis of vasculature for liver surgical planning. IEEE Trans. Med. Imaging 2002, 21, 1344–1357. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Guindalini, F.D.; Botelho, M.P.; Harmath, C.B.; Sandrasegaran, K.; Miller, F.H.; Salem, R.; Yaghmai, V. Assessment of liver tumor response to therapy: Role of quantitative imaging. Radiographics 2013, 33, 1781–1800. [Google Scholar] [CrossRef]
- Padma, S.; Martinie, J.B.; Iannitti, D.A. Liver tumor ablation: Percutaneous and open approaches. J. Surg. Oncol. 2009, 100, 619–634. [Google Scholar] [CrossRef]
- Vijayan, S.; Reinertsen, I.; Hofstad, E.F.; Rethy, A.; Hernes, T.A.N.; Langø, T. Liver deformation in an animal model due to pneumoperitoneum assessed by a vessel-based deformable registration. Minim. Invasive Ther. Allied Technol. 2014, 23, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Lange, T.; Eulenstein, S.; Hünerbein, M.; Schlag, P.M. Vessel-based non-rigid registration of MR/CT and 3D ultrasound for navigation in liver surgery. Comput. Aided Surg. 2003, 8, 228–240. [Google Scholar] [CrossRef] [PubMed]
- Dawkins, R.; Davis, N. The Selfish Gene; Macat Library: London, UK, 2017. [Google Scholar]
- Gardner, A.; Welch, J.J. A formal theory of the selfish gene. J. Evol. Biol. 2011, 24, 1801–1813. [Google Scholar] [CrossRef]
- Cohn, D.L. Optimal systems: I. The vascular system. Bull. Math. Biophys. 1954, 16, 59–74. [Google Scholar] [CrossRef]
- Karch, R.; Neumann, F.; Neumann, M.; Schreiner, W. Staged growth of optimized arterial model trees. Ann. Biomed. Eng. 2000, 28, 495–511. [Google Scholar] [CrossRef] [PubMed]
- Carpenter, B.; Lin, Y.; Stoll, S.; Raffai, R.L.; McCuskey, R.; Wang, R. VEGF is crucial for the hepatic vascular development required for lipoprotein uptake. Development 2005, 132, 3293–3303. [Google Scholar] [CrossRef] [Green Version]
- Schwen, L.O.; Preusser, T. Analysis and algorithmic generation of hepatic vascular systems. Int. J. Hepatol. 2012, 2012, 357687. [Google Scholar] [CrossRef]
- McCulloh, K.A.; Sperry, J.S.; Adler, F.R. Water transport in plants obeys Murray’s law. Nature 2003, 421, 939–942. [Google Scholar] [CrossRef]
- Murray, C.D. The physiological principle of minimum work: I. The vascular system and the cost of blood volume. Proc. Natl. Acad. Sci. USA 1926, 12, 207. [Google Scholar] [CrossRef] [Green Version]
- Murray, C.D. The physiological principle of minimum work: II. Oxygen exchange in capillaries. Proc. Natl. Acad. Sci. USA 1926, 12, 299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sutera, S.P.; Skalak, R. The history of Poiseuille’s law. Annu. Rev. Fluid Mech. 1993, 25, 1–20. [Google Scholar] [CrossRef]
- Acheson, D.; Acheson, F.D. Elementary Fluid Dynamics; Oxford University Press: Oxford, UK, 1990. [Google Scholar]
- Hamarneh, G.; Jassi, P. VascuSynth: Simulating vascular trees for generating volumetric image data with ground-truth segmentation and tree analysis. Comput. Med. Imaging Graph. 2010, 34, 605–616. [Google Scholar] [CrossRef] [Green Version]
- Jassi, P.; Hamarneh, G. Vascusynth: Vascular tree synthesis software. Insight J. 2011. [Google Scholar] [CrossRef]
- Zhang, J.; Chang, W.; Wu, F.; Kong, D. Pixel-RRT*: A Novel Skeleton Trajectory Search Algorithm for Hepatic Vessels. In Proceedings of the 2020 Digital Image Computing: Techniques and Applications (DICTA), Melbourne, Australia, 29 November–2 December 2020; pp. 1–8. [Google Scholar]
- Du, G.; Cao, X.; Liang, J.; Chen, X.; Zhan, Y. Medical image segmentation based on u-net: A review. J. Imaging Sci. Technol. 2020, 64, 20508. [Google Scholar] [CrossRef]
- Erdt, M.; Raspe, M.; Suehling, M. Automatic hepatic vessel segmentation using graphics hardware. In International Workshop on Medical Imaging and Virtual Reality; Springer: Berlin/Heidelberg, Germany, 2008; pp. 403–412. [Google Scholar]
- Conversano, F.; Franchini, R.; Demitri, C.; Massoptier, L.; Montagna, F.; Maffezzoli, A.; Malvasi, A.; Casciaro, S. Hepatic vessel segmentation for 3D planning of liver surgery: Experimental evaluation of a new fully automatic algorithm. Acad. Radiol. 2011, 18, 461–470. [Google Scholar] [CrossRef]
- Yan, Q.; Wang, B.; Zhang, W.; Luo, C.; Xu, W.; Xu, Z.; Zhang, Y.; Shi, Q.; Zhang, L.; You, Z. Attention-Guided Deep Neural Network With Multi-Scale Feature Fusion for Liver Vessel Segmentation. IEEE J. Biomed. Health Inform. 2020, 25, 2629–2642. [Google Scholar] [CrossRef] [PubMed]
- Luu, H.M.; Klink, C.; Moelker, A.; Niessen, W.; Van Walsum, T. Quantitative evaluation of noise reduction and vesselness filters for liver vessel segmentation on abdominal CTA images. Phys. Med. Biol. 2015, 60, 3905. [Google Scholar] [CrossRef] [PubMed]
- Kitrungrotsakul, T.; Han, X.H.; Iwamoto, Y.; Lin, L.; Foruzan, A.H.; Xiong, W.; Chen, Y.W. VesselNet: A deep convolutional neural network with multi pathways for robust hepatic vessel segmentation. Comput. Med. Imaging Graph. 2019, 75, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Su, J.; Liu, Z.; Zhang, J.; Sheng, V.S.; Song, Y.; Zhu, Y.; Liu, Y. DV-Net: Accurate liver vessel segmentation via dense connection model with D-BCE loss function. Knowl.-Based Syst. 2021, 232, 107471. [Google Scholar] [CrossRef]
- Lu, S.; Huang, H.; Liang, P.; Chen, G.; Xiao, L. Hepatic vessel segmentation using variational level set combined with non-local robust statistics. Magn. Reson. Imaging 2017, 36, 180–186. [Google Scholar] [CrossRef]
- Goceri, E.; Shah, Z.K.; Gurcan, M.N. Vessel segmentation from abdominal magnetic resonance images: Adaptive and reconstructive approach. Int. J. Numer. Methods Biomed. Eng. 2017, 33, e2811. [Google Scholar] [CrossRef] [PubMed]
- Marcan, M.; Pavliha, D.; Music, M.M.; Fuckan, I.; Magjarevic, R.; Miklavcic, D. Segmentation of hepatic vessels from MRI images for planning of electroporation-based treatments in the liver. Radiol. Oncol. 2014, 48, 267–281. [Google Scholar] [CrossRef] [Green Version]
- Liu, M.; Vanguri, R.; Mutasa, S.; Ha, R.; Liu, Y.C.; Button, T.; Jambawalikar, S. Channel width optimized neural networks for liver and vessel segmentation in liver iron quantification. Comput. Biol. Med. 2020, 122, 103798. [Google Scholar] [CrossRef] [PubMed]
- Goceri, E. Automatic labeling of portal and hepatic veins from MR images prior to liver transplantation. Int. J. Comput. Assist. Radiol. Surg. 2016, 11, 2153–2161. [Google Scholar] [CrossRef] [PubMed]
- Ivashchenko, O.V.; Rijkhorst, E.J.; Ter Beek, L.C.; Hoetjes, N.J.; Pouw, B.; Nijkamp, J.; Kuhlmann, K.F.; Ruers, T.J. A workflow for automated segmentation of the liver surface, hepatic vasculature and biliary tree anatomy from multiphase MR images. Magn. Reson. Imaging 2020, 68, 53–65. [Google Scholar] [CrossRef]
- Thomson, B.R.; Nijkamp, J.; Ivashchenko, O.; van der Heijden, F.; Smit, J.N.; Kok, N.F.; Kuhlmann, K.F.; Ruers, T.J.; Fusaglia, M. Hepatic vessel segmentation using a reduced filter 3D U-Net in ultrasound imaging. arXiv 2019, arXiv:1907.12109. [Google Scholar]
- Thomson, B.R.; Smit, J.N.; Ivashchenko, O.V.; Kok, N.F.; Kuhlmann, K.F.; Ruers, T.J.; Fusaglia, M. MR-to-US registration using multiclass segmentation of hepatic vasculature with a reduced 3D U-Net. In International Conference on Medical Image Computing and Computer-Assisted Intervention; Springer: Cham, Switzerland, 2020; pp. 275–284. [Google Scholar]
- Mishra, D.; Chaudhury, S.; Sarkar, M.; Manohar, S.; Soin, A.S. Segmentation of vascular regions in ultrasound images: A deep learning approach. In Proceedings of the 2018 IEEE International Symposium on Circuits and Systems (ISCAS), Florence, Italy, 27–30 May 2018; pp. 1–5. [Google Scholar]
- Kawajiri, S.; Zhou, X.; Zhang, X.; Hara, T.; Fujita, H.; Yokoyama, R.; Kondo, H.; Kanematsu, M.; Hoshi, H. Automated segmentation of hepatic vessels in non-contrast X-ray CT images. Radiol. Phys. Technol. 2008, 1, 214–222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chu, P.; Pang, Y.; Cheng, E.; Zhu, Y.; Zheng, Y.; Ling, H. Structure-aware rank-1 tensor approximation for curvilinear structure tracking using learned hierarchical features. In International Conference on Medical Image Computing and Computer-Assisted Intervention; Springer: Cham, Switzerland, 2016; pp. 413–421. [Google Scholar]
- Heimann, T.; Van Ginneken, B.; Styner, M.A.; Arzhaeva, Y.; Aurich, V.; Bauer, C.; Beck, A.; Becker, C.; Beichel, R.; Bekes, G.; et al. Comparison and evaluation of methods for liver segmentation from CT datasets. IEEE Trans. Med. Imaging 2009, 28, 1251–1265. [Google Scholar] [CrossRef] [PubMed]
- Bilic, P.; Christ, P.F.; Vorontsov, E.; Chlebus, G.; Chen, H.; Dou, Q.; Fu, C.W.; Han, X.; Heng, P.A.; Hesser, J.; et al. The liver tumor segmentation benchmark (lits). arXiv 2019, arXiv:1901.04056. [Google Scholar]
- Kavur, A.E.; Gezer, N.S.; Barış, M.; Aslan, S.; Conze, P.H.; Groza, V.; Pham, D.D.; Chatterjee, S.; Ernst, P.; Özkan, S.; et al. CHAOS challenge-combined (CT-MR) healthy abdominal organ segmentation. Med. Image Anal. 2021, 69, 101950. [Google Scholar] [CrossRef]
- Simpson, A.L.; Antonelli, M.; Bakas, S.; Bilello, M.; Farahani, K.; Van Ginneken, B.; Kopp-Schneider, A.; Landman, B.A.; Litjens, G.; Menze, B.; et al. A large annotated medical image dataset for the development and evaluation of segmentation algorithms. arXiv 2019, arXiv:1902.09063. [Google Scholar]
- Soler, L.; Hostettler, A.; Agnus, V.; Charnoz, A.; Fasquel, J.; Moreau, J.; Osswald, A.; Bouhadjar, M.; Marescaux, J. 3D Image Reconstruction for Comparison of Algorithm Database: A Patient Specific Anatomical and Medical Image Database; IRCAD: Strasbourg, France, 2010. [Google Scholar]
- Erickson, B.; Kirk, S.; Lee, Y.; Bathe, O.; Kearns, M.; Gerdes, C.; Rieger-Christ, K.; Lemmerman, J. Radiology data from the cancer genome atlas liver hepatocellular carcinoma [TCGA-LIHC] collection. Cancer Imaging Arch. 2016. Available online: https://wiki.cancerimagingarchive.net/display/Public/TCGA-LIHC (accessed on 16 February 2022).
- Cheema, M.N.; Nazir, A.; Yang, P.; Sheng, B.; Li, P.; Li, H.; Wei, X.; Qin, J.; Kim, J.; Feng, D.D. Modified GAN-CAED to Minimize Risk of Unintentional Liver Major Vessels Cutting by Controlled Segmentation Using CTA/SPET-CT. IEEE Trans. Ind. Inform. 2021, 17, 7991–8002. [Google Scholar] [CrossRef]
- Montaña-Brown, N.; Ramalhinho, J.; Allam, M.; Davidson, B.; Hu, Y.; Clarkson, M.J. Vessel segmentation for automatic registration of untracked laparoscopic ultrasound to CT of the liver. Int. J. Comput. Assist. Radiol. Surg. 2021, 16, 1151–1160. [Google Scholar] [CrossRef] [PubMed]
- Dice, L.R. Measures of the amount of ecologic association between species. Ecology 1945, 26, 297–302. [Google Scholar] [CrossRef]
- Bradley, A.P. The use of the area under the ROC curve in the evaluation of machine learning algorithms. Pattern Recognit. 1997, 30, 1145–1159. [Google Scholar] [CrossRef] [Green Version]
- Metz, C.E. Basic principles of ROC analysis. In Seminars in Nuclear Medicine; Elsevier: Amsterdam, The Netherlands, 1978; Volume 8, pp. 283–298. [Google Scholar]
- Burke, D.S.; Brundage, J.F.; Redfield, R.R.; Damato, J.J.; Schable, C.A.; Putman, P.; Visintine, R.; Kim, H.I. Measurement of the false positive rate in a screening program for human immunodeficiency virus infections. N. Engl. J. Med. 1988, 319, 961–964. [Google Scholar] [CrossRef]
- Brejl, M.; Sonka, M. Object localization and border detection criteria design in edge-based image segmentation: Automated learning from examples. IEEE Trans. Med. Imaging 2000, 19, 973–985. [Google Scholar] [PubMed]
- Ibanez, L. The ITK Software Guide. 2003. Available online: http://www.itk.org/ItkSoftwareGuide.pdf (accessed on 16 February 2022).
- Schroeder, W.J.; Avila, L.S.; Hoffman, W. Visualizing with VTK: A tutorial. IEEE Comput. Graph. Appl. 2000, 20, 20–27. [Google Scholar] [CrossRef] [Green Version]
- Wolf, I.; Vetter, M.; Wegner, I.; Nolden, M.; Bottger, T.; Hastenteufel, M.; Schobinger, M.; Kunert, T.; Meinzer, H.P. The medical imaging interaction toolkit (MITK): A toolkit facilitating the creation of interactive software by extending VTK and ITK. In Medical Imaging 2004: Visualization, Image-Guided Procedures, and Display; SPIE: Bellingham, WA, USA, 2004; Volume 5367, pp. 16–27. [Google Scholar]
- Moccia, S.; De Momi, E.; El Hadji, S.; Mattos, L.S. Blood vessel segmentation algorithms—Review of methods, datasets and evaluation metrics. Comput. Methods Programs Biomed. 2018, 158, 71–91. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kirbas, C.; Quek, F. A review of vessel extraction techniques and algorithms. ACM Comput. Surv. (CSUR) 2004, 36, 81–121. [Google Scholar] [CrossRef]
- Ciecholewski, M.; Kassjański, M. Computational methods for liver vessel segmentation in medical imaging: A review. Sensors 2021, 21, 2027. [Google Scholar] [CrossRef] [PubMed]
- Rudyanto, R.D.; Kerkstra, S.; Van Rikxoort, E.M.; Fetita, C.; Brillet, P.Y.; Lefevre, C.; Xue, W.; Zhu, X.; Liang, J.; Öksüz, I.; et al. Comparing algorithms for automated vessel segmentation in computed tomography scans of the lung: The VESSEL12 study. Med. Image Anal. 2014, 18, 1217–1232. [Google Scholar] [CrossRef] [Green Version]
- Frangi, A.F.; Niessen, W.J.; Vincken, K.L.; Viergever, M.A. Multiscale vessel enhancement filtering. In International Conference on Medical Image Computing And Computer-Assisted Intervention; Springer: Berlin/Heidelberg, Germany, 1998; pp. 130–137. [Google Scholar]
- Pock, T.; Janko, C.; Beichel, R.; Bischof, H. Multiscale medialness for robust segmentation of 3d tubular structures. In Proceedings of the Computer Vision Winter Workshop, Zell an der Pram, Austria, 2–4 February 2005; Volume 2005. [Google Scholar]
- Kass, M.; Witkin, A.; Terzopoulos, D. Snakes: Active contour models. Int. J. Comput. Vis. 1988, 1, 321–331. [Google Scholar] [CrossRef]
- Pratondo, A.; Chui, C.K.; Ong, S.H. Robust edge-stop functions for edge-based active contour models in medical image segmentation. IEEE Signal Process. Lett. 2015, 23, 222–226. [Google Scholar] [CrossRef]
- Sethian, J.A. A fast marching level set method for monotonically advancing fronts. Proc. Natl. Acad. Sci. USA 1996, 93, 1591–1595. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Matuszewski, B.J.; Shark, L.K.; Moore, C.J. Medical image segmentation using new hybrid level-set method. In Proceedings of the 2008 Fifth International Conference Biomedical Visualization: Information Visualization in Medical and Biomedical Informatics, London, UK, 9–11 July 2008; pp. 71–76. [Google Scholar]
- Alhonnoro, T.; Pollari, M.; Lilja, M.; Flanagan, R.; Kainz, B.; Muehl, J.; Mayrhauser, U.; Portugaller, H.; Stiegler, P.; Tscheliessnigg, K. Vessel segmentation for ablation treatment planning and simulation. In International Conference on Medical Image Computing and Computer-Assisted Intervention; Springer: Berlin/Heidelberg, Germany, 2010; pp. 45–52. [Google Scholar]
- Wan, S.Y.; Higgins, W.E. Symmetric region growing. IEEE Trans. Image Process. 2003, 12, 1007–1015. [Google Scholar]
- Adams, R.; Bischof, L. Seeded region growing. IEEE Trans. Pattern Anal. Mach. Intell. 1994, 16, 641–647. [Google Scholar] [CrossRef] [Green Version]
- Sangsefidi, N.; Foruzan, A.H.; Dolati, A. Balancing the data term of graph-cuts algorithm to improve segmentation of hepatic vascular structures. Comput. Biol. Med. 2018, 93, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Jegelka, S.; Bilmes, J. Submodularity beyond submodular energies: Coupling edges in graph cuts. In Proceedings of the CVPR 2011, Colorado Springs, CO, USA, 20–25 June 2011; pp. 1897–1904. [Google Scholar]
- Boykov, Y.; Funka-Lea, G. Graph cuts and efficient ND image segmentation. Int. J. Comput. Vis. 2006, 70, 109–131. [Google Scholar] [CrossRef] [Green Version]
- Keshwani, D.; Kitamura, Y.; Ihara, S.; Iizuka, S.; Simo-Serra, E. TopNet: Topology preserving metric learning for vessel tree reconstruction and labelling. In International Conference on Medical Image Computing and Computer-Assisted Intervention; Springer: Cham, Switzerland, 2020; pp. 14–23. [Google Scholar]
- Huang, Q.; Sun, J.; Ding, H.; Wang, X.; Wang, G. Robust liver vessel extraction using 3D U-Net with variant dice loss function. Comput. Biol. Med. 2018, 101, 153–162. [Google Scholar] [CrossRef]
- Ronneberger, O.; Fischer, P.; Brox, T. U-net: Convolutional networks for biomedical image segmentation. In International Conference on Medical Image Computing and Computer-Assisted Intervention; Springer: Cham, Switzerland, 2015; pp. 234–241. [Google Scholar]
- Isensee, F.; Petersen, J.; Klein, A.; Zimmerer, D.; Jaeger, P.F.; Kohl, S.; Wasserthal, J.; Koehler, G.; Norajitra, T.; Wirkert, S.; et al. nnu-net: Self-adapting framework for u-net-based medical image segmentation. arXiv 2018, arXiv:1809.10486. [Google Scholar]
- Paetzold, J.C.; Shit, S.; Ezhov, I.; Tetteh, G.; Ertürk, A.; Munich, H.Z.; Menze, B. clDice—A novel connectivity-preserving loss function for vessel segmentation. In Proceedings of the 33rd Conference on Neural Information Processing Systems (NeurIPS 2019), Vancouver, Canada, 8–14 December 2019. [Google Scholar]
- Chu, J.; Chen, Y.; Zhou, W.; Shi, H.; Cao, Y.; Tu, D.; Jin, R.; Xu, Y. Pay more attention to discontinuity for medical image segmentation. In International Conference on Medical Image Computing and Computer-Assisted Intervention; Springer: Cham, Switzerland, 2020; pp. 166–175. [Google Scholar]
- Wang, Y.; Wei, X.; Liu, F.; Chen, J.; Zhou, Y.; Shen, W.; Fishman, E.K.; Yuille, A.L. Deep distance transform for tubular structure segmentation in ct scans. In Proceedings of the IEEE/CVF Conference on Computer Vision and Pattern Recognition, Seattle, WA, USA, 13–19 June 2020; pp. 3833–3842. [Google Scholar]
- Rajpurkar, P.; Chen, E.; Banerjee, O.; Topol, E.J. AI in health and medicine. Nat. Med. 2022, 28, 31–38. [Google Scholar] [CrossRef]
- Krig, S. Computer Vision Metrics; Springer: Berlin, Germany, 2016. [Google Scholar]
- Remy-Jardin, M.; Remy, J.; Artaud, D.; Deschildre, F.; Duhamel, A. Diffuse infiltrative lung disease: Clinical value of sliding-thin-slab maximum intensity projection CT scans in the detection of mild micronodular patterns. Radiology 1996, 200, 333–339. [Google Scholar] [CrossRef]
- Sorantin, E.; Halmai, C.; Erdohelyi, B.; Palágyi, K.; Nyúl, L.G.; Ollé, K.; Geiger, B.; Lindbichler, F.; Friedrich, G.; Kiesler, K. Spiral-CT-based assessment of tracheal stenoses using 3-D-skeletonization. IEEE Trans. Med. Imaging 2002, 21, 263–273. [Google Scholar] [CrossRef]
- Jang, B.K.; Chin, R.T. Analysis of thinning algorithms using mathematical morphology. IEEE Trans. Pattern Anal. Mach. Intell. 1990, 12, 541–551. [Google Scholar] [CrossRef]
- Lee, T.C.; Kashyap, R.L.; Chu, C.N. Building skeleton models via 3-D medial surface axis thinning algorithms. CVGIP Graph. Model. Image Process. 1994, 56, 462–478. [Google Scholar] [CrossRef]
- Guo, X.; Xiao, R.; Zhang, T.; Chen, C.; Wang, J.; Wang, Z. A novel method to model hepatic vascular network using vessel segmentation, thinning, and completion. Med. Biol. Eng. Comput. 2020, 58, 709–724. [Google Scholar] [CrossRef]
- Homann, H. Implementation of a 3D Thinning Algorithm. Insight J. 2007. Available online: https://hodad.bioen.utah.edu/devbuilds/biomesh3d/FEMesher/references/ITKbinaryThinningImageFilter3D.pdf (accessed on 16 February 2022).
- Culjak, I.; Abram, D.; Pribanic, T.; Dzapo, H.; Cifrek, M. A brief introduction to OpenCV. In Proceedings of the 2012 35th International Convention MIPRO, Opatija, Croatia, 21–25 May 2012; pp. 1725–1730. [Google Scholar]
- Ogniewicz, R.L.; Ilg, M. Voronoi skeletons: Theory and applications. CVPR 1992, 92, 63–69. [Google Scholar]
- Mady, A.M.; Omar, K. A comparative study of Voronoi algorithm construction in thinning. In Proceedings of the 2011 International Conference on Electrical Engineering and Informatics, Bandung, Indonesia, 17–19 July 2011; pp. 1–5. [Google Scholar]
- Grevera, G.J. Distance transform algorithms and their implementation and evaluation. In Deformable Models; Springer: New York, NY, USA, 2007; pp. 33–60. [Google Scholar]
- Pudney, C. Distance-ordered homotopic thinning: A skeletonization algorithm for 3D digital images. Comput. Vis. Image Underst. 1998, 72, 404–413. [Google Scholar] [CrossRef]
- Zhang, T.Y.; Suen, C.Y. A fast parallel algorithm for thinning digital patterns. Commun. ACM 1984, 27, 236–239. [Google Scholar] [CrossRef]
- Saeed, K.; Tabędzki, M.; Rybnik, M.; Adamski, M. K3M: A universal algorithm for image skeletonization and a review of thinning techniques. Int. J. Appl. Math. Comput. Sci. 2010, 20, 317–335. [Google Scholar] [CrossRef]
- Dijkstra, E.W. A note on two problems in connexion with graphs. Numer. Math. 1959, 1, 269–271. [Google Scholar] [CrossRef] [Green Version]
- Park, K.; Sim, S.; Bae, H. Vessel estimated time of arrival prediction system based on a path-finding algorithm. Marit. Transp. Res. 2021, 2, 100012. [Google Scholar] [CrossRef]
- Hart, P.E.; Nilsson, N.J.; Raphael, B. A formal basis for the heuristic determination of minimum cost paths. IEEE Trans. Syst. Sci. Cybern. 1968, 4, 100–107. [Google Scholar] [CrossRef]
- He, J.; Pan, C.; Yang, C.; Zhang, M.; Wang, Y.; Zhou, X.; Yu, Y. Learning hybrid representations for automatic 3D vessel centerline extraction. In International Conference on Medical Image Computing and Computer-Assisted Intervention; Springer: Cham, Switzerland, 2020; pp. 24–34. [Google Scholar]
- Liu, M.; Ren, J. A Global Path Planning Approach based on Improved A* Algorithm for Intelligent Vessel Navigating. In Proceedings of the 2021 International Conference on Security, Pattern Analysis, and Cybernetics (SPAC), Chengdu, China, 18–20 June 2021; pp. 244–248. [Google Scholar]
- Zhang, Z.; Fang, C.; Zhang, J.; Qian, J.; Su, Q. Traffic Guidance Based on LSTM Neural Network and Dual Tracking Dijkstra Algorithm. In Proceedings of the 2020 IEEE 3rd International Conference on Information Systems and Computer Aided Education (ICISCAE), Dalian, China, 27–29 September 2020; pp. 497–503. [Google Scholar]
- Yonetani, R.; Taniai, T.; Barekatain, M.; Nishimura, M.; Kanezaki, A. Path planning using neural a* search. In Proceedings of the 38th International Conference on Machine Learning, PMLR, Virtual Conference, 18–24 July 2021; Volume 139, pp. 12029–12039. [Google Scholar]
- LaValle, S.M. Rapidly-Exploring Random Trees: A New tool For Path Planning. Computer ence Dept. 1998. Available online: https://www.cs.csustan.edu/~xliang/Courses/CS4710-21S/Papers/06%20RRT.pdf (accessed on 16 February 2022).
- Karaman, S.; Frazzoli, E. Sampling-based algorithms for optimal motion planning. Int. J. Robot. Res. 2011, 30, 846–894. [Google Scholar] [CrossRef]
- Klemm, S.; Oberländer, J.; Hermann, A.; Roennau, A.; Schamm, T.; Zollner, J.M.; Dillmann, R. RRT*-Connect: Faster, asymptotically optimal motion planning. In Proceedings of the 2015 IEEE international conference on robotics and biomimetics (ROBIO), Zhuhai, China, 6–9 December 2015; pp. 1670–1677. [Google Scholar]
- Gammell, J.D.; Srinivasa, S.S.; Barfoot, T.D. Informed RRT*: Optimal sampling-based path planning focused via direct sampling of an admissible ellipsoidal heuristic. In Proceedings of the 2014 IEEE/RSJ International Conference on Intelligent Robots and Systems, Chicago, IL, USA, 14–18 September 2014; pp. 2997–3004. [Google Scholar]
- Naderi, K.; Rajamäki, J.; Hämäläinen, P. RT-RRT* a real-time path planning algorithm based on RRT. In Proceedings of the 8th ACM SIGGRAPH Conference on Motion in Games, Paris, France, 16–18 November 2015; pp. 113–118. [Google Scholar]
- Chen, Y.; Yue, X.; Zhong, C.; Wang, G. Functional region annotation of liver CT image based on vascular tree. BioMed Res. Int. 2016, 2016, 5428737. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, M.; Lee, J.; Chung, J.W.; Shin, Y.G. Accurate liver vessel segmentation via active contour model with dense vessel candidates. Comput. Methods Programs Biomed. 2018, 166, 61–75. [Google Scholar] [CrossRef]
- Pan, J.; Zhang, J.; Luo, S.; Zhang, J.; Liang, Y. Automatic annotation of liver computed tomography images based on a vessel-skeletonization method. Int. J. Imaging Syst. Technol. 2020, 30, 704–715. [Google Scholar] [CrossRef]
- Kim, J.; Lee, J.; Chung, J.W.; Shin, Y.G. Locally adaptive 2D–3D registration using vascular structure model for liver catheterization. Comput. Biol. Med. 2016, 70, 119–130. [Google Scholar] [CrossRef]
- Zhao, Y.; Qi, H.; Li, D.; Jin, J.; Wang, Q. Biopsy Needle Puncture Path Planning Method based on 3D Ultrasound Images. In Proceedings of the 2018 14th IEEE International Conference on Signal Processing (ICSP), Beijing, China, 12–16 August 2018; pp. 1169–1174. [Google Scholar]
- Sangsefidi, N.; Foruzan, A.H.; Dolati, A.; Chen, Y.W. Incorporating a locally estimated appearance model in the graphcuts algorithm to extract small hepatic vessels. In Proceedings of the 2017 IEEE International Conference on Image Processing (ICIP), Beijing, China, 17–20 September 2017; pp. 2324–2328. [Google Scholar]
- Dagon, B.; Baur, C.; Bettschart, V. A framework for intraoperative update of 3D deformable models in liver surgery. In Proceedings of the 2008 30th Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Vancouver, BC, Canada, 20–25 August 2008; pp. 3235–3238. [Google Scholar]
- Drechsler, K.; Laura, C.O. Hierarchical decomposition of vessel skeletons for graph creation and feature extraction. In Proceedings of the 2010 IEEE international conference on bioinformatics and biomedicine (BIBM), Hong Kong, China, 18–21 December 2010; pp. 456–461. [Google Scholar]
- Merveille, O.; Naegel, B.; Talbot, H.; Najman, L.; Passat, N. 2D filtering of curvilinear structures by ranking the orientation responses of path operators (RORPO). Image Process. On Line 2017, 7, 246–261. [Google Scholar] [CrossRef] [Green Version]
- Ibragimov, B.; Toesca, D.; Chang, D.; Koong, A.; Xing, L. Combining deep learning with anatomical analysis for segmentation of the portal vein for liver SBRT planning. Phys. Med. Biol. 2017, 62, 8943. [Google Scholar] [CrossRef]
- Sato, Y.; Nakajima, S.; Atsumi, H.; Koller, T.; Gerig, G.; Yoshida, S.; Kikinis, R. 3D multi-scale line filter for segmentation and visualization of curvilinear structures in medical images. In CVRMed-MRCAS’97; Springer: Berlin/Heidelberg, Germany, 1997; pp. 213–222. [Google Scholar]
- Mueller, D. Fast marching minimal path extraction in ITK. Insight J. 2008, 1–8. [Google Scholar] [CrossRef]
- Wang, L.; Schnurr, A.K.; Zidowitz, S.; Georgii, J.; Zhao, Y.; Razavi, M.; Schwier, M.; Hahn, H.K.; Hansen, C. Segmentation of hepatic artery in multi-phase liver CT using directional dilation and connectivity analysis. In Medical Imaging 2016: Computer-Aided Diagnosis; International Society for Optics and Photonics: Bellingham, WA, USA, 2016; Volume 9785, p. 97851P. [Google Scholar]
- Wu, Z.; Guo, X.; Huang, X.; Huang, S. A liver vessel skeleton line reconstruction method based on linear interpolation. In Proceedings of the 2013 International Conference on Virtual Reality and Visualization, Xi’an, China, 14–15 September 2013; pp. 257–260. [Google Scholar]
- Kang, X.; Zhao, Q.; Sharma, K.; Shekhar, R.; Wood, B.J.; Linguraru, M.G. Automatic labeling of liver veins in CT by probabilistic backward tracing. In Proceedings of the 2014 IEEE 11th International Symposium on Biomedical Imaging (ISBI), Beijing, China, 29 April–2 May 2014; pp. 1115–1118. [Google Scholar]
- Cui, H.; Xia, Y. Automatic coronary centerline extraction using gradient vector flow field and fast marching method from CT images. IEEE Access 2018, 6, 41816–41826. [Google Scholar] [CrossRef]
- Sethian, J.A. Fast marching methods. SIAM Rev. 1999, 41, 199–235. [Google Scholar] [CrossRef]
- Qian, K.; Cao, S.; Bhattacharya, P. Skeletonization of gray-scale images by gray weighted distance transform. In Visual Information Processing VI; SPIE: Bellingham, WA, USA, 1997; Volume 3074, pp. 224–228. [Google Scholar]
- Sethian, J.A.; Vladimirsky, A. Fast methods for the Eikonal and related Hamilton–Jacobi equations on unstructured meshes. Proc. Natl. Acad. Sci. USA 2000, 97, 5699–5703. [Google Scholar] [CrossRef] [Green Version]
- Soille, P. Generalized geodesy via geodesic time. Pattern Recognit. Lett. 1994, 15, 1235–1240. [Google Scholar] [CrossRef]
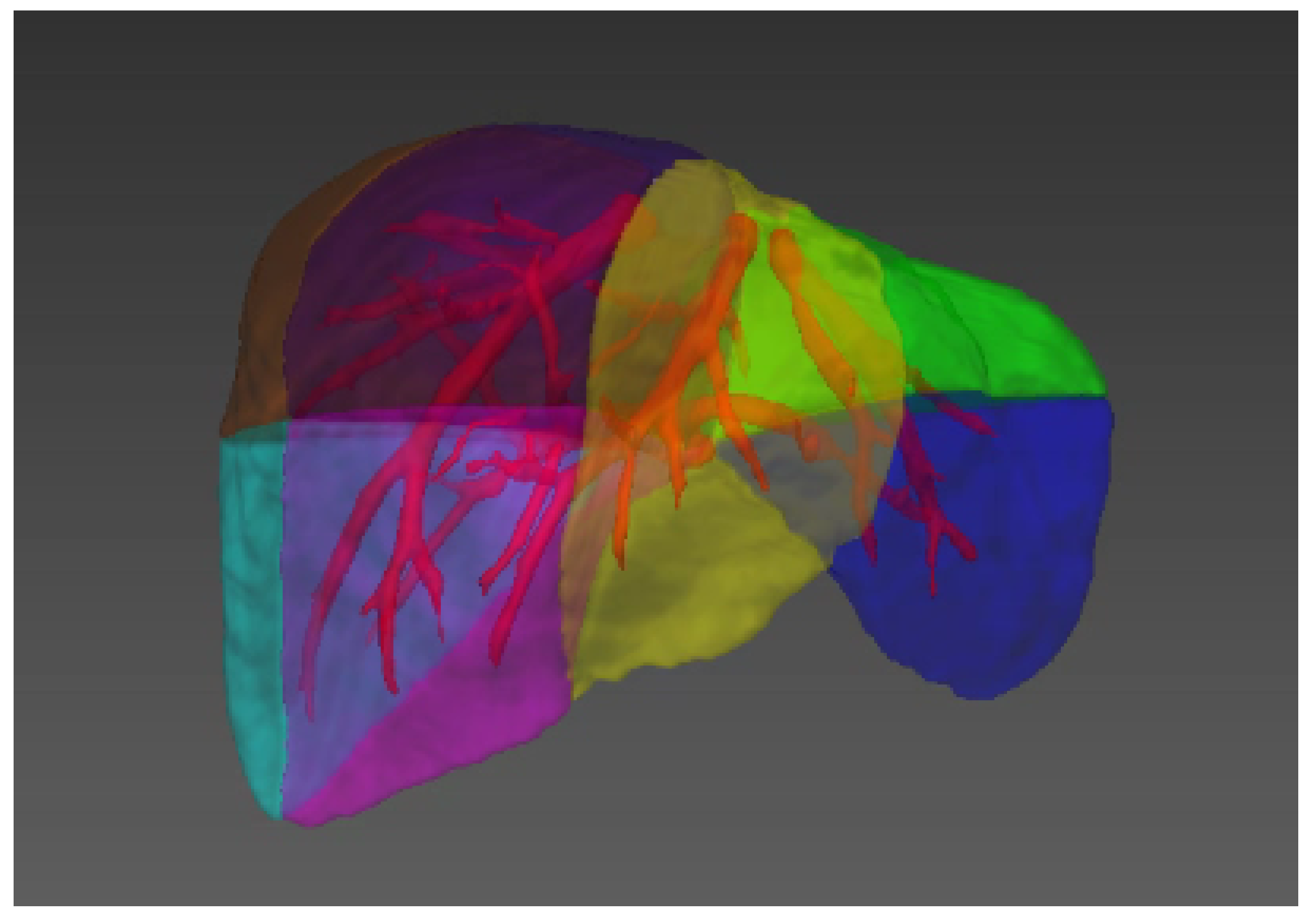
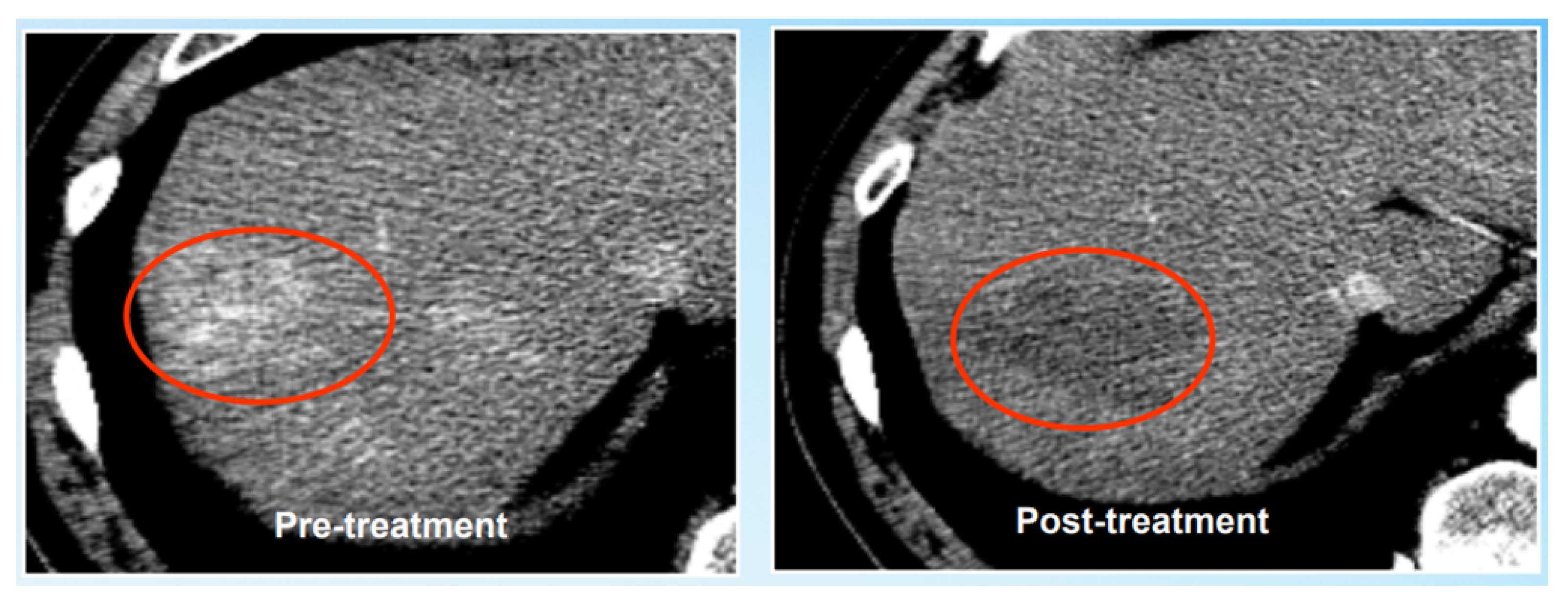

| Imaging System | Imaging Method | Imaging Basis | Advantage |
|---|---|---|---|
| CT | Mathematics reconstruction | Absorption coefficient | High density resolution |
| MRI | Mathematics reconstruction | A variety of parameters | Multiple functions |
| US | Mathematics reconstruction | Acoustic impedance interface | Safe, dynamic and repetitive |
| OCT | Mathematics reconstruction | Based on interferometer principle | High resolution |
| PET | Mathematics reconstruction | Using positron radionuclide labeling | Accurate location and high clinical value |
| X-ray | Transmission projection | Density and thickness | Strong penetrability |
| Name | Time | Modality | File Format | Number |
|---|---|---|---|---|
| MICCAI-Sliver07 [56] | 2007 | CT | MetaImage | 20 Training + 10 Testing |
| LiTS [57] | 2017 | CT | Nifti | 130 Training + 70 Testing |
| CHAOS [58] | 2019 | CT+MR | DICOM | 40 CT+120 MRI |
| Vascular Synthesizer [35] | 2013 | 3D synthetic data | MetaImage | 120 |
| MSD-Task08 [59] | 2018 | CT | Nifti | 303 Training + 140 Testing |
| 3D-IRCADb-01 [60] | 2010 | CT | DICOM | 20 Training + 2 Testing |
| TCGA-LIHC [61] | 2016 | CT+MRI+PET | DICOM | 237 |
| Metrics | Formula | Description |
|---|---|---|
| Dice [64] | Similarity between two sample sets. | |
| Accuracy [65] | Proportion of detected true samples that are actually true. | |
| Sensitivity; recall; true positive rate (TPR) [66] | Proportion of positives that are correctly identified. | |
| Specificity [66] | Proportion of negatives that are correctly identified. | |
| False positive rate () [67] | Ratio of the number of negative samples wrongly categorized as positive () to the total number of actual negative samples. | |
| False negative rate () [67] | Ratio of the number of positive samples wrongly categorized as negative () to the total number of actual positive samples. | |
| Root mean standard error () [68] | Measure of the average squared difference between the result R and the actual value T (ground truth), where denotes the distances from points R to points T. | |
| Hausdorff distance (HD) [60] | Overlapping index, which measures the largest Euclidean distance between two contours A and B and vice versa, computed over all pixels of each curve. |
| Methods | Datasets | Dice (%) | Accuracy (%) | Sensitivity (%) |
|---|---|---|---|---|
| Paetzold et al., 2019 [92] | Vascular Synth | 98.73 | 99.94 | - |
| Wang et al., 2020 [94] | MSD-Task08 | 63.43 | - | - |
| Kitrungrotsakul et al., 2019 [43] | 3D-IRCADb-01 | 87.9 | - | 91.8 |
| Pock et al., 2005 [77] | non-public | - | 54.0 | - |
| Huang et al., 2018[89] | 3D-IRCADb-01 and Sliver07 | 66.5 | 96.9 | 75.8 |
| Isensee et al., 2018 [91] | MSD-Task08 | 63.00 | - | - |
| Keshwani et al., 2020 [88] | 3D-IRCADb-01 | 92.0 | - | 96.0 |
| Sangsefidi et al., 2018 [85] | Vascular Synth | 93.73 | 93.74 | 93.68 |
| Frangi et al., 1998 [76] | 3D-IRCADb-01 | 66.4 | - | 61.8 |
| Alhonnoro et al., 2010 [82] | non-public | - | 87.0 | - |
| Ronneberger et al., 2015 [90] | 3D-IRCADb-01 | 72.3 | - | 75.8 |
| Lu et al., 2017 [45] | non-public | 72.74 | - | - |
| Jegelka et al., 2011 [86] | 3D-IRCADb-01 | 75.0 | - | 77.6 |
| Chu et al., 2020 [93] | self-collected | 90.17 | - | - |
| Boykov et al., 2006 [87] | 3D-IRCADb-01 | 33.4 | - | 41.6 |
| References | Methods | Datasets | Metrics Results | Pros and Cons |
|---|---|---|---|---|
| Lebre et al., 2018 [16] | 3D thinning | 3D-IRCADb-01 | accuracy = 0.97 specificity = 0.98 sensitivity = 0.69 | full-auto, but affected by vessel segmentation |
| Chen et al., 2016 [122] | 3D thinning | non-public | visualization | full-auto, but affected by vessel segmentation |
| Chung et al., 2018 [123] | distance ordered thinning | non-public | Dice = 0.96 | full-auto, but affected by vessel segmentation |
| Pan et al., 2020 [124] | 3D iterative thinning | non-public | visualization | full-auto, but affected by vessel segmentation |
| Zhang et al., 2020 [37] | Pixel-RRT* | Sliver07 and LiTS | HD = 4.816, 2.829, 6.241, 5.984 visualization | ensure continuity, but semi-auto |
| Sangsefidi et al., 2018 [85] | axes enhancement and thinning | Vascular Synth | Dice = 0.93 specificity = 0.94 sensitivity = 0.93 | full-auto, but affected by vessel segmentation |
| Yan et al., 2017 [18] | distance transform | non-public | visualization | full-auto, but affected by vessel segmentation |
| Alirr et al., 2020 [125] | fast marching method | 3D-IRCADb-01 | distance error = 1.65, 1.77 mm | full-auto, but affected by vessel segmentation |
| Zhao et al., 2018 [126] | path planning | non-public | cosine angle = 73.76 arc length = 234.19 mm | ensure continuity, but not strict center axes |
| Sangsefidi et al., 2017 [127] | axes enhancement and threshold | Vascular Synth | Dice = 0.93 TPR = 0.96 | full-auto, but weak robustness |
| Dagon et al., 2008 [128] | geodesic distance transform | non-public | visualization | full-auto, but too many hyperparameters |
| Drechsler et al., 2010 [129] | 3D thinning | non-public | visualization | full-auto, but affected by vessel segmentation |
| Merveille et al., 2017 [130] | 3D thinning | 3D-IRCADb-01 | accuracy = 0.90 specificity = 0.97 sensitivity = 0.20 | full-auto, but affected by vessel segmentation |
| Ibragimov et al., 2017 [131] | distance ordered thinning | non-public | Dice = 0.83 | full-auto, but affected by vessel segmentation |
| Sato et al., 1997 [132] | 3D thinning | 3D-IRCADb-01 | accuracy = 0.89 specificity = 0.97 sensitivity = 0.24 | full-auto, but affected by vessel segmentation |
| Mueller et al., 2008 [37,133] | fast marching method | Sliver07 and LiTS | HD = 69.311, 3.162, 81.025, 81.025 visualization | ensure continuity, but conventional grid search |
| Wang et al., 2016 [134] | thinning and connection cost computation | non-public | skeleton coverage = 0.55 mean symmetrical distance = 12.7 mm | full-auto, but affected by vessel pre-segmentation |
| Wu et al., 2013 [135] | 3D thinning and linear interpolation | non-public | visualization | full-auto, but affected by vessel pre-segmentation |
| Kang et al., 2014 [136] | Laplacian-based contraction | non-public | accuracy = 0.97 | full-auto, but not strict single voxel |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Wu, F.; Chang, W.; Kong, D. Techniques and Algorithms for Hepatic Vessel Skeletonization in Medical Images: A Survey. Entropy 2022, 24, 465. https://doi.org/10.3390/e24040465
Zhang J, Wu F, Chang W, Kong D. Techniques and Algorithms for Hepatic Vessel Skeletonization in Medical Images: A Survey. Entropy. 2022; 24(4):465. https://doi.org/10.3390/e24040465
Chicago/Turabian StyleZhang, Jianfeng, Fa Wu, Wanru Chang, and Dexing Kong. 2022. "Techniques and Algorithms for Hepatic Vessel Skeletonization in Medical Images: A Survey" Entropy 24, no. 4: 465. https://doi.org/10.3390/e24040465
APA StyleZhang, J., Wu, F., Chang, W., & Kong, D. (2022). Techniques and Algorithms for Hepatic Vessel Skeletonization in Medical Images: A Survey. Entropy, 24(4), 465. https://doi.org/10.3390/e24040465






