Thermodynamics in Ecology—An Introductory Review
Abstract
1. Introduction
2. History
3. The Thermodynamic Laws
3.1. The First Law of Thermodynamics
3.2. The Second Law of Thermodynamics
4. Some Fundamental Concepts
- (a)
- the various types of systems and
- (b)
- the relations between energy form and quality
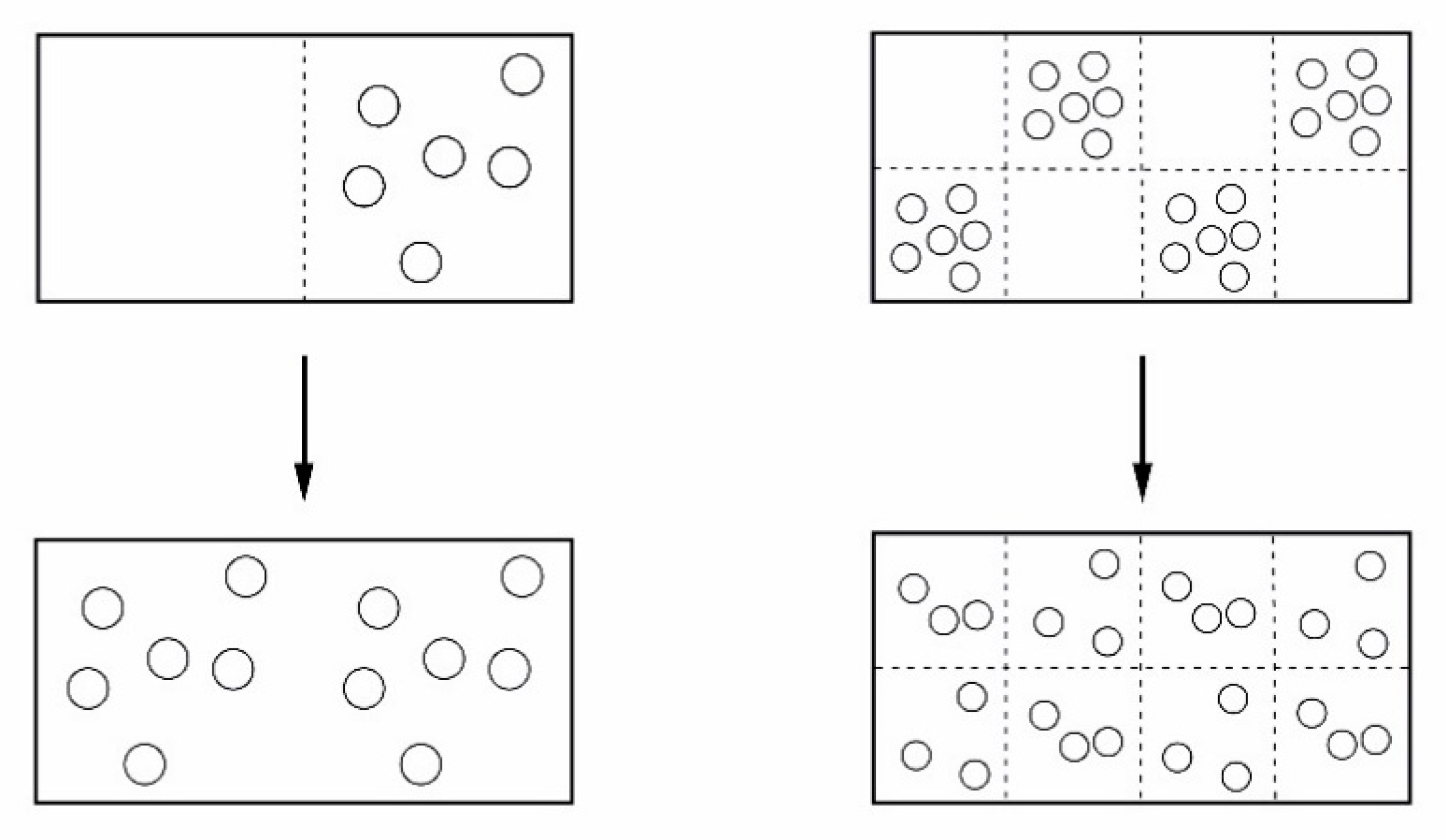
4.1. Types of Systems
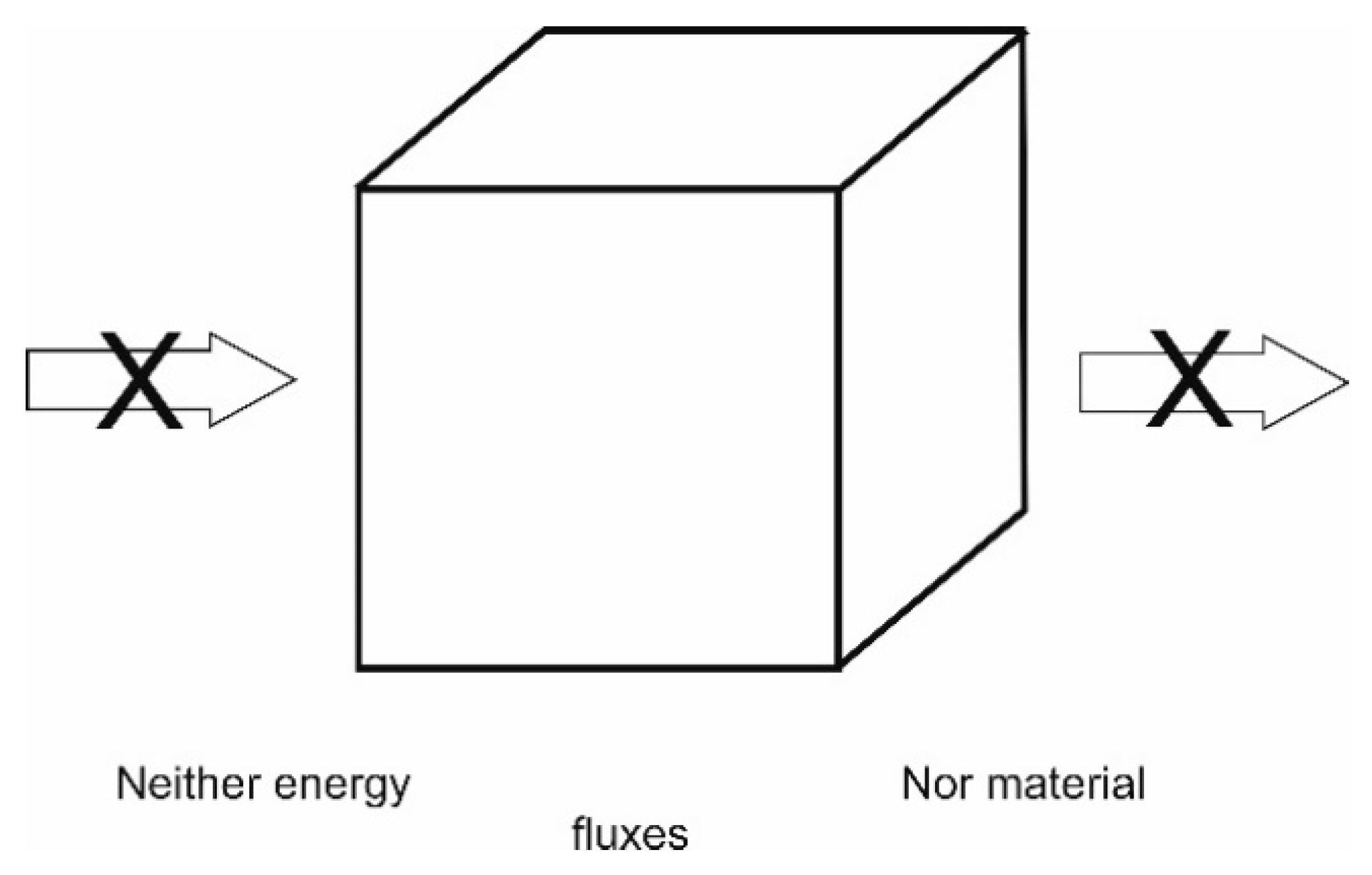
4.1.1. Isolated Systems—Or Adiabatic Systems
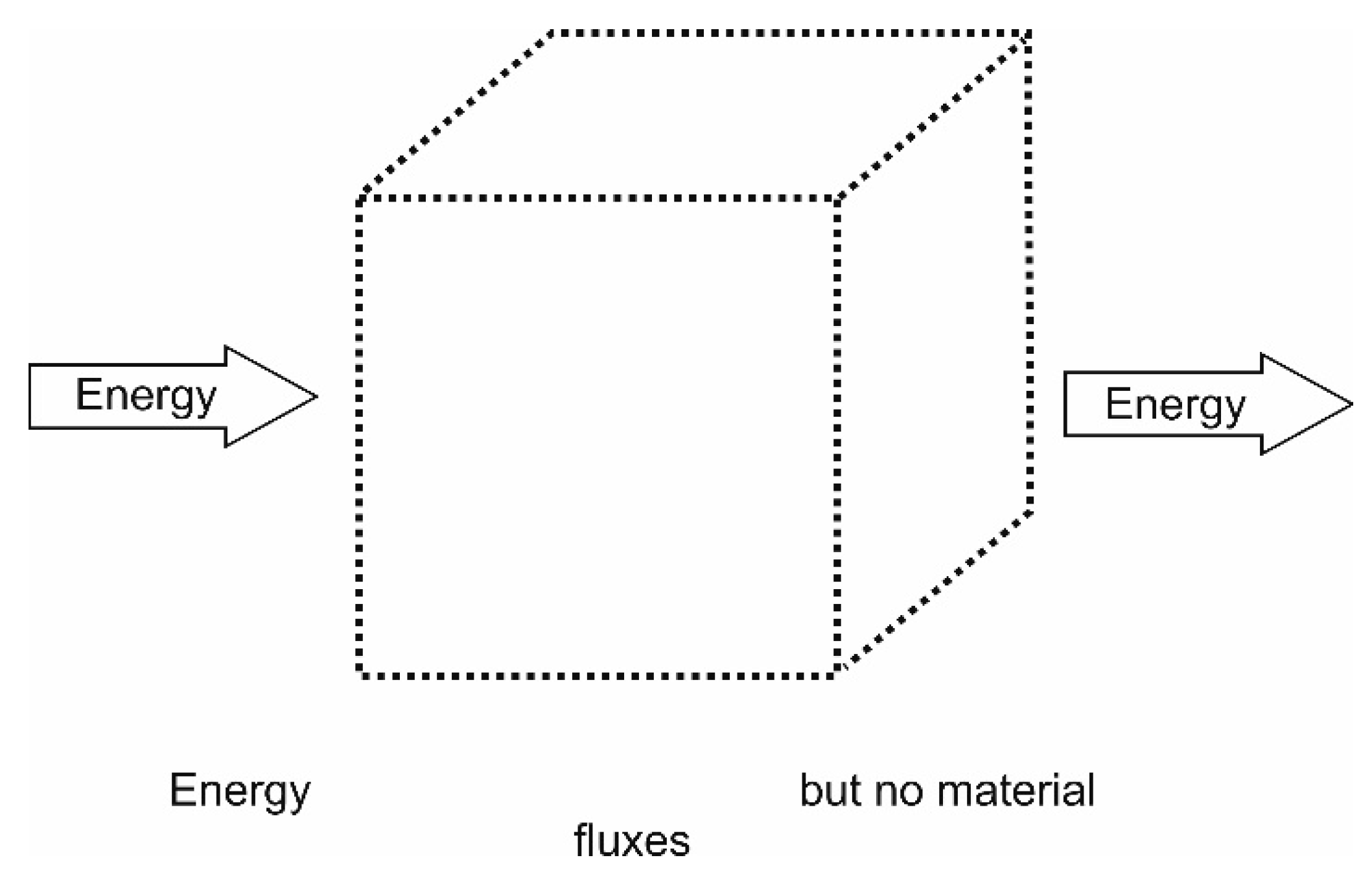
4.1.2. Closed Systems
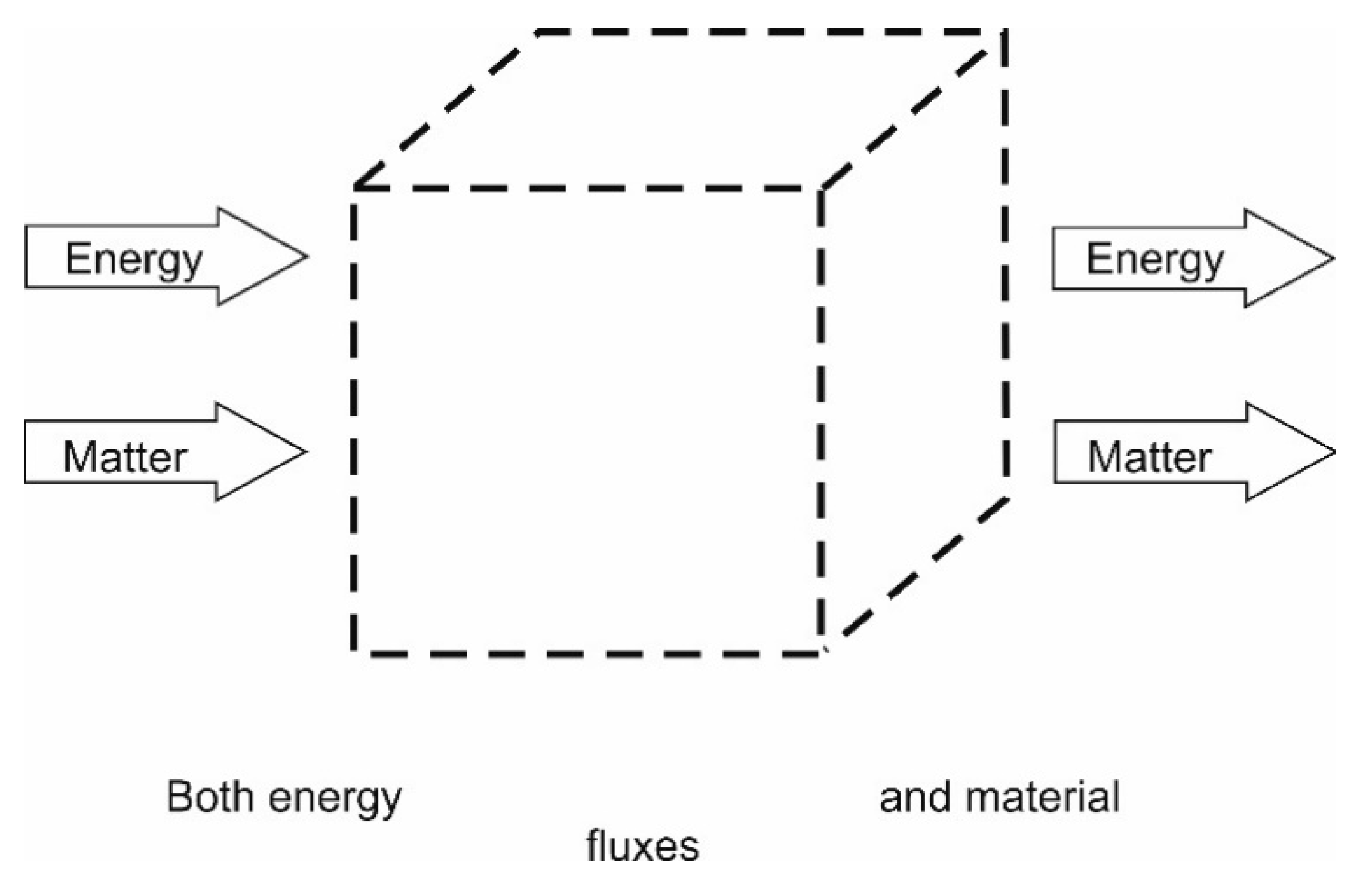
4.1.3. Open Systems
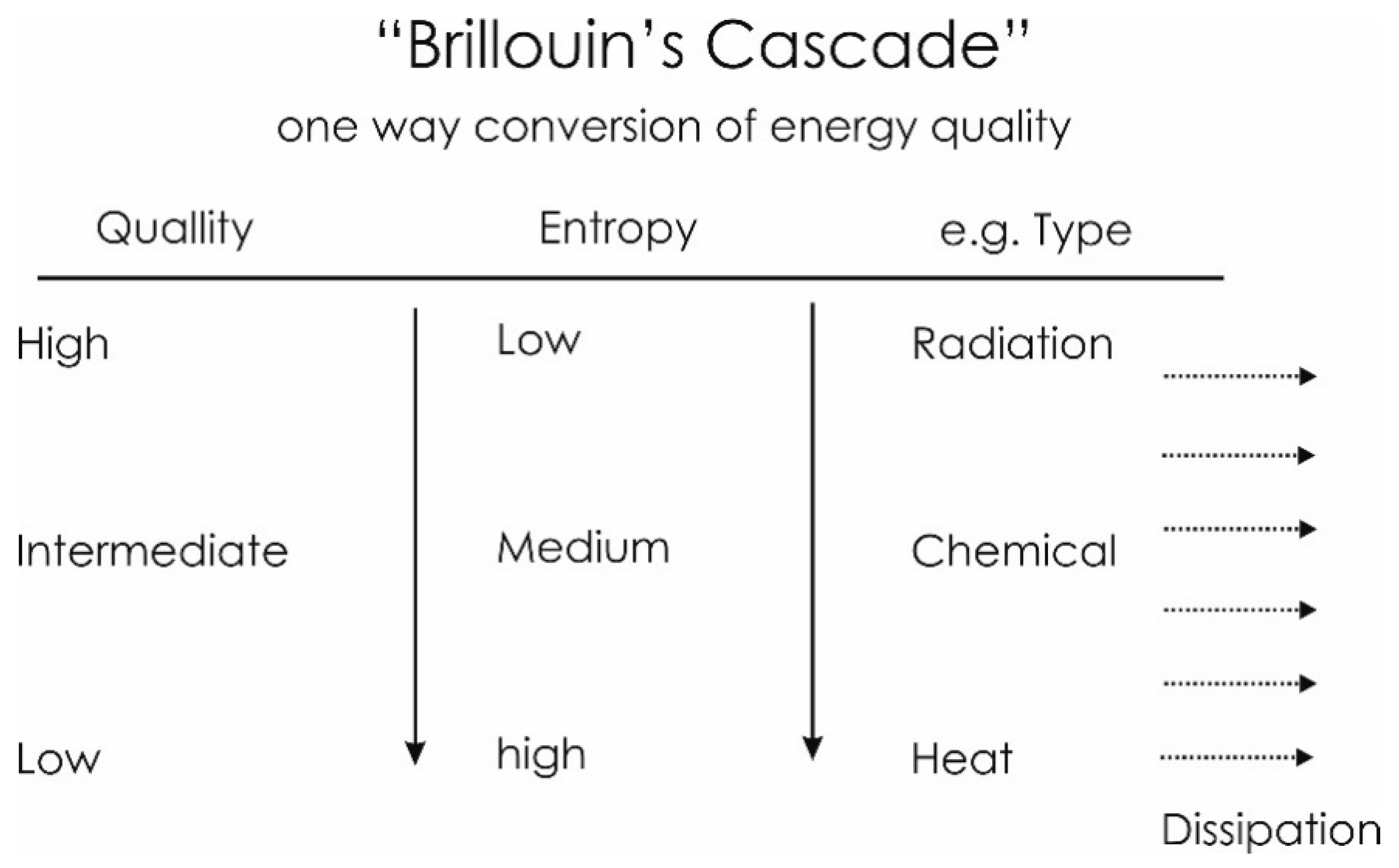
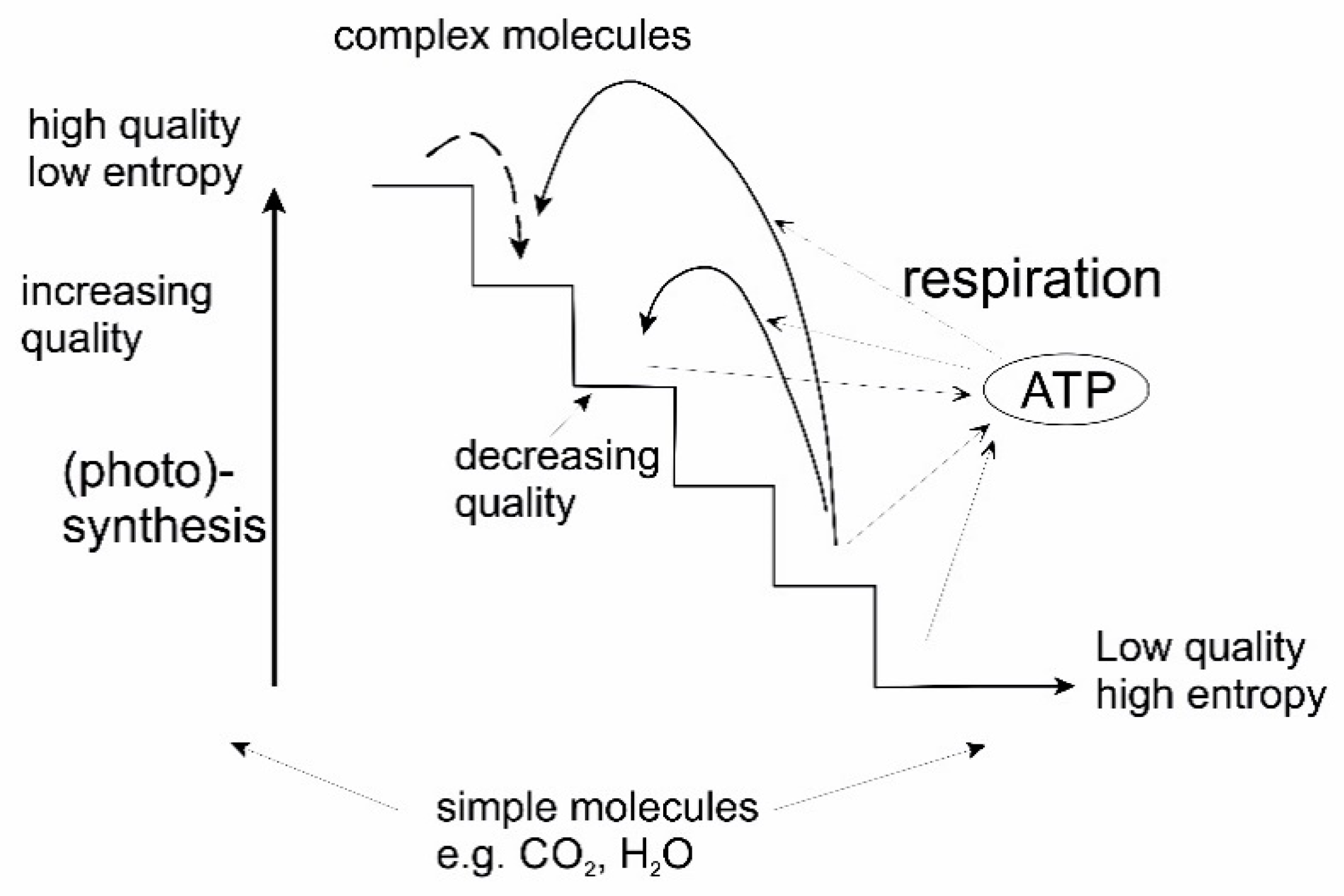
4.2. Energy: Form and Quality
5. Far from Equilibrium Thermodynamics
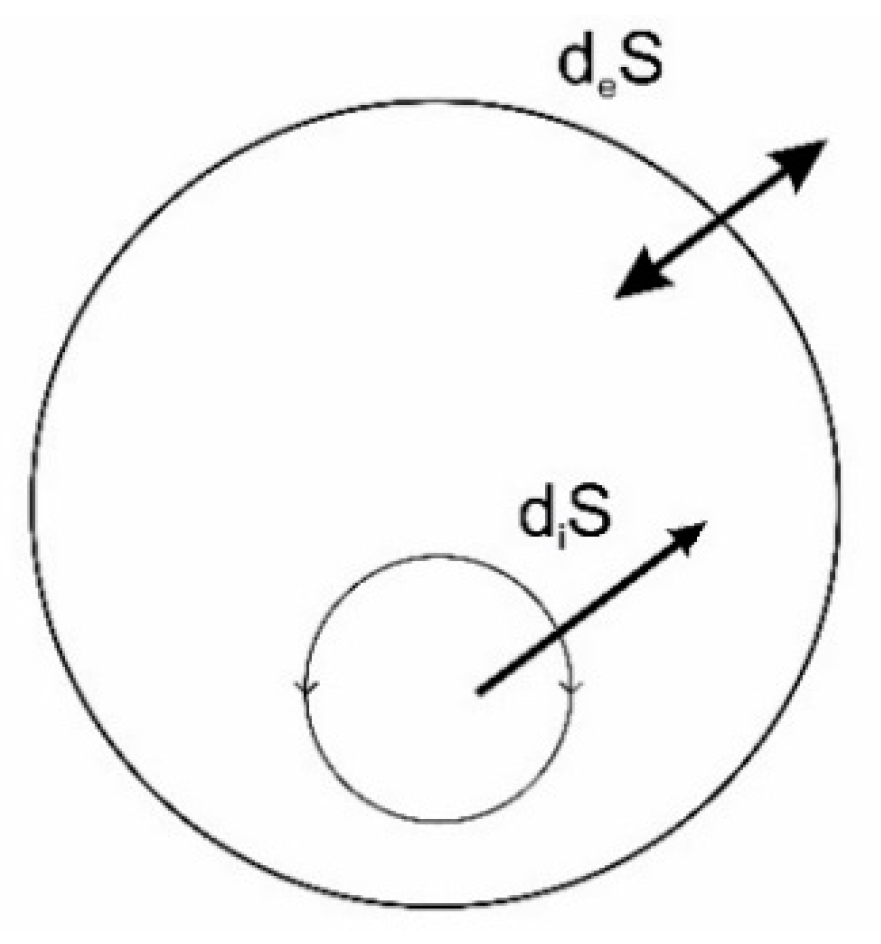
5.1. Dissipative Structures
5.2. Minimum Specific Dissipation
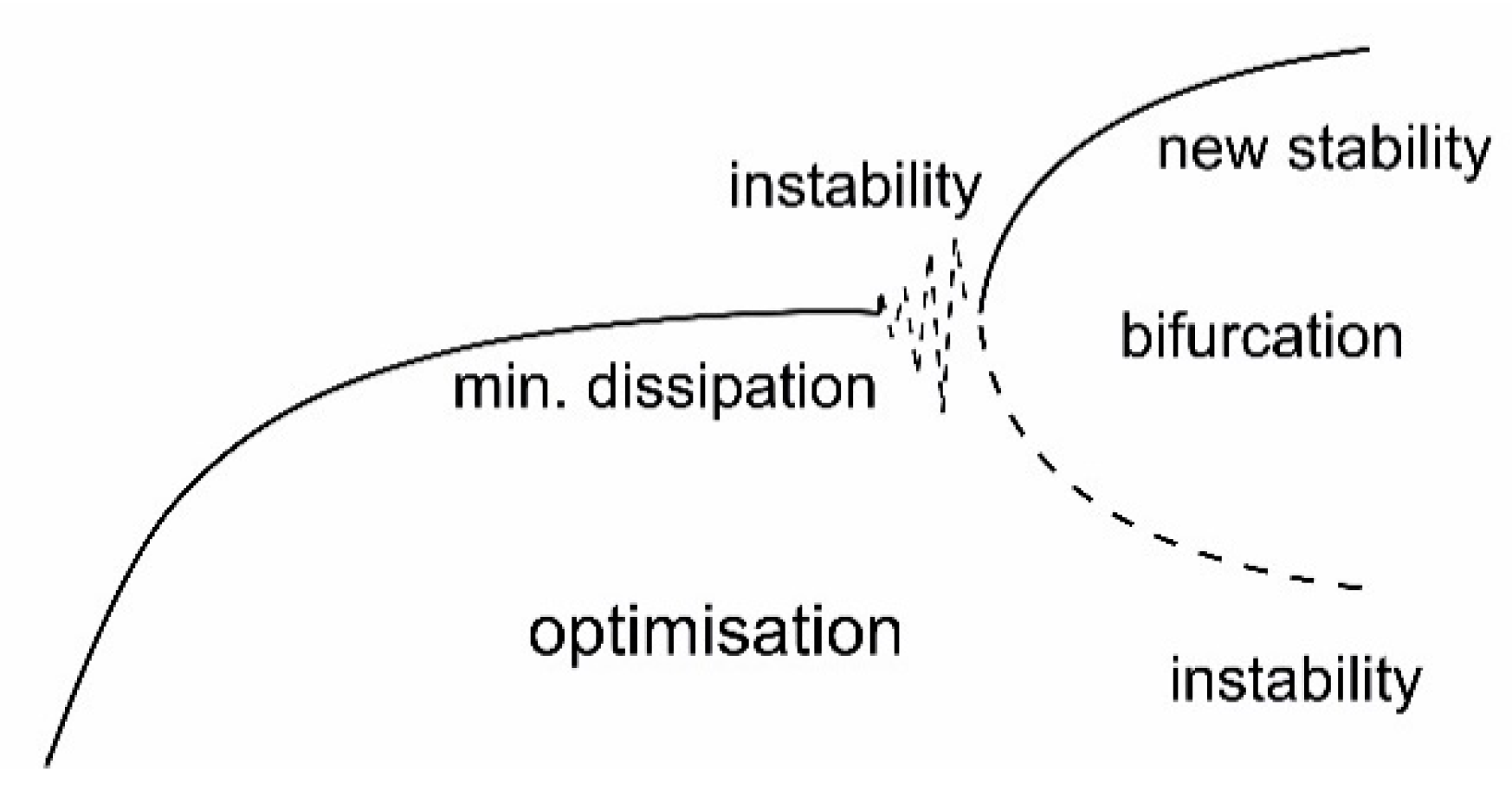
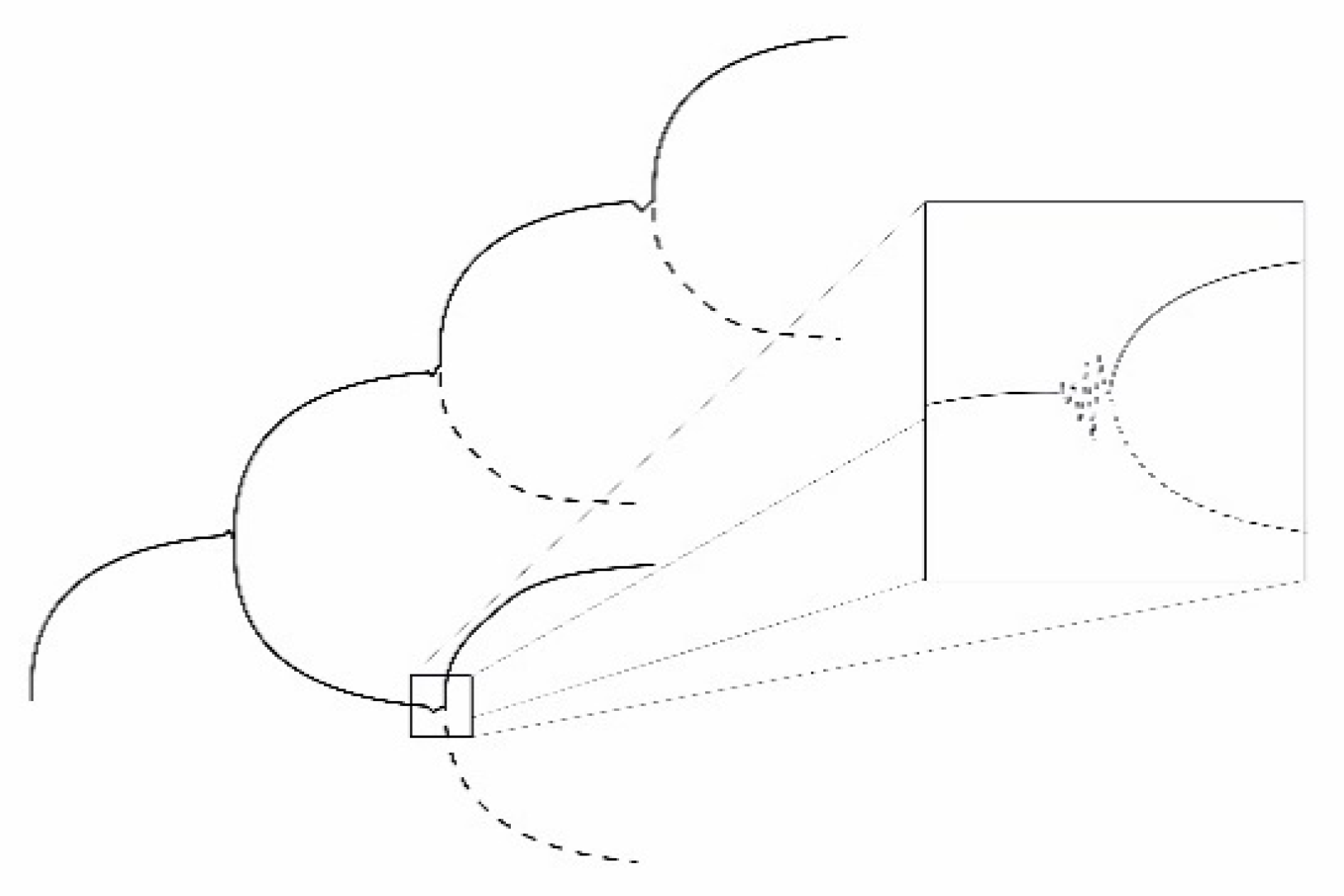
5.3. Evolution through Instabilities
5.4. An Ecological Law of Thermodynamics?
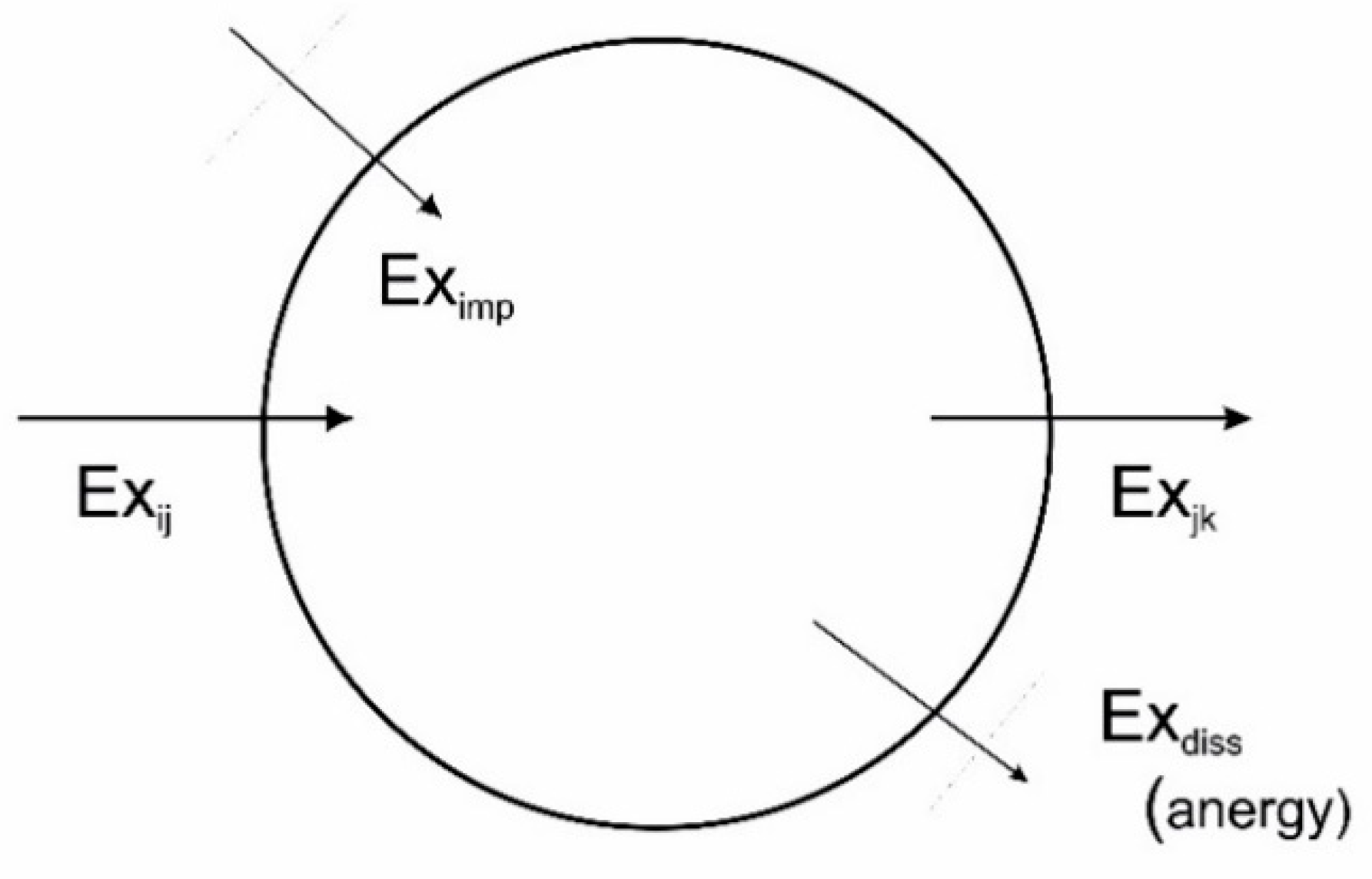
6. Exergetics
6.1. Thermodynamic Information
6.2. Exergy Optimization
- (1)
- Living systems—all biological systems as well as ecosystems are open systems in the sense that they import and exchange both energy and matter with the environment in which they are embedded
- (2)
- As the imported energy through metabolism is used for driving irreversible processes it is at the same time
- (a)
- converted into lower quality/value energy forms exporting dissipated energy to the surrounding environment, and/or
- (b)
- built into intermediate, chemical energy compounds, thereby
- (3)
- Building up structures through processes such as auto-poiesis, autocatalysis, and self-organization driven by the energy and material gradient of the system.
7. Application of Thermodynamics to Ecology
7.1. Entropy of Biological Systems
7.1.1. Entropy and Developmental Biology
7.1.2. Entropy and Organisms
7.1.3. Entropy of Ecosystems
7.1.4. Other Studies
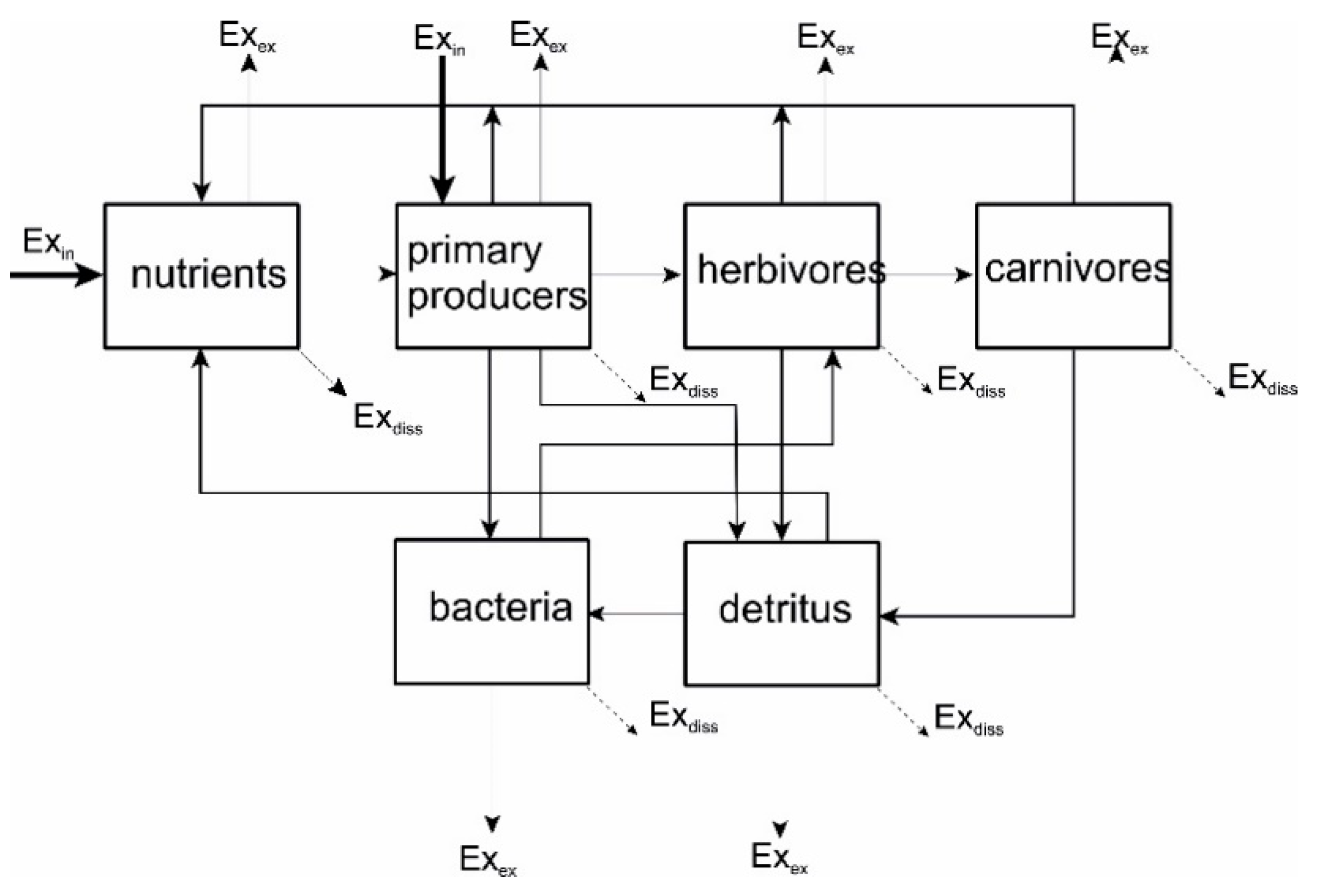
7.2. Exergy and Ecosystems
7.3. Exergy Storage
7.3.1. The Classical Approach
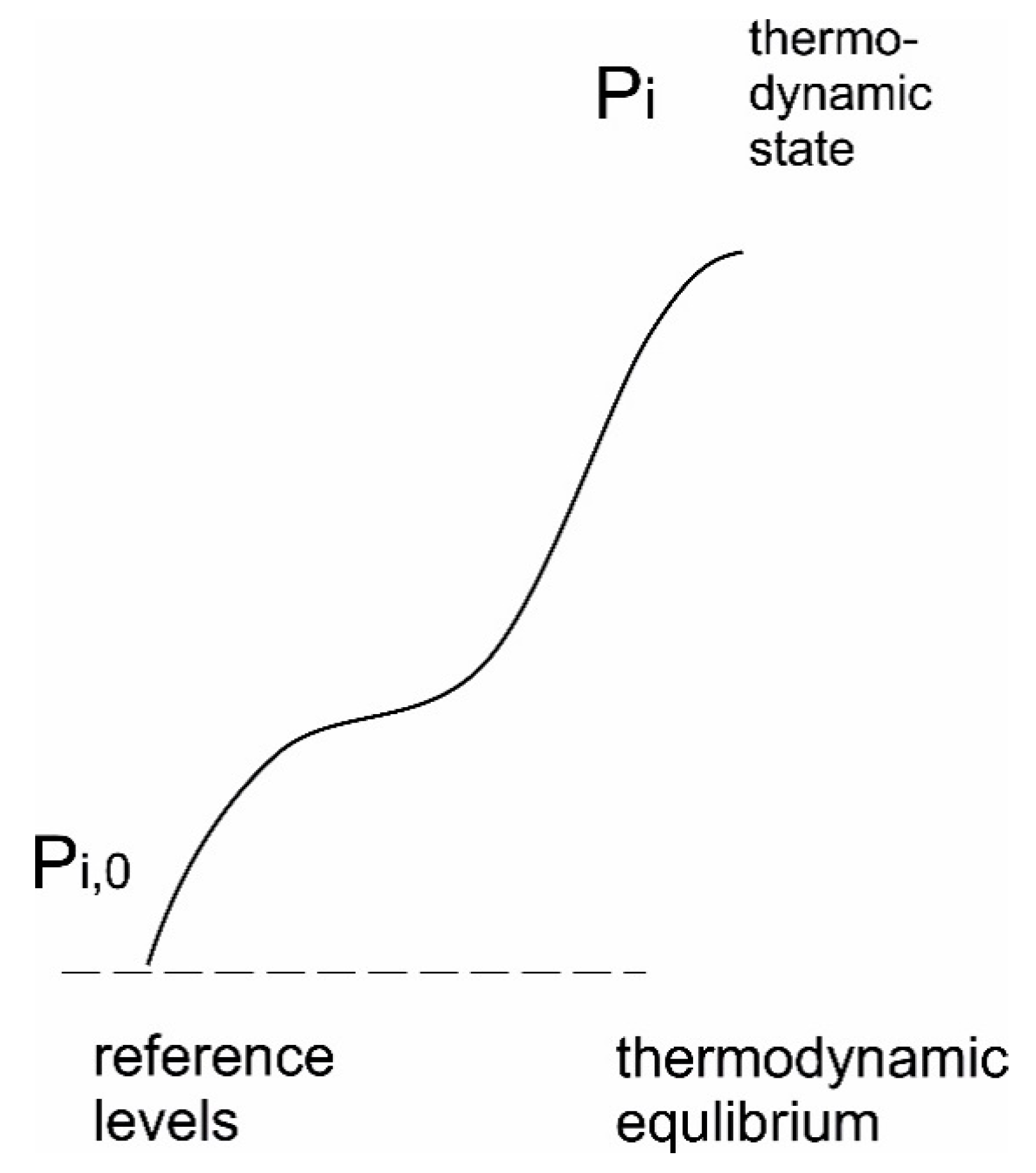
7.3.2. Internal Exergy
7.3.3. Exergy Indices
7.4. Application of Exergy Storage
7.4.1. Observation and Evaluation
7.4.2. Goal Functions
7.4.3. Comparisons to Other Ecosystem Theories
7.5. Exergy Degradation
“When an isolated (sic!) system performs a process after the removal of a series of internal constraints, it will reach a unique state of equilibrium: this state of equilibrium is independent of the order in which the constraints are removed”.
“As systems are moved away from equilibrium, they will utilize all avenues available to counter/resist the applied gradients. As the applied gradients increase, so does the system’s ability to oppose further the movement from equilibrium”[39]
7.6. Results of Exergy Degradation
7.6.1. Remote Sensing, Global
7.6.2. Landscapes and Regional Scale
7.7. Storage or Degradation
8. Discussion and Future
- (a)
- Problems related to science of physics - the science of thermodynamics and particular its extension into the far-from-equilibrium domain of conglomerate systems is still a relatively new discipline and in many ways in opposition to the Newtonian and determinist worldview still held by many scientists. As a consequence, many discussions are still taking place within the area.
- (b)
- Problems of transfer—whenever a scientific theory is transferred (reduced) to another area problems are to be expected. Does the theory, or the transfer of it, hold at all, for the whole set of systems or for parts of it, i.e., is the transfer to new conditions or domains valid?
- (c)
- Problems of application—after theoretical transfer problems of practical application appear. This in brief deals with both problems of measuring as well as how to proof the validity of such theories after transfer. Insofar, we must take much of the above statements as conjectures although much evidence of at least some important thermodynamic features of ecosystems has been gathered.
- (a)
- Most important to establishing a connection between thermodynamics and biology seems to be the necessary extension of the validity of thermodynamics into far-from-equilibrium conditions. The traditional point taken, stated in a very simplified form, would argue that thermodynamics only deals with ideal gases at conditions close to thermodynamic equilibrium. Whatever variety, or nuances, of this attitude will be taken, it will bring the transfer and application of thermodynamics into deep trouble. If one stands hard on the point that thermodynamics as science is valid only to “ideal gases close to (real) thermodynamic equilibrium, not only will the situation in biology and ecology be in deep trouble, so would a large part of the physical and engineering sciences as the universality and role of the second law together with its penetration into all other physical disciplines vanishes.
- (b)
- With the last points we implicitly address the problem of transfer to biological sciences and also to ecology as presented above. At this point it should be clear that not all problems come from the transfer alone, they existed already.
- (c)
- This brings us back to yet another problem in the application of the exergy principle. None of the above presented approaches is able to measure entropy, exergy or any kind of thermodynamic balance, directly. We have no entropy syringe or exergy meter to put on our system. This means that we are not able to fulfil the Cartesian demand of “making everything measurable”. In short, we will be forced to work with inductive or abductive based methods. Except, if we accept indirect measurements, calculation or modelling as valid methods for this purpose, which seems to be our only way out of this dilemma at the moment.
9. Summary and Conclusions
- (a)
- Thermodynamics in organizational levels and hierarchy perspectives
- (b)
- Thermodynamics of Earth and the biosphere
- (c)
- Entropy production vs. entropy state, exergy storage
- (d)
- Thermodynamic synergism
- (e)
- Ecological time-space thermodynamics
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Odum, E.P. Fundamentals of Ecology, 1st ed.; Saunders: Philadelphia, PA, USA, 1953. [Google Scholar]
- Odum, E.P. Fundamentals of Ecology, 2nd ed.; Saunders: Philadelphia, PA, USA, 1959. [Google Scholar]
- Odum, H.T. Environment, Power and Society; Wiley Interscience: Hoboken, NJ, USA, 1971. [Google Scholar]
- Higashi, M.; Burns, T.P. Theoretical Studies of Ecosystems: The Network Perspective; Cambridge University Press: Cambridge, UK, 1991; ISBN 9780521361385. [Google Scholar]
- Ulanowicz, R.E. Growth and Development—Ecosystems Phenomenology; Springer: Berlin/Heidelberg, Germany, 1986. [Google Scholar]
- Ulanowicz, R.E. Ecology, the Ascendent Perspective; Columbia University Press: New York, NY, USA, 1997; ISBN 9780231108287. [Google Scholar]
- Jørgensen, S.E. Integration of Ecosystem Theories: A Pattern; Springer: Dordrecht, The Netherlands, 1992; ISBN 9401051879. [Google Scholar]
- Jørgensen, S.E. Integration of Ecosystem Theories: A pattern, 3rd ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 2002; ISBN 1402007558. [Google Scholar]
- Ulanowicz, R.E. An hypothesis on the development of natural communities. J. Theor. Biol. 1980, 85, 223–245. [Google Scholar] [CrossRef]
- Ulanowicz, R.E.; Platt, T. Ecosystem theory for biological oceanography. Can. Bull. Fish. Aquat. Sci. 1985, 213, 65–75. [Google Scholar]
- Patten, B.C. Systems approach to the concept of environment. Ohio J. Sci. 1978, 78, 206–222. [Google Scholar]
- Müller, F.; Leupelt, M. Eco Targets, Goal Functions, and Orientors; Springer: Berlin/Heidelberg, Germany, 1998. [Google Scholar]
- May, R. Stability and Complexity in Model. Ecosystems; Princeton University Press: Princeton, NJ, USA, 1973. [Google Scholar]
- Schneider, E.D.; Kay, J.J. Order from disorder: The thermodynamics of complexity in biology. In What Is Life: The Next Fifty Years: Reflections on the Future of Biology; Murphy, M.P., O’Neill, L.A.J., Eds.; Cambridge University Press: Cambridge, UK, 1995. [Google Scholar]
- Kay, J.J.; Schneider, E.D. Embracing complexity—The challenge of the ecosystem approach. Alternatives 1994, 20, 32–38. [Google Scholar]
- Nielsen, S.N. Reductions in ecology and thermodynamics. On the problems arising when shifting the concept of exergy to other hierarchical levels and domains. Ecol. Indic. 2019, 100, 118–134. [Google Scholar] [CrossRef]
- Dyke, J.; Kleidon, A. The maximum entropy production principle: Its theoretical foundations and applications to the earth system. Entropy 2010, 12, 613–630. [Google Scholar] [CrossRef]
- Kleidon, A. Nonequilibrium thermodynamics and maximum entropy production in the Earth system: Applications and implications. Naturwissenschaften 2009, 96, 653–677. [Google Scholar] [CrossRef]
- Schymanski, S.J.; Kleidon, A.; Stieglitz, M.; Narula, J. Maximum entropy production allows a simple representation of heterogeneity in semiarid ecosystems. Pilos. Trans. R Soc. Lond. B. Biol. Sci. 2010, 365, 1449–1455. [Google Scholar] [CrossRef]
- Svirezhev, Y.M. Virtual biospheres: Complexity versus simplicity. Ann. N. Y. Acad. Sci. 1999, 879, 368–382. [Google Scholar] [CrossRef]
- Odum, E.P. The emergence of ecology as a new integrative discipline. Science 1977, 195, 1289–1293. [Google Scholar] [CrossRef]
- Odum, H.T.; Pinkerton, R.C. Time’s speed regulator: The optimum efficiency for maximum power output in physical and biological systems. Am. Sci. 1955, 43, 331–343. [Google Scholar]
- Odum, H.T. Pulsing, Power and Hierarchy. In Energetics and Systems; Mitsch, W.J., Ragade, R.K., Bosserman, R.W., Dillon, J.A., Jr., Eds.; Ann Arbor Science: Ann Arbor, MI, USA, 1982; p. 132. [Google Scholar]
- Lotka, A.J. Natural selection as a physical principle. Proc. Natl. Acad. Sci. USA 1922, 8, 151–154. [Google Scholar] [CrossRef] [PubMed]
- Odum, H.T. Self-organization, transformity, and information. Science 1988, 242, 1132–1139. [Google Scholar] [CrossRef] [PubMed]
- Odum, H.T. Environmental Accounting: EMERGY and Environmental Decision Making; Wiley and Sons: Hoboken, NJ, USA, 1996; ISBN 9780471114420. [Google Scholar]
- Mauersberger, P. On the role of entropy in water quality modelling. Ecol. Model. 1979, 7, 191–199. [Google Scholar] [CrossRef]
- Mauersberger, P. Entropy control of complex ecological processes. In Complex Ecology: The Part-Whole Relation in Ecosystems; Patten, B.C., Jørgensen, S.E., Eds.; Prentice Hall: Upper Saddle River, NJ, USA, 1995; p. 705. [Google Scholar]
- Prigogine, I.; Wiame, J.M. Biologie et thermodynamique des phénomènes irréversibles. Experientia 1946, 2, 451–453. [Google Scholar] [CrossRef] [PubMed]
- Glansdorff, P.; Prigogine, I. Thermodynamic Theory of Structure, Stability and Fluctuations; Wiley-Interscience: Hoboken, NJ, USA, 1971; ISBN 0471302805. [Google Scholar]
- Aoki, I. Entropy flows and entropy productions in the earth’s surface and in the earth’s atmosphere. J. Phys. Soc. Jpn. 1988, 57, 3262–3269. [Google Scholar] [CrossRef]
- Aoki, I.; Mizushima, T. Biomass diversity and stability of food webs in aquatic ecosystems. Ecol. Res. 2001, 16, 65–71. [Google Scholar] [CrossRef]
- Aoki, I. Entropy and exergy in the development of living systems: A case study of lake-ecosystems. J. Phys. Soc. Jpn. 1998, 67, 2132–2139. [Google Scholar] [CrossRef]
- Aoki, I. Monthly variations of entropy production in Lake Biwa. Ecol. Model. 1990, 51, 227–232. [Google Scholar] [CrossRef]
- Aoki, I. Entropy balance in Lake Biwa. Ecol. Model. 1987, 37, 235–248. [Google Scholar] [CrossRef]
- Jaynes, E.T. Where do we stand on maximum entropy? In The Maximum Entropy Formalism; Levine, R.D., Tribus, M., Eds.; MIT Press: Cambridge, MA, USA, 1979; p. 498. [Google Scholar]
- Mejer, H.F.; Jørgensen, S.E. Exergy and ecological buffer capacity. In Proceedings of the Conference on Ecological Modelling, Copenhagen, Denmark, 28 August–2 September 1979; pp. 829–846. [Google Scholar]
- Jørgensen, S.E.; Mejer, H.F. Exergy as key function in ecological models. In Energy and Ecological Modelling. Developments in Environmental Modelling 1; Mitsch, W.J., Bosserman, R.W., Klopatek, J.M., Eds.; Elservier: Amsterdam, The Netherlands, 1981; p. 839. [Google Scholar]
- Schneider, E.D.; Kay, J.J. Life as a manifestation of the second law of thermodynamics. Math. Comput. Model. 1994, 19, 25–48. [Google Scholar] [CrossRef]
- Schneider, E.D.; Kay, J.J. Complexity and thermodynamics. Towards a new ecology. Futures 1994, 26, 626–647. [Google Scholar] [CrossRef]
- Schneider, E.D.; Kay, J.J. Exergy degradation. Thermodynamics and the development of Ecosystems. In Proceedings of the International Conference ENSEC ’93 Energy systems and Ecology, Cracow, Poland, 59 July 1993. [Google Scholar]
- Martyushev, L.M.; Seleznev, V.D. Maximum entropy production principle in physics, chemistry and biology. Phys. Rep. 2006, 426, 1–45. [Google Scholar] [CrossRef]
- Martyushev, L.M. The maximum entropy production principle: Two basic questions. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 1333–1334. [Google Scholar] [CrossRef] [PubMed]
- Ulanowicz, R.E. Formal agency in ecosystem development. In Theoretical Studies of Ecosystems. The Network Perspective; Higashi, M., Burns, T.P., Thomas, P., Eds.; Cambridge University Press: Cambridge, UK, 1991; p. 364. [Google Scholar]
- Ulanowicz, R.E. Trophic flow networks as indicators of ecosystem stress. In Food Webs: Integration of Patterns and Dynamics; Polis, G., Winemiller, K., Eds.; Chapmann-Hall: New York, NY, USA, 1995. [Google Scholar]
- Patten, B.C. On the quantitative dominance of indirect effects in ecosystems. In Analysis of Ecological Systems: State-of-the-Art in Ecological Modelling: Developments in Environmental Modelling 5; Lauenroth, W.K., Skogerboe, G.V., Flug, M., Eds.; Elsevier: Amsterdam, The Netherlands, 1983; p. 992. [Google Scholar]
- Fath, B.D.; Patten, B.C. A utility goal function based on network synergism. In Eco Targets, Goal Functions, and Orientors; Müller, F., Leupelt, M., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; p. 619. [Google Scholar]
- Straškraba, M. Natural control mechanisms in models of aquatic ecosystems. Ecol. Model. 1979, 6, 305–321. [Google Scholar] [CrossRef]
- Margalef, R. Perspectives in Ecological Theory; University of Chicago Press: Chicago, IL, USA, 1968; ISBN 9780226505053. [Google Scholar]
- von Bertalanffy, L. General System Theory. Foundations, Development, Applications, Revised ed.; George Braziller: New York, NY, USA, 1969. [Google Scholar]
- Mathes, K.; Breckling, B.; Ekschmitt, K. Systemtheorie in der Ökologie; Springer: Berlin/Heidelberg, Germany, 1996. [Google Scholar]
- Jørgensen, S.E.; Fath, B.D.; Bastianoni, S.; Marques, J.C.; Müller, F.; Nielsen, S.N.; Patten, B.C.; Tiezzi, E.; Ulanowicz, R.E. A New Ecology—Systems Perspective; Elsevier: Amsterdam, The Netherlands, 2007; ISBN 9780444531605. [Google Scholar]
- Nielsen, S.N.; Fath, B.D.; Bastianoni, S.; Marques, J.C.; Müller, F.; Patten, B.C.; Ulanowicz, R.E.; Jørgensen, S.E.; Tiezzi, E. A New Ecology: Systems Perspective; Elsevier: Amsterdam, The Netherlands, 2019; ISBN 9780444637574. [Google Scholar]
- Hall, C.A.S. Maximum Power. The Ideas and Applications of H.T. Odum; University of Colorado Press: Boulder, CO, USA, 1995. [Google Scholar]
- Lotka, A.J. Contribution to the energetics of evolution. Proc. Natl. Acad. Sci. USA 1922, 8, 147–151. [Google Scholar] [CrossRef]
- Lotka, A.J. Elements of Physical Biology; Williams and Wilkins Company: Philadelphia, PA, USA, 1925. [Google Scholar]
- Bastianoni, S.; Marchettini, N.; Panzieri, M.; Tiezzi, E. Environmental sustainability indicators: Thermodynamic aspects. Ann. Chim. 1998, 88, 755–760. [Google Scholar]
- Bastianoni, S.; Marchettini, N. Emergy/exergy ratio as a measure of the level of organization of systems. Ecol. Model. 1997, 99, 33–40. [Google Scholar] [CrossRef]
- Odum, H.T. Systems Ecology: An. Introduction; Wiley and Sons: Hoboken, NJ, USA, 1983; ISBN 0471652776. [Google Scholar]
- Odum, H.T. Maximum power and efficiency: A rebuttal. Ecol. Model. 1983, 20, 71–82. [Google Scholar] [CrossRef]
- Kangas, P. Contributions of H.T. Odum to ecosystem simulation modelling. In Maximum Power. The Ideas and Applications of H.T. Odum; University of Colorado Press: Boulder, CO, USA, 1995; p. 393. [Google Scholar]
- Leontief, W. Quantitative input and output relations in the economic system of the United States. Rev. Econ. Stat. 1936, 18, 105–125. [Google Scholar] [CrossRef]
- Leontief, W. Input-Output Analysis: Foundations and Extensions; Oxford University Press: Oxford, UK, 1966. [Google Scholar]
- Hannon, B. The structure of ecosystems. J. Theor. Biol. 1973, 41, 535–546. [Google Scholar] [CrossRef]
- Hannon, B. Total energy costs in ecosystems. J. Theor. Biol. 1979, 80, 271–293. [Google Scholar] [CrossRef]
- Finn, J.T. Measures of ecosystem structure and function derived from analysis of flows. J. Theor. Biol. 1976, 56, 363–380. [Google Scholar] [CrossRef]
- Finn, J.T. Flow analysis of models of the Hubbard brook ecosystem. Ecology 1980, 61, 562–571. [Google Scholar] [CrossRef]
- Onsager, L. Reciprocal relations in irreversible processes. I. Phys. Rev. 1931, 37, 405–426. [Google Scholar] [CrossRef]
- Onsager, L. Reciprocal relations in irreversible processes. II. Phys. Rev. 1931, 38, 2265–2279. [Google Scholar] [CrossRef]
- Prigogine, I. Etude Thermodynamique des Phènomènes Irreversibles. Ph.D. Thesis, Free University of Bruxelles, Bruxelles, Belgium, 1947. [Google Scholar]
- Prigogine, I.; Nicolis, G. On symmetry-breaking instabilities in dissipative systems. J. Chem. Phys. 1967, 46, 3542–3550. [Google Scholar] [CrossRef]
- Prigogine, I. Time, structure, and fluctuations. Science 1978, 201, 777–785. [Google Scholar] [CrossRef]
- Prigogine, I. Order out of chaos. In Energetics and Systems; Mitsch, W.J., Ragade, R.K., Bosserman, R.W., Dillon, J.A., Jr., Eds.; Ann Arbor Science: Ann Arbor, MI, USA, 1982; p. 132. [Google Scholar]
- Nicolis, G.; Prigogine, I. Self-Organization in Nonequilibrium Systems: From Dissipative Structures to Order Through Fluctuations; Wiley and Sons: Hoboken, NJ, USA, 1977; ISBN 9780471024019. [Google Scholar]
- Prigogine, I.; Nicolis, G. Biological order, structure and instabilities. Quart Rev. Biophys. 1971, 4, 107–148. [Google Scholar] [CrossRef]
- Martyushev, L.M. Minimal time, Weibull distribution and maximum entropy production principle: Comment on “Redundancy principle and the role of extreme statistics in molecular and cellular biology” by Z. Phys. Life Rev. 2019, 28, 83–84. [Google Scholar] [CrossRef]
- Prigogine, I.; Stengers, I. Order Out of Chaos: Man’s New Dialogue with Nature; Bantam Books: New York, NY, USA, 1984; ISBN 0553343637. [Google Scholar]
- Nicolis, G.; Prigogine, I. Exploring Complexity: An Introduction; W.H. Freeman: New York, NY, USA, 1989; ISBN 9780716718598. [Google Scholar]
- Skene, K.R. Life’s a gas: A thermodynamic theory of biological evolution. Entropy 2015, 17, 5522–5548. [Google Scholar] [CrossRef]
- Baranowski, B. Some thermodynamical comments on the origin and evolution of life. Viva Orig. 1981, 9, 49–54. [Google Scholar]
- Berry, S. Entropy, irreversibility and evolution. J. Theor. Biol. 1995, 175, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Brooks, D.; Wiley, E.O. Evolution as Entropy—Toward a Unified Theory of Biology, 2nd ed.; University of Chicago Press: Chicago, IL, USA, 1988. [Google Scholar]
- Elitzur, A.C. Life’s emergence is not an axiom: A reply to Yockey. J. Theor. Biol. 1996, 180, 175–180. [Google Scholar] [CrossRef]
- Wesley, J.P. Ecophysics—The Application of Physics to Ecology; Thomas: Springfield, IL, USA, 1974. [Google Scholar]
- Wesley, J.P. Life and thermodynamic ordering of earth’s surface. Evol. Theory 1989, 9, 45–56. [Google Scholar]
- Wesley, J.P. Selected Topics in Advanced Fundamental Physics; Benjamin Wesley: Blumberg, Germany, 1991. [Google Scholar]
- Weber, B.H.; Depew, D.J.; Smith, J.D. Entropy, Information, and Evolution: New Perspectives on Physical and Biological Evolution; MIT Press: Cambridge, MA, USA, 1988; ISBN 9780262231329. [Google Scholar]
- Michaelian, K. Thermodynamic stability of ecosystems. J. Theor. Biol. 2005, 237, 323–335. [Google Scholar] [CrossRef]
- Michaelian, K. Microscopic dissipative structuring and proliferation at the origin of life. Heliyon 2017, 3. [Google Scholar] [CrossRef]
- Kompanichenko, V.N. Inversion concept of the origin of life. Orig. Life Evol. Biosph. 2012, 42, 153–178. [Google Scholar] [CrossRef]
- Skene, K.R. Thermodynamics, ecology and evolutionary biology: A bridge over troubled water or common ground? Acta Oecol. 2017, 85, 116–125. [Google Scholar] [CrossRef]
- Aoki, I. Entropy principle for the evolution of living systems and the universe from bacteria to the universe. J. Phys. Soc. Jpn. 2018, 87. [Google Scholar] [CrossRef]
- Hansen, L.D.; Popovic, M.; Tolley, H.D.; Woodfield, B. Laws of evolution parallel the laws of thermodynamics. J. Chem. Thermodyn. 2018, 124, 141–148. [Google Scholar] [CrossRef]
- Tiezzi, E. Steps Towards and Evolutionary Physics; WIT Press: Southampton, UK, 2006. [Google Scholar]
- Lewontin, R.C. The meaning of stability. Bookhaven Symp. Biol. 1969, 22, 13–24. [Google Scholar]
- Alimov, A.F. Structural and functional characteristics of aquatic animal communities. Int. Rev. Gesamten Hydrobiol. Hydrogr. 1991, 76, 169–182. [Google Scholar] [CrossRef]
- Kiflemariam, M. Dialectics of biological dimensionality and biological diversity. COENOSES 1993, 8, 149–158. [Google Scholar]
- Livingston, R.J. Diurnal and seasonal fluctuations of organisms in a North Florida Estuary. Estuar. Coast. Mar. Sci. 1976, 4, 373–400. [Google Scholar] [CrossRef]
- Westfall, R.H.; Theron, G.K.; Rooyen, N. Objective classification and analysis of vegetation data. Plant. Ecol. 1997, 132, 137–154. [Google Scholar] [CrossRef]
- Margalef, R. Our biosphere. In Excellence in Ecology; Ecology Institute: Odendorf/Luhe, Germany, 1997. [Google Scholar]
- Marques, J.C. Diversity, biodiversity, conservation, and sustainability. Sci. World J. 2001, 1, 534–543. [Google Scholar] [CrossRef]
- Marques, J.C. Diversity and biodiversity: What might be their significance in the scope of sustainable development? Bol. Mus. Mun. Funchal 2001, 6, 349–361. [Google Scholar]
- Brookhaven National Laboratory. Diversity and stability in ecological systems. Brookhaven Symp. Biol. 1969, 22, 1–264. [Google Scholar]
- Leinster, T.; Meckes, M. Maximizing diversity in biology and beyond. Entropy 2016, 18, 88. [Google Scholar] [CrossRef]
- Newman, E.A.; Kennedy, M.C.; Falk, D.A.; McKenzie, D. Scaling and complexity in landscape ecology. Front. Ecol. Evol. 2019, 7, 293. [Google Scholar] [CrossRef]
- Gao, P.; Li, Z. Computation of the Boltzmann entropy of a landscape: A review and a generalization. Landsc. Ecol. 2019, 34, 2183–2196. [Google Scholar] [CrossRef]
- Nielsen, S.N. Towards an ecosystem semiotics. Some basic aspects for a new research programme. Ecol. Complex. 2007, 4, 93–101. [Google Scholar] [CrossRef]
- Nielsen, S.N. Second order cybernetics and semiotics in ecological systems—Where complexity really begins. Ecol. Model. 2015, 319, 119–129. [Google Scholar] [CrossRef]
- Maze, J.; Scagel, R.K. A different view of plant morphology and the evolution of form. Syst. Bot. 1983, 8, 469–472. [Google Scholar]
- Meysman, F.J.R.; Bruers, S. A thermodynamic perspective on food webs: Quantifying entropy production within detrital-based ecosystems. J. Theor. Biol. 2007, 249, 124–139. [Google Scholar] [CrossRef] [PubMed]
- Popović, M. Entropy change of open thermodynamic systems in self-organizing processes. Therm. Sci. 2014, 18, 1425–1432. [Google Scholar] [CrossRef]
- Demetrius, L.A.; Gundlach, V.M. Directionality theory and the entropic principle of natural selection. Entropy 2014, 16, 5428–5522. [Google Scholar] [CrossRef]
- Yen, J.D.L.; Paganin, D.M.; Thomson, J.R.; Mac Nally, R. Thermodynamic extremization principles and their relevance to ecology. Austral. Ecol. 2014, 39, 619–632. [Google Scholar] [CrossRef]
- Yen, J.D.L.; Paganin, D.M.; Thomson, J.R.; Mac Nally, R. Thermodynamics predicts density-dependent energy use in organisms and ecological communities. Phys. Rev. E Stat. Nonlinear Soft Matter Phys. 2015, 91, 042708. [Google Scholar] [CrossRef]
- Marsland, R.; Cui, W.; Goldford, J.; Sanchez, A.; Korolev, K.; Mehta, P. Available energy fluxes drive a transition in the diversity, stability, and functional structure of microbial communities. PLoS Comput. Biol. 2019, 15, e1006793. [Google Scholar] [CrossRef] [PubMed]
- Martyushev, L.M.; Nazarova, A.S.; Seleznev, V.D. On the problem of the minimum entropy production in the nonequilibrium stationary state. J. Phys. A Math. Theor. 2007, 40, 371–380. [Google Scholar] [CrossRef]
- Martyushev, L.M. Entropy and entropy production: Old misconceptions and new breakthroughs. Entropy 2013, 15, 1152–1170. [Google Scholar] [CrossRef]
- Morowitz, H.J. Foundations of Bioenergetics; Academic Press: Cambridge, MA, USA, 1978. [Google Scholar]
- Carnot, S. Reflections on the Motive Power of Fire, and on Machines Fitted to Develop that Power. In The Second Law of Thermodynamics. Benchmark Papers on Energy 5; Kestin, J., Ed.; Dowden, Hutchinson and Ross: Stroudsburg, PA, USA, 1976; p. 329. [Google Scholar]
- Clausius, R. Über verschiedene für die anwendung bequeme formen der hauptgleichungen der mechanische wärmetheorie. In The Second Law of Thermodynamics. Benchmark Papers on Energy 5; Kestin, J., Ed.; Dowden, Hutchinson and Ross: Stroudsburg, PA, USA, 1976; p. 329. [Google Scholar]
- Boltzmann, L. The second law of thermodynamics, Populare Schriften, Essay no. 3, address to the Imperial Academy of Sciences 1886. In Theoretical Physics and Philosophical Problems, Selected Writings of L. Boltzmann; D. Reidel: Dordrecht, The Netherlands, 1905. [Google Scholar]
- Gibbs, J.W. On the equilibrium of heterogeneous substances. In The Second Law of Thermodynamics. Benchmark Papers on Energy 5; Kestin, J., Ed.; Dowden, Hutchinson and Ross: Stroudsburg, PA, USA, 1976; p. 329. [Google Scholar]
- Schrödinger, E. What Is Life; Cambridge University Press: Cambridge, UK, 1944. [Google Scholar]
- Nicolis, G.; Prigogine, I. Fluctuations in nonequilibrium systems. Proc. Natl. Acad. Sci. USA 1971, 68, 2102–2107. [Google Scholar] [CrossRef]
- Glansdorff, P.; Nicolis, G.; Prigogine, I. The thermodynamic stability theory of non-equilibrium states. Proc. Natl. Acad. Sci. USA 1974, 71, 197–199. [Google Scholar] [CrossRef]
- Leff, H.S.; Rex, A.F. Maxwell’s Demon: Entropy, Information, Computing; Adam Hilger: London, UK, 1990; ISBN 0750300566. [Google Scholar]
- Brillouin, L. Science and Information Theory; Academic Press: Cambridge, MA, USA, 1960. [Google Scholar]
- Thomson, W. On a universal tendency in nature to the dissipation of mechanical energy. Reprinted from: Mathematical and Physical Papers of William Thomson. In The Second Law of Thermodynamics. Benchmark Papers on Energy 5; Kestin, J., Ed.; Dowden, Hutchinson and Ross: Stroudsburg, PA, USA, 1976; p. 329. [Google Scholar]
- Schrödinger, E. Statistical Thermodynamics, 2nd ed.; Dover Publications: Mineola, NY, USA, 1952. [Google Scholar]
- Kestin, J.; Dorfman, J.R. A Course in Statistical Thermodynamics; Academic Press: Cambridge, MA, USA, 1971; ISBN 0124053505. [Google Scholar]
- Lambert, F.L. Disorder—A cracked crutch for supporting entropy discussions. J. Chem. Educ. 2002, 79, 187. [Google Scholar] [CrossRef]
- Lambert, F.L. The misinterpretation of entropy as “disorder”. J. Chem. Educ. 2012, 89, 310. [Google Scholar] [CrossRef]
- Kirwan, A.D. Quantum and ecosystem entropies. Entropy 2008, 10, 58–70. [Google Scholar] [CrossRef]
- Velasco, R.M.; García-Colín, L.S.; Uribe, F.J. Entropy production: Its role in non-equilibrium thermodynamics. Entropy 2011, 13, 82–116. [Google Scholar] [CrossRef]
- Baverstock, K. Life as physics and chemistry: A system view of biology. Prog. Biophys. Mol. Biol. 2013, 111, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Popović, M.E. Research in entropy wonderland: A review of the entropy concept. Therm. Sci. 2018, 22, 1163–1178. [Google Scholar] [CrossRef]
- Rodríguez, R.A.; Herrera, A.M.; Otto, R.; Delgado, J.D.; Fernández-Palacios, J.M.; Arévalo, J.R. Ecological state equation. Ecol. Model. 2012, 224, 18–24. [Google Scholar] [CrossRef]
- Nielsen, S.N. Thermodynamic constraints of life as downward causation in ecosystems. Cybern. Hum. Knowing 2009, 16, 27–49. [Google Scholar]
- Lad, F.; Sanfilippo, G.; Agrò, G. The duality of entropy/extropy, and completion of the Kullback information complex. Entropy 2018, 20, 593. [Google Scholar] [CrossRef]
- Carr-Chellman, A.; Kitchel, A.; Freeman, S. Negentropy: Energy creating tools for organizational development. TechTrends 2020, 64, 275–279. [Google Scholar] [CrossRef]
- Bejan, A. Fundamentals of exergy analysis, entropy generation minimization, and the generation of flow architecture. Int. J. Energy Res. 2002, 26. [Google Scholar] [CrossRef]
- Županović, P.; Kuić, D.; Lošić, Ž.B.; Petrov, D.; Juretić, D.; Brumen, M. The maximum entropy production principle and linear irreversible processes. Entropy 2010, 12, 996–1005. [Google Scholar] [CrossRef]
- Harte, J.; Newman, E.A. Maximum information entropy: A foundation for ecological theory. Trends Ecol. Evol. 2014, 29, 384–389. [Google Scholar] [CrossRef]
- Chapman, E.J.; Childers, D.L.; Vallino, J.J. How the second law of thermodynamics has informed ecosystem ecology through its history. Bioscience 2016, 66, 27–39. [Google Scholar] [CrossRef]
- Brummer, A.B.; Newman, E.A. Derivations of the core functions of the maximum entropy theory of ecology. Entropy 2019, 21, 712. [Google Scholar] [CrossRef]
- Shannon, C.E.; Weaver, W. The Mathematical Theory of Communication; University of Illinois Press: Champaign, IL, USA, 1949; ISBN 9780252725487. [Google Scholar]
- Tribus, M.; Shannon, P.T.; Evans, R.B. Why thermodynamics is a logical consequence of information theory. AIChE J. 1966, 12, 244–248. [Google Scholar] [CrossRef]
- Wicken, J.S. Evolution, Thermodynamics, and Information: Extending the Darwinian Program; Oxford University Press: Oxford, UK, 1987; ISBN 0195043189. [Google Scholar]
- O’Connor, M.I.; Pennell, M.W.; Altermatt, F.; Matthews, B.; Melián, C.J.; Gonzalez, A. Principles of ecology revisited: Integrating information and ecological theories for a more unified science. Front. Ecol. Evol. 2019, 7, 219. [Google Scholar] [CrossRef]
- Ryan, J.P. Information—Entropy interfaces and different levels of biological organization. J. Theor. Biol. 1980, 84, 31–48. [Google Scholar] [CrossRef]
- Stonier, T. Towards a general theory of information II: Information and entropy. Aslib. Proc. 1989, 41, 41–55. [Google Scholar] [CrossRef]
- Stonier, T. Information and the Internal Structure of the Universe: An Exploration into Information Physics; Springer: Berlin/Heilderbelg, Germany, 1990; ISBN 3540195998. [Google Scholar]
- Chaitin, G.J. Information, Randomness and Incompleteness: Papers on Algorithmic Information Theory; World Scientific: Singapore, 1987; ISBN 9971504804. [Google Scholar]
- Chaitin, G.J. Information-Theoretic Incompleteness; World Scientific: Singapore, 1992; ISBN 9810236956. [Google Scholar]
- Morowitz, H.J. Some order-disorder considerations in living systems. Bull. Math. Biophys. 1955, 17, 81–86. [Google Scholar] [CrossRef]
- Papentin, F. On order and complexity. I. General considerations. J. Theor. Biol. 1980, 87, 421–456. [Google Scholar] [CrossRef]
- Papentin, F. On order and complexity. II. Application to chemical and biochemical structures. J. Theor. Biol. 1982, 95, 225–245. [Google Scholar] [CrossRef]
- Hinegardner, R.; Engelberg, J. Biological complexity. J. Theor. Biol. 1983, 104, 7–20. [Google Scholar] [CrossRef]
- Russel, L.D.; Adebiyi, G.A. Classical Thermodynamics; Saunders College Publishing: Philadelphia, PA, USA, 1993. [Google Scholar]
- Bejan, A. Advanced Engineering Thermodynamics; Wiley Interscience: Hoboken, NJ, USA, 1997. [Google Scholar]
- Cerbe, G.; Hoffmann, H.-J. Einführung in Die Thermodynamik; Carl Hanser GmbH and Co.: München, Germany, 1996. [Google Scholar]
- Denbigh, K. The Thermodynamics of the Steady State; Methuen: London, UK, 1951. [Google Scholar]
- Denbigh, K. The Principles of Chemical Equilibrium; Cambridge University Press: Cambridge, UK, 1971. [Google Scholar]
- Hatsopoulos, G.N.; Keenan, J.H. Principles of General Thermodynamics; Wiley and Sons: Hoboken, NJ, USA, 1965; ISBN 9780471359999. [Google Scholar]
- Kestin, J. A Course in Thermodynamics; Blaisdell: Waltham, MA, USA, 1966. [Google Scholar]
- Morowitz, H.J. Energy Flow in Biology: Biological Organization as a Problem in Thermal Physics; Ox Bow Press: Woodbridge, CT, USA, 1979; ISBN 0918024137. [Google Scholar]
- Perrot, P. A to Z of Thermodynamics; Oxford University Press: Oxford, UK, 1998. [Google Scholar]
- Anderson, G.M. Thermodynamics of Natural Systems; John Wiley and Sons Inc.: Hoboken, NJ, USA, 1996. [Google Scholar]
- Morowitz, H.J. Entropy for Biologists—An Introduction to Thermodynamics, 3rd Print; Academic Press: Cambridge, MA, USA, 1972. [Google Scholar]
- Finn, C.B.P. Thermal Physics (Physics and Its Applications); CRC Press: Boca Raton, FL, USA, 1993; ISBN 0748743790. [Google Scholar]
- Evans, R.B. A Proof that Essergy is the Only Consistent Measure of Potential Work (for Chemical Systems); Hayer School of Engineering Dartmouth College: Hanover, NH, USA, 1969. [Google Scholar]
- Wall, G. Exergy—A Usefull Concept; Chalmers University of Technology: Göteborg, Sweden, 1986. [Google Scholar]
- Kay, J.J. Self-Organisation in Living Systems; University of Waterloo: Waterloo, ON, Canada, 1984. [Google Scholar]
- Allen, P.M. Ecosystem theory for Biological Oceanography. In Canadian Bulletin of Fisheries and Aquatic Sciences, 123; Ulanowicz, R.E., Platt, T., Eds.; NRC Research Press: Ottawa, ON, Canada, 1985; pp. 3–26. [Google Scholar]
- Ebeling, W.; Engel, A.; Feistel, R. Physik der Evolutionsprozesse: Mit 18 Tabellen; Walter de Gruyter: Berlin, Germany, 1990; ISBN 3055006224. [Google Scholar]
- Ebeling, W.; Volkenstein, M.V. Entropy and the evolution of biological information. Phys. A Stat. Mech. Appl. 1990, 163, 398–402. [Google Scholar] [CrossRef]
- Garby, L.; Larsen, P.S. Bioenergetics; Cambridge University Press: Cambridge, UK, 2008; ISBN 0521066352. [Google Scholar]
- Müller, F.; Nielsen, S.N. Thermodynamische systemauffassungen in der ökologie. In Systemtheorie in der Ökologie; Mathes, K., Breckling, B., Ekschmitt, K., Eds.; Ecomed: Landsberg, Germany, 1996; p. 128. [Google Scholar]
- Ahern, J.E. The Exergy Method of Energy Systems Analysis; Wiley and Sons: Hoboken, NJ, USA, 1980; ISBN 0471054941. [Google Scholar]
- Brzustowski, T.A.; Golem, P.J. Second law analysis of energy processes part I: Exergy—An introduction. Trans. Can. Soc. Mech. Eng. 1978, 4, 209–218. [Google Scholar] [CrossRef]
- Lindemann, R.L. The Trophic-dynamic aspect of ecology. Ecology 1942, 23, 399–418. [Google Scholar] [CrossRef]
- Kestin, J. The Second Law of Thermodynamics. Benchmark Papers on Energy 5; Dowden, Hutchinson and Ross: Stroudsburg, PA, USA, 1976. [Google Scholar]
- Gibbs, J.W. Elementary Principles in Statistical Mechanics; Scribner: New York, NY, USA, 1902. [Google Scholar]
- Katchalsky, A.; Curran, P.F. Nonequilibrium Thermodynamics in Biophysics. Harvard Books in Biophysics 1; Harvard University Press: Cambridge, MA, USA, 1967. [Google Scholar]
- Popovic, M. Comparative study of entropy and information change in closed and open thermodynamic systems. Thermochim. Acta 2014, 598, 77–81. [Google Scholar] [CrossRef]
- Vernadsky, V.I. Biosphere: Complete Annotated Edition; Copernicus Books: New York, NY, USA, 1998; ISBN 9780387982687. [Google Scholar]
- Svirezhev, Y. Exergy of the biosphere. Ecol. Model. 1997, 96, 309–310. [Google Scholar] [CrossRef]
- Jørgensen, S.E.; Svirezhev, Y.M. Towards a Thermodynamic Theory for Ecological Systems; Elsevier: Amsterdam, The Netherlands, 2004; ISBN 9780080441672. [Google Scholar]
- Svirezhev, Y.M.; Steinborn, W.H.; Pomaz, V.L. Exergy of solar radiation: Global scale. Ecol. Model. 2003, 169, 339–346. [Google Scholar] [CrossRef]
- Nielsen, S.N. Application of Maximum Exergy in Structural Dynamic Models; National Environmental Research Institute/Royal Danish School of Pharmacy: Copenhagen, Denmark, 1992. [Google Scholar]
- Fernandez-Gonzalez, N.; Huber, J.A.; Vallino, J.J. Microbial communities are well adapted to disturbances in energy input. mSystems 2016, 1. [Google Scholar] [CrossRef]
- Chukova, Y.P. Dissipative functions of the processes of interactions of electromagnetic radiation with biological objects. Biophysics (Oxf.) 1989, 34, 975–978. [Google Scholar]
- Morel, R.E.; Flick, G. Onsager’s principle: A unifying bio-theme. J. Theor. Biol. 1989, 136, 171–175. [Google Scholar] [CrossRef]
- Prigogine, I. Introduction to Thermodynamics of Irreversible Processes; Interscience Publishers: Geneva, Switzerland, 1968; ISBN 9780470699287. [Google Scholar]
- Lurié, D.; Wagensberg, J. Non-equilibrium thermodynamics and biological growth and development. J. Theor. Biol. 1979, 78, 241–250. [Google Scholar] [CrossRef]
- Gould, S.J. Punctuated Equilibrium; Belknap Press of Harvard University Press: Cambridge, MA, USA, 2007; ISBN 9780674024441. [Google Scholar]
- Kauffman, S. The Origins of Order: Self-Organization and Selection in Evolution; Oxford University Press: Oxford, UK, 1993. [Google Scholar]
- Müller, F. State-of-the-art in ecosystem theory. Ecol. Model. 1997, 100, 135–161. [Google Scholar] [CrossRef]
- Müller, F. Gradients in ecological systems. Ecol. Model. 1998, 108, 3–21. [Google Scholar] [CrossRef]
- Müller, F.; Fath, B.D. The physical basis of ecological goal functions—An integrative discussion. In Eco Targets, Goal Functions, and Orientors; Müller, F., Leupelt, M., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; p. 619. [Google Scholar]
- Müller, F.; Fath, B.D. The physical basis of ecological goal functions—Fundamentals, problems and questions. In Eco Targets, Goal Functions, and Orientors; Müller, F., Leupelt, M., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; p. 619. [Google Scholar]
- Ulanowicz, R.E. The propensities of evolving systems. In Evolution Order and Complexity; Khalil, E.L., Boulding, K., Eds.; Routledge: London, UK, 1996. [Google Scholar]
- Ulanowicz, R.E. Network orientors: Theoretical and philosophical considerations why ecosystems may exhibit a propensitiy to increase in ascendency. In Eco Targets, Goal Functions, and Orientors; Müller, F., Leupelt, M., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; p. 619. [Google Scholar]
- Nielsen, S.N.; Jørgensen, S.E. Goal functions, orientors and indicators (GoFOrIt’s) in ecology. Application and functional aspects-Strengths and weaknesses. Ecol. Indic. 2013, 28, 31–47. [Google Scholar] [CrossRef]
- Keenan, J.H. Availability and irreversibility in thermodynamics. Brit. J. Appl. Phys. 1951, 2, 183–192. [Google Scholar] [CrossRef]
- Gaggioli, R.A. Thermodynamics: Second Law Analysis. ACS Symposium Series; American Chemical Society: Washington, DC, USA, 1980; Volume 122, ISBN 0-8412-0541-8. [Google Scholar]
- Gaggioli, R.A. Principles of thermodynamics. In Thermodynamics: Second Law Analysis; American Chemical Society: Washington, DC, USA, 1980; pp. 3–13. [Google Scholar]
- Bošnaković, F. Die Praktische Bedeutung der Exergie. BWK 1961, 11, 481. [Google Scholar]
- Bruges, E.A. Second law analysis. Energy 1954, 341–344. [Google Scholar]
- Fadden, W. A way to pinpoint Energy Loss. Consult. Eng. 1962, 18–19, 106–110. [Google Scholar]
- Grassmann, P. Die Exergie das Flussbild der technisch nutzbaren Leistung. Alg. Wärmetechnik 1959, 9, 79–86. [Google Scholar]
- Szargut, J.; Morris, D.R.; Steward, F.R. Exergy Analysis of Thermal, Chemical, and Metallurgical Processes; Hemisphere: Washington, DC, USA, 1988; ISBN 9783540188643. [Google Scholar]
- Rotstein, E. The exergy balance: A Diagnostic tool for energy optimisation. J. Food. Sci. 1983, 48, 945–950. [Google Scholar] [CrossRef]
- Forcitini, D.; Rotstein, E.; Urbicain, M.J. Heat recovery and exergy balance in a tomato paste plant. J. Food. Sci. 1985, 50, 934–939. [Google Scholar] [CrossRef]
- Hellström, D. An exergy analysis for a wastewater treatment plant-an estimation of the consumption of physical resources. Water Environ. Res. 1997, 69, 44–51. [Google Scholar] [CrossRef]
- Wall, G. Exergy conversion in the Japanese society. Energy 1990, 15, 435–444. [Google Scholar] [CrossRef]
- Wall, G. Exergy use in the Swedish society 1994. In Thermodynamic Analysis and Improvement of Energy Systems; Ruixan, C., Al, E., Eds.; Beijing World, Chinese Society of Engineering Thermophysics and American Society of Mechanical Engineers: Beijing, China, 1997. [Google Scholar]
- Nielsen, S.N.; Jørgensen, S.E. Sustainability analysis of a society based on exergy studies—A case study of the island of Samsø (Denmark). J. Clean. Prod. 2015, 96, 12–29. [Google Scholar] [CrossRef]
- Sciubba, E. From engineering economics to extended exergy accounting: A possible path from monetary to resource-based costing. J. Ind. Ecol. 2004, 8, 19–40. [Google Scholar] [CrossRef]
- Sciubba, E. An exergy-based Ecological Indicator as a measure of our resource use footprint. Int. J. Exergy 2012, 10, 239–266. [Google Scholar] [CrossRef]
- Evans, R.B.; Crellin, G.L.; Tribus, M. Thermoeconomic considerations of Seawater Demineralization. In Principles of Desalination; Spiegler, K.S., Ed.; Academic Press: Cambridge, MA, USA, 1966; p. 566. [Google Scholar]
- Kay, J.J. A nonequilibrium thermodynamic framework for discussing ecosystem integrity. Environ. Manag. 1991, 15, 483–495. [Google Scholar] [CrossRef]
- Kay, J.J.; Schneider, E.D. Thermodynamics and measure of ecosystems integrity. In Ecological Indicators Vol 1. Proceedings of the International Symposium on Ecological Indicators Fort Lauderdale Florida; McKenzie, D.H., Hyatt, D.E., McDonalds, V.J., Eds.; Elservier: Amsterdam, The Netherlands, 1992. [Google Scholar]
- Schneider, E.D. Thermodynamics, ecological succession, and natural selection: A common thread. In Entropy Information and Evolution. New Perspectives on Physical and Biological Evolution; Weber, B.H., Depew, D.J., Smith, J.D., Eds.; MIT Press: Bradford, UK, 1988; p. 376. [Google Scholar]
- Schneider, E.D.; Kay, J.J. Nature abhors a gradient. In Proceedings of the 33rd Annual Meeting of the International Society for the Systems Sciences, Edinburgh, UK, 2–7 July 1989; International Society for the Systems Sciences: Edinburgh, UK, 1989. [Google Scholar]
- Schneider, E.D.; Sagan, D. Into the Cool—Energy Flow, Thermodynamics and Life; University of Chicago Press: Chicago, IL, USA, 2005. [Google Scholar]
- Aoki, I. Holological study of lakes from an entropy viewpoint—Lake Mendota. Ecol. Model. 1989, 45, 81–93. [Google Scholar] [CrossRef]
- Jørgensen, S.E. The shifts in species composition and ecological modelling in hydrobiology. Hydrobiologia 1992, 239, 115–129. [Google Scholar] [CrossRef]
- Jørgensen, S.E. Development of models able to account for changes in species composition. Ecol. Model. 1992, 62, 195–208. [Google Scholar] [CrossRef]
- Nielsen, S.N. Strategies for structural-dynamic modelling. Ecol. Model. 1992, 63, 91–101. [Google Scholar] [CrossRef]
- Jørgensen, S.E.; Padisák, J. Does the intermediate disturbance hypothesis comply with thermodynamics? Hydrobiologia 1996, 323, 9–21. [Google Scholar] [CrossRef]
- Nielsen, S.N.; Ulanowicz, R.E. On the consistency between thermodynamical and network approaches to ecosystems. Ecol. Model. 2000, 132, 23–31. [Google Scholar] [CrossRef]
- Reynolds, C.S. The state of freshwater ecology. Freshw. Biol. 1998, 39, 741–753. [Google Scholar] [CrossRef]
- Jørgensen, S.E.; Nielsen, S.N.; Mejer, H. Emergy, environ, exergy and ecological modelling. Ecol. Model. 1995, 77, 99–109. [Google Scholar] [CrossRef]
- Bendoricchio, G.; Jørgensen, S.E. Exergy as goal function of ecosystems dynamic. Ecol. Model. 1997, 102, 5–15. [Google Scholar] [CrossRef]
- Marques, J.C.; Pardal, M.Â.; Nielsen, S.N.; Jørgensen, S.E. Analysis of the properties of exergy and biodiversity along an estuarine gradient of eutrophication. Ecol. Model. 1997, 102, 155–167. [Google Scholar] [CrossRef]
- Marques, J.C.; Nielsen, S.N.; Jørgensen, S.E. Applying thermodynamic orientors: The use of exergy as an indicator in environmental management. In Eco Targets, Goal Functions, and Orientors; Müller, F., Leupelt, M., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; p. 619. [Google Scholar]
- Xu, F.-L. Modelling the Effects of Ecological Engineering on Ecosystem Health of the Lake Chao, a Shallow Eutrophic Chinese Lake; Royal Danish School of Pharmacy: Copenhagen, Denmark, 1997. [Google Scholar]
- Xu, F.-L. Exergy and structural exergy as ecological indicators for the development state of the Lake Chaohu ecosystem. Ecol. Model. 1997, 99, 41–49. [Google Scholar] [CrossRef]
- Wall, G. Exergy—A usefull Concept Within Ressource Accounting; Chalmers University of Technology: Goteborg, Sweden, 1977. [Google Scholar]
- Eriksson, B.; Eriksson, K.E.; Wall, G. Basic Thermodynamics of Energy Conversions and Energy Use; Institute of Theoretical Physics: Goteborg, Sweden, 1976. [Google Scholar]
- Tessera, M.; Hoelzer, G.A. On the thermodynamics of multilevel evolution. BioSystems 2013, 113, 140–143. [Google Scholar] [CrossRef]
- Imai, Y. Power balance equation of membrane transport system: A basis for network thermodynamics. Membrane 1996, 21, 264–269. [Google Scholar] [CrossRef]
- Mikulecky, D.C. Network thermodynamics: A simulation and modeling method based on the extension of thermodynamic thinking into the realm of highly organized systems. Math. Biosci. 1984, 72, 157–179. [Google Scholar] [CrossRef]
- Mikulecky, D.C. Network thermodynamics: A unifying approach to dynamic nonlinear living systems. In Theoretical Studies of Ecosystems. The Network Perspective; Higashi, M., Burns, T.P., Thomas, P., Eds.; Cambridge University Press: Cambridge, UK, 1991; p. 364. [Google Scholar]
- Riggs, J.E. Aging and mortality: Manifestations of increasing informational entropy of the genome? Mech. Ageing Dev. 1993, 66, 249–256. [Google Scholar] [CrossRef]
- Samaras, T.T.; Stroms, L.H. Impact of height and weight on life span. Bull. WHO 1992, 70, 259–267. [Google Scholar]
- Toussaint, O.; Houbion, A.; Remacle, J. Aging as a multi-step process characterized by a lowering of entropy production leading the cell to a sequence of defined stages. II. Testing some predictions on aging human fibroblasts in culture. Mech. Ageing Dev. 1992, 65, 65–83. [Google Scholar] [CrossRef]
- Toussaint, O.; Houbion, A.; Remacle, J. Effects of modulations of the energetic metabolism on the mortality of cultured cells. BBA Bioenerg. 1994, 1186, 209–220. [Google Scholar] [CrossRef]
- van Rooij, J.C.G.M.; Plomp, R. The effect of linguistic entropy on speech perception in noise in young and elderly listeners. J. Acoust. Soc. Am. 1991, 90, 2985–2991. [Google Scholar] [CrossRef] [PubMed]
- Schultz, T.R.; Coddington, M. Development of the concepts of energy conservation and entropy. J. Exp. Child. Psychol. 1981, 13, 131–153. [Google Scholar] [CrossRef]
- Zotin, A.I. Thermodynamic Aspects of Developmental Biology; Karger Publishers: Berlin, Germany, 1972; ISBN 3805514115. [Google Scholar]
- Lamprecht, I.; Zotin, A.I. Thermodynamics and Kinetics of Biological Processes; Walter de Gruyter: Berlin, Germany, 1983; ISBN 3110082004. [Google Scholar]
- Lamprecht, I.; Zotin, A.I. Thermodynamics of Biological Processes; Walter de Gruyter: Berlin, Germany, 1978; ISBN 3110073129. [Google Scholar]
- Lamprecht, I.; Zotin, A.I. Thermodynamics and Regulation of Biological Processes; Walter de Gruyter: Berlin, Germany, 1985; ISBN 0899250076. [Google Scholar]
- Gladyshev, G.P. Dynamic trends in biological evolution: Model and reality. Biol. Bull. 1995, 22, 1–9. [Google Scholar]
- Gladyshev, G.P. Thermodynamic direction of biological evolution: Model and reality. Biol. Bull. 1996, 23, 315–322. [Google Scholar]
- Wolkenstein, M.W. Entropie und Information; Harry Deutsch: Frankfurt, Germany, 1986. [Google Scholar]
- Straškraba, M. Cybernetic formulation of control in ecosystems. Ecol. Model. 1983, 18, 85–97. [Google Scholar] [CrossRef]
- Mauersberger, P.; Straškraba, M. Two approaches to generalized ecosystem modelling: Thermodynamic and cybernetic. Ecol. Model. 1987, 39, 161–169. [Google Scholar] [CrossRef]
- Straškraba, M. Aquatic ecosystems. An operational research approaches. Ecol. Model. 1982, 17, 319–321. [Google Scholar] [CrossRef]
- Mauersberger, P. From a theory of local processes in aquatic ecosystems to a theory at the ecosystem scale. Sci. Total Environ. 1996, 183, 99–106. [Google Scholar] [CrossRef]
- Mauersberger, P. Rates of primary production, respiration and grazing in accordance with the balances of energy and entropy. Ecol. Model. 1982, 17, 1–10. [Google Scholar] [CrossRef]
- Svirezhev, Y. Thermodynamic orientors: How to use thermodynamic concepts in ecology. In Eco Targets, Goal Functions, and Orientors; Müller, F., Leupelt, M., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; p. 619. [Google Scholar]
- Svirezhev, Y.M. Thermodynamics and ecology. Ecol. Model. 2000, 132, 11–22. [Google Scholar] [CrossRef]
- Aoki, I. Entropy balance of white-tailed deer during a winter night. Bull. Math. Biol. 1987, 49, 321–327. [Google Scholar] [CrossRef]
- Fonck, C.; Jaffé, K. On the energetic cost of sociality. Physiol. Behav. 1996, 59, 713–719. [Google Scholar] [CrossRef]
- Jaffe, K.; Hebling-Beraldo, M.J. Oxygen consumption and the evolution of order: Negentropy criteria applied to the evolution of ants. Experientia 1993, 49, 587–592. [Google Scholar] [CrossRef]
- Aoki, I. Entropy budgets of soybean and bur oak leaves at night. Physiol. Plant. 1987, 71, 293–295. [Google Scholar] [CrossRef]
- Aoki, I. Entropy budgets of deciduous plant leaves and a theorem of oscillating entropy production. Bull. Math. Biol. 1987, 49, 449–460. [Google Scholar] [CrossRef]
- Aoki, I. Entropy budget of conifer branches. Bot. Mag. Tokyo 1989, 102, 133–141. [Google Scholar] [CrossRef]
- Dewar, R.C. Maximum entropy production and plant optimization theories. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 1429–1435. [Google Scholar] [CrossRef]
- Franklin, O.; Johansson, J.; Dewar, R.C.; Dieckmann, U.; Mcmurtrie, R.E.; Brännström, A.; Dybzinski, R. Modeling carbon allocation in trees: A search for principles. Tree Physiol. 2012, 32, 648–666. [Google Scholar] [CrossRef]
- Bertram, J.; Dewar, R.C. Statistical patterns in tropical tree cover explained by the different water demand of individual trees and grasses. Ecology 2013, 94, 2138–2144. [Google Scholar] [CrossRef] [PubMed]
- Franklin, O.; Harrison, S.; Dewar, R.; Farrior, C.; Brännström, A.; Dieckmann, U.; Pietsch, S.; Falster, D.; Cramer, W.; Loreau, M. Organizing principles for vegetation dynamics. Nat. Plants 2020, 6, 444–453. [Google Scholar] [CrossRef] [PubMed]
- Luvall, J.C.; Holbo, H.R. Thermal remote sensing methods in landscape ecology. In Quantitative Methods in Landscape Ecology. Ecological Studies 82; Turner, M., Gardner, R.H., Eds.; Springer: Berlin/Heidelberg, Germany, 1991. [Google Scholar]
- Aoki, I. Eco-physiology of a Lizard (Scleropus occidentalis) from an entropy viewpoint. Physiol. Ecol. Jpn. 1988, 25, 27–38. [Google Scholar]
- Aoki, I. Entropy production in human life span: A thermodynamical measure for aging. Age (Omaha) 1994, 17, 29–31. [Google Scholar] [CrossRef]
- Aoki, I. Entropy flow and entropy production in the human body in basal conditions. J. Theor. Biol. 1989, 141, 11–21. [Google Scholar] [CrossRef]
- Aoki, I. Effects of exercise and chills on entropy production in human body. J. Theor. Biol. 1990, 145, 421–428. [Google Scholar] [CrossRef]
- Aoki, I. Entropy principle for human development, growth and aging. J. Theor. Biol. 1991, 150, 215–223. [Google Scholar] [CrossRef]
- Aoki, I. Entropy physiology of swine-A macroscopic viewpoint. J. Theor. Biol. 1992, 157, 363–371. [Google Scholar] [CrossRef]
- Aoki, I. Entropy laws in ecological networks at steady state. Ecol. Model. 1988, 42, 289–303. [Google Scholar] [CrossRef]
- Aoki, I. Exergy analysis of network systems at steady state. Ecol. Model. 1992, 62, 183–193. [Google Scholar] [CrossRef]
- Aoki, I. Inclusive Kullback index—A macroscopic measure in ecological systems. Ecol. Model. 1993, 66, 289–299. [Google Scholar] [CrossRef]
- Jørgensen, S.E.; De Bernardi, R. The application of a model with dynamic structure to simulate the effect of mass fish mortality on zooplankton structure in Lago di Annone. Hydrobiologia 1997, 356, 87–96. [Google Scholar] [CrossRef]
- Marques, J.C.; Pardal, M.A.; Nielsen, S.N.; Jørgensen, S.E. Thermodynamic orientors: Exergy as a holistic ecosystem indicator: A case study. In Eco Targets, Goal Functions, and Orientors; Müller, F., Leupelt, M., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; p. 619. [Google Scholar]
- Nielsen, S.N.; Jørgensen, S.E.; Marques, J.C. Case Studies: Modelling approaches for the practical application of ecological goal functions. In Eco Targets, Goal Functions, and Orientors; Müller, F., Leupelt, M., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; p. 619. [Google Scholar]
- Fonseca, J.C.; Marques, J.C.; Paiva, A.A.; Freitas, A.M.; Madeira, V.M.C.; Jørgensen, S.E. Nuclear DNA in the determination of weighing factors to estimate exergy from organism’s biomass. Ecol. Model. 2000, 126, 179–189. [Google Scholar] [CrossRef]
- Fonseca, J.C.; Pardal, M.A.; Azeiteiro, U.M.; Marques, J.C. Estimation of ecological exergy using weighing parameters determined from DNA contents of organisms—A case study. Hydrobiologia 2002, 475–476, 79–90. [Google Scholar] [CrossRef]
- Ludovisi, A. Energy degradation and ecosystem development: Theoretical framing, indicators definition and application to a test case study. Ecol. Indic. 2012, 20, 204–212. [Google Scholar] [CrossRef]
- Jørgensen, S.E.; Ludovisi, A.; Nielsen, S.N. The free energy and information embodied in the amino acid chains of organisms. Ecol. Model. 2010, 221, 2388–2392. [Google Scholar] [CrossRef]
- Herendeen, R. Energy intensity, residence time, exergy, and ascendency in dynamic ecosystems. Ecol. Model. 1989, 48, 19–44. [Google Scholar] [CrossRef]
- Nielsen, S.N. Application of exergy in structural- dynamic modelling. Verh. Internat. Verein. Limnol. 1990, 24, 641–645. [Google Scholar]
- Nielsen, S.N. Recent developments in structural dynamic models. In Environmental Models: Emissions and Consequences. Risø International Conference 22–25 May 1989: Developments in Environmental Modelling 15; Fenhann, J., Larsen, H., Mackenzie, G., Rasmussen, B., Eds.; Elsevier: Amsterdam, The Netherlands, 1990. [Google Scholar]
- Nielsen, S.N. Modelling structural dynamical changes in a Danish shallow lake. Ecol. Model. 1994, 73, 13–30. [Google Scholar] [CrossRef]
- Nielsen, S.N. Optimization of exergy in a structural dynamic model. Ecol. Model. 1995, 77, 111–122. [Google Scholar] [CrossRef]
- Vihervaara, P.; Franzese, P.P.; Buonocore, E. Information, energy, and eco-exergy as indicators of ecosystem complexity. Ecol. Model. 2019, 395, 23–27. [Google Scholar] [CrossRef]
- Flindt, M.R.; Kamp-Nielsen, L.; Marques, J.C.; Pardal, M.A.; Bocci, M.; Bendoricchio, G.; Salomonsen, J.; Nielsen, S.N.; Jørgensen, S.E. Description of the three shallow estuaries: Mondego River (Portugal), Roskilde Fjord (Denmark) and the Lagoon of Venice (Italy). Ecol. Model. 1997, 102, 17–31. [Google Scholar] [CrossRef]
- Connell, J.H. Diversity in tropical rain forests and coral reefs. Science 1978, 199, 1302–1310. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.-L. Ecosystem health assessment of Lake Chao, a shallow eutrophic Chinese lake. Lakes Reservoirs. Res. Manag. 1996, 2, 101–109. [Google Scholar]
- Coffaro, G. Modelling Primary Producers’ Dynamics in the Lagoon of Venice. Ph.D. Thesis, Royal Danish School of Pharmacy, Copenhagen, Denmark, 1996. [Google Scholar]
- Jørgensen, S.E. Ecological network theory. Ecol. Model. 2007, 208, 1–2. [Google Scholar] [CrossRef]
- Jørgensen, S.E.; Ulanowicz, R. Network calculations and ascendency based on eco-exergy. Ecol. Model. 2009, 220, 1893–1896. [Google Scholar] [CrossRef]
- Jørgensen, S.E.; Nielsen, S.N. Use of eco-exergy in ecological networks. Ecol. Model. 2014, 293, 202–209. [Google Scholar] [CrossRef]
- Christensen, V. Network analysis of trophic interactions in aquatic ecosystems. In Eco Targets, Goal, and Orientors; Springer: Berlin/Heidelberg, Germany, 1992; pp. 243–254. [Google Scholar]
- Salomonsen, J. Examination of properties of exergy, power and ascendency along a eutrophication gradient. Ecol. Model. 1992, 62, 171–181. [Google Scholar] [CrossRef]
- Carathéodory, C. Investigations into the foundations of thermodynamics. In The Second Law of Thermodynamics. Benchmark Papers on Energy 5; Kestin, J., Ed.; Dowden, Hutchinson and Ross: Stroudsburg, PA, USA, 1976; p. 329. [Google Scholar]
- Hatsopoulos, G.N.; Keenan, J.H. A single axiom for classical thermodynamics. J. Appl. Mech. 1962, 29, 193–199. [Google Scholar] [CrossRef]
- Bass, B. Applying thermodynamic orientors: Goal functions in the holling figure-eight model. In Eco Targets, Goal Functions, and Orientors; Müller, F., Leupelt, M., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; p. 619. [Google Scholar]
- Jørgensen, S.E.; Nielsen, S.N. Thermodynamic Orientors: Exergy as a goal function in ecological modelling and as an ecological indicator for the description of ecosystem development. In Eco Targets, Goal Functions, and Orientors; Müller, F., Leupelt, M., Eds.; Springer: Berlin/Heidelberg, Germany, 1998; p. 619. [Google Scholar]
- Saint-Béat, B.; Baird, D.; Asmus, H.; Asmus, R.; Bacher, C.; Pacella, S.R.; Johnson, G.A.; David, V.; Vézina, A.F.; Niquil, N. Trophic networks: How do theories link ecosystem structure and functioning to stability properties? A review. Ecol. Indic. 2015, 52, 458–471. [Google Scholar] [CrossRef]
- Holbo, H.R.; Luvall, J.C. Modeling surface temperature distributions in forest landscapes. Remote Sens. Environ. 1989, 27, 11–24. [Google Scholar] [CrossRef]
- Luvall, J.C.; Holbo, H.R. Measurements of short-term thermal responses of coniferous forest canopies using thermal scanner data. Remote Sens. Environ. 1989, 27, 1–10. [Google Scholar] [CrossRef]
- Swenson, R.; Turvey, M.T. Thermodynamic reasons for perception-action cycles. Ecol. Psychol. 1991, 3, 317–348. [Google Scholar] [CrossRef]
- Levine, R.D.; Tribus, M. The Maximum Entropy Formalism; MIT Press: Cambridge, MA, USA, 1979. [Google Scholar]
- Liebig, J. Chemistry in Its Application to Agriculture and Physiology; Wiley and Putnam: New York, NY, USA, 1847. [Google Scholar]
- Holling, C.S. The resilience of terrestrial ecosystems: Local surprise and global change. In Sustainable Development of the Biosphere.; Clark, W.C., Munn, R.E., Eds.; IIASA, Cambridge University Press: Cambridge, UK, 1986. [Google Scholar]
- Hull, D. Philosophy of Biological Science; Prentice Hall College Div.: Upper Saddle River, NJ, USA, 1974. [Google Scholar]
- Kuhn, T.S. The Structure of Scientific Revolutions; University of Chicago Press: Chicago, IL, USA, 1962; ISBN 0226458083. [Google Scholar]
- Georgescu-Roegen, N. The Entropy Law and the Economic Process; Harvard University Press: Cambridge, MA, USA, 1971; ISBN 0674281640. [Google Scholar]
- Georgescu-Roegen, N. Energy and Economic Myths; Pergamon Press: Oxford, UK, 1976. [Google Scholar]
- Bastianoni, S. A definition of “pollution” based on thermodynamic goal functions. Ecol. Model. 1998, 113, 163–166. [Google Scholar] [CrossRef]
- Nielsen, S.N. What has modern ecosystem theory to offer to cleaner production, industrial ecology and society? The views of an ecologist. J. Clean. Prod. 2007, 15, 1639–1653. [Google Scholar] [CrossRef]
- Nielsen, S.N.; Müller, F. Understanding the functional principles of nature—Proposing another type of ecosystem service. Ecol. Model. 2009, 220, 1913–1925. [Google Scholar] [CrossRef]
- Rifkin, J. Entropy: Into the Greenhouse World; Bantam Books: New York, NY, USA, 1989; ISBN 9780553347173. [Google Scholar]
- Tiezzi, E. Tempi Storici, Tempi Biologici; Garzanti: Milano, Italy, 1984. [Google Scholar]
- Tiezzi, E. The End of Time; WIT Press: Southampton, UK, 2002. [Google Scholar]
- Kleidon, A. Non-equilibrium thermodynamics, maximum entropy production and Earth-system evolution. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 2010, 368, 181–196. [Google Scholar] [CrossRef] [PubMed]
- Volk, T.; Pauluis, O. It is not the entropy you produce, rather, how you produce it. Philos. Trans. R Soc. Lond. B Biol. Sci. 2010, 365, 1317–1322. [Google Scholar] [CrossRef] [PubMed]
- Kleidon, A.; Malhi, Y.; Cox, P.M. Maximum entropy production in environmental and ecological systems. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 1297–1302. [Google Scholar] [CrossRef]
- Kleidon, A. How does the Earth system generate and maintain thermodynamic disequilibrium and what does it imply for the future of the planet? Trans. R. Soc. A 2012, 370, 1012–1040. [Google Scholar] [CrossRef]
- Dyke, J.G.; Gans, F.; Kleidon, A. Towards understanding how surface life can affect interior geological processes: A non-equilibrium thermodynamics approach. Earth Syst. Dyn. 2011, 2, 139–160. [Google Scholar] [CrossRef]
- Kleidon, A.; Fraedrich, K.; Kunz, T.; Lunkeit, F. The atmospheric circulation and states of maximum entropy production. Geophys. Res. Lett. 2003, 30, 2223. [Google Scholar] [CrossRef]
- Dhara, C.; Renner, M.; Kleidon, A. Broad climatological variation of surface energy balance partitioning across land and ocean predicted from the maximum power limit. Geophys. Res. Lett. 2016, 43, 7686–7693. [Google Scholar] [CrossRef]
- Kleidon, A.; Renner, M.; Porada, P. Estimates of the climatological land surface energy and water balance derived from maximum convective power. Hydrol. Earth Syst. Sci. 2014, 18, 2201–2218. [Google Scholar] [CrossRef]
- Kleidon, A.; Renner, M. Thermodynamic limits of hydrologic cycling within the Earth system: Concepts, estimates and implications. Hydrol. Earth Syst. Sci. 2013, 17, 2873–2892. [Google Scholar] [CrossRef]
- Kleidon, A.; Renner, M. Diurnal land surface energy balance partitioning estimated from the thermodynamic limit of a cold heat engine. Earth Syst. Dynam. 2018, 9, 1127–1140. [Google Scholar] [CrossRef]
- Simoncini, E.; Virgo, N.; Kleidon, A. Geoscientific instrumentation methods and data systems quantifying drivers of chemical disequilibrium: Theory and application to methane in the Earth’s atmosphere. Earth Syst. Dynam. 2013, 4, 317–331. [Google Scholar] [CrossRef]
- Dickman, R.; Motai, R. Inconsistencies in steady-state thermodynamics. Phys. Rev. E 2014, 89, 32134. [Google Scholar] [CrossRef]
- Vallino, J.J.; Algar, C.K. The thermodynamics of marine biogeochemical cycles: Lotka revisited. Annu. Rev. Mar. Sci. 2016, 8, 333–356. [Google Scholar] [CrossRef]
- Vallino, J.J. Ecosystem biogeochemistry considered as a distributed metabolic network ordered by maximum entropy production. Philos. Trans. R. Soc. B Biol. Sci. 2010, 365, 1417–1427. [Google Scholar] [CrossRef]
- Vallino, J.J. Earth system dynamics differences and implications in biogeochemistry from maximizing entropy production locally versus globally. Earth Syst. Dynam 2011, 2, 69–85. [Google Scholar] [CrossRef]
- Vallino, J.J.; Huber, J.A. Using maximum entropy production to describe microbial biogeochemistry over time and space in a meromictic pond. Front. Environ. Sci. 2018, 6, 100. [Google Scholar] [CrossRef]
- Algar, C.K.; Vallino, J.J. Predicting microbial nitrate reduction pathways in coastal sediments. Aquat. Microb. Ecol. 2014, 71, 223–238. [Google Scholar] [CrossRef]
- Aller, R.C. Benthic fauna and biogeochemical processes in marine sediments: The role of burrow structures. In Nitrogen Cycling in Coastal Marine Environments; Blackburn, T.H., Sørensen, J., Eds.; John Wiley and Sons: Chichester, UK, 1988; p. 338. [Google Scholar]
- Dewar, R.C.; Porté, A. Statistical mechanics unifies different ecological patterns. J. Theor. Biol. 2008, 251, 389–403. [Google Scholar] [CrossRef]
- Bertram, J.; Dewar, R.C. Combining mechanism and drift in community ecology: A novel statistical mechanics approach. Theor. Ecol. 2015, 8, 419–435. [Google Scholar] [CrossRef]





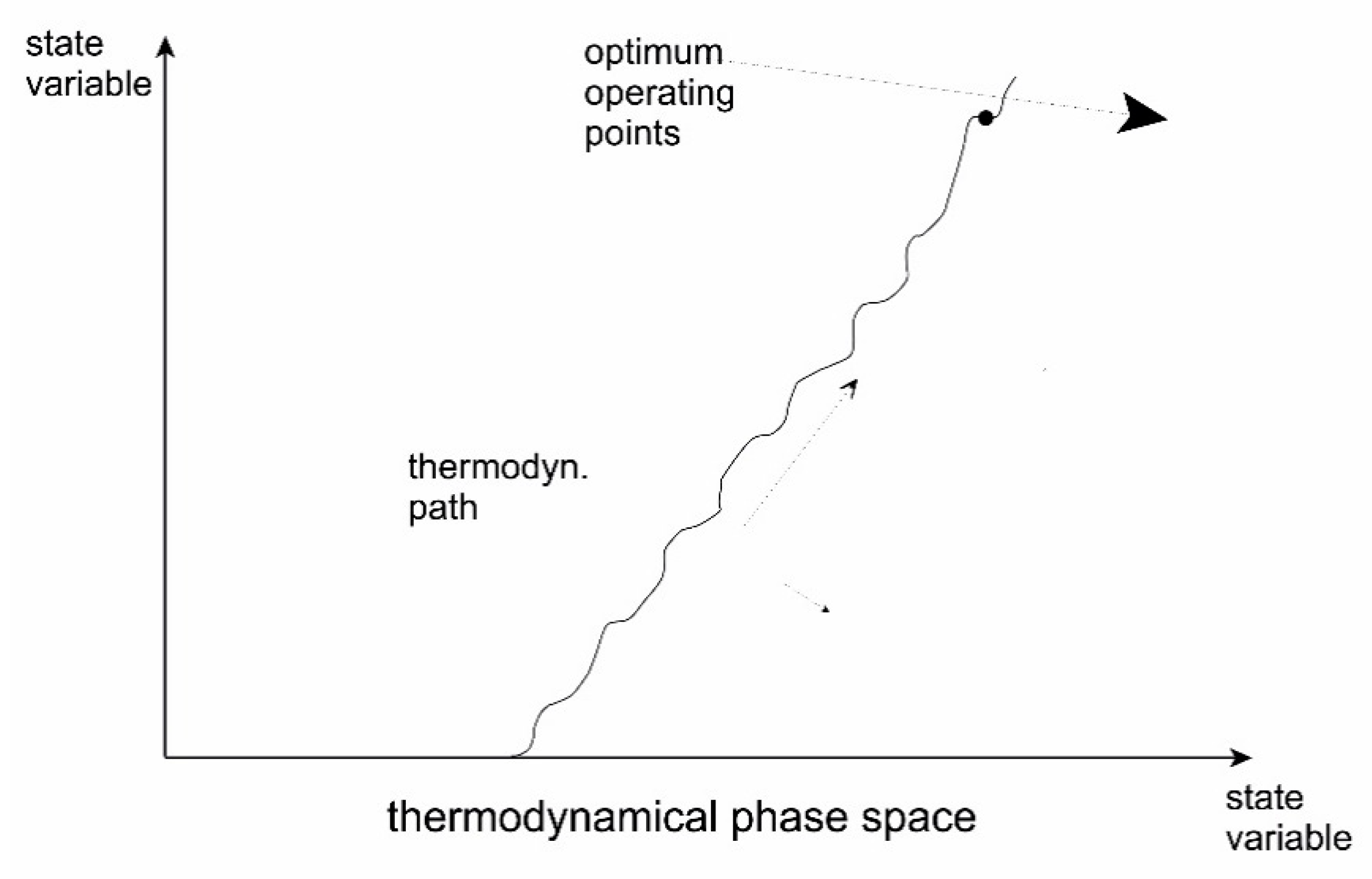
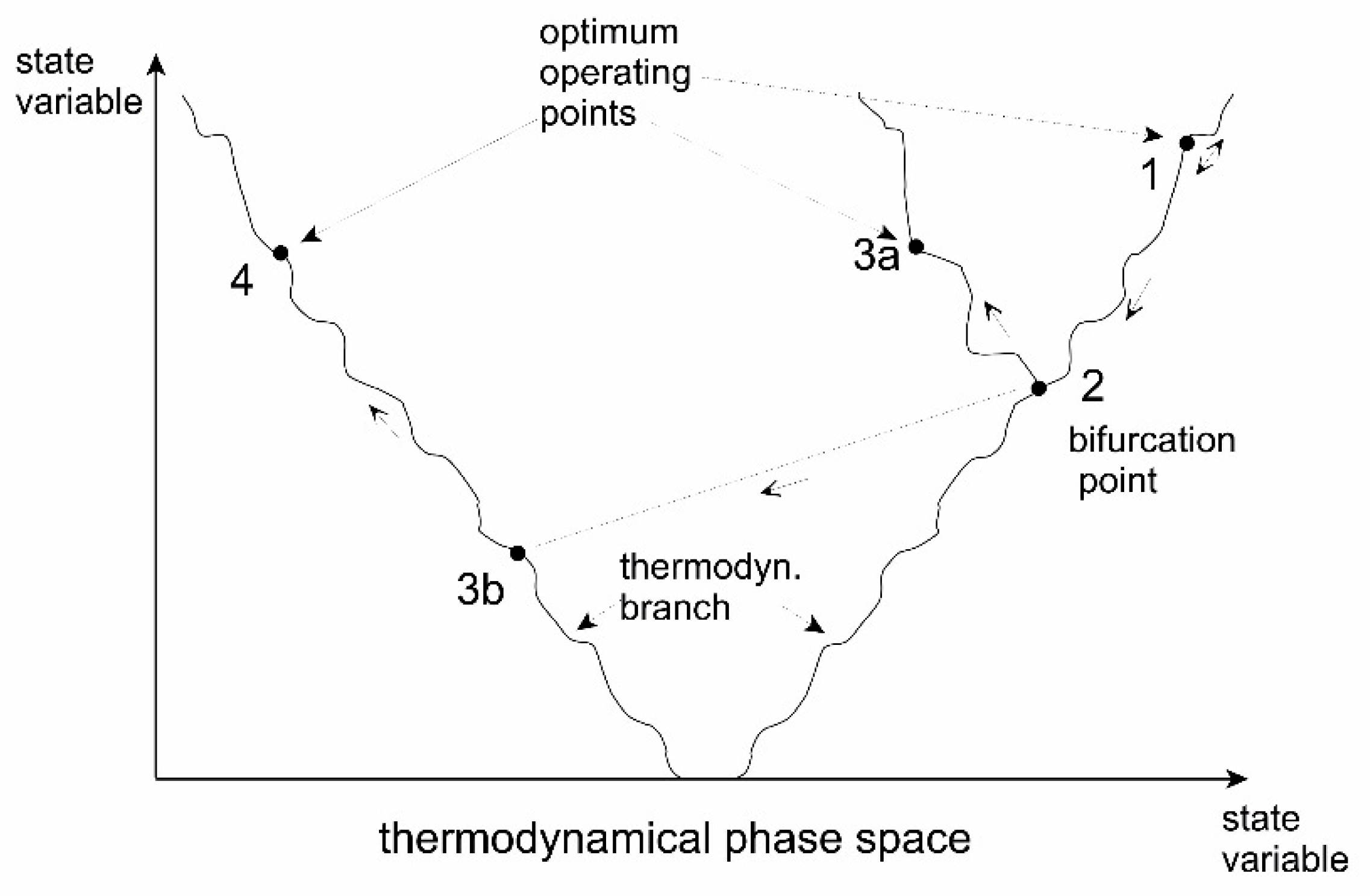
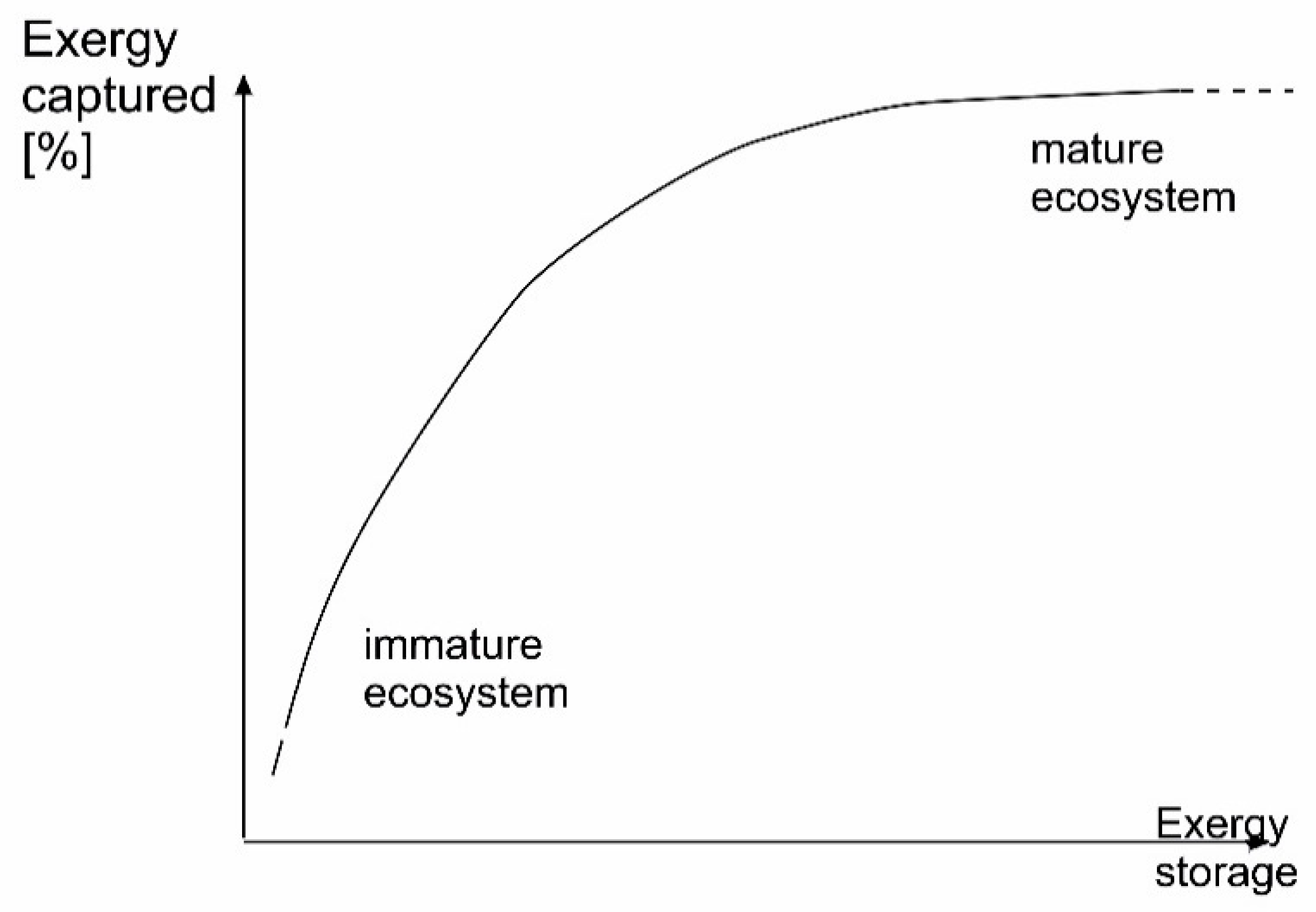
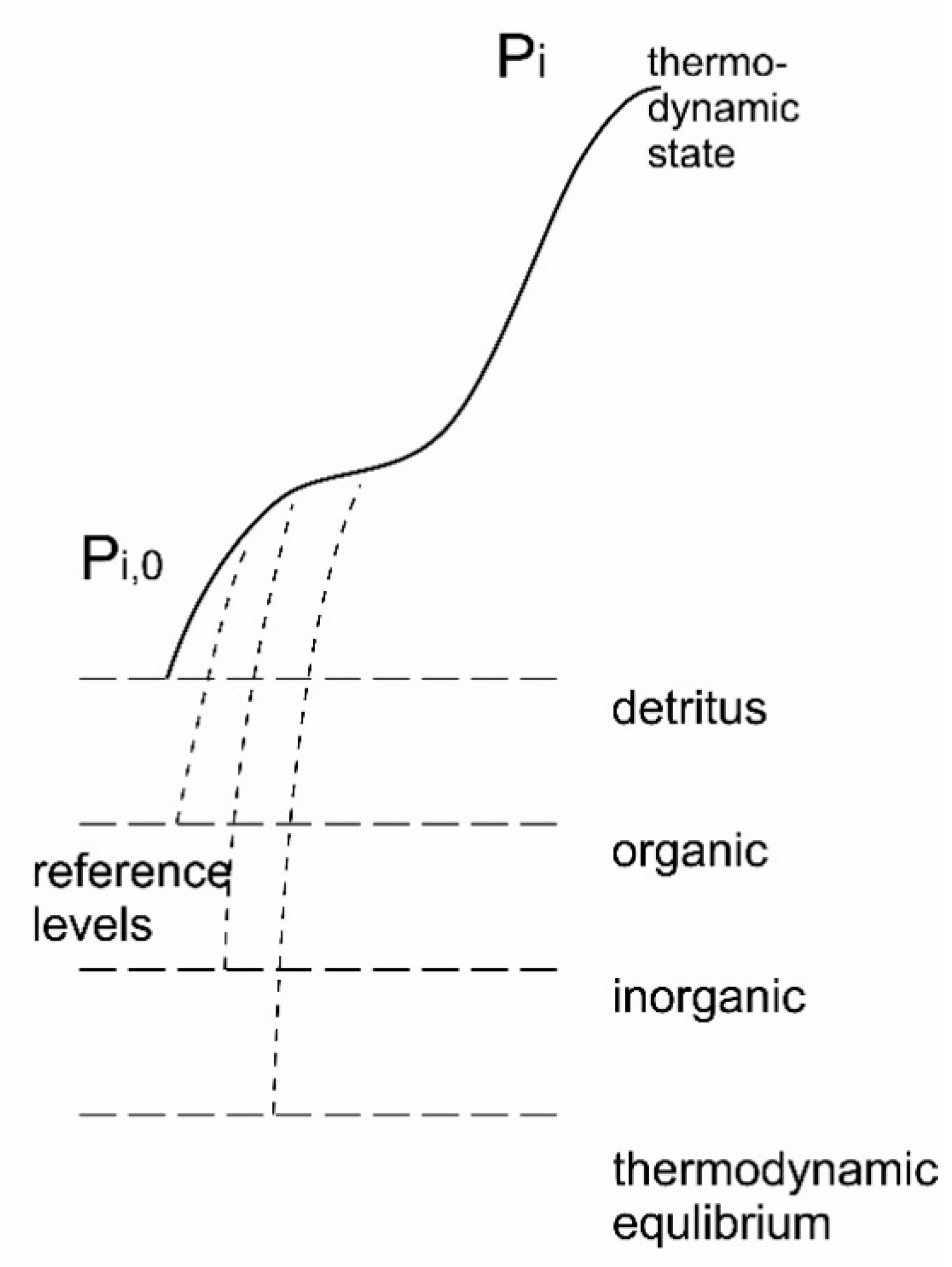
| Variant | Origin (Major References) | Remarks |
|---|---|---|
| phenomenology of 24 principles during undisturbed development of naturals systems towards climax society | Odum, E.P. [1,2] | Principle 23 and 24 are referring to decrease in entropy and increase in information of the ecosystem, respectively |
| emergent properties | Odum, E.P. [21] | The study of emergent properties of ecosystems is proposed as research strategy |
| maximum (useful) power | Odum, H.T. [3,22,23,24] | The idea originating in Lotka’s papers from the early 1920′ies |
| eMergy | Odum, H.T. [25,26] | |
| minimum dissipation/entropy | Mauersberger, P. [27,28,29,30] | minimum dissipation as extremal principle for aquatic ecosystems |
| entropy | Aoki, I. [31,32,33,34,35,36] | |
| maximum exergy (storage) | Jørgensen, S.E. [7,37,38] | the exergy function derived was shown to relate to buffer capacity and proposed as a holistic indicator and goal function— exergy optimization of ecosystems recently proposed as an ecological law of thermodynamics |
| maximum exergy degradation | Schneider, E & Kay, J.J. [14,15,39,40,41] | maximum exergy degradation proposed as driving mechanism, exergy degradation as indicator of ecosystem integrity |
| maximum entropy production | Martyushev [42,43] | validity of maximum entropy production from physics to biology |
| Ascendency | Ulanowicz, R.E. [5,6,44,45] | ecosystems as they grow and develop show an increase in ascendency, flows serve as orientor and “stress” indicator |
| Utility and indirect effect | Patten, B.C. [4,46,47] | Ecosystems flows serve to increase quantitative and qualitative utility of the system Indirect flows are dominating over direct effects by several orders of magnitude |
| Biomass (maximum) | Straskraba, M. [48] Margalef, R [49] | Biomass as goal function Endosomatic and exosomatic causes |
| Year(s) | Event | Ref/Source |
|---|---|---|
| 1789–1791 | Lavoiser and Sequin discovers food combustion leading to formation of CO2 and H2O with a parallel release of heat | after Morowitz [118] |
| 1824 | One of the earliest works of Sadi Carnot Betrachtungen über die Bewegende Kraft des Feuers, appears | Carnot 1824 [119] |
| 1865 | Clausius’ formulation of the first and second law | Clausius 1865 [120] |
| 1872 | Boltzmann search for the so-called H-theorem leading to Boltzmann’s formula | Boltzmann 1872 [121] |
| 1878 | Gibbs’ extension of the Boltzmann equation | Gibbs 1878 [122] |
| 1944 | Schrödinger states that living organisms are feeding on negentropy and formulates his order form order and order from disorder principles | Schrödinger 1944 [123] |
| 1946 | Establishment of far from equilibrium thermodynamics by Prigogine and co-workers (1) understanding of systems as dissipative structures (2) formulation of the minimum dissipation principle (3) evolution through instabilities and bifurcations | Prigogine, 1947 [70] Prigogine and Wiame, 1946 [29] Prigogine and Nicolis, 1971 [124] Prigogine and Stengers [77] Glansdorff and Prigogine, 1971 [125] Nicolis and Prigogine, 1977 [74] |
| 1867 | Maxwell’s demon violating the second law | Leff and Rex, 1990 [126] |
| 1967 | Brillouin, closer connection to information theory | Brillouin 1960 [127] |
| Year(s) | Event | Main Ref/Source |
|---|---|---|
| 1922, 1925 | Lotka proposes that living organisms compete for energy | after Morowitz [118] |
| 1944 | Schrödinger’s states that living organisms are feeding on negentropy and formulates his order form order and order from disorder principles | Schrödinger [123] |
| 1976 | Exergy proposed as important factor | Jørgensen and Mejer, 1981 [38] Mejer and Jørgensen, 1979 [37] |
| 1979 | Exergy relates to buffer capacity | Jørgensen and Mejer, 1981 [38] |
| 1984 | Exergy degradation | Kay, 1984, 1991 [173,222] Kay and Schneider, 1992, [223] Schneider, 1988, [224] Schneider and Kay, 1994a,b,c [15,39,225,226] |
| 1987, 1989 | Entropy analysis of lake ecosystems | Aoki [35,227] |
| 1990–1992 | changes in ecosystems are generally accompanied by increases in exergy (storage) | Nielsen, 1992 [190] Jørgensen, 1992 [228] |
| 1992 | exergy storage used as goal function | Jørgensen, 1992, 1997 [229] Nielsen, 1992 [230] |
| exergy relates to: intermediate disturbance hypothesis chaos ascendency the exergy “cushion” | Jørgensen and Padisak, [231] Jørgensen, [228] Nielsen and Ulanowicz [232] Reynolds [233] | |
| 1995 | New exergy index and specific exergy proposed based on (1) informational content of genome and (2) reference at detritus level | Jørgensen et al. [234] Bendoricchio and Jørgensen [235] |
| 1997 | Specific exergy covers other perspectives than the other exergy | Marques et al. [236,237] Xu [238,239] |
| 1997 | emergy/exergy ratios | Bastianoni and Marchettini, [58] |
| (a) Architecture | (b) Biological | (c) Ecological |
|---|---|---|
| castle, cathedral | biosphere | top carnivore |
| manor, mansion | ecosystem | carnivore |
| house | societies | herbivore |
| stable of bricks | populations | primary producers |
| pile of bricks | organisms | bacteria |
| bricks | organs | nutrients |
| clay | cells | |
| molecules | cell organelles | |
| proteins, enzymes | ||
| amino acids | ||
| organic molecules | ||
| inorganic molecules | ||
| atoms |
| Organism | Number of Information Genes | Weighting Factor |
|---|---|---|
| Detritus | 0 | 1 |
| Minimal Cell | 470 | 2.3 |
| Bacteria | 600 | 2.7 |
| Algae | 850 | 3.3 |
| Yeast | 2000 | 6 |
| Fungi | 3000 | 10 |
| Sponges | 9000 | 26 |
| Plants, trees | 10,000–30,000 | 30–90 |
| Worms | 10,000–100,000 | 30–300 |
| Insects | 10,000–15,000 | 30–45 |
| Zooplankton | 10,000–50,000 | 30–150 |
| Crustaceans | 100,000 | 300 |
| Fish | 100,000–120,000 | 300–350 |
| Birds | 120,000 | 350 |
| Amphibians | 120,000 | 350 |
| Reptiles | 120,000 | 350 |
| Mammals | 140,000 | 400 |
| Humans | 250,000 | 700 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nielsen, S.N.; Müller, F.; Marques, J.C.; Bastianoni, S.; Jørgensen, S.E. Thermodynamics in Ecology—An Introductory Review. Entropy 2020, 22, 820. https://doi.org/10.3390/e22080820
Nielsen SN, Müller F, Marques JC, Bastianoni S, Jørgensen SE. Thermodynamics in Ecology—An Introductory Review. Entropy. 2020; 22(8):820. https://doi.org/10.3390/e22080820
Chicago/Turabian StyleNielsen, Søren Nors, Felix Müller, Joao Carlos Marques, Simone Bastianoni, and Sven Erik Jørgensen. 2020. "Thermodynamics in Ecology—An Introductory Review" Entropy 22, no. 8: 820. https://doi.org/10.3390/e22080820
APA StyleNielsen, S. N., Müller, F., Marques, J. C., Bastianoni, S., & Jørgensen, S. E. (2020). Thermodynamics in Ecology—An Introductory Review. Entropy, 22(8), 820. https://doi.org/10.3390/e22080820





