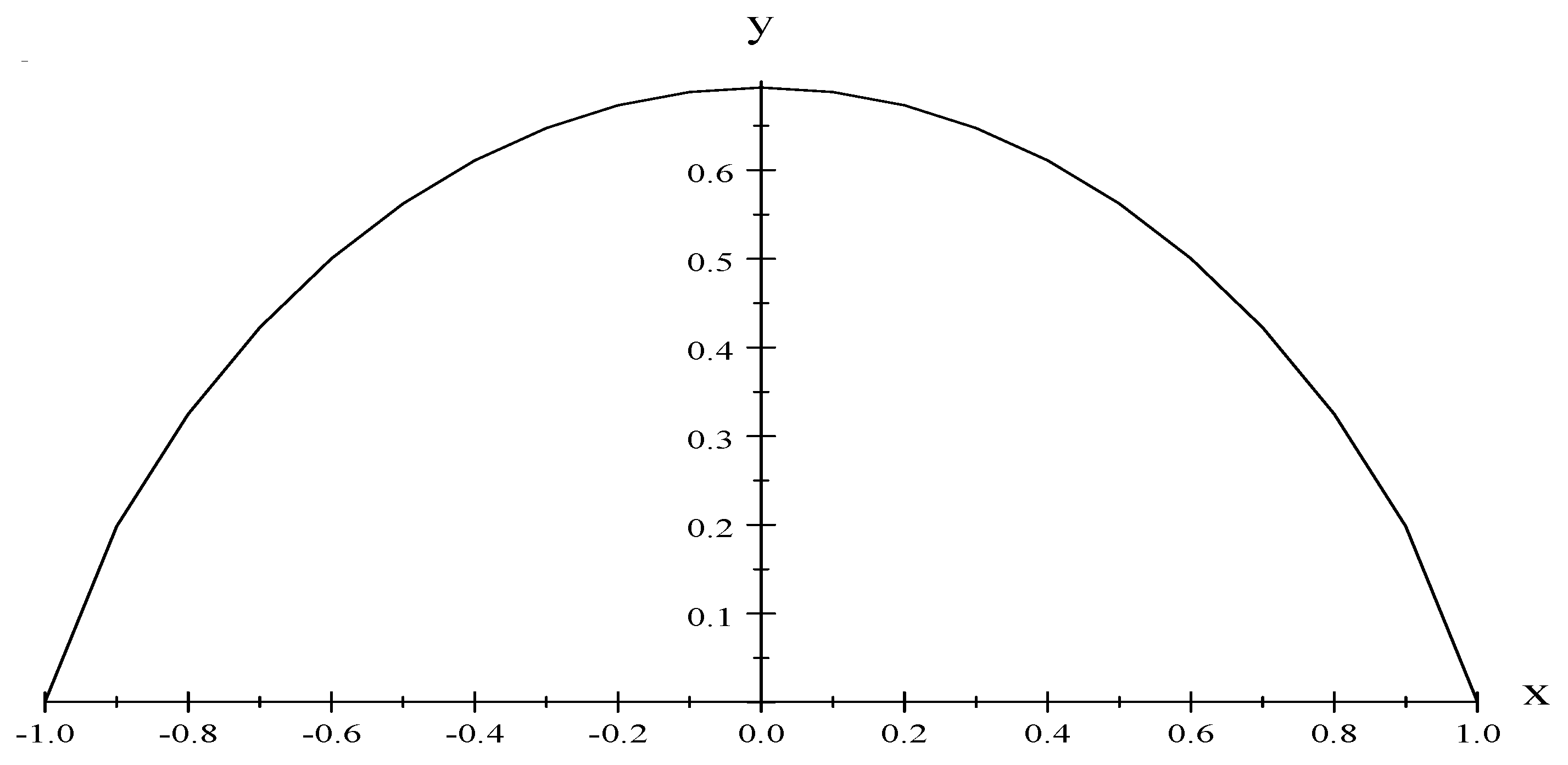In order to apply the methods described in
Section 2 and
Section 3, some examples are analyzed. First, we start with the ideal gas with
N variable. All the classical results are recovered for this case. The completeness method permits obtaining certain novel identities. Then, an unconventional equation of state is studied and procuring the other equation of state in order to get a thermodynamic system. The rubber band with variable length is described giving a correct thermodynamic set of equations. Starting with the correct expression of the entropy, the paramagnetic solid system is exposed in order to find the correct expressions of the
equations. Finally, a equation proposed by Kelly [
7] is analyzed, adding a equation in order to find a complete thermodynamic system.
4.2. Unconventional System: A Particular Case
As we mentioned in the Introduction, it has been shown that for thermodynamic systems with unconventional state equations, the Carnot theorem is valid [
6]. Apparently, the Carnot theorem is a universal property for a large number of systems despite not behaving physically.
Let us consider the following equation of state,
with
. We have to note that the isothermal compressibility
,
is negative and for most substances
. In statistical mechanics, it is required that
in order to have low fluctuations which give sense to it. Moreover, Van Hove’s theorem [
1] proves that by means of regular intermolecular forces,
must be positive within the framework of statistical mechanics. However, as we shall see, we can have a thermodynamic system without matching the classical proposal of statistical mechanics. A discussion about this point is conducted at the end of the Hessian method.
On the other hand, as we do not have another equation of state, we use our method for proposing it. First, we know that
Then, we can propose that
U is such that
which shows that the internal energy is always negative. Note that in order to differentiate our system with respect to regular systems, we do not incorporate the ideal gas limit term
in order to have a completely different unconventional system.
4.2.1. Calculation of Entropy with the N Variable
From Equation (
32), by integrating the Gibbs–Duhem relation, we can calculate the chemical potential obtaining
where
and
. On the other hand, by applying the Euler relation, we arrive at
Therefore, the entropy is
where
We obtain an entropy that complies with the first-degree homogeneous property. However, it has to be noted that entropy can be negative. This implies that we obtain a restriction in the value of the chemical potential
of this system. Indeed, It is important to note that if we rewrite
S, as
, using Equation (
35) in the following form, we have
Therefore, in order to have a positive entropy , we need to have a negative chemical potential, .
4.2.2. Hessian Method
We can obtain the internal energy as a fundamental equation using Equation (
36)
Since from Equation (
36),
we can assure that the internal energy is a real negative quantity. First, note that the temperature
We can calculate the Hessian by noting that Shwarz’s theorem is fulfilled. Then, from the the Hessian, we obtain the characteristic polynomial,
We can see that one of the eigenvalues is
, and the other two are,
with
and
We know that
, and it must be fulfilled that
, so for
, we can see
so
. Rewriting
, we have
so
.
However, this system will be an unstable system according to the Le Chatelier–Braun principle [
4] because the isothermal compressibility is negative (see Equation (
33)). However, the no-null eigenvalues are real, which means that the relaxation times
[
9] are real. This means that this system is an unstable thermodynamic system that may after a perturbation come back to a equilibrium state [
10], but the effects of the instability will bring it to a position of increasing volume.
4.2.3. Entropy Method
By looking at Equations (
32), (
34), (
36) and (
39), we have already proven that the items
A,
F,
G,
H, and
I are fulfilled. Therefore, the set of equations represents a thermodynamic system.
4.2.4. Completeness Method
In general, we can express the
equations of this system using Equation (
15). In this case, we need just three independent
equations. We choose the 1-, 2-, and 3-equations.
From Equations (
15) and (
16), we need to know
and
, that is:
with
. We arrive to the 1-equation,
with
.
From Equations (
15) and (
16), we need to know
y
, that is:
Finally, the 2-equation is
From Equations (
15) and (
16), we need to know
,
, and
, that is:
We need to analyze
; we know that
and
, we see that
, and
, so
is real. Therefore, the 3-equation is
with
.
Thermodynamic Identities
With the three independent
equations, it is possible to calculate the remaining
equations and in this way obtain different thermodynamic identities. In this case, it is possible from the 1-equation and 2-equation to deduce the 7-equation, which can be calculated previously with the formulas of the method developed in
Section 3. We have
Then, if we substitute the already calculated coefficients in
(remember that
), we have
Note that it can be proven that this system complies with the Carnot theorem.
4.3. The Rubber Band
The rubber band is normally described using two equations of motion which consider the following constraints:
- (i)
An analogue of the mole number might be associated with the number of monomer units in the rubber band, but in this first approach, this mole number is not considered as a variable and is taken as a constant. It has to be highlighted that for the ideal gas, we can consider the number of particles as a constant, but the entropy will not be a homogeneous function of degree one. The same happens with the entropy of the rubber band when the number of monomer is not taken as a variable.
- (ii)
In the rubber band, the length L and the tension play a role analogue to the volume V and minus the pressure in the ideal gas, respectively.
- (iii)
represents the unstretched length of the rubber band, is the elastic limit length, and c and b are characteristic constants.
The equations of state for this model are:
and
By considering the different representations from the variables
T,
, and
L, we can obtain the
equations, to check the validity of the entropic relations and deduce the entropy. Everything functions like a thermodynamic system unless the entropy does not satisfy the first-degree homogeneous property; that is, the obtained entropy is
which is obviously not a first-degree homogeneous function as happens with the ideal gas when the number of particles
N is taken as a constant. It has to be noted that Callen [
4] says that these equations have been constructed on the basis only of the most qualitative of information. They do not include any numbers of monomers. Our purpose in this section is to give an expression of the entropy which meets the first-degree homogeneous requirement. However, as in the ideal gas, we are obligated to consider including a quantity related with the number of monomer. Let us consider that this number of monomer can be described by
N in a linear way,
being the length of each monomer when the rubber band measures
and
the length of each monomer when the rubber band measures
L and is composed of
N monomers.
Therefore, let us propose the following equations of state with
N variable:
with
Then, we need to propose another equation of state. We propose it using the property that
U is a function of state and therefore,
and
Consequently, we arrive to our final proposal for the internal energy as
where
, and it coincides with the internal energy when the number of monomers are constant equal to
.
4.3.1. Calculation of Entropy with N Variable
The state equations for this case are Equations (
50) and (
53)
From these expressions, using the Gibbs–Duhem method, we obtain the entropy,
where,
The expression for the entropy shows that is a first-degree homogeneous function. Moreover, must be such that . Therefore, this will limit the validity of the system by giving a range of temperature such that .
We can explicitly write the energy in the following way
4.3.2. Hessian Method
We rewrite our energy as
with
,
,
y
.
First, it is necessary to calculate the temperature,
This shows the requirement that .
Further, it is clear that the internal energy accomplishes Schwarz’s theorem.
Then, the characteristic polynomial of the Hessian is
We can see that one of the eigenvalues is
and the other two are
with
and
Consequently, the requirement for the Hessian is satisfied.
4.3.3. Entropy Method
By looking at Equations (
50), (
53), (
54) and (
57), we have already proven that the items
A,
G,
C,
Hm and
I are fulfilled. Therefore, the set of equations represents a thermodynamic system.
4.3.4. Completeness Method
In general, we can express all the
equations of this system using Equation (
15). Before continuing, let us give the chemical potential as:
with
As in the case of the ideal gas from the ten equations, we just need to choose three linearly independent equations. Let us choose the 1-equation, the 2-equation, and the 3-equation.
From Equations (
15) and (
16), we need
and
. We have
and
From Equations (
15) and (
16), we need to know
and
. We have
and
Hence,
with
.
From Equations (
15) and (
16), we need to know
,
and
. We have
and
We know that with the three above
equations, we can generate all the other
equations. We deal with the 7-equation in order to
determine some interesting identities. The 7-equation is:
with
From Equations (
15) and (
16), we need to know
,
and
. We have
Therefore,
with
Thermodynamic Identities
With the three linearly independent
equations, it is possible to calculate the remaining
equations and in this way obtain different thermodynamic identities. In this case, it is possible from the 1-equation, Equation (
58), and the 2-equation, Equation (
60), to deduce the 7-equation, Equations (
63) and (
64), using the method previously described in
Section 3; we obtain
Finally, a Mayer-like equation for the rubber band is obtained:
4.4. The Paramagnetic Solid
There exists an interesting discussion about the
equations for a paramagnetic solid [
11]. Indeed, according to Kittel [
12] “a great deal of unnecessary confusion exists as to how to write the First Law of Thermodynamics for a magnetic system”. Callen [
4] gave a proposal for a simple paramagnetic system described by the internal energy as:
where
M represents the magnetization, and
and
are positive constants. However, he said that this model does not describe any particular known system. Moreover, in
Appendix C, he treated the paramagnetic solid by including the solenoid which produces the magnetic field, redefining the energy of the system by putting
However, he needed to give a redefinition of the energy by subtracting the energy stored in the volume by the magnetic field. The most appropriate treatment is given by Barrett and Macdonald [
13], who studied the system in a more direct way and through statistical physics obtain an expression for the work done by the magnetic field in a paramagnetic solid. Barrett and Macdonald mentioned there are two forms for the work done when the magnetic field
B and the magnetization
M change, that is:
where the form
applies when the mutual field energy is included in the system and the form
when it is not. Although he claimed that there are thermodynamic systems with no fundamental thermodynamic equation, he arrived to the conclusion that the work done by the magnetic field for a system where the mutual field is not included as
Of course, for a fundamental equation in the
S representation, the work must be written as the product of an intensive quantity times the differential of an extensive quantity, which is not expressed in Equation (
69) (note that there is a misprint in the Barrett and Macdonald [
13] article of the sign in the expression for the temperature). The good expression is:
From Equation (
70), he arrived to the correct expression for the entropy:
or in a different form
being
and consequently
which implies an infinite temperature. These contradicts Equation (
70), where the temperature is well-defined. Therefore, we need to obtain another representation to express the thermodynamic system. The problem is based on that the
equation is written as
which implies
where
does not represent minus the chemical potential over the temperature
(we use this notation for this case in order to not be confused with the magnetic
, and
X represents an extensive quantity which has to be defined below). That is, these identities derived from Equation (
76) are obtained by considering that the number of particles is constant and can be corrected by calculating the chemical potential using the Gibbs–Duhem relation. However, due to Equations (
73)–(
75), we can conclude that the magnetization
M is not a good extensive variable.
In order to solve this inconsistency, let us define a set of extensive variables for the paramagnetic solid:
U the internal energy,
N the number of particles and
where
G is defined as an extensive special variable which will help to obtain an expression for the entropy as a fundamental equation; that is,
, if it exists, will represent a fundamental equation and, as a consequence, a thermodynamic system. In order to verify that Equation (
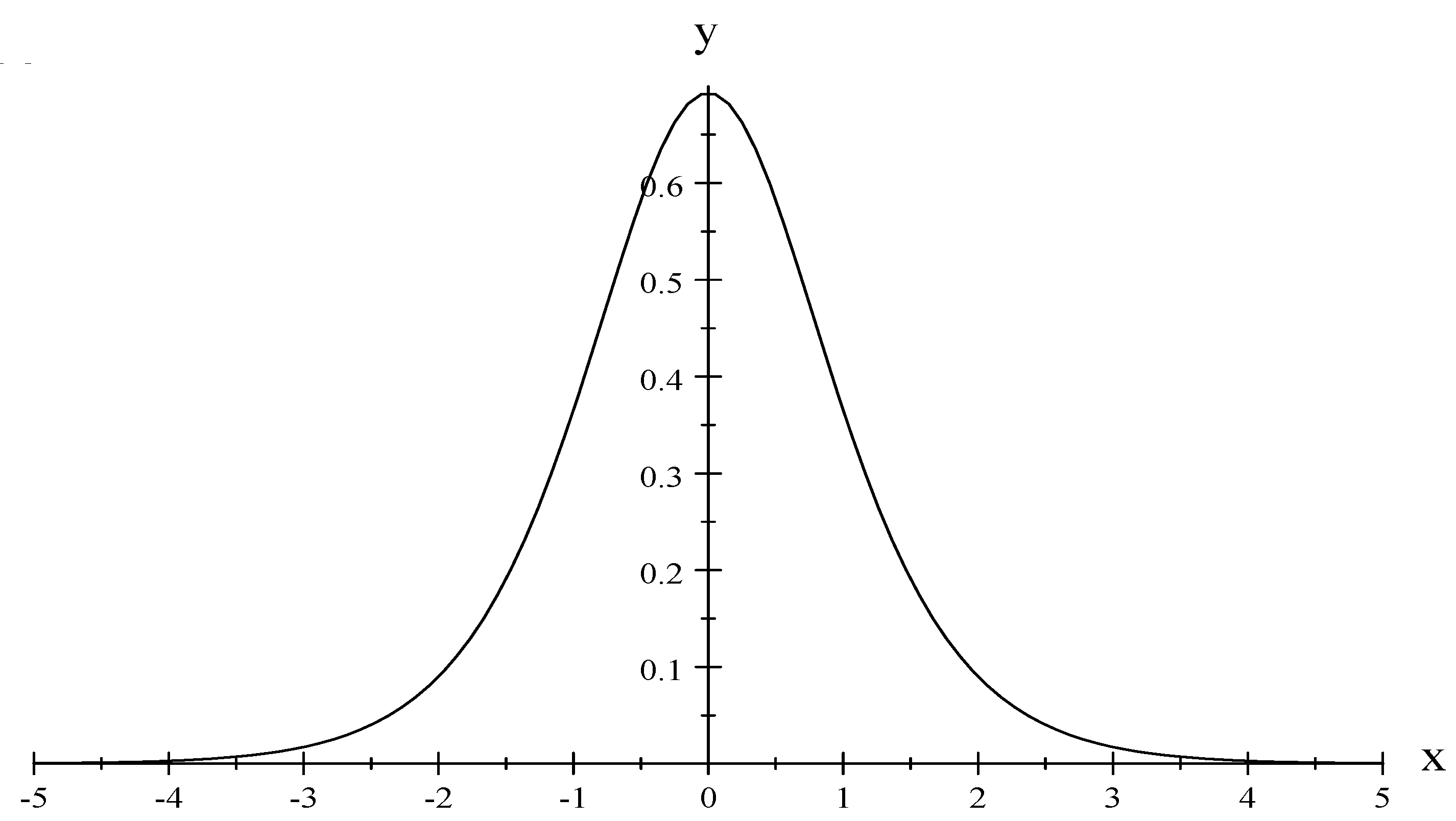
73) represents the entropy of the paramagnetic solid, let us begin by taking the result derived by Greiner et al. [
14] for
, and we have that (see
Figure 1):
where
. Using Equation (
70) in Equation (
78), we obtain Equation (
73). Introducing in Equation (
73)
U,
G and
N, and using the other equation of state,
where
, we have (see
Figure 2),
This expression for the entropy is now in the form of a fundamental equation, , and it can be used to directly obtain the equation, the equations of state, and in particular the chemical potential .
Using Equation (
79), we obtain
which represents the equation of state as expected, Equation (72). Note that if
m is negative, we have a negative temperature. For now, we consider
, and we postpone the discussion of the possibility of having
below in this subsection. Further,
Using Equation (
82), we arrive at
We can now express
as
Finally, the
equation can be written as
If we consider the case where
N is constant, we arrive at
which coincides with Equation (
76) obtained by Barrett and Macdonald [
13].
Finally, we have shown how to construct a fundamental equation for the paramagnetic solid with the
N variable. It has to be highlighted that when
N is not constant and we want to represent the
equation in the representation
U,
B and
N, we arrive at
Note that represents and not minus the chemical potential ().
4.4.1. Hessian Method
In order to calculate the Hessian, we need to express
. However, an inspection of Equation (
80) shows that it is not possible to write explicitly
, and consequently, it is not possible to directly obtain the Hessian. In
Appendix C, the Hessian for the paramagnetic solid is calculated. Since we have all the entries for the Hessian and by putting
and
, the characteristic polynomial is:
We can see that one of the eigenvalues is
as it is required, and the other two are
with
and
Therefore, the system satisfies the thermodynamic requirements.
4.4.2. Entropy Method
We have already proven item
I, that is, the entropy is a first-degree homogeneous function (
80), and items
A and
G are satisfied by looking at Equations (
79), (
82) and (
80). Therefore, we just need to prove conditions
H (
) and
G (
) are satisfied, that is, for condition
H, we have shown in the Hessian that
are positive if
m is positive, which is equivalent to
[
5]. However, from a statistical point of view, it is possible to have a negative temperature which comes from an initial oriented magnetization due to use a contrary direction of the magnetic field and suddenly changes the direction of it. However, this represents a non-equilibrium system [
14,
15]. Therefore, we can consider that the entropy is positive in a regular situation with
m positive (
). For condition
G, it is clear that all the first and second derivatives exist. Further, note that obtaining Equation (
73) is left as an exercise (Problem 2.4) in Reif’s book [
16] and the discussion of negative temperatures can be found in Problem (3.2).
4.4.3. Completeness Method
In general, we can express the
equations of this system using Equation (
15). In this case, we know that we only need three linearly independent equations. Thus, we choose the 1-equation, the 2-equation, and the 5-equation. That is, in these three equations, all the differentials of the variables
T,
m,
,
, and
N are included in one of the equations as it is required to obtain the three independent equations.
From Equations (
15) and (
16), we need to know
,
, and
, that is:
From Equations (
15) and (
16), we need to know
,
, and
, that is:
From Equations (
15) and (
16), we need to know
and
. We have
and
Then, by putting
and
, we obtain
Thermodynamic Identities
With the three linearly independent
equations, it is possible to calculate the rest of the
equations and in this way obtain different thermodynamic identities. In this case, it is possible from the 1-equation and the 2-equation to deduce the 7-equation, which can be calculated using the method previously described in
Section 3, that is:
Then, by putting
, and remembering that
, we have
Or, in an equivalent form, we obtain a Mayer-like relation:
4.5. The Kelly Plasma Equation
For more than 70 years, there has been much interest in plasma physics due to its applications in Tokamaks and Astrophysics. However, under nonrelativistic conditions, the equations of state for an ideal gas state are used as the first approximation to calculate the equations of balance, the moment equations, and thermodynamic flows [
17,
18]. Nevertheless, among other proposals, Kelly [
7] gave an equation of state for a plasma with different species relating the pressure
P, the number of particles for each species
, the volume
V, and the temperature
T. However, one equation of state is not enough to describe a system composed of many species without giving another equation of state. This point was partially corrected by Wergeland [
19] by including an expression for the corrected internal energy due to Debye and Hückel theory, where the energy is obtained by making an average of the Coulomb energy.
Our purpose in this subsection consists of using our method to complete the set of equations, as we did in
Section 4.2 (Unconventional System: A Particular Case) and in
Section 4.3 (The Rubber Band), obtaining the Debye–Hückel–Wergeland corrected total internal energy. Since in many situations in plasma physics, it is just necessary to consider only the electrons, we analyze the Kelly equations for one species. We obtain the entropy for a plasma composed of electrons where the effect of the ions is considered just in the equations of state. We obtain the entropy as a function of the volume
V, the number of electrons
N, and the temperature
T. Since it is not possible to explicitly write the entropy as a function of the internal energy
U, the volume
V, and the number of electrons
N, that is, the entropy as a fundamental equation, we develop a method to obtain the Hessian in order to check the viability of the system similar to what we have done in
Section 4.2 (Unconventional System: A Particular Case). Indeed, in plasma physics, it is very important to know the relaxation times which are related with the eigenvalues of the Hessian. We also analyze the first-degree homogeneous property of the entropy using a technique exposed in
Section 2.4. We also show that the temperature
T is positive. The
equations can be obtained from the results obtained for calculating the Hessian.
The Kelly equation of state for a system with different species is:
If we consider just one species (electrons) but keeping the interaction with the ions of the system, we have
where
It is necessary to note that
D is Debye’s length and
represents the number of particles contained in Debye’s sphere. The contribution of the pressure that corrects the ideal gas is
Following Wergeland [
19], the contribution of the energy should be
We note that
which has the form of the equation of state for bosons and fermions. Let us analyze Equation (
104); we have
Finally, we can write the total pressure as
and the total energy as
It is interesting that we can obtain the heat capacity
, that is:
Due to the similarity with the relationship between energy and pressure in the case of photons, we can propose a correction for the entropy given by the following expression
Note that due to the expression of the corrected energy in our case (see Equation (
107)), there is a factor
and not a factor
as it happens in the photon case. Thus, we can propose the total entropy as
where
represents the entropy for a null charge,
, that is, the ideal gas. It has to be noted that although
is formally the entropy of the ideal gas, it cannot be substituted in another functionality, that is, the expression is good for our purpose only when it is a function of the volume
V, the temperature
T, and the number of particles
N. Therefore,
In order to verify that this proposed entropy describes the system, it is necessary to check that we can obtain the equations of state. For this purpose, let us calculate the Helmholtz free energy
A, that is:
Now, we can calculate the pressure
P,
and the equation of state is verified. Using Equations (
111) and (
112), the equation of state for
U can also be verified.
4.5.1. Hessian Method
In order to calculate the Hessian, we need to express
. However, an inspection of Equation (
111) shows that it is not possible to write explicitly
, and consequently, it is not possible to directly obtain the Hessian. In
Appendix D, the Hessian for the Kelly plasma is calculated. Once the Hessian is obtained and by putting
and
, the characteristic polynomial is:
Therefore, we found an eigenvalue
, and the other two eigenvalues are:
with
and
4.5.2. Entropy Method
The items
A,
F, and
G can be verified by looking at Equations (
106), (
107) and (A32). Although we have not been able to deduce the entropy as a fundamental equation for this system, we can apply the technique developed in
Section 2.4 in order to verify item
I. Therefore, we need to prove Equation (
A1) in our case (see
Appendix A), that is:
It is necessary to prove that the temperature is positive (item
H). We know from Equations (
102), (
103) and (
104) that:
which implies that
.
On the other hand, due to the fact that that the number of particles contained in a Debye sphere is big, the isothermal compressibility is positive,
Therefore, the Kelly completed plasma represents a stable system.