1. Introduction
Plant disease epidemiology studies how diseases affect plant populations and how to combat plant diseases. Using spatial and temporal plant epidemiology models can provide useful statistical and mathematical data about disease transmission. In the mid-20th century, plant epidemiological models became prominent [
1]. Examples of actual uses of this type of model include cassava mosaic disease [
2], pine wilt disease [
3], and potato late blight [
4]. Later, new methods for studying nonlinear dynamics and numerical simulations helped solve complex ecological problems [
5,
6]. This accelerated the creation of more realistic and complicated plant disease models.
An essential part of the plant epidemiological system is modeling the interactions between infected and healthy plant populations, either directly or via a vector. Infected vectors feed on healthy plants, infecting them. Similarly, non-infected vectors become infected by diseased plants. The vector-borne plant disease is classified as persistent, semipersistent, or non-persistent based on the infectious agent’s residence period in the vector [
7,
8]. The vector ingests viruses while feeding on infected plant sap in persistent transmission. The salivary glands then release the viruses into the plant tissue as they penetrate the digestive system. The persistent mode of transmission differs from the other two because it takes a long time for a vector to become infected with the virus and become infectious [
7,
9]. In the case of vectors, this time lag is referred to as the latent phase of infection.
The latent period in plants is similar to the time it takes for a healthy plant to become infected following infection [
10]. The incubation period (or incubation time) is the time it takes for symptoms to manifest following infection [
1]. Depending on the plant species, the incubation period varies [
11]. Incubation durations for beet mosaic virus (BMV), African cassava mosaic virus (ACMV), tobacco mosaic virus (TMV) and bean golden mosaic virus (BGMV) are 7–15 days [
12], 3–5 weeks [
13], 5 h [
14], and 5–6 days [
15], respectively. The incubation and latent periods in plants are distinct. However, the expression of disease symptoms correlates with disease transmission [
16]. Furthermore, determining the latent period is challenging, whereas observing disease signs is straightforward. So our model development analysis considers the incubation period.
Among the most frequent vector-borne viral diseases affecting crops, leaf curl disease and mosaic disease are two of the most common. The whitefly (
Bemisia sp.), which transmits several viral infections to Jatropha, cassava, tomato, tobacco, cotton, and other plants, is a hemipteran vector. Most of the disease is systematically spread by whiteflies, meaning that a latent period is frequently observed [
17]. Unfortunately, information on the latent and incubation time of infection for various persistently transmitted diseases is lacking in the literature. Due to the variety of viral agents and host plant species, both delay methods have varying effects on disease severity. It also differs between whitefly species and host plants. These delays may vary due to genetic complexity, climate fluctuation, phenotypic heterogeneity, and plasticity [
18]. The plant incubation period is usually longer than the latent period in vectors. For example, ACMV has a 6-hour latent period and a 3–5 week incubation period [
13].
Ordinary differential equations (ODEs) models cannot account for the incubation or latent period. However, models based on delay differential equations (DDEs) allow system integration. It can represent a system’s dynamics when its evolution depends on prior events. When time lag responses exist, delays are one of the most powerful mathematical modeling tools [
19]. DDE models are more sophisticated than ODE models but more realistic. Prey–predator mathematical models with delay differential equations are commonly employed [
20,
21]. Delay can teach us dynamic phenomena, such as instability, oscillations, and bifurcation.
Van der Plank [
1] used DDE to delay plant epidemics. Cooke [
22] proposed a model with an incubation time state variable for vector-borne diseases. Wang et al. [
23] discussed wheat starch and gluten’s thermal characteristics and interactions. Zhang [
24] added the plant incubation period to a Meng and Li [
25] plant disease model, causing modifications in the model’s dynamics. Munyasya et al. [
26] proposed an integrated on-site and off-site rainwater-harvesting system that enhances rainfed maize output for better climate change adaption. Buonomo and Cerasuolo [
27] presented and analyzed a soil-borne plant disease dynamics model. Miao [
28] suggested an accuracy of space-for-time substitution for predicting vegetation status after shrub restoration.
An ODE model of the impact of replanting and roguing on eliminating plant disease latency comprises a compartment for latently diseased plant populations [
29]. The model does not consider any vector compartment, but it includes classes of latently infected, healthy, post-infection, and infectious plants. Holt et al. [
2] proposed a model with infected plants, healthy vectors, and susceptible vectors but no delays. The vector-borne plant disease model [
30] was modified by Jackson and Chen [
31] by delaying plant incubation and vector latent periods. The threshold value for delay-induced destabilization was determined by observing changes in system solution dynamics. Li et al. used an updated model [
31] to analyze Hopf bifurcation, which included incubation and latent period characteristics [
32].
Banerjee and Takeuchi [
33] identified several critical elements of the dynamics that could lead to false findings. A long wait can stabilize or cure a system Buonomo, and Cerasuolo [
27]. Transcritical bifurcations, periodic oscillations, and stability switches can be revealed if the vector-borne plant disease models’ parameters change [
2,
27,
34]. The undelayed model analysis cannot be ignored [
31,
32]. A mathematical model (
1) with parameters given in
Table 1 [
2,
35], which was previously analyzed by Basir et al. [
35] for persistent vector-borne viral plant disease dynamics for the effect of both latent period and incubation delay of the dynamics of the deceased. This model is numerically analyzed using a gradient-based numerical technique. Numerous studies claimed that gradient-based techniques, such as RK-4, take up much more computer time than soft computing methods with comparable accuracy and that it is difficult to produce accurate global estimates of the truncation error [
36,
37]. For instance, at each step of the RK-4 method, the derivative must be evaluated
n times. Here, ’
n’ is the order of accuracy of the RK-4 method, which is a significant drawback of gradient-based algorithms [
38]. Moreover, RK-4 suffers from divergence for complex systems [
39]. Failure in the case of singularity is another hurdle in using these gradient-based numerical techniques. Keeping these disadvantages in mind, the authors of this paper aimed to suggest an alternative gradient-free approach that can handle problems, such as model (
1), with accuracy and reliability. The key features of this study are outlined as follows:
In this paper, we analyzed an established mathematical model (
1) for persistent vector-borne viral plant disease dynamics, which is presented in
Section 2. The set of parameters substituted in the model is for the case of cassava mosaic disease.
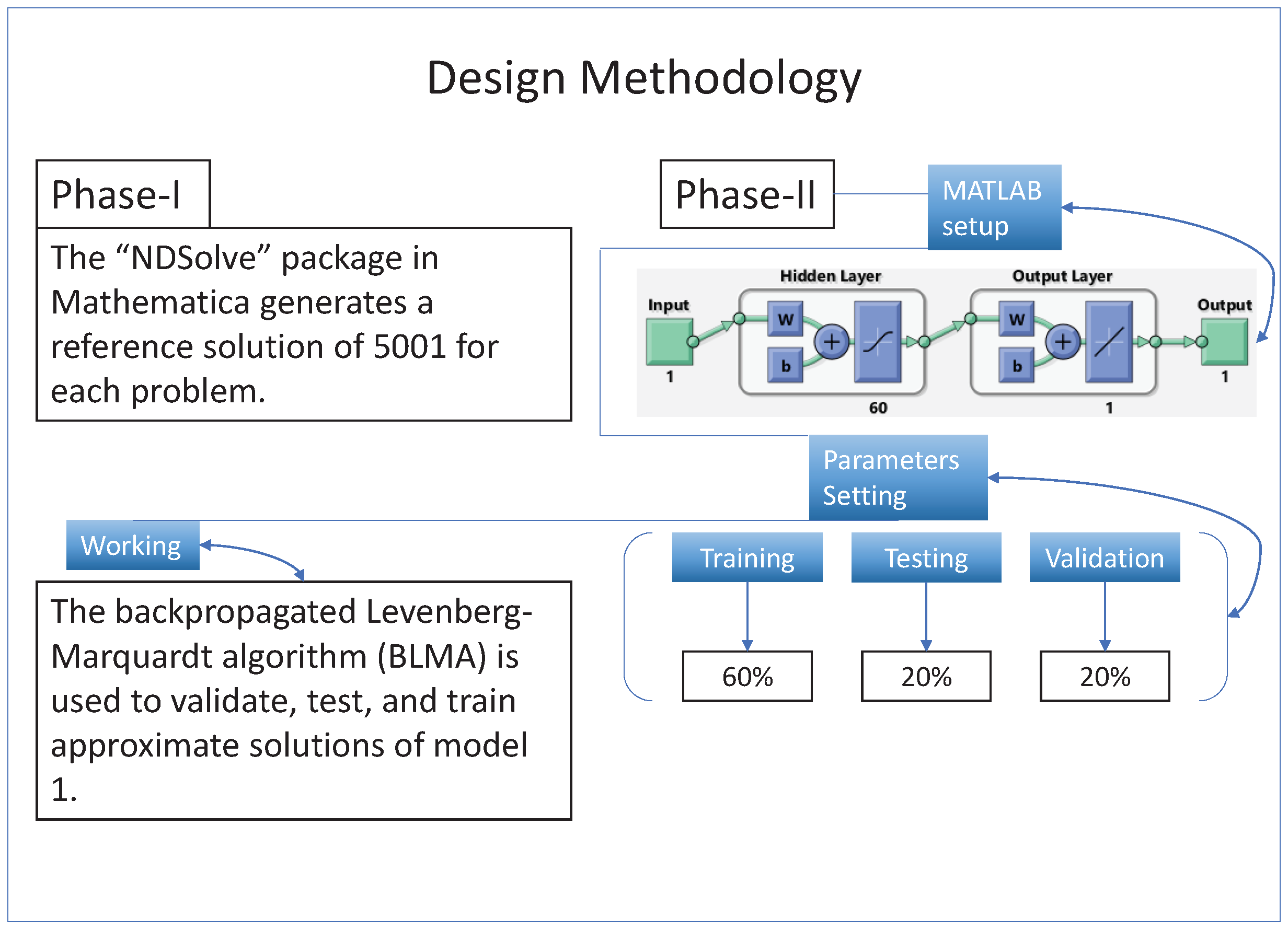
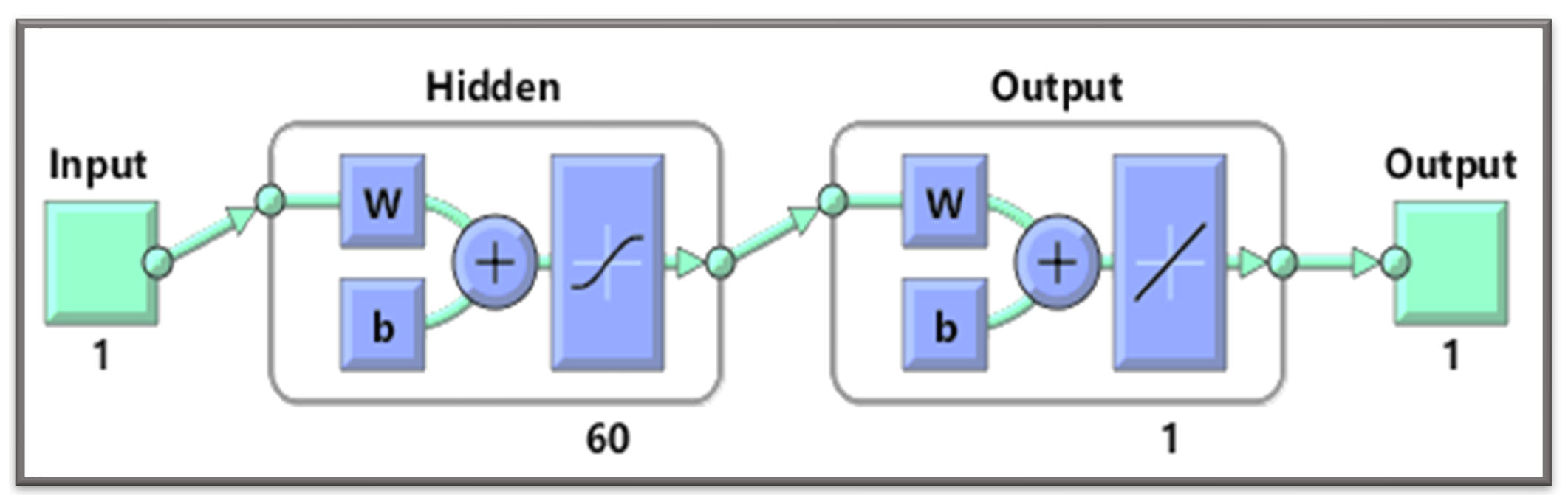
A gradient-free intelligent design of a two-layer artificial neural network architecture and the Levenberg—Marquardt algorithm is utilized to formulate surrogate solutions. A state-of-the-art numerical method is used to calculate reference solutions for establishing the accuracy, validity, and reliability of NN-BLMA; see
Section 3.
The impact of variations in parameters, such as plant mortality and vector mortality rate, on the model of persistent vector-borne viral plant disease dynamics is observed through the surrogate solutions formulated by the designed NN-BLMA; see
Section 4. Graphical analysis for the convergence of NN-BLMA is carried out based on mean square error, regression analysis plots, and error histograms. Moreover, statistical values are tabulated to show the accuracy and reliability of the designed technique.
Table 1.
Parameters’ description and their numerical values.
Table 1.
Parameters’ description and their numerical values.
| Parameters | Description | Values | Unit |
|---|
| r | Net growth rate of plants | 0.3 | |
| K | Carrying capacity | 1 | |
| Infected vector to healthy plant disease transmission rate | 0.025 | |
| Plants natural mortality rate | 0.1 | |
| Mortality of infected plants | 0.01 | |
| Vector population’s overall growth rate due to immigration or births | 40 | |
| Transmission rate between diseased vector and healthy plant | 0.03 | |
| d | Vector mortality rate | 0.1 | |
2. Problem Formulation
This section develops a mathematical model for persistent vector-borne viral plant disease dynamics. The model considers plant and vector populations without explicitly including the mosaic virus. signifies healthy plants, while the infected plants are represented by , represents uninfected, and W(t) represents the infected whiteflies population.
Due to restricted plantation space and natural resources, logistic growth
r and carrying capacity
K are considered for healthy plants [
2]. A healthy plant becomes infected when it comes into contact with an infected vector. When an infected vector and a susceptible plant are present,
is the transmission rate, and
is the number of sensitive individuals moving from the susceptible compartment to the infected compartment.
An insect pest, such as a whitefly, shifts its host in response to changing biological and environmental conditions. They generally move between fields of crops [
40,
41]. They breed in the fields. The Holling type III survival curve describes their life course because of the high death rate they experience early on [
41]. Whiteflies (adults and nymphs) can transmit illness.
Crops are typically planted and reaped at specific times of the year. Most crops are reaped a few months after they are produced. A few vectors travel from close or distant patches and reproduce in the vegetation. Vectors grow by migrating from another patch because of reproducing in the same patch or vegetative area. For the same reason, seasonal fluctuations in vector populations are ignored [
35].
An open system is considered in this model. Assume
is the rate of vector birth and migration into the system. No vertical virus transmission is allowed, and a vector cannot infect another vector. Viruses do not destroy or defend vectors. The vector retains the virus and does not recover. However, the infective insects do not get sick from the virus [
31]. Let the mortality rate of plants and vectors be represented by
and
d, respectively. Infection-related plant death is expected to be higher than average plant mortality.
is the infection-related mortality of infected plants. Thus, the overall plant mortality rate is
. Consider
to be the conversion between uninfected vectors (i.e.,
Q) and the infected plant (i.e.,
I). So,
signifies entering the number of uninfected vectors
Q into the infected vectors
W compartment.
In truth, both plant and vector infection takes time. Let be the healthy plant’s incubation time following successful infection. At time t, the disease transmission is given by the expression , where the positive constants described previously are and . The term denotes the chance of a healthy plant surviving through the incubation time , i.e., the number of susceptible plants that came into touch with an infected vector at time and lived up to time t to become infected plants.
Again the latent period in a vector is
. At time
t, the expression
describes the transmission of infection, where
reflects the vector’s survival probability across the latent time
. The number of uninfected vectors met an infected vector at time
and survived until time
t to become infected [
35]. Based on the given assumptions, the mathematical model is
The initial biological conditions are
, , , ; , ,
The parameters used in the model (
1) assigned some numerical values for solving the model numerically, and
Table 1 shows its description and numerical values.
4. Numerical Experimentation and Discussion
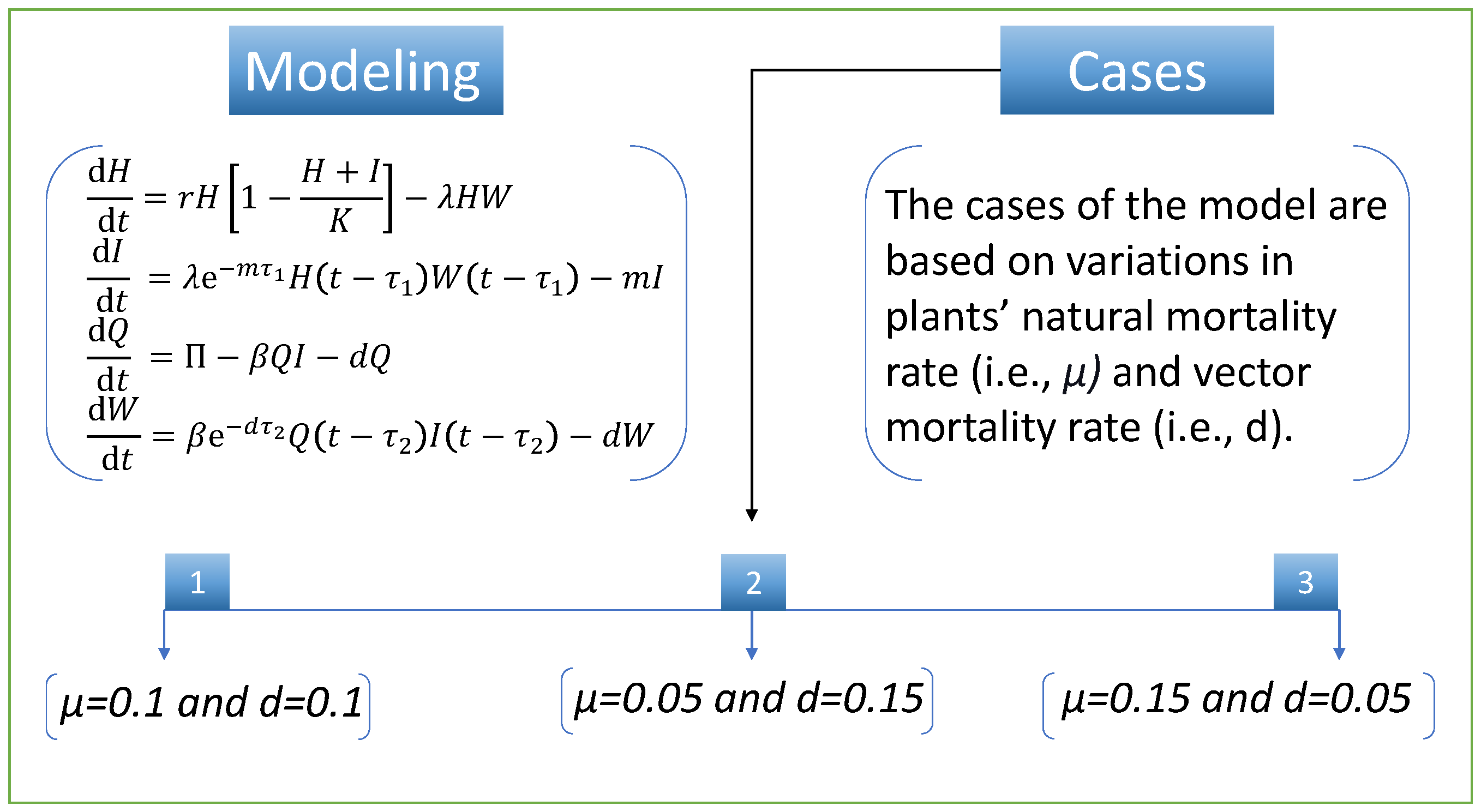
To study the design algorithms’ performance and efficiency, we discuss various cases of the nonlinear model of vector-borne viral plant disease dynamics. The cases are based on variation in two parameters (i.e., plants’ natural mortality rate,
, and vector mortality rate,
d). We set the same numerical value for both parameters in the first case. In case two, there is a slight decrease in the
parameter and a slight increase in the parameter
d, while in the third case, there is an increase in the parameter
and a decrease in the
d parameter compared with the first case.
Figure 3 illustrates the mathematical model and the cases detail for vector-borne viral plant disease dynamics.
The design technique generates output data sets with probabilities of
of the sample data for testing,
for training, and
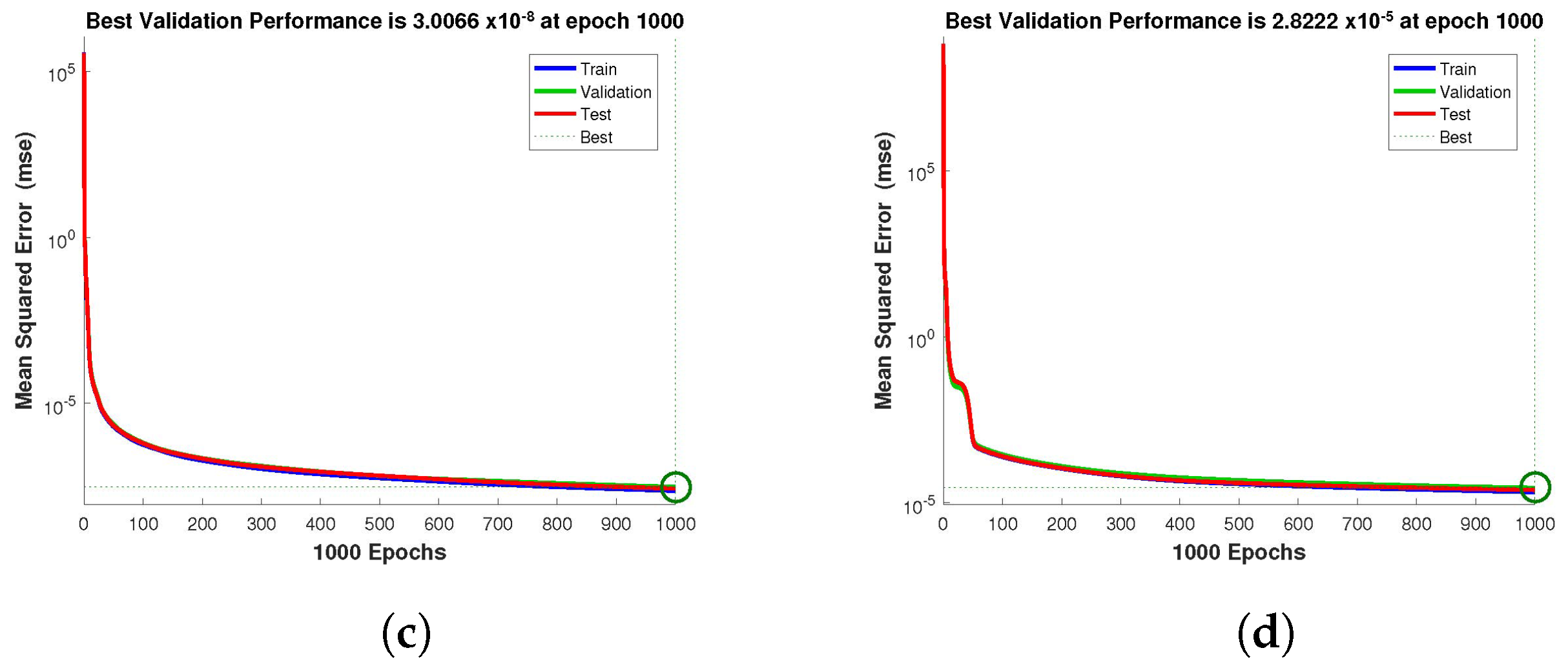
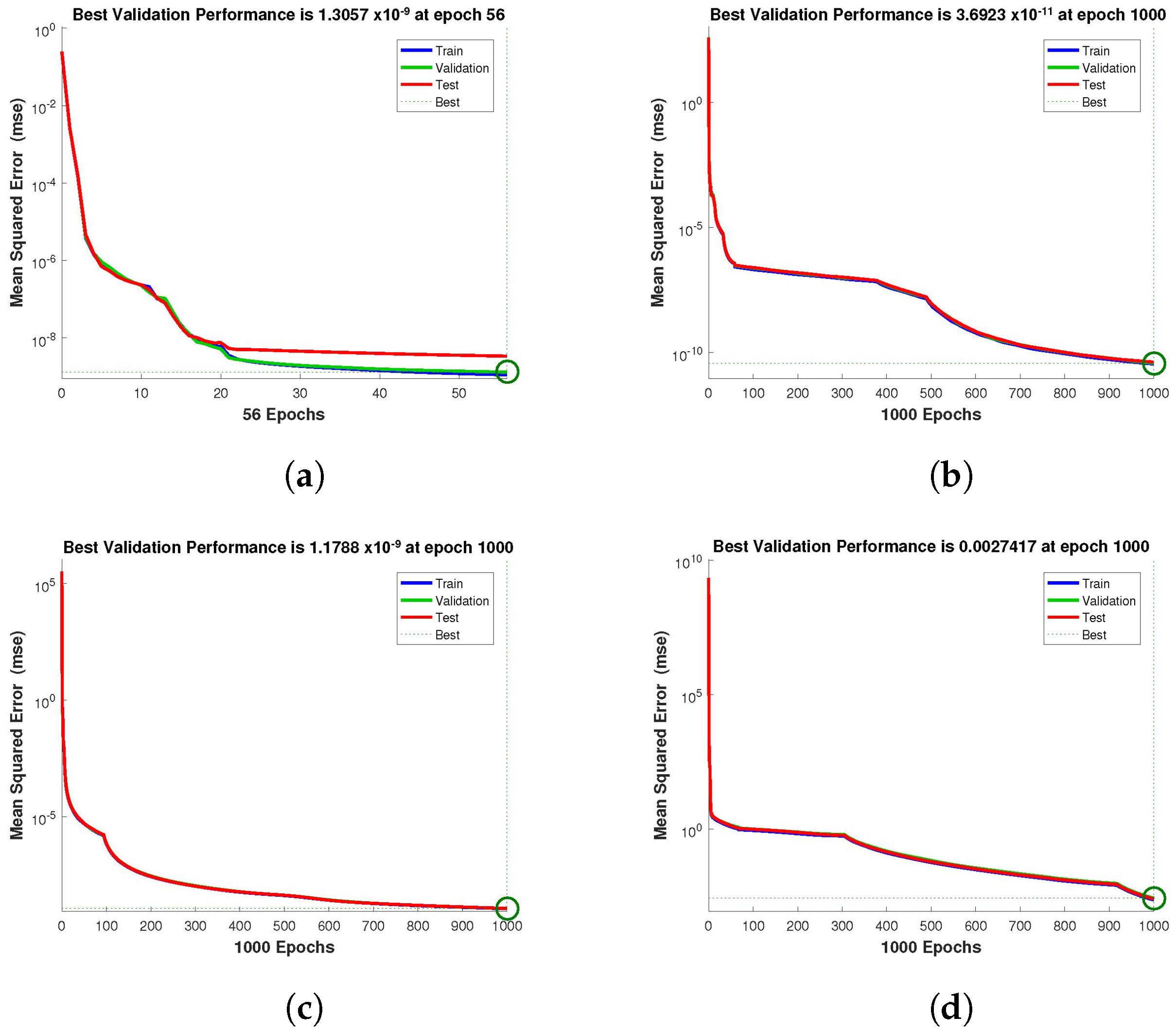
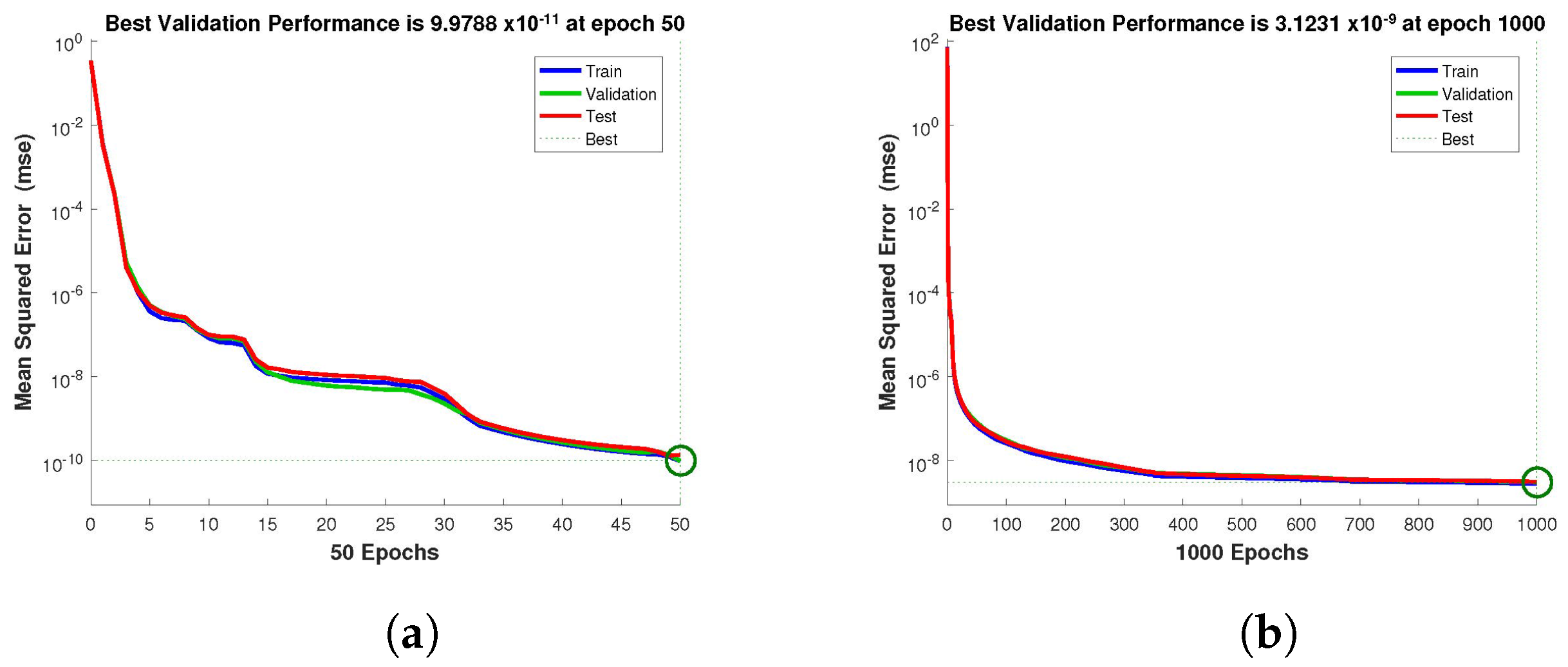
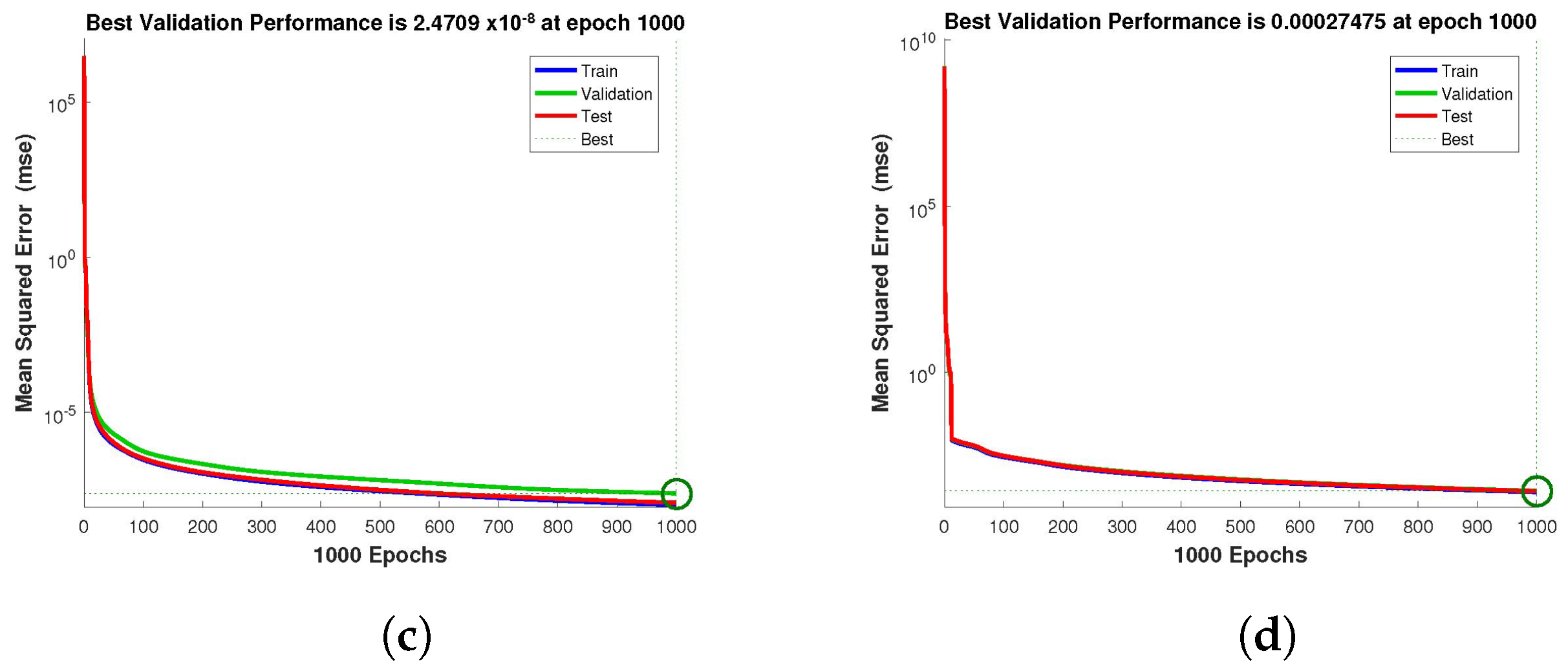
for validation. The performance graph of the design technique shows us its mean squared error (MSE).
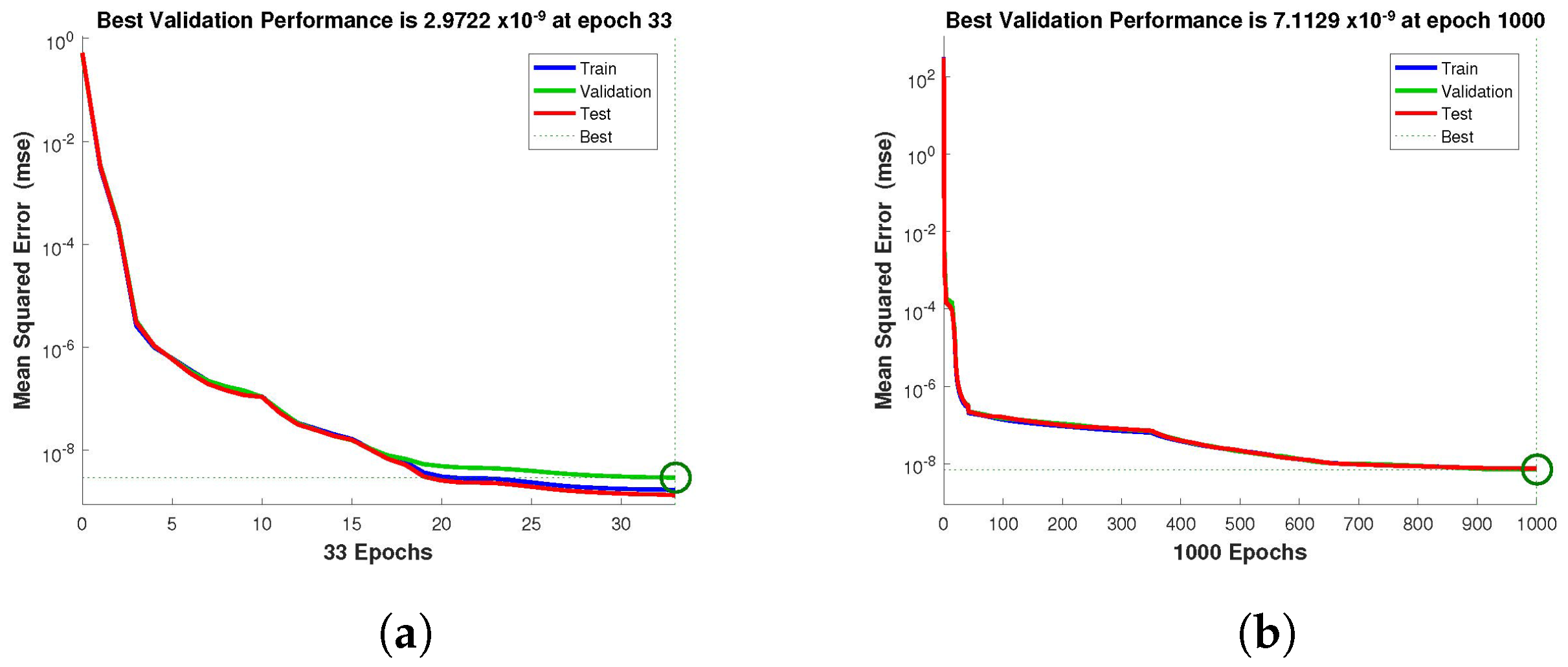
Figure 4,
Figure 5 and
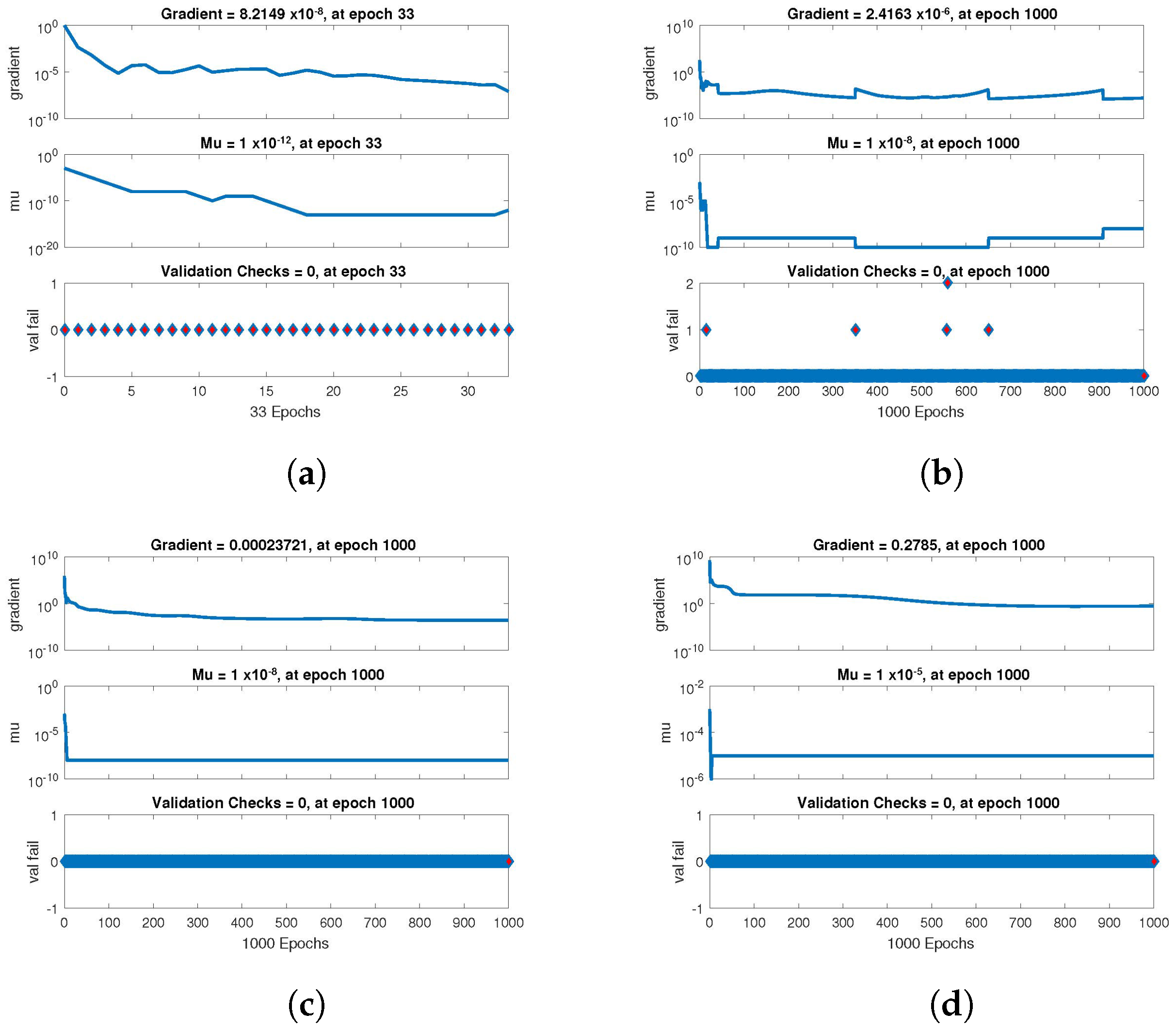
Figure 6 depict the best validation performance provided by the design technique because the error is minimized after some epochs of training but may increase on the validation data set as the network begins to overfit the training data. The training is halted after six consecutive rises in the validation error, and the best performance is picked from the epoch with the lowest validation error. The case 1 performance values are in the range of
,
,
and
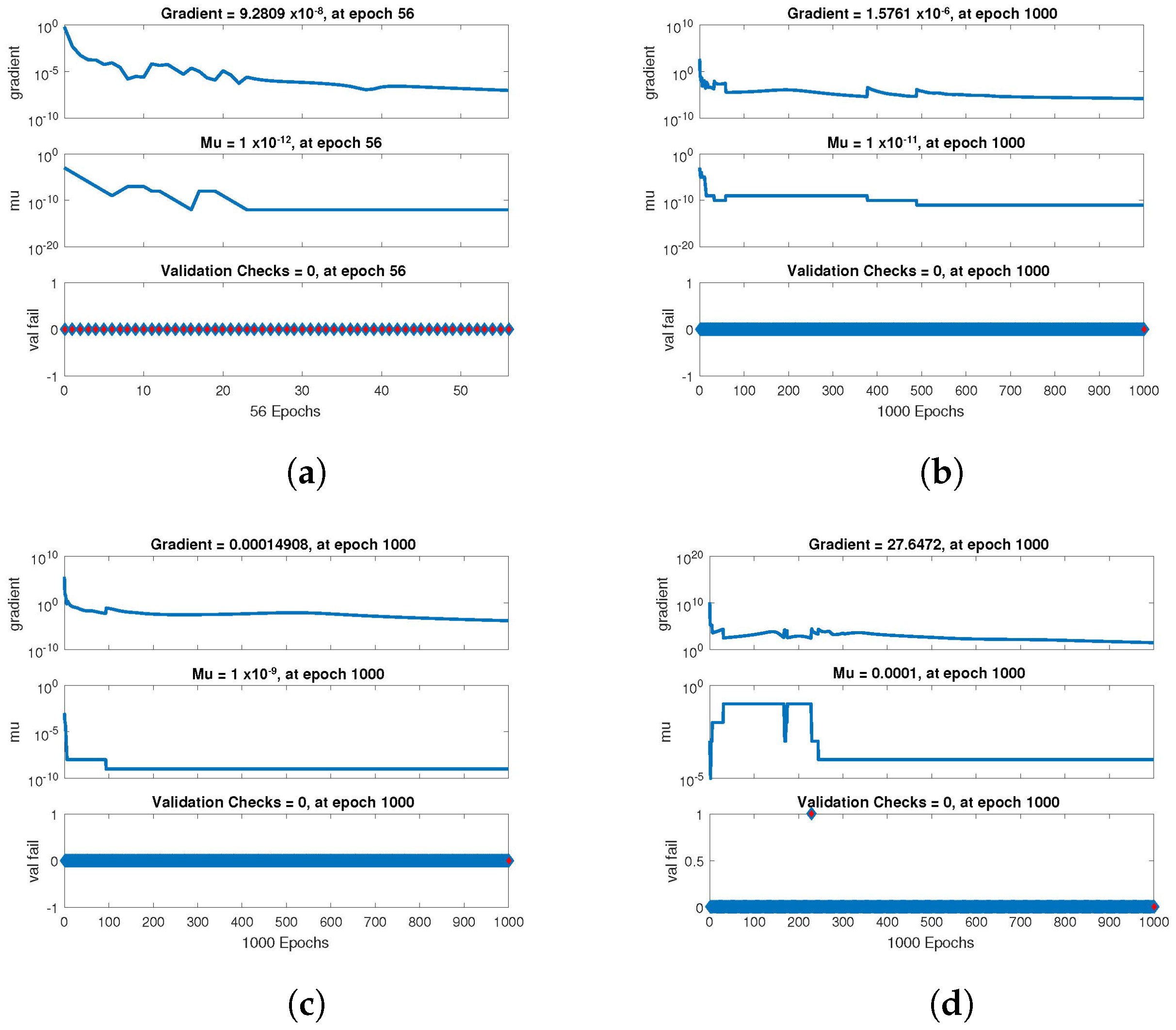
. Similarly, the case 2 and case 3 performance values are in the range of
,
,
,
, and
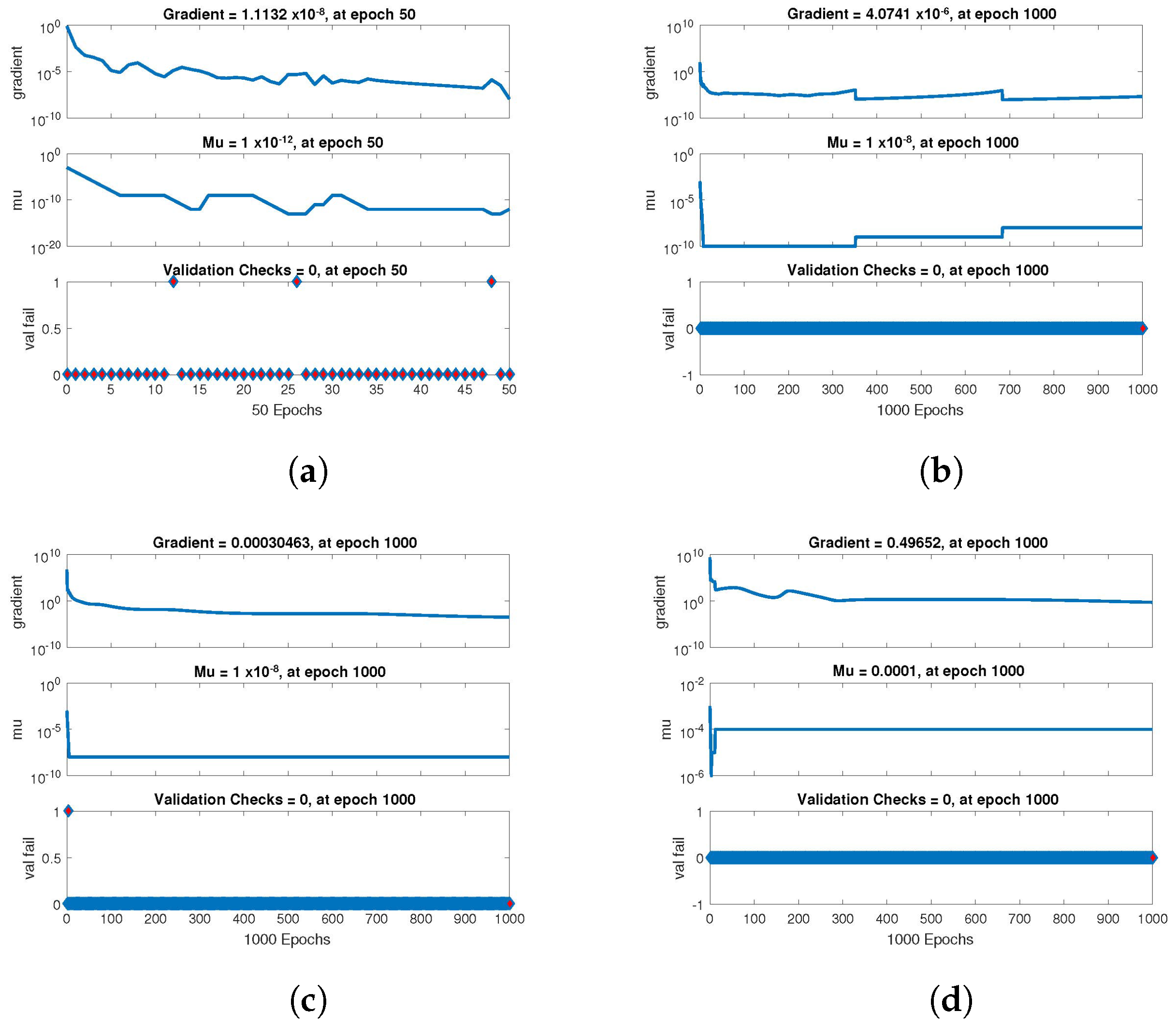
,
,
,
, respectively.
The statistical performance of all the cases in gradient, mu, and validation failures are illustrated in
Figure 7,
Figure 8 and
Figure 9. The gradient values for the case 1 lie in between
,
,
and
, whereas the values for case 2 and case 3 are
,
,
,
, and
,
,
,
, respectively. The mu values for all the cases lie in the range
to
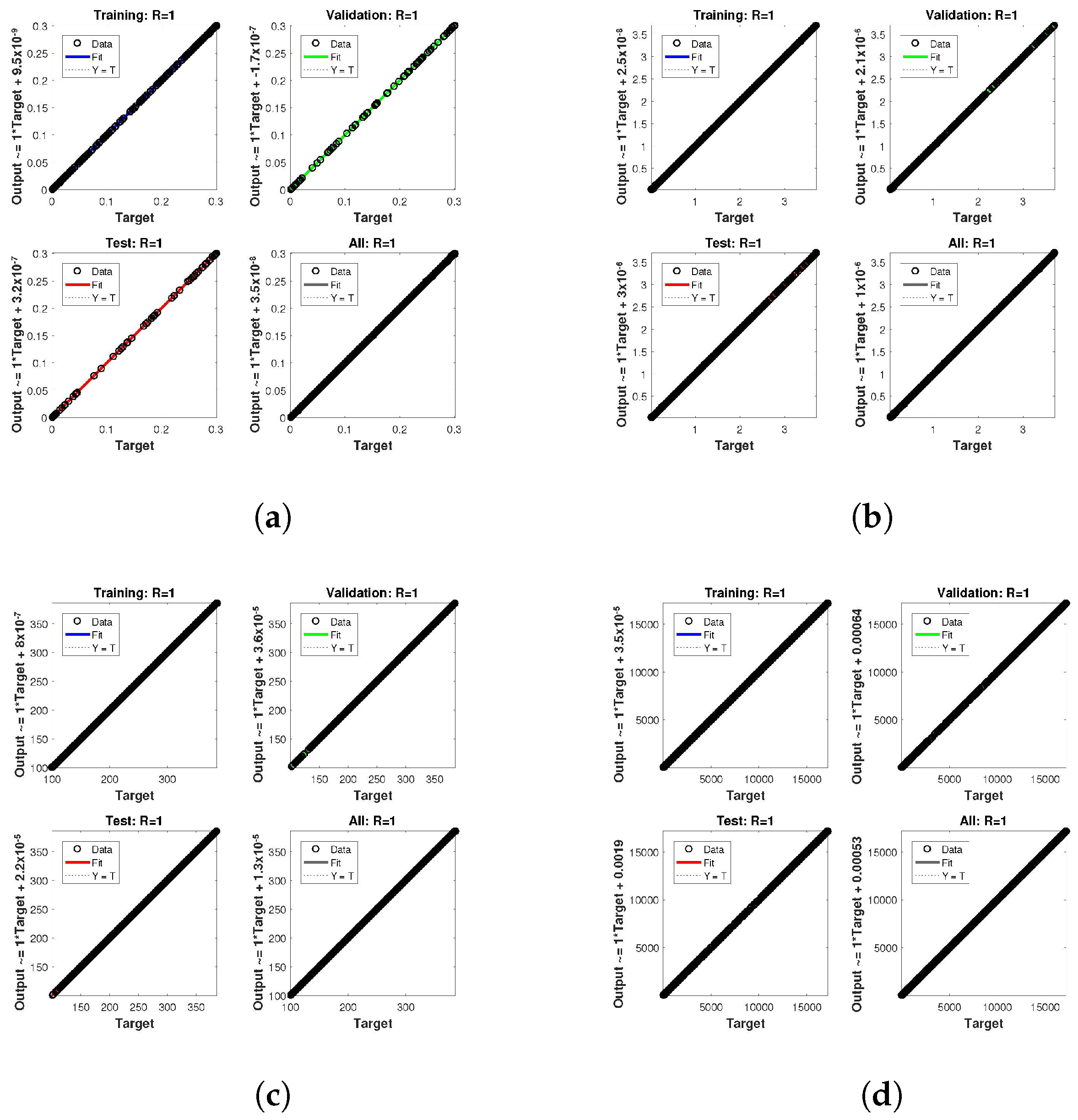
. The network output concerning the target for the training, validation, and test sets is shown on the regression plot. The data must fall on a 45-degree line where the network outputs and targets are equal for a perfect match. When the data fall on a 45 degree, the regression plot gives us a value of
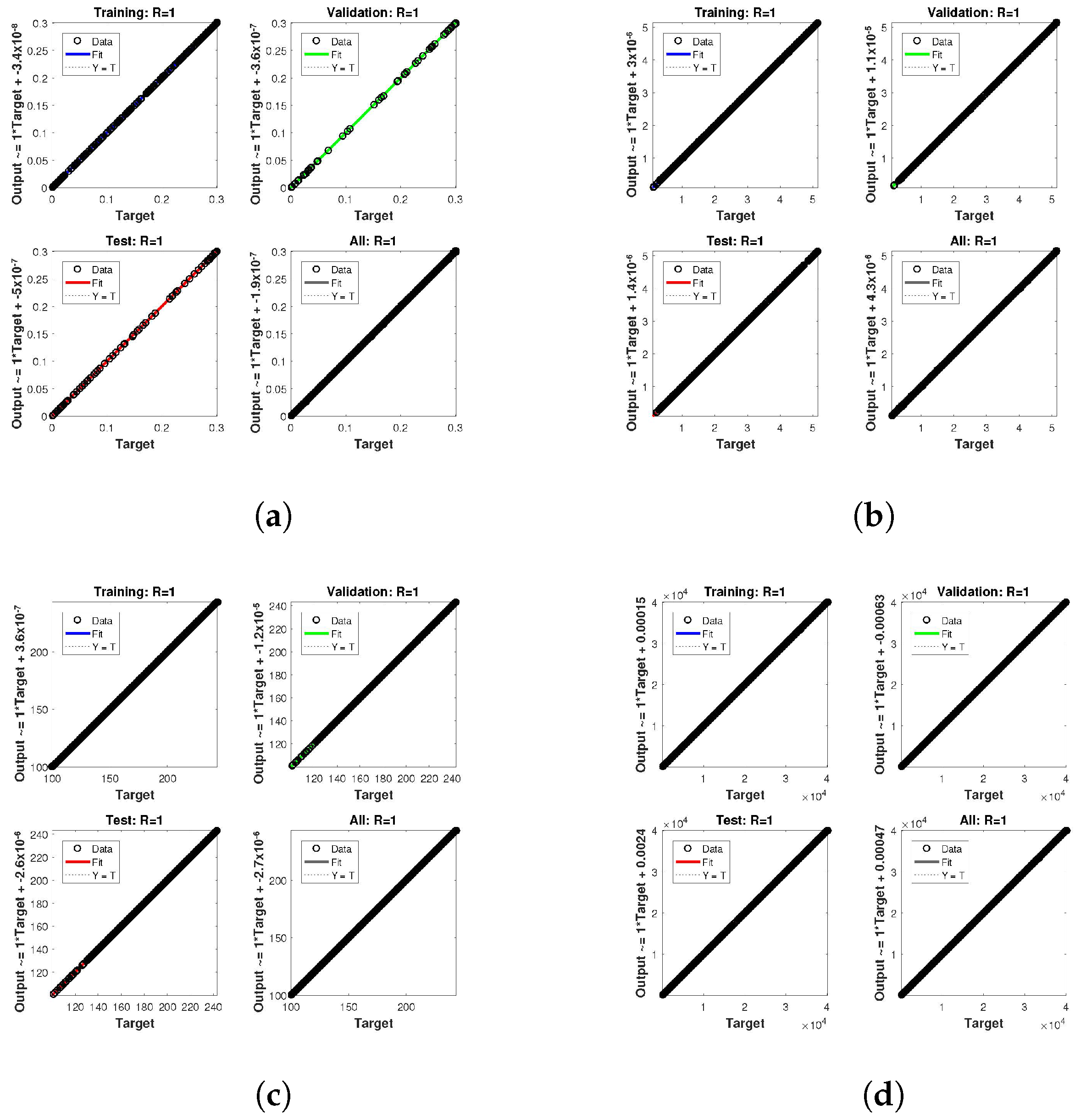
. This article shows the regression analysis of all the cases in
Figure 10,
Figure 11 and
Figure 12. From the figures, regression values are 1 for all cases, which perfectly matches the network and the targets.
The tables below provide the data information provided by the computing system. The tables show the best performance values in training, testing, validation, etc.
Table 3 displays the best performance data for case 1, while the best performance data for case 2 and case 3 are displayed in
Table 4 and
Table 5, respectively. These tables also show the hidden neuron count, iterations, and time spent.
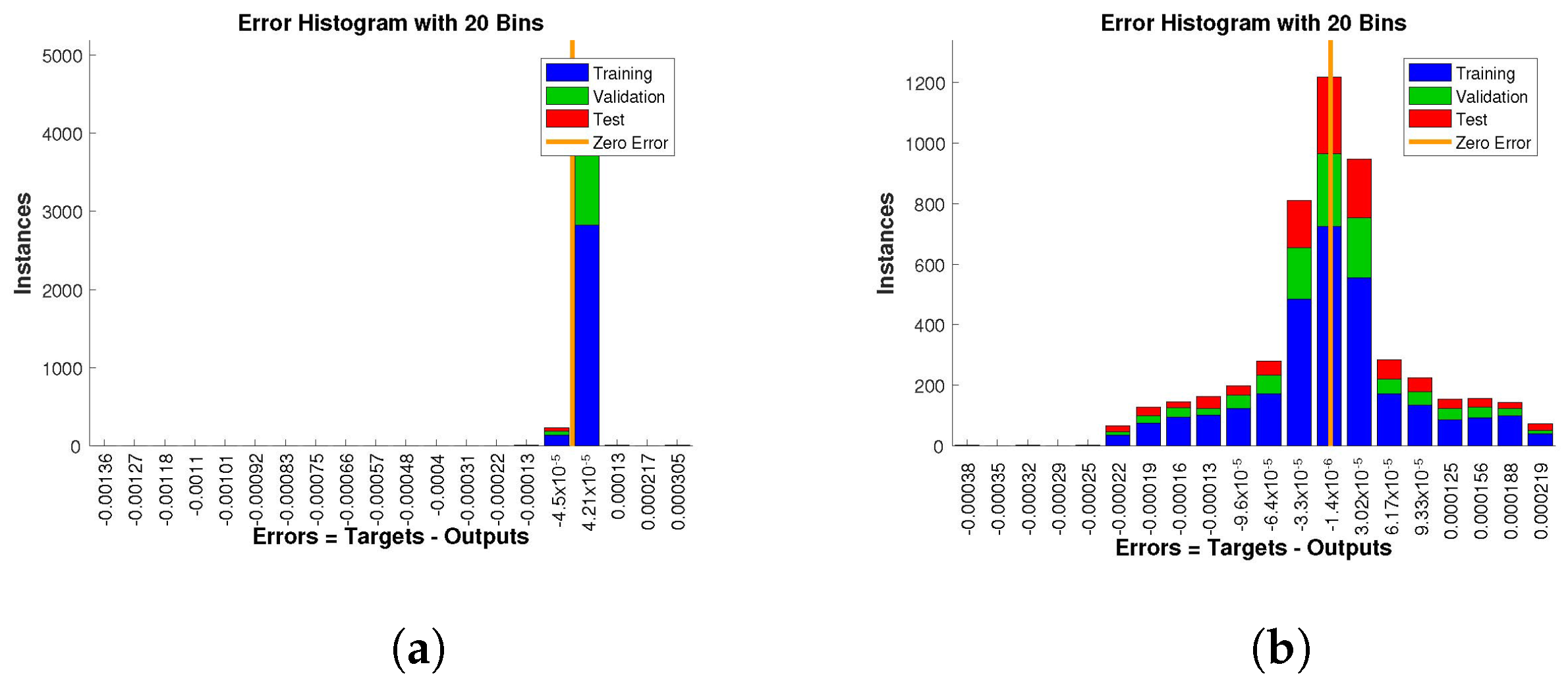
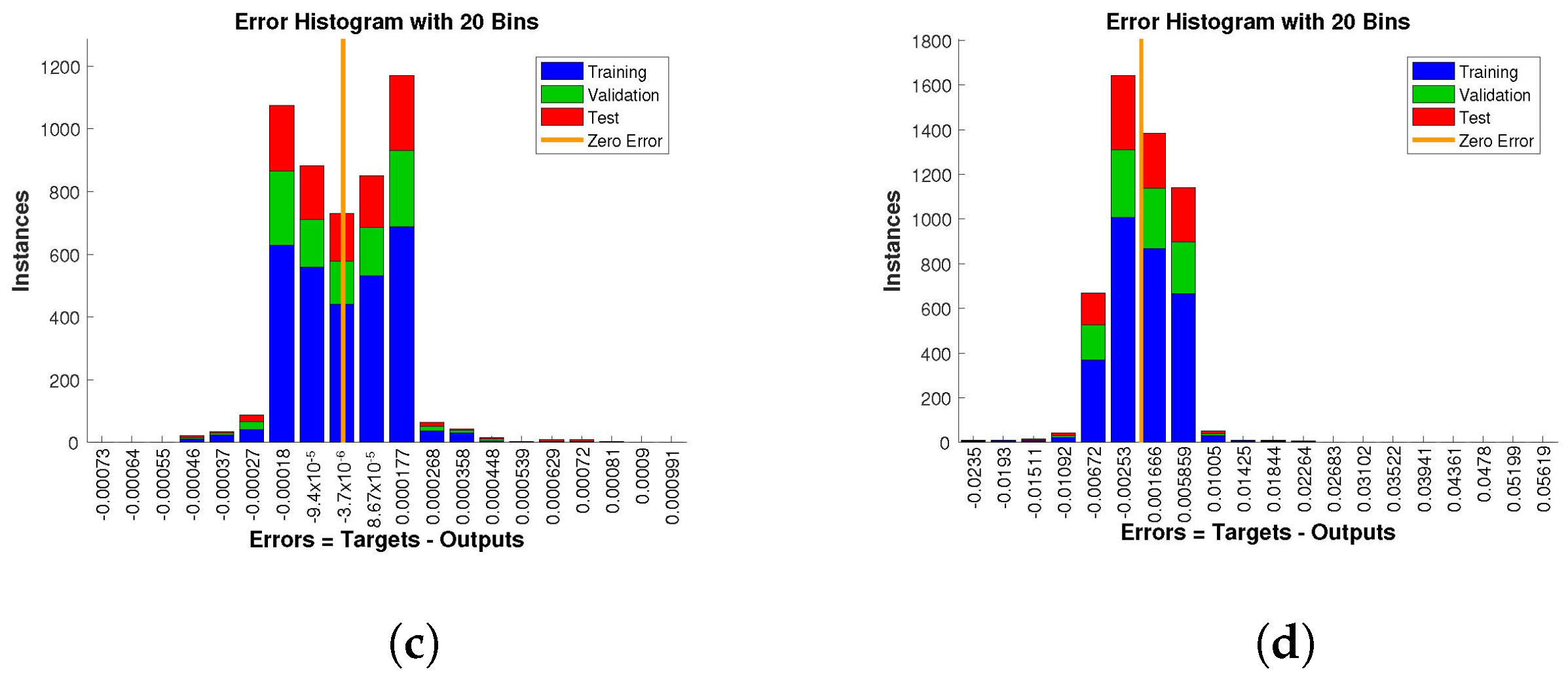
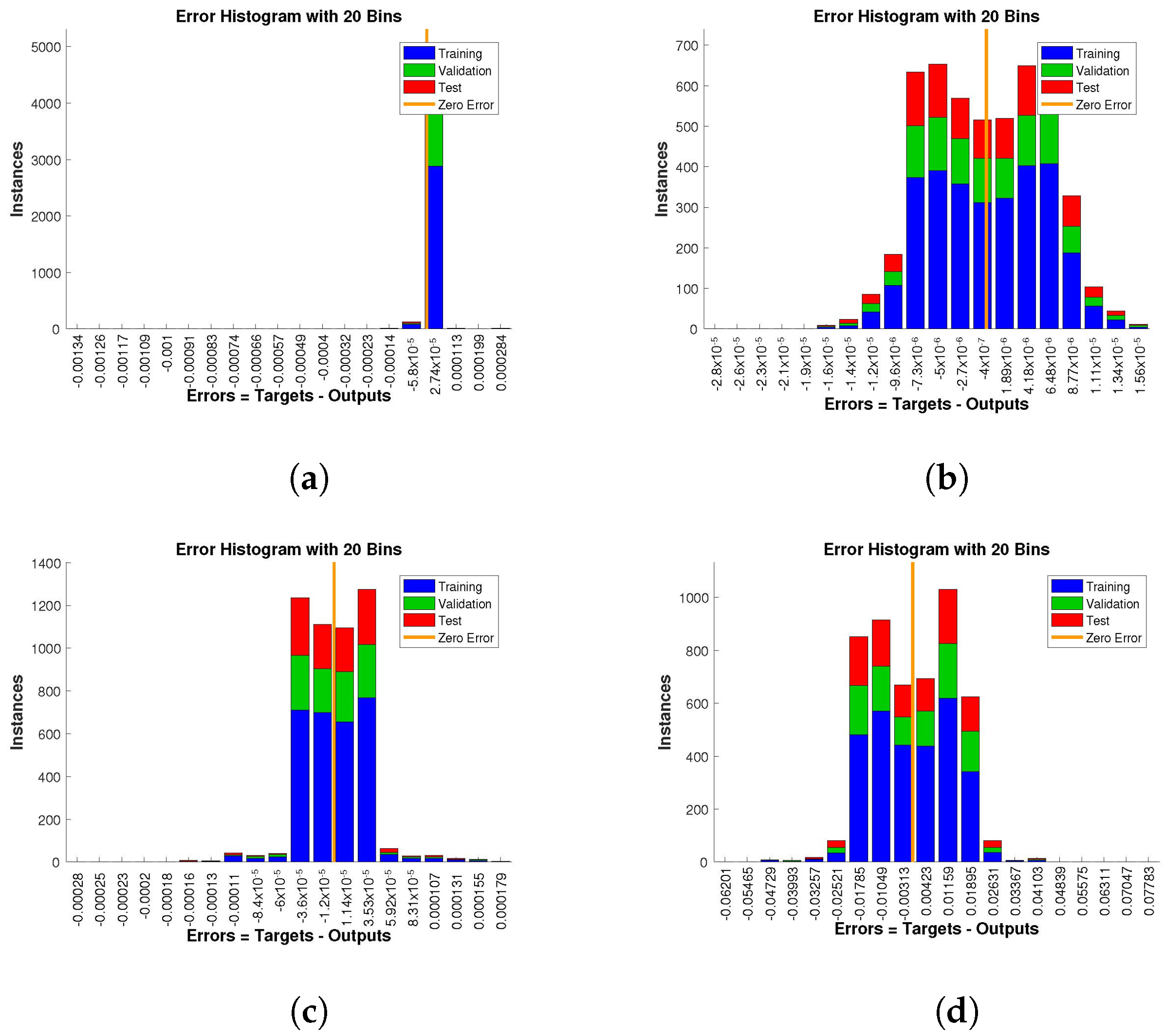
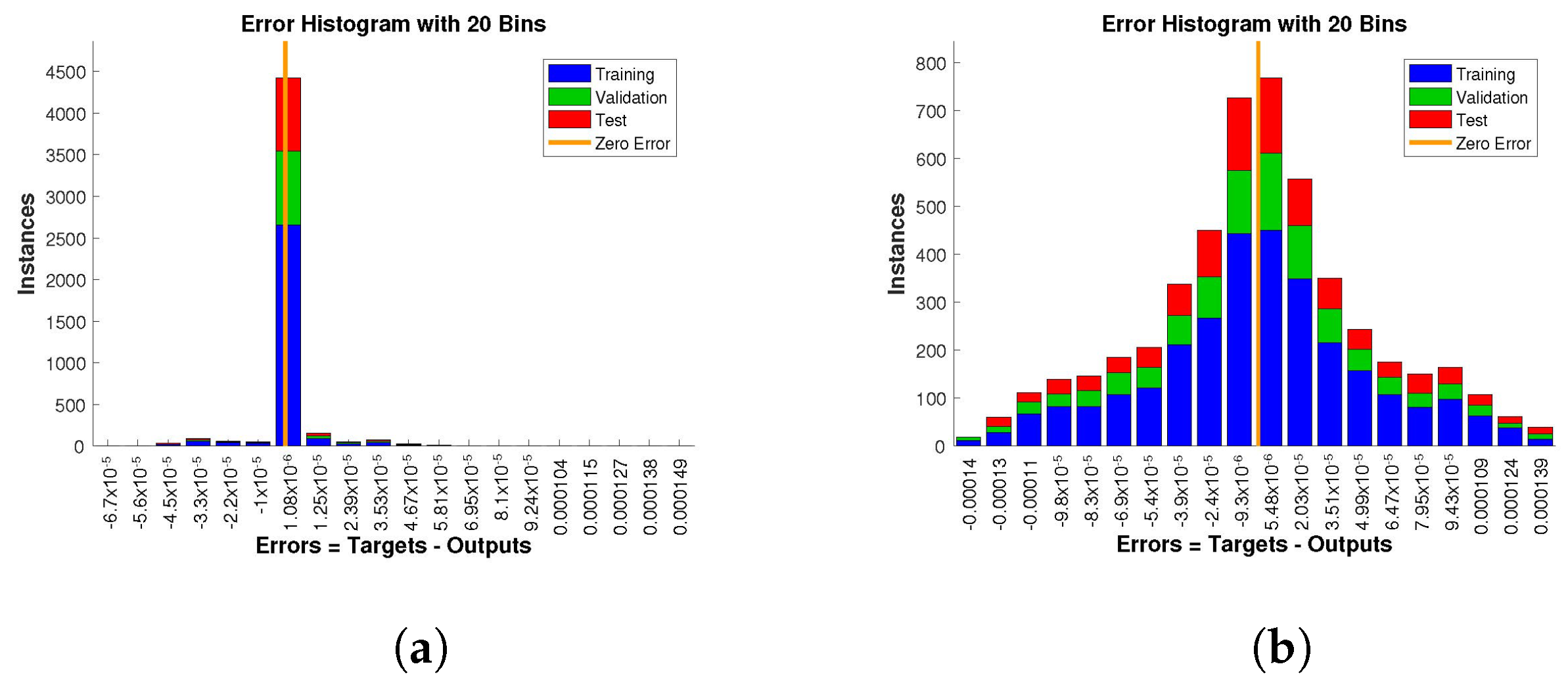
The histogram of errors between targets and outputs after training a neural network is shown in
Figure 13,
Figure 14 and
Figure 15. Different color bars show the errors in the training, validation, and testing data. The error bars in which most of the points lie are very close to the zero error line, which means targets and the outputs are well matched and have the fewest errors, which shows the accuracy of our design technique. The error values for case 1 lie in the range
to
,
to
,
to
, and
to
. For case 2 and case 3 the error values lie in the range
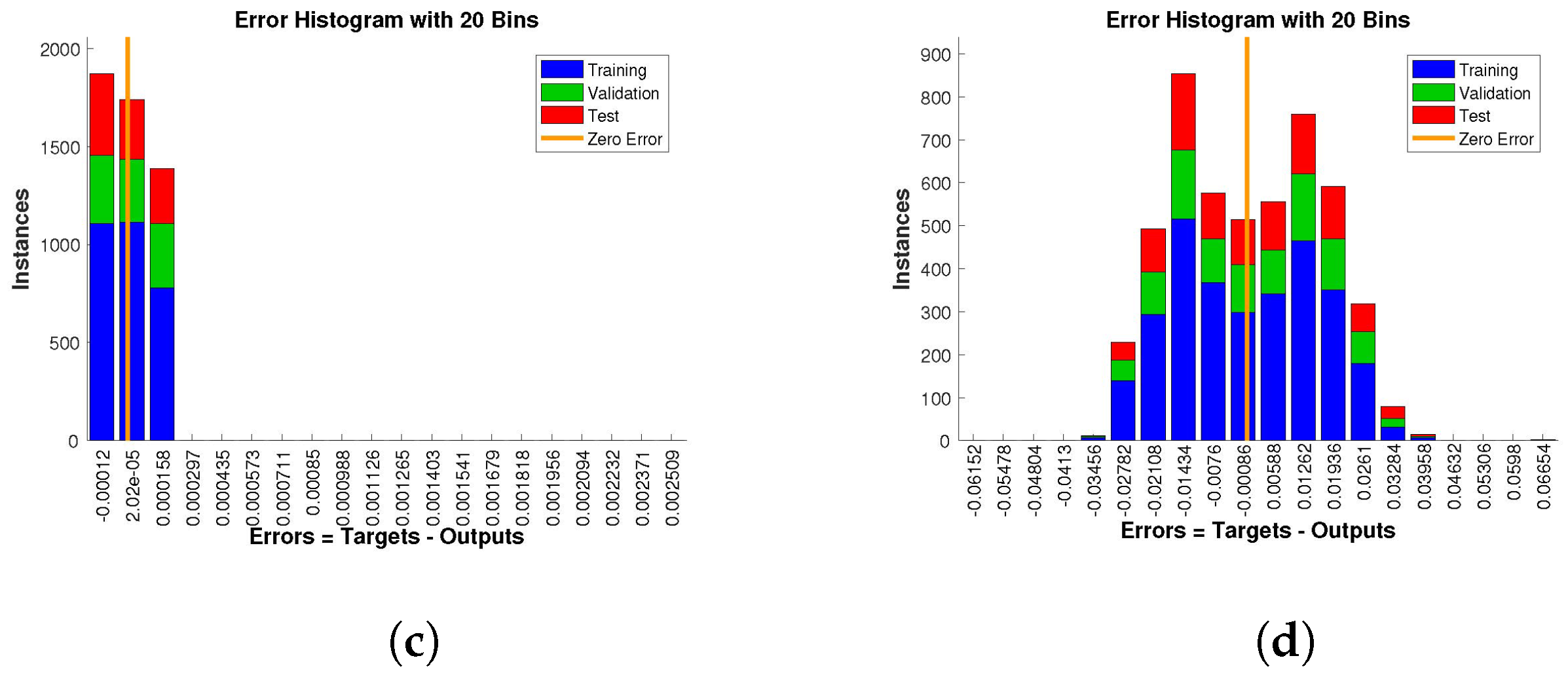
to
,
to
,
to
,
to
,
to
,
to
,
to
,
to
, and
to
, respectively.
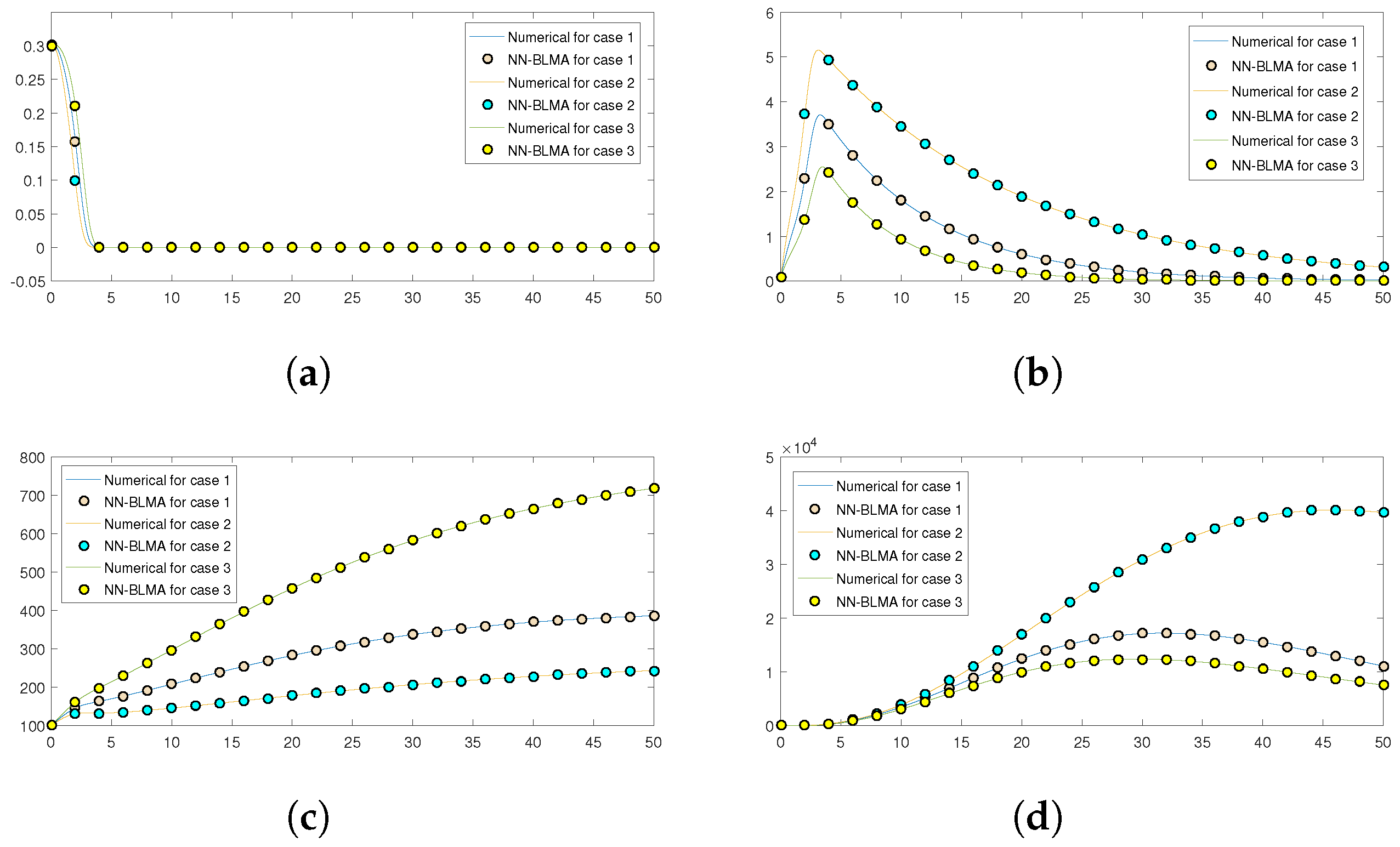
Further,
Figure 16 compares the numerical solution of the model obtained by the “NDSolve” package in Mathematica (targets) to the solution obtained by executing NN-BLMA (outputs). The solid lines show the solution obtained by solving the model numerically by the “NDSolve” package in Mathematica, while the circles show the solution by NN-BLMA. In the figure, we see that the solutions obtained from NN-BLMA come exactly on the targets’ solutions lines, which shows how accurate our design technique is. These figures also indicate the model’s variation due to some parameters in the model. It is obvious from the figures that healthy plants and uninfected whiteflies rise when there is an increase in plant mortality rate and a drop in vector mortality rate. In contrast, a drop in plant mortality rate and an increase in vector mortality rate leads to a rise in infected plants and whiteflies. The comparison of statistical data given by the ’NDSlove’ package in Mathematica with the outputs of NN-BLMA is illustrated in the tables below.
Table 6 illustrates the comparative analysis of both the solutions for case 1, while the comparison for case 2 and case 3 are illustrated in
Table 7 and
Table 8, respectively.