Voronoi Decomposition of Cardiovascular Dependency Structures in Different Ambient Conditions: An Entropy Study
Abstract
1. Introduction
- To propose a method that enables an application of multiscale entropy to an arbitrary number of signals and to analyze the outcome;
- To compare the results of the classical multiscale method and the proposed method when applicable, i.e., in a case of two-dimensional signals;
- To test whether the proposed method recognizes the changes of dependency level (coupling strength, level of interaction) of joint multivariate signals in different biomedical experiments.
2. Materials and Methods
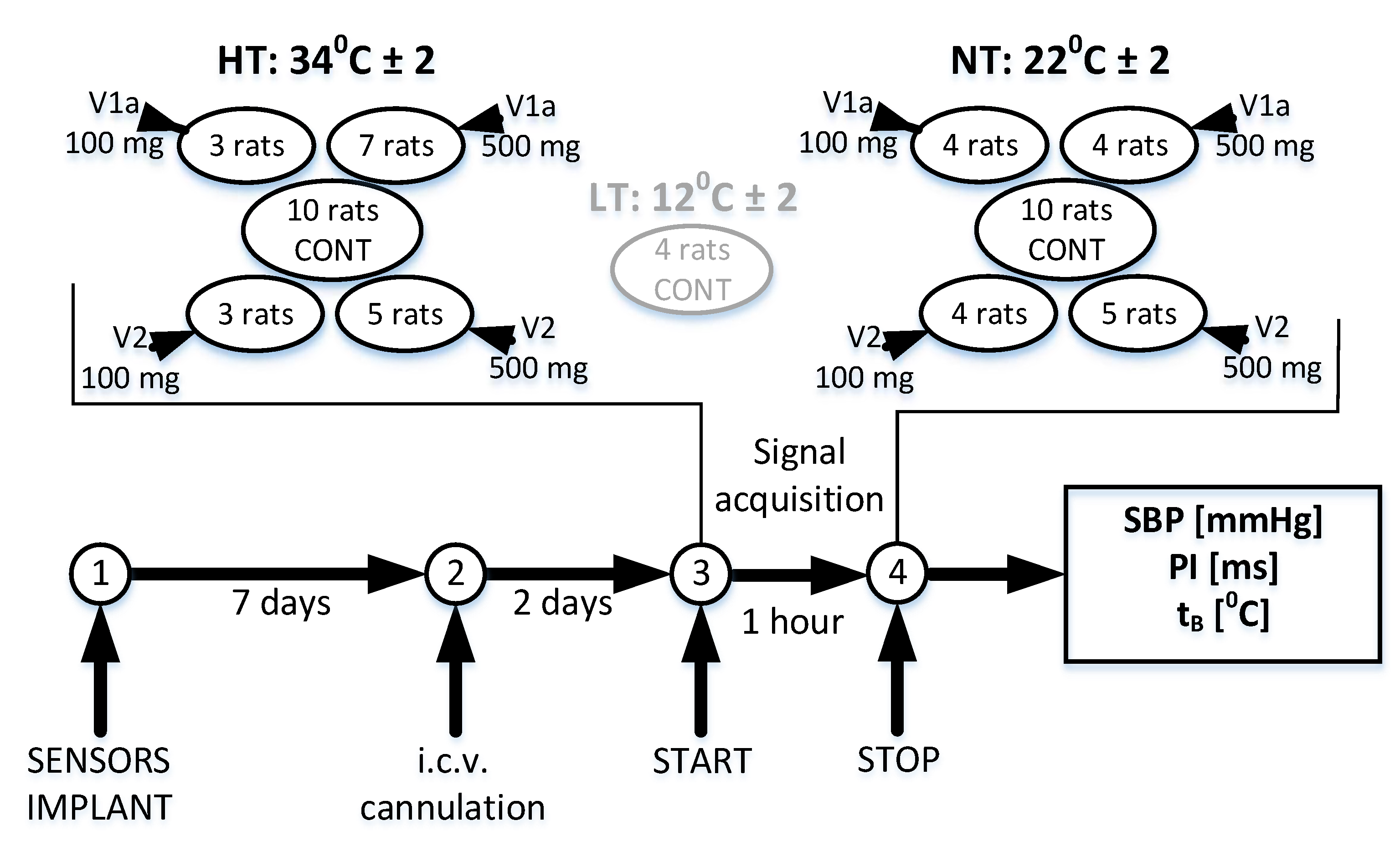
2.1. Experimental Setting and Signal Acquisition
2.2. Signal Pre-Processing
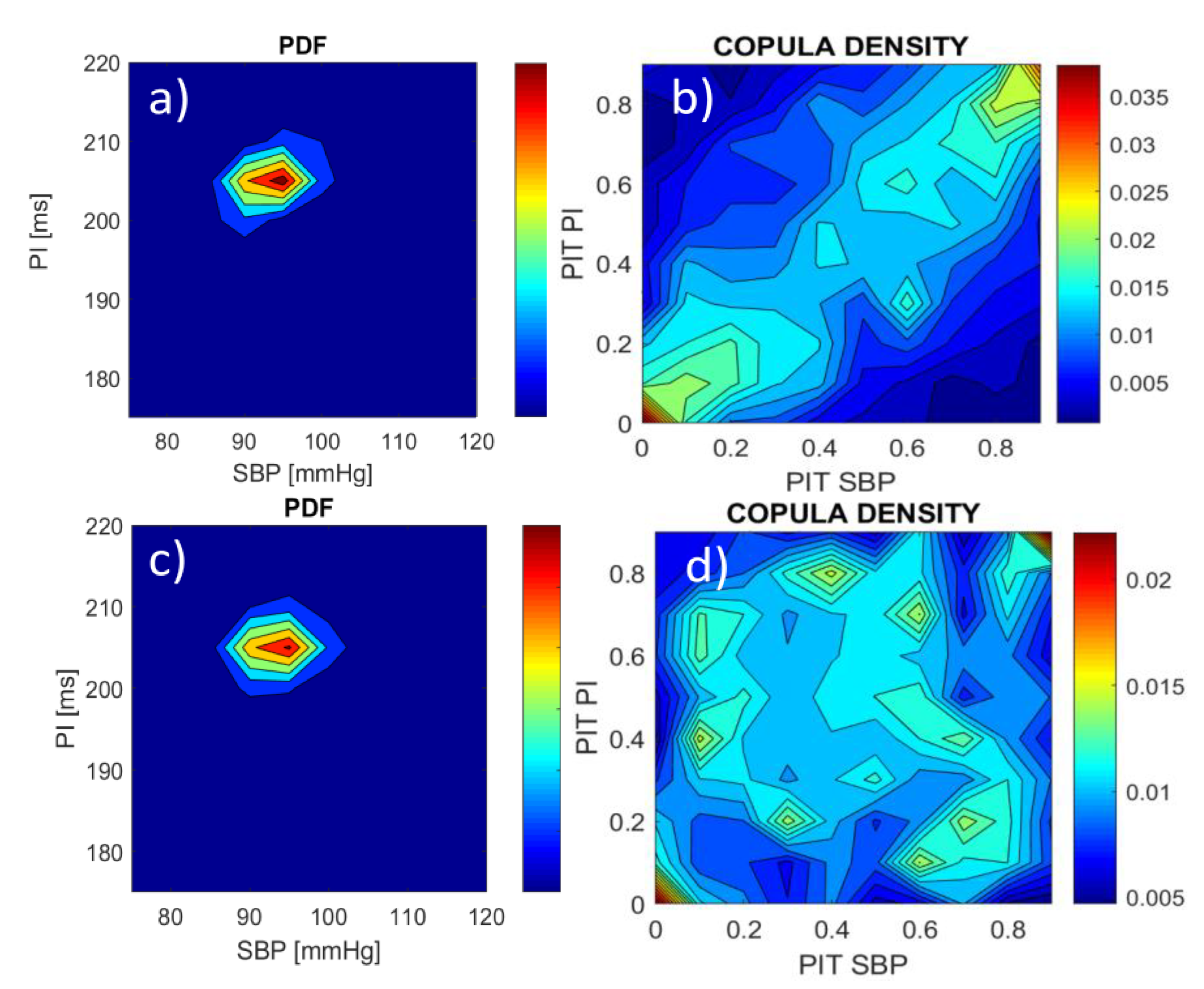
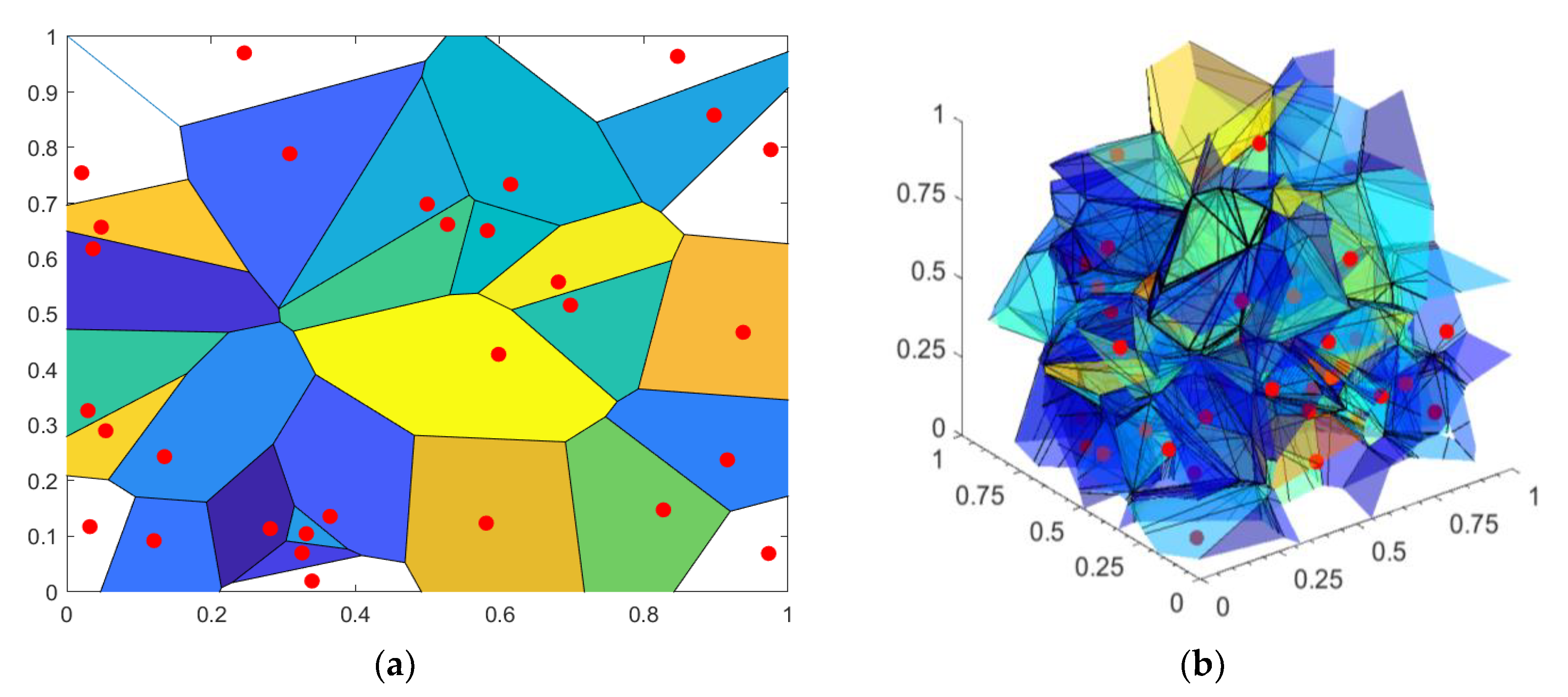
2.3. Copula Density, Voronoi Regions and Dependency Time Series
- (a)
- The surface/volume of is inversely proportional to the dependency level of the point . An increased density of dependency structures in [0 1]D space implies a decrease of available space between the points.
- (b)
- The region is shaped like the best distance separation of the point , so its surface/volume is unambiguously calculated and unique, without a necessity to include any thresholds.
3. Results
3.1. Source Signal Analysis
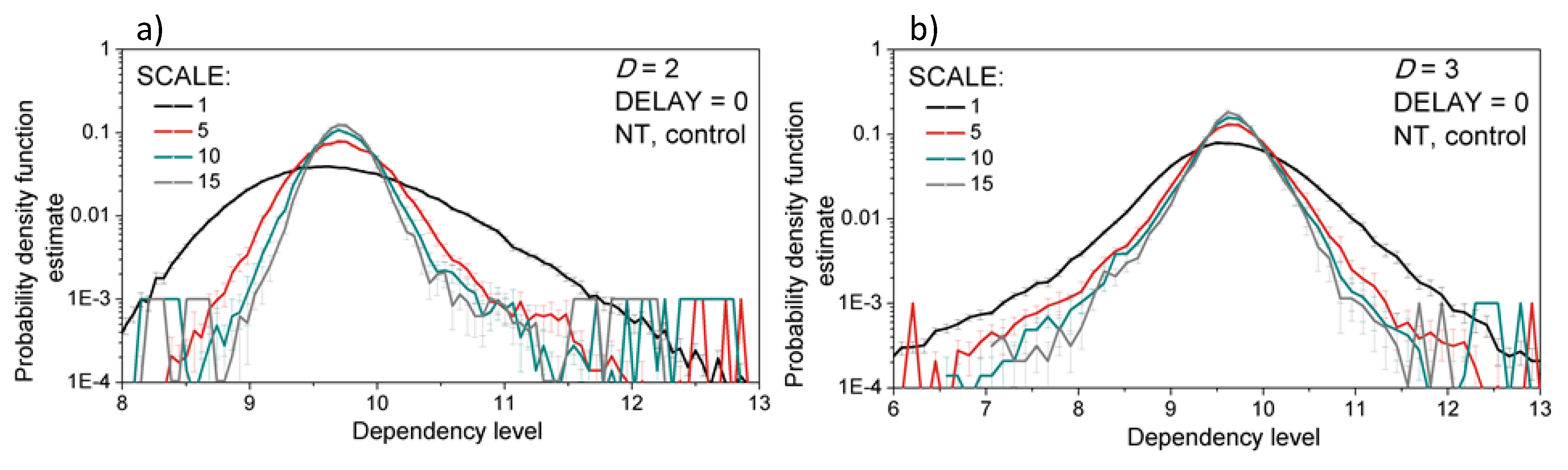
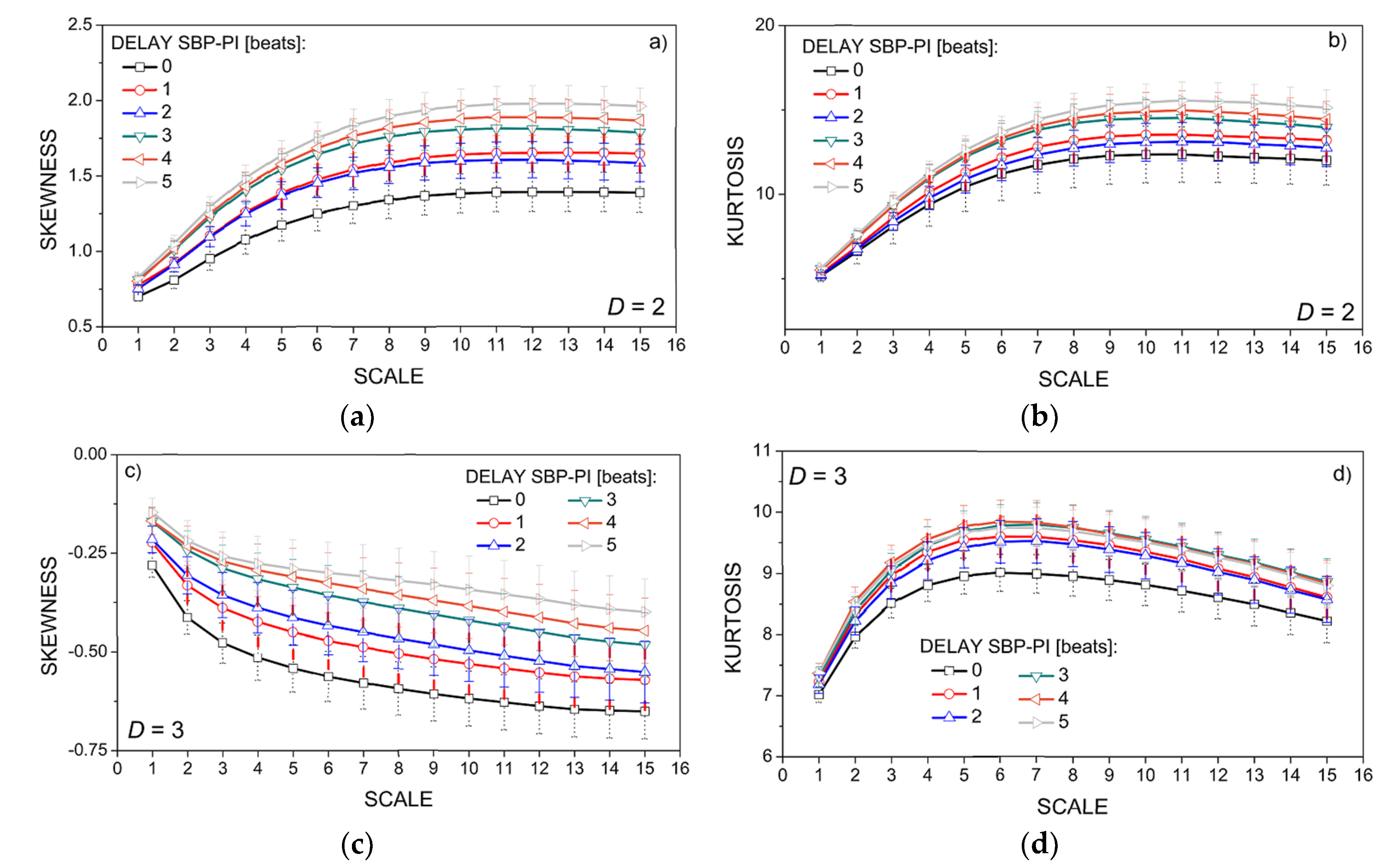
3.2. Properties of the Dependency Time Series
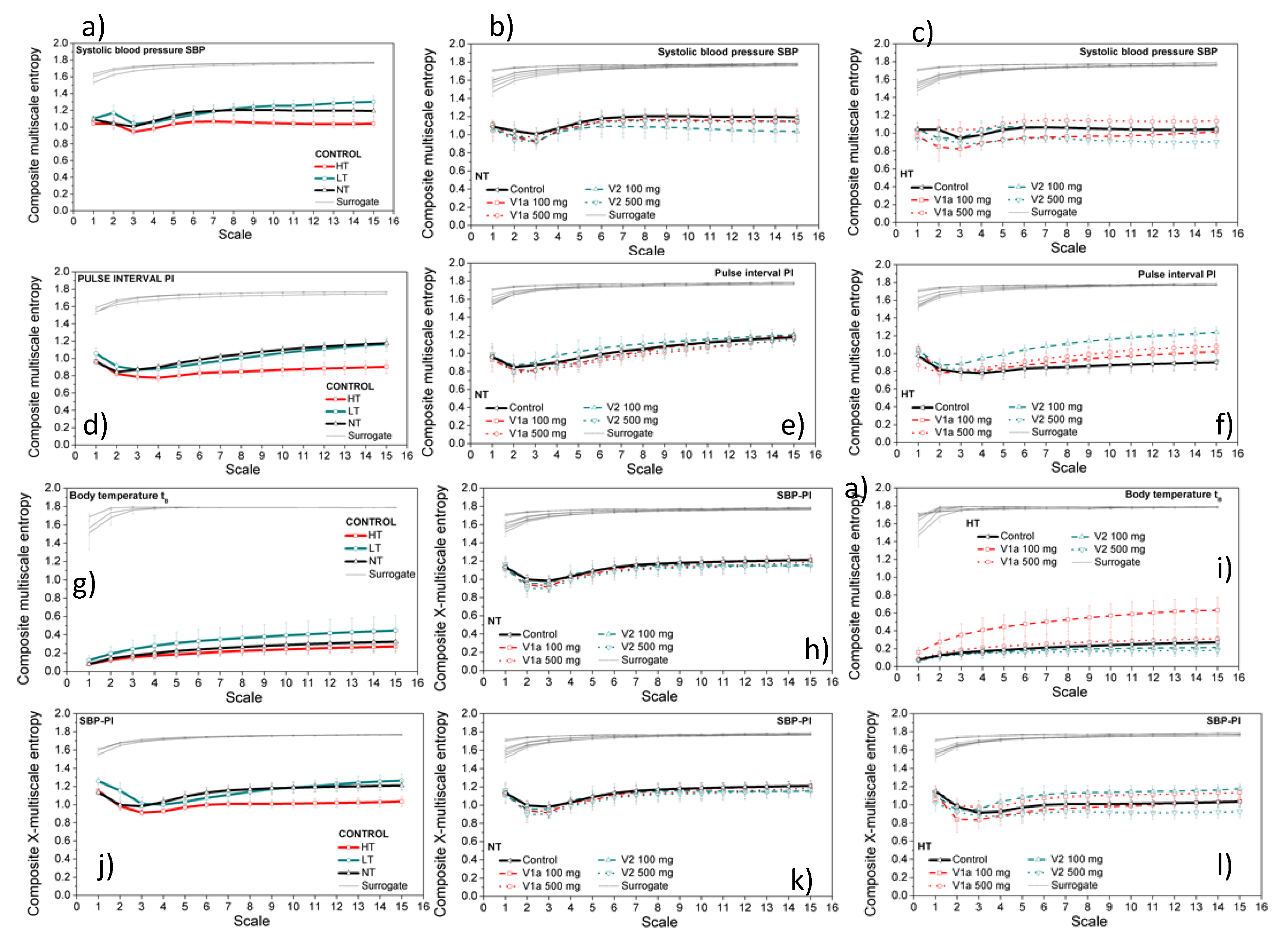
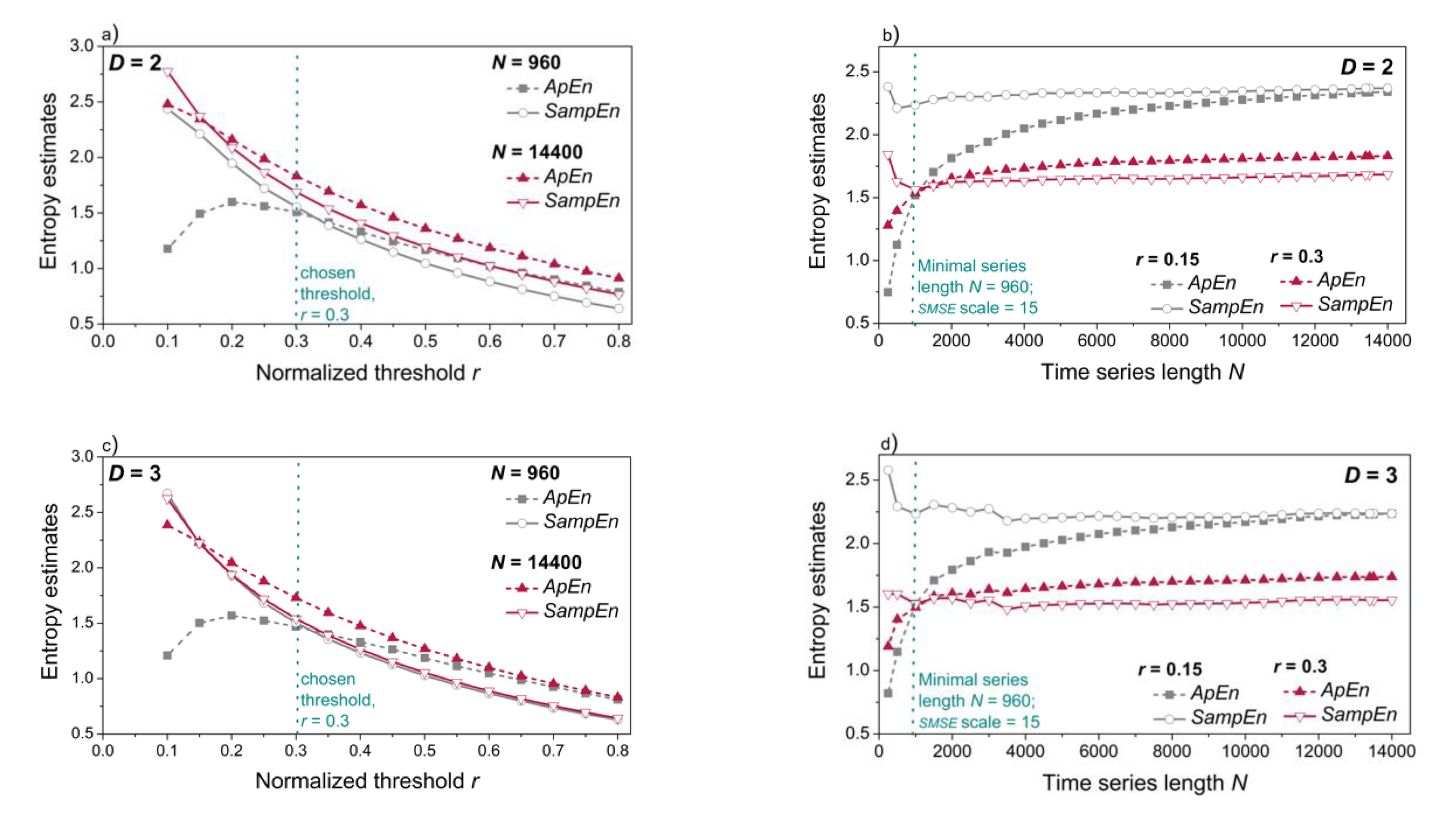
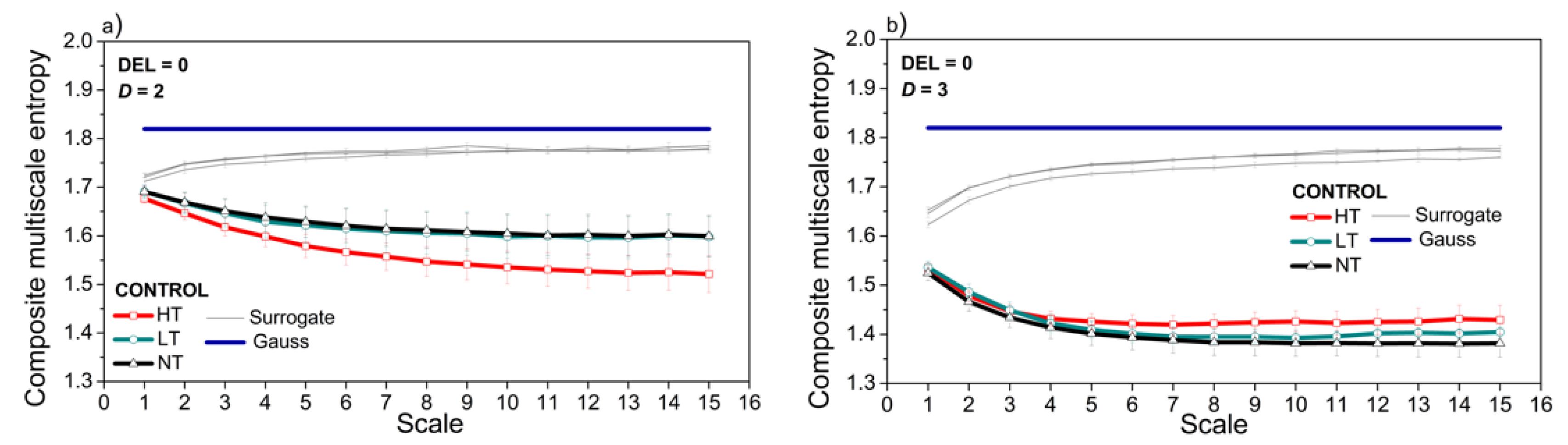
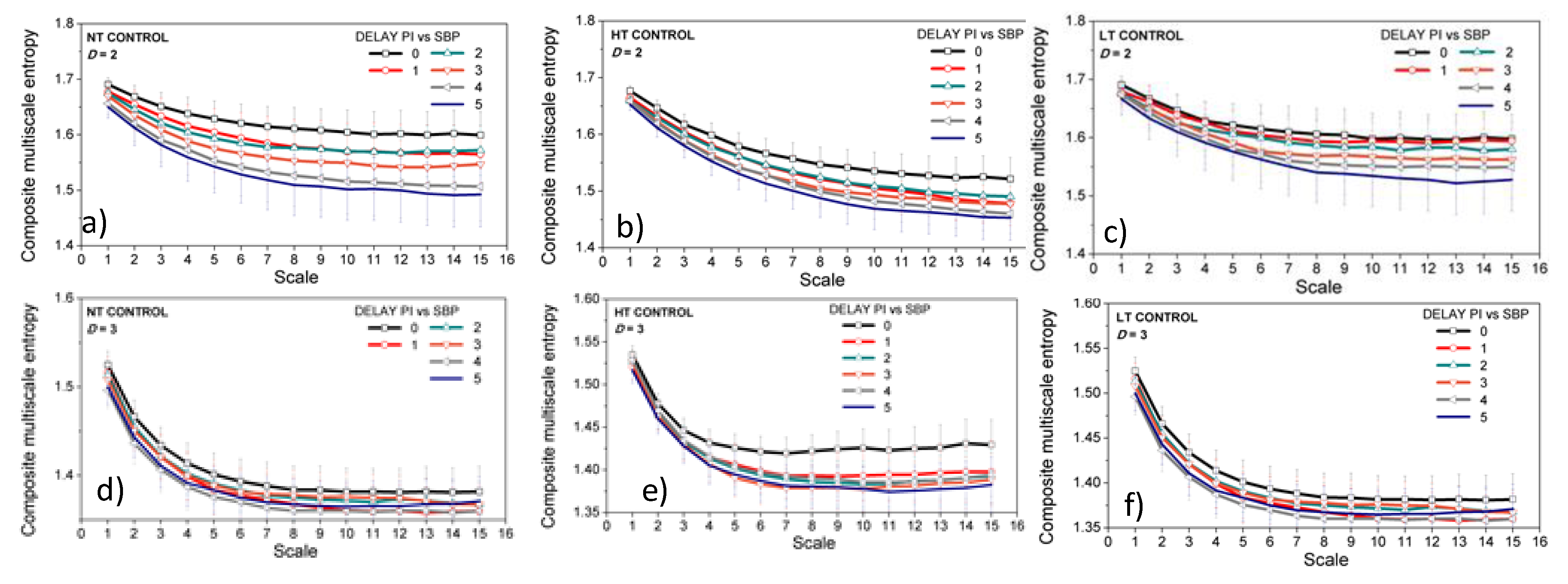
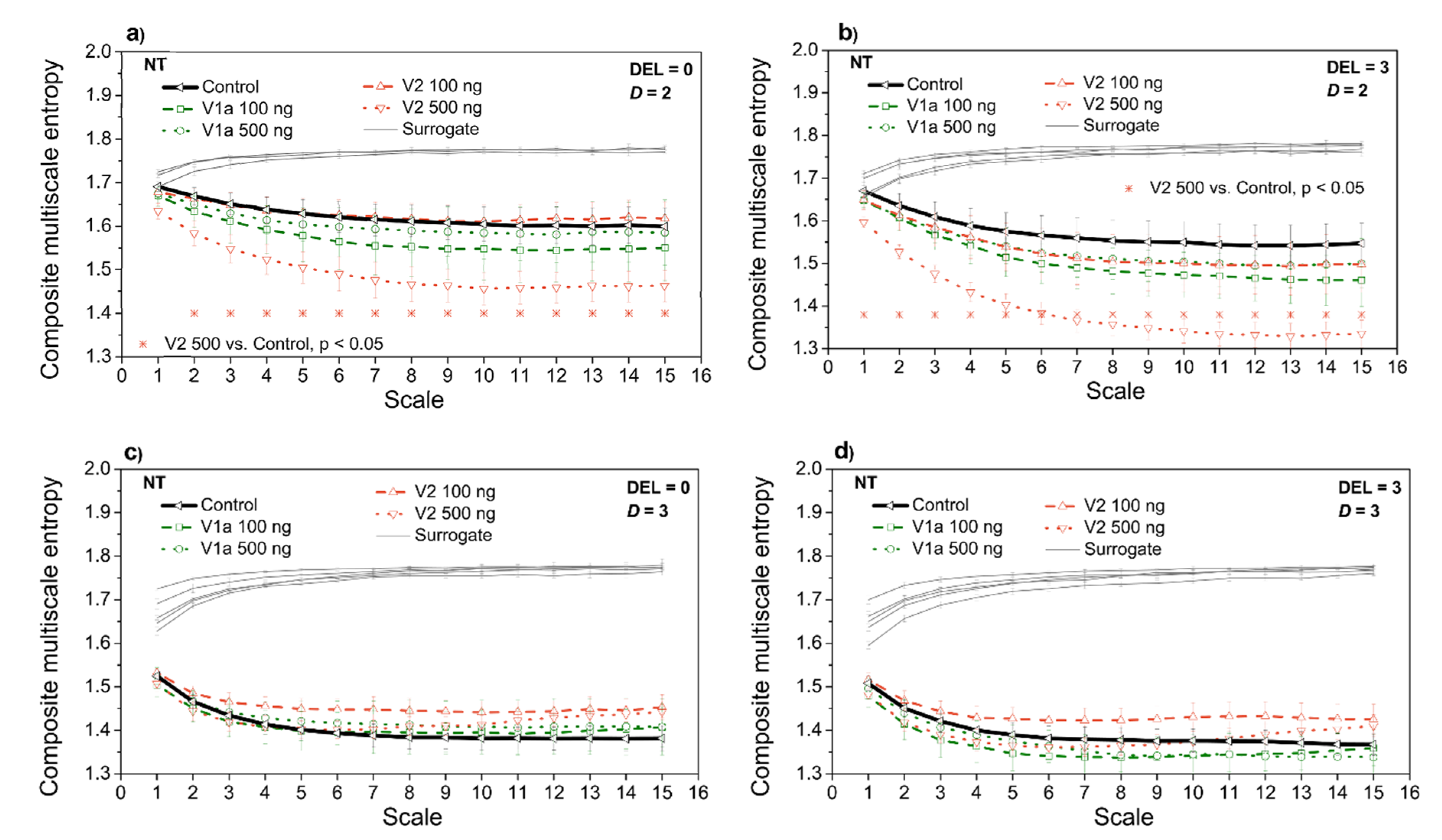
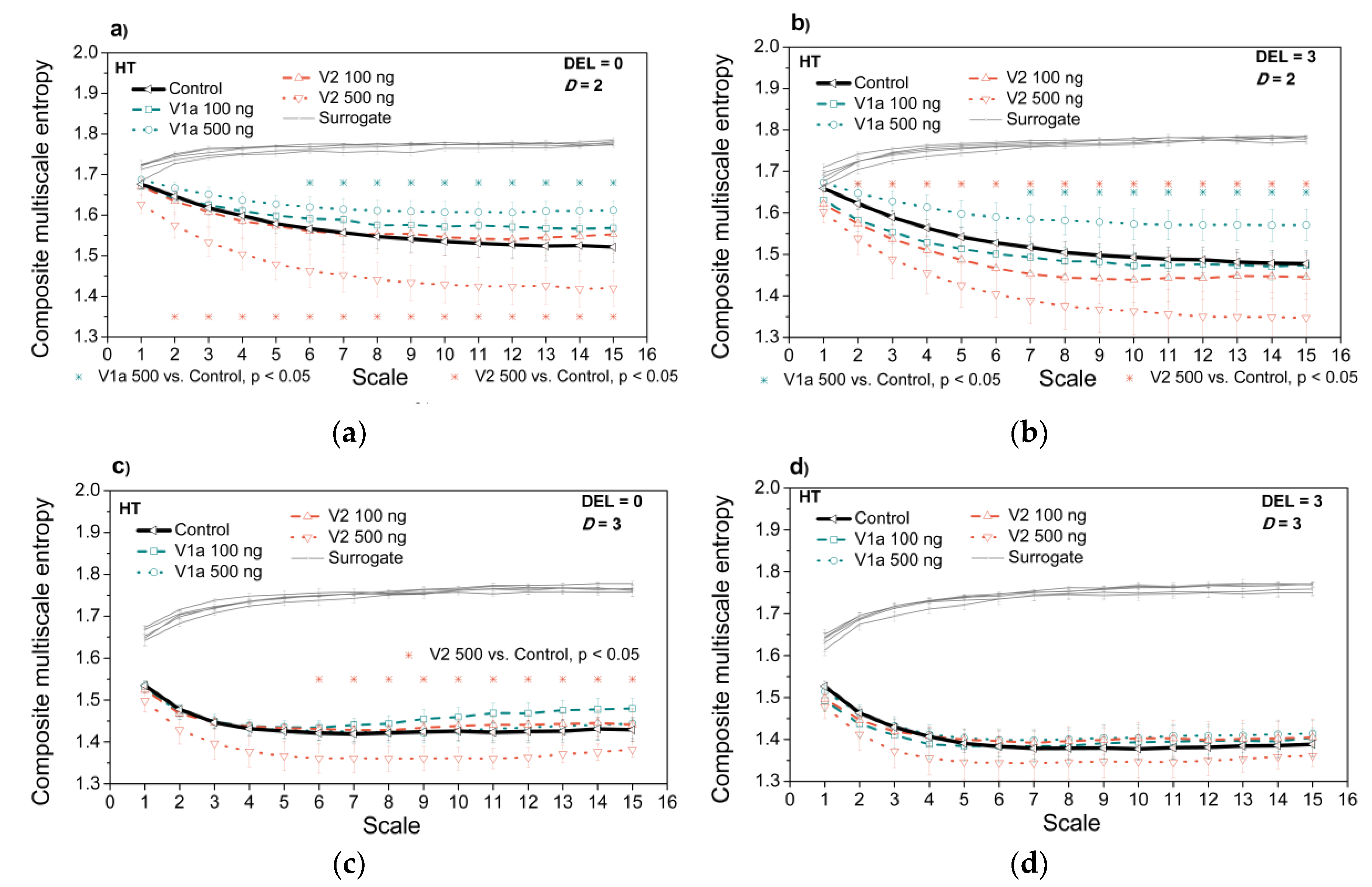
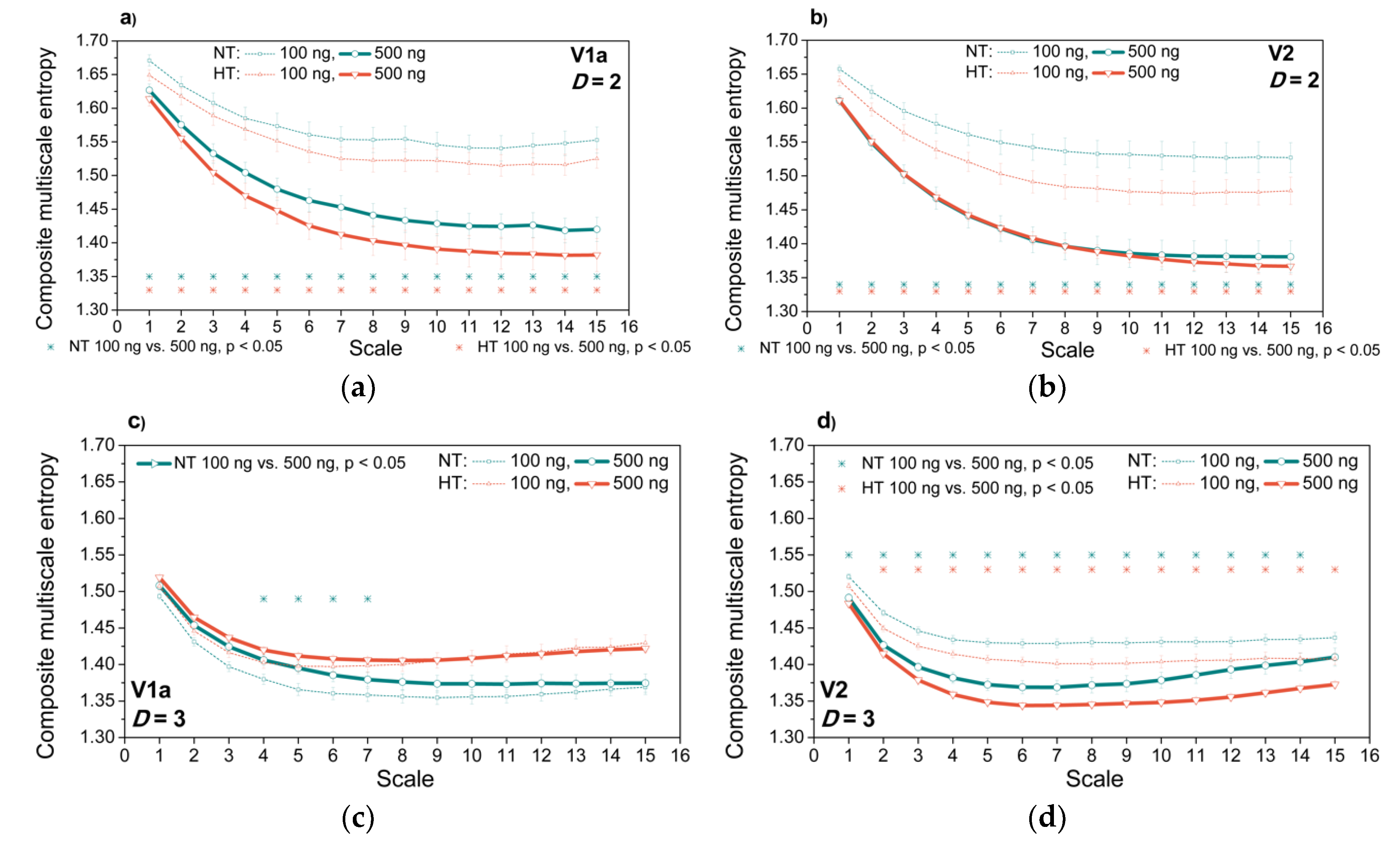
3.3. Entropy Analysis of the Dependency Time Series
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Entropy Concepts in Brief
References
- Pincus, S.M. Approximate entropy as a measure of system complexity. Proc. Natl. Acad. Sci. USA 1991, 88, 2297–2301. [Google Scholar] [CrossRef] [PubMed]
- Pincus, S.M.; Huang, W.M. Approximate entropy: Statistical properties and applications. Commun. Stat. Theory Methods 1992, 21, 3061–3077. [Google Scholar] [CrossRef]
- Richman, J.S.; Moorman, J.R. Physiological time-series analysis using approximate entropy and sample entropy. Am. J. Physiol. Heart Circ. Physiol. 2000, 278, 2039–2049. [Google Scholar] [CrossRef] [PubMed]
- Yentes, J.M.; Hunt, N.; Schmid, K.K.; Kaipust, J.P.; McGrath, D.; Stergiou, N. The appropriate use of approximate entropy and sample entropy with short data sets. Ann. Biomed. Eng. 2013, 41, 349–365. [Google Scholar] [CrossRef]
- Pincus, S.; Singer, B.H. Randomness and degrees of irregularity. Proc. Natl. Acad. Sci. USA 1996, 93, 2083–2088. [Google Scholar] [CrossRef]
- Pincus, S.M.; Mulligan, T.; Iranmanesh, A.; Gheorghiu, S.; Godschalk, M.; Veldhuis, J.D. Older males secrete luteinizing hormone and testosterone more irregularly, and jointly more asynchronously than younger males. Proc. Natl. Acad. Sci. USA 1996, 93, 14100–14105. [Google Scholar] [CrossRef]
- Delgado-Bonal, A.; Marshak, A. Approximate Entropy and Sample Entropy: A Comprehensive Tutorial. Entropy 2019, 21, 541. [Google Scholar] [CrossRef]
- Costa, M.; Goldberger, A.L.; Peng, C.K. Multiscale entropy analysis of complex physiologic time series. Phys. Rev. Lett. 2002, 89, 068102. [Google Scholar] [CrossRef]
- Costa, M.; Goldberger, A.L.; Peng, C.K. Multiscale entropy analysis of biological signals. Phys. Rev. E 2005, 71, 021906. [Google Scholar] [CrossRef]
- Wu, S.D.; Wu, C.W.; Lin, S.G.; Wang, C.C.; Lee, K.Y. Time series analysis using composite multiscale entropy. Entropy 2013, 15, 1069–1084. [Google Scholar] [CrossRef]
- Lin, T.K.; Chien, Y.H. Performance Evaluation of an Entropy-Based Structural Health Monitoring System Utilizing Composite Multiscale Cross-Sample Entropy. Entropy 2019, 21, 41. [Google Scholar] [CrossRef]
- Castiglioni, P.; Parati, G.; Faini, A. Information-Domain Analysis of Cardiovascular Complexity: Night and Day Modulations of Entropy and the Effects of Hypertension. Entropy 2019, 21, 550. [Google Scholar] [CrossRef]
- Marwaha, P.; Sunkaria, R.M. Cardiac variability time–series analysis by sample entropy and multiscale entropy. Int. J. Med. Eng. Inform. 2015, 7, 1–14. [Google Scholar] [CrossRef]
- Chen, C.; Jin, Y.; Lo, I.L.; Zhao, H.; Sun, B.; Zhao, Q.; Zhang, X.D. Complexity Change in Cardiovascular Disease. Int. J. Biol. Sci. 2017, 13, 1320–1328. [Google Scholar] [CrossRef]
- Li, X.; Yu, S.; Chen, H.; Lu, C.; Zhang, K.; Li, F. Cardiovascular autonomic function analysis using approximate entropy from 24-h heart rate variability and its frequency components in patients with type 2 diabetes. J. Diabetes Investig. 2015, 6, 227–235. [Google Scholar] [CrossRef]
- Krstacic, G.; Gamberger, D.; Krstacic, A.; Smuc, T.; Milicic, D. The Chaos Theory and Non-linear Dynamics in Heart Rate Variability in Patients with Heart Failure. In Proceedings of the Computers in Cardiology, Bologna, Italy, 14–17 September 2008; pp. 957–959. [Google Scholar]
- Storella, R.J.; Wood, H.W.; Mills, K.M.; Kanters, J.K.; Højgaard, M.V.; Holstein-Rathlou, N.H. Approximate entropy and point correlation dimension of heart rate variability in healthy subjects. Integr. Physiol. Behav. Sci. 1998, 33, 315–320. [Google Scholar] [CrossRef]
- Boskovic, A.; Loncar-Turukalo, T.; Sarenac, O.; Japundžić-Žigon, N.; Bajić, D. Unbiased entropy estimates in stress: A parameter study. Comput. Biol. Med. 2012, 42, 667–679. [Google Scholar] [CrossRef]
- Ryan, S.M.; Goldberger, A.L.; Pincus, S.M.; Mietus, J.; Lipsitz, L.A. Gender- and Age-Related Differences in Heart Rate Dynamics: Are Women More Complex Than Men? J. Am. Coll. Cardiol. 1994, 24, 1700–1707. [Google Scholar] [CrossRef]
- Wang, S.Y.; Zhang, L.F.; Wang, X.B.; Cheng, J.H. Age dependency and correlation of heart rate variability, blood pressure variability, and baroreflex sensitivity. J. Gravit. Physiol. 2002, 7, 145–146. [Google Scholar]
- Sklar, A. Fonctions de Répartition à n Dimensions et Leurs Marges; Institut de Statistique del Universit’e de Paris: Paris, France, 1959; Volume 8, pp. 229–231. [Google Scholar]
- Claeys, M.J.; Rajagopalan, S.; Nawrot, T.S.; Brook, R.D. Climate and environmental triggers of acute myocardial infarction. Eur. Heart J. 2017, 38, 955–960. [Google Scholar] [CrossRef]
- Akselrod, S.; Gordon, D.; Ubel, F.A.; Shannon, D.C.; Bezger, A.C.; Cohen, R.J. Power spectrum analysis of heart rate fluctuations: A quantitative probe of beat-to-beat cardiovascular control. Science 1981, 213, 220–222. [Google Scholar] [CrossRef] [PubMed]
- Kinugasa, H.; Hirayanagi, K. Effects of skin surface cooling and heating on autonomic nervous activity and baroreflex sensitivity in humans. Exp. Physiol. 1999, 84, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Japundžić-Žigon, N. Effects of nonpeptide V1a and V2 antagonists on blood pressure fast oscillations in conscious rats. Clin. Exp. Hypertens. 2001, 23, 277–292. [Google Scholar] [CrossRef] [PubMed]
- Japundžić-Žigon, N.; Milutinović, S.; Jovanović, A. Effects of nonpeptide and selective V1 and V2 antagonists on blood pressure short-term variability in spontaneously hypertensive rats. J. Pharmacol. Sci. 2004, 95, 47–55. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Milutinović, S.; Murphy, D.; Japundžić-Žigon, N. The role of central vasopressin receptors in the modulation of autonomic cardiovascular controls: A spectral analysis study. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2006, 291, 1579–1591. [Google Scholar] [CrossRef][Green Version]
- Milutinović-Smiljanić, S.; Šarenac, O.; Lozić-Djurić, M.; Murphy, D.; Japundžić-Žigon, N. Evidence for the involvement of central vasopressin V1b and V2 receptors in stress-induced baroreflex desensitization. Br. J. Pharmacol. 2013, 169, 900–908. [Google Scholar] [CrossRef]
- Oosting, J.; Struijker-Boudier, H.A.; Janssen, B.J. Validation of a continuous baroreceptor reflex sensitivity index calculated from spontaneous fluctuations of blood pressure and pulse interval in rats. J. Hypertens. 1997, 15, 391–399. [Google Scholar] [CrossRef]
- Schreiber, T.; Schmitz, A. Surrogate time series. Phys. D Nonlinear Phenom. 1999, 142, 346–382. [Google Scholar] [CrossRef]
- Theiler, J.; Eubank, S.; Longtin, A.; Galdrikian, B.; Farmer, J.D. Testing for nonlinearity in time series: The method of surrogate data. Phys. D Nonlinear Phenom. 1992, 58, 77–94. [Google Scholar] [CrossRef]
- Papoulis, A.; Pillai, S.U. Probability, Random Variables, and Stochastic Processes; McGraw-Hill: New York, NY, USA, 1984. [Google Scholar]
- Wessel, N.; Voss, A.; Malberg, H.; Ziehmann, C.H.; Voss, H.U.; Schirdewan, A.; Meyerfeldt, U.; Kurths, J. Nonlinear analysis of complex phenomena in cardiological data. Herzschrittmacherther. Elektrophysiol. 2000, 11, 159–173. [Google Scholar] [CrossRef]
- Tarvainen, M.P.; Ranta-aho, P.O.; Karjalainen, P.A. An advanced detrending approach with application to HRV analysis. IEEE Trans. Biomed. Eng. 2002, 42, 172–174. [Google Scholar] [CrossRef] [PubMed]
- Angus, J.E. The Probability Integral Transform and Related Results. SIAM Rev. 1994, 36, 652–654. [Google Scholar] [CrossRef]
- Cherubini, U.; Luciano, E.; Vecchiato, W. Copula Methods in Finance; Wiley Finance Series; Wiley: Chichester, UK, 2004. [Google Scholar]
- Malevergne, Y.; Sornette, D. Extreme Financial Risks; Springer: Berlin/Heidelberg, Germany, 2006. [Google Scholar]
- McNeil, A.J.; Frey, R.; Embrechts, P. Quantitative Risk Management: Concepts, Techniques, and Tools; Princeton Series in Finance; Princeton University Press: Princeton, NJ, USA, 2005. [Google Scholar]
- Schönbucker, P. Credit Derivatives Pricing Models: Models, Pricing, Implementation; Wiley Finance Series; Wiley: Chichester, UK, 2003. [Google Scholar]
- Genest, C.; Favre, A.C. Everything you always wanted to know about copula modeling but were afraid to ask. J. Hydrol. Eng. 2007, 12, 347–368. [Google Scholar] [CrossRef]
- Salvadori, G.; DeMichele, C.; Kottegoda, N.T.; Rosso, R. Extremes in Nature: An Approach Using Copulas; Springer: Dordrecht, The Netherlands, 2007; Volume 56. [Google Scholar]
- Vandenberghe, S.; Verhoest, N.E.C.; De Baets, B. Fitting bivariate copulas to the dependence structure between storm characteristics: A detailed analysis based on 105 years 10 min rainfall. Water Resour. Res. 2010, 46. [Google Scholar] [CrossRef]
- Yan, X.; Clavier, L.; Peters, G.; Septier, F.; Nevat, I. Modeling dependence in impulsive interference and impact on receivers. In Proceedings of the 11th MCM COST IC1004, Krakow, Poland, 24–26 September 2014. [Google Scholar]
- Kumar, P.; Shoukri, M.M. Copula based prediction models: An application to an aortic regurgitation study. BMC Med. Res. Methodol. 2007, 7, 21. [Google Scholar] [CrossRef] [PubMed]
- Bansal, R.; Hao, X.J.; Liu, J.; Peterson, B.S. Using Copula distributions to support more accurate imaging-based diagnostic classifiers for neuropsychiatric disorders. Magn. Reson. Imaging 2014, 32, 1102–1113. [Google Scholar] [CrossRef]
- Jovanovic, S.; Skoric, T.; Sarenac, O.; Milutinovic-Smiljanic, S.; Japundzic-Zigon, N.; Bajic, D. Copula as a dynamic measure of cardiovascular signal interactions. Biomed. Signal Process. Control 2018, 43, 250–264. [Google Scholar] [CrossRef]
- Laude, D.; Baudrie, V.; Elghozi, J.L. Tuning of the sequence technique. IEEE Eng. Med. Biol. Mag. 2009, 28, 30–34. [Google Scholar]
- Bajic, D.; Loncar-Turukalo, T.; Stojicic, S.; Sarenac, O.; Bojic, T.; Murphy, J.; Paton, R.; Japundzic-Zigon, N. Temporal analysis of the spontaneous baroreceptor reflex during mild emotional stress in the rat. Stress Int. J. Biol. Stress 2009, 13, 142–154. [Google Scholar] [CrossRef]
- Laude, D.; Elghozi, J.L.; Girard, A.; Bellard, E.; Bouhaddi, M.; Castiglioni, P.; Cerutti, C.; Cividjian, A.; Di Rienzo, M.; Fortrat, J.O.; et al. Comparison of various techniques used to estimate spontaneous baroreflex sensitivity (the EUROBAVAR study). Am. J. Physiol. Regul. Integr. Comp. Physiol. 2004, 286, 226–231. [Google Scholar] [CrossRef]
- Тasic, T.; Jovanovic, S.; Mohamoud, O.; Skoric, T.; Japundzic-Zigon, N.; Bajićc, D. Dependency structures in differentially coded cardiovascular time series. Comput. Math. Methods Med. 2017, 2017, 2082351. [Google Scholar] [CrossRef] [PubMed]
- Bernard, W. Silverman: Density Estimation for Statistics and Data Analysis; CRC Press: Boca Raton, FL, USA, 1986. [Google Scholar]
- Voronoi, G. Nouvelles applications des paramètres continuos à la théorie des formes quadratiques. J. Reine Angew. Math. 1908, 133, 198–287. [Google Scholar] [CrossRef]
- Skoric, T.; Sarenac, O.; Milovanovic, B.; Japundzic-Zigon, N.; Bajic, D. On Consistency of Cross-Approximate Entropy in Cardiovascular and Artificial Environments. Complexity 2017, 2017, 8365685. [Google Scholar] [CrossRef]
- Shannon, C.E. Communications in the presence of noise. Proc. IRE 1949, 37, 10–21. [Google Scholar] [CrossRef]
- Bendat, J.S.; Piersol, A.G. Random Data Analysis and Measurement Procedures; Wiley Series in Probability and Statistics; Wiley: New York, NY, USA, 1986. [Google Scholar]
- Lu, S.; Chen, X.; Kanters, J.K.; Solomon, I.C.; Chon, K.H. Automatic selection of the threshold value r for approximate entropy. IEEE Trans. Biomed. Eng. 2008, 55, 1966–1972. [Google Scholar]
- Chon, K.H.; Scully, C.G.; Lu, S. Approximate entropy for all signals. IEEE Eng. Med. Biol. Mag. 2009, 28, 18–23. [Google Scholar] [CrossRef]
- Castiglioni, P.; Di Rienzo, M. How the threshold “r” influences approximate entropy analysis of heart-rate variability. In Proceedings of the Conference Computers in Cardiology, Bologna, Italy, 14–17 September 2008; Volume 35, pp. 561–564. [Google Scholar]
- Liu, C.; Shao, P.; Li, L.; Sun, X.; Wang, X.; Liu, F. Comparison of different threshold values r for approximate entropy: Application to investigate the heart rate variability between heart failure and healthy control groups. Physiol. Meas. 2011, 32, 167–180. [Google Scholar] [CrossRef]
- Restrepo, J.F.; Schlotthauer, G.; Torres, M.E. Maximum approximate entropy and r threshold: A new approach for regularity changes detection. Phys. A Stat. Mech. Appl. 2014, 409, 97–109. [Google Scholar] [CrossRef]
- Yang, Y.; Gordon, J. Ambient temperature limits and stability of temperature regulation in telemetered male and female rats. J. Therm. Biol. 1996, 21, 353–363. [Google Scholar] [CrossRef]


| Ambient Temperature (°C) | Drug | SBP | (mmHg) | PI | (ms) | tB | (°C) |
|---|---|---|---|---|---|---|---|
| NT 22 ± 2 | Control | 112.81 | ±19.54 | 179.22 | ±33.22 | 38.07 | ±0.29 |
| V1a, 100 mg | 115.62 | ±12.17 | 173.74 | ±20.69 | 38.42 | ±0.10 | |
| V1a, 500 mg | 110.28 | ±15.35 | 184.77 | ±28.39 | 38.05 | ±0.10 | |
| V2, 100 mg | 119.98 | ±16.53 | 184.79 | ±38.30 | 38.54 | ±0.38 | |
| V2, 500 mg | 108.61 | ±14.79 | 176.16 | ±4.04 | 38.33 | ±0.41 | |
| HT 34 ± 2 | Control | 107.26 | ±4.19 | 188.63 | ±8.95 | 38.27 | ±0.34 |
| V1a, 100 mg | 107.90 | ±10.52 | 197.08 | ±21.63 | 38.52 | ±0.26 | |
| V1a, 500 mg | 110.40 | ±10.07 | 177.21 | ±16.34 | 38.57 | ±0.57 | |
| V2, 100 mg | 113.26 | ±15.41 | 193.14 | ±30.65 | 38.01 | ±0.37 | |
| V2, 500 mg | 114.28 | ±6.14 | 184.23 | ±12.97 | 38.33 | ±0.47 | |
| LT 12 ± 2 | Control | 115.22 | ±5.23 | 164.54 | ±24.31 | 37.51 | ±0.43 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bajic, D.; Skoric, T.; Milutinovic-Smiljanic, S.; Japundzic-Zigon, N. Voronoi Decomposition of Cardiovascular Dependency Structures in Different Ambient Conditions: An Entropy Study. Entropy 2019, 21, 1103. https://doi.org/10.3390/e21111103
Bajic D, Skoric T, Milutinovic-Smiljanic S, Japundzic-Zigon N. Voronoi Decomposition of Cardiovascular Dependency Structures in Different Ambient Conditions: An Entropy Study. Entropy. 2019; 21(11):1103. https://doi.org/10.3390/e21111103
Chicago/Turabian StyleBajic, Dragana, Tamara Skoric, Sanja Milutinovic-Smiljanic, and Nina Japundzic-Zigon. 2019. "Voronoi Decomposition of Cardiovascular Dependency Structures in Different Ambient Conditions: An Entropy Study" Entropy 21, no. 11: 1103. https://doi.org/10.3390/e21111103
APA StyleBajic, D., Skoric, T., Milutinovic-Smiljanic, S., & Japundzic-Zigon, N. (2019). Voronoi Decomposition of Cardiovascular Dependency Structures in Different Ambient Conditions: An Entropy Study. Entropy, 21(11), 1103. https://doi.org/10.3390/e21111103






