Multivariate Matching Pursuit Decomposition and Normalized Gabor Entropy for Quantification of Preictal Trends in Epilepsy
Abstract
1. Introduction
2. Materials and Methods
2.1. Univariate Matching Pursuit Decomposition (MP)
2.2. Multivariate Matching Pursuit Decomposition (MMP)
2.3. MMP-Based Gabor Measures of Complexity
3. Results
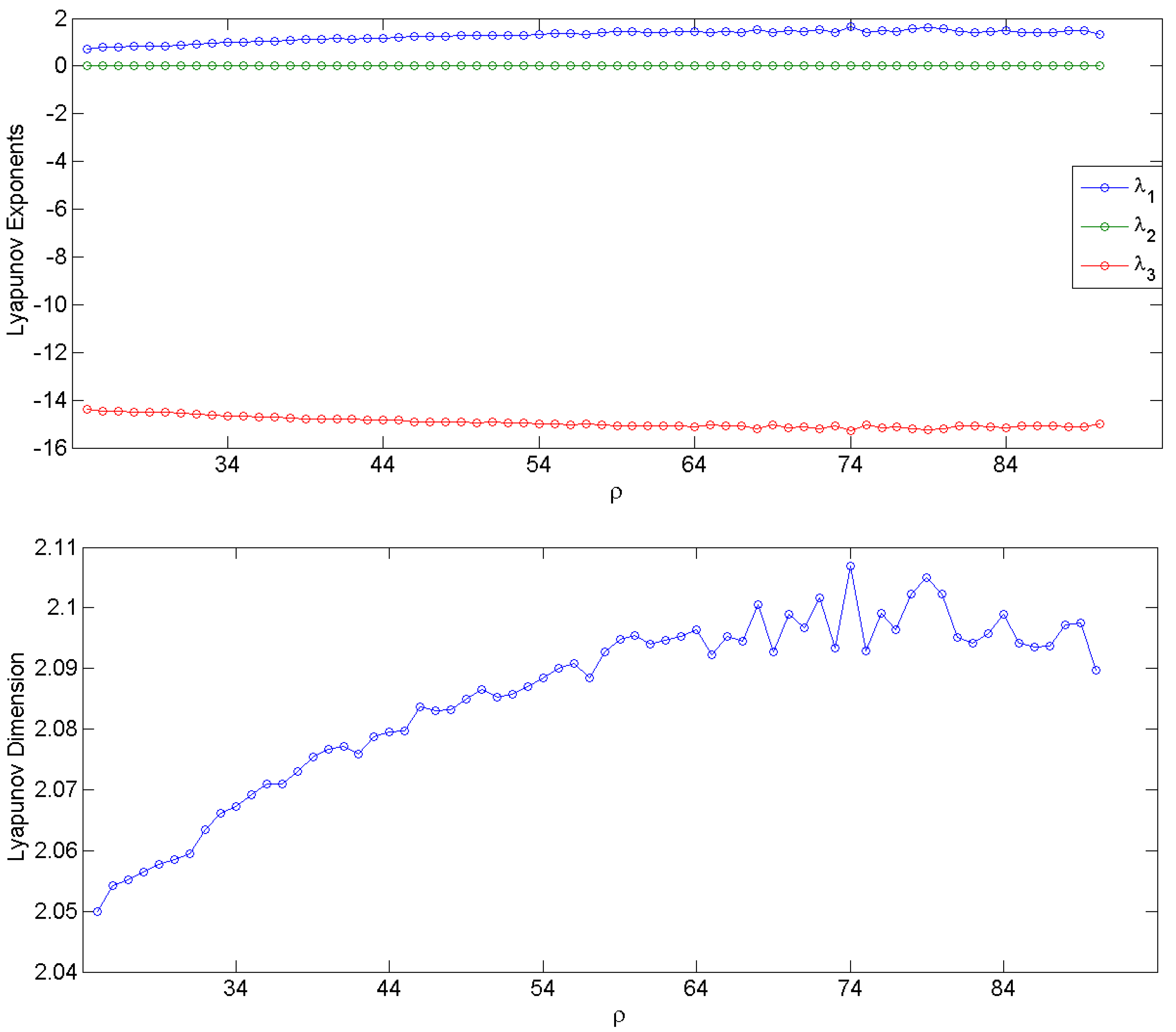
3.1. Simulation Data and Gabor Measures of Complexity
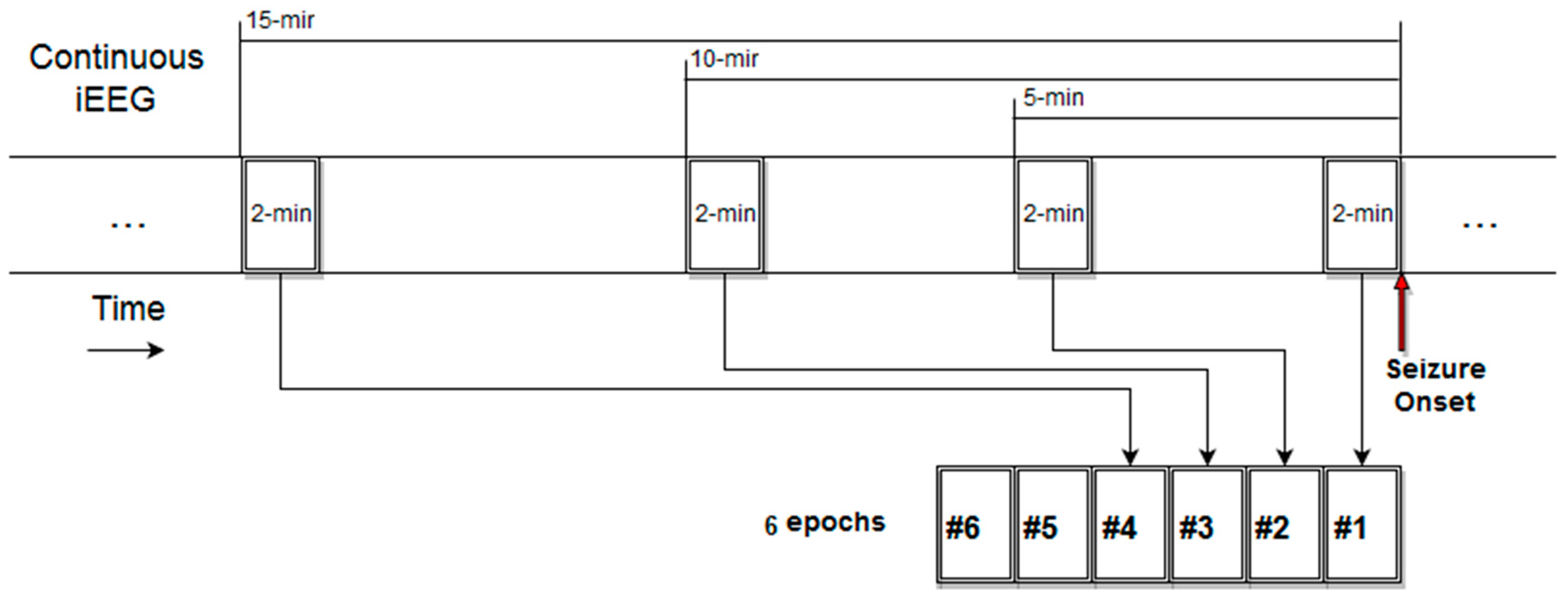
3.2. Intracranial EEG Data
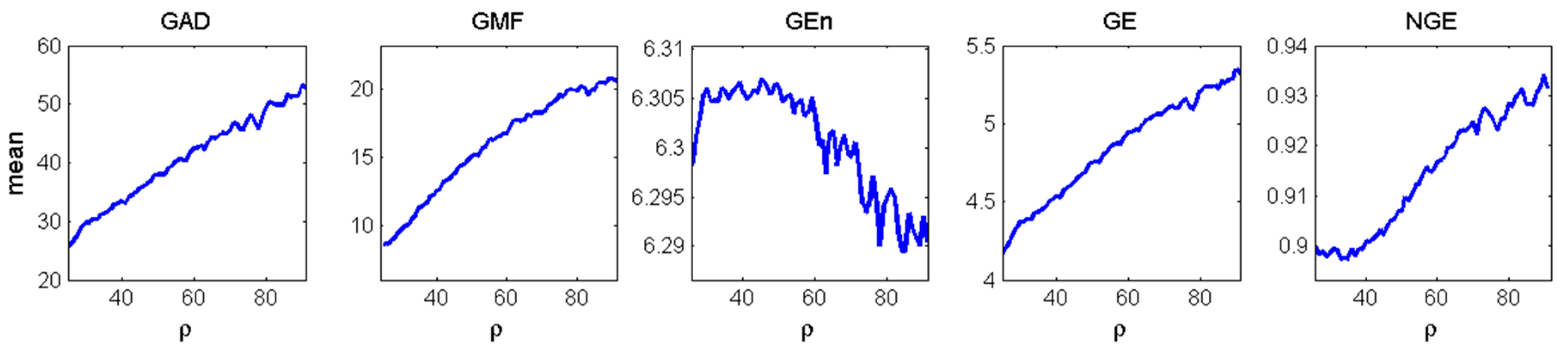
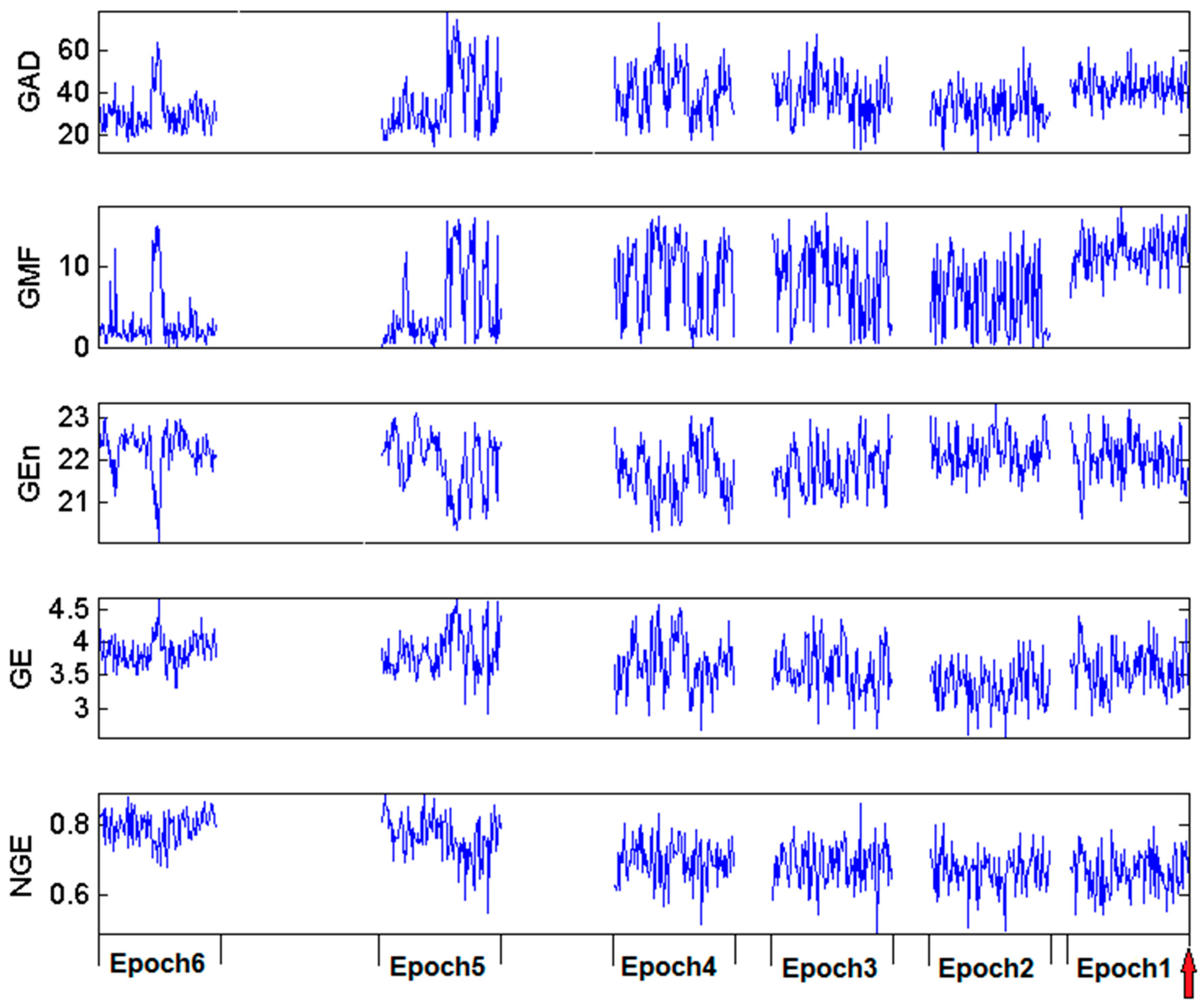
3.3. Estimation and Trend Analysis of Gabor Measures of Complexity from the EEG data
4. Discussion
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Northrop, R. Signals and Systems Analysis in Biomedical Engineering; CRC Press: Boca Raton, FL, USA, 2003. [Google Scholar]
- Bar-Yam, Y. Dynamics of Complex Systems; Perseus Books: Cambridge, MA, USA, 1997. [Google Scholar]
- Lopez-Ruiz, R.; Mancini, H.L.; Calbet, X. A statistical measure of complexity. Phys. Lett. A 1995, 209, 321–326. [Google Scholar] [CrossRef]
- Shannon, C. A mathematical theory of communication. Bell Syst. Tech. J. 1948, 27, 379–423. [Google Scholar] [CrossRef]
- Pincus, S. Approximate entropy as a measure of system complexity. Proc. Natl. Acad. Sci. USA 1991, 88, 2297–2301. [Google Scholar] [CrossRef] [PubMed]
- Maragos, P.; Sun, F. Measuring the fractal dimension of signals: Morphological covers and iterative optimization. IEEE Trans. Signal Proc. 1993, 41, 108–121. [Google Scholar] [CrossRef]
- Peng, C.; Buldyrev, S.; Havlin, S.; Simons, M.; Stanley, H.; Goldberger, A. Mosaic organization of DNA nucleotides. Phys. Rev. E 1994, 49, 1685–1689. [Google Scholar] [CrossRef]
- Ishizaki, R.; Shinba, T.; Mugishima, G.; Haraguchi, H.; Inoue, M. Time-series analysis of sleep-wake stage of rat EEG using time-dependent pattern entropy. Phys. A Stat. Mech. Appl. 2008, 387, 3145–3154. [Google Scholar] [CrossRef]
- Ivanov, P.; Amaral, L.; Goldberger, A.; Havlin, S.; Rosenblum, M.; Struzik, Z.; Stanley, H. Multifractality in human heartbeat dynamics. Nature 1999, 399, 461–465. [Google Scholar] [CrossRef] [PubMed]
- Goldberger, A.; Peng, C.; Lipsitz, L. What is physiologic complexity and how does it change with aging and disease? Neurobiol. Aging 2002, 23, 23–26. [Google Scholar] [CrossRef]
- Hornero, R.; Ortiz, T.; Lopez-Ibor, J. Analysis of brain complexity and mental disorders. Actas Esp. Psiquiatr. 2010, 38, 229–238. [Google Scholar]
- Vaillancourt, D.; Newell, K. Changing complexity in human behavior and physiology through aging and disease. Neurobiol. Aging 2002, 23, 1–11. [Google Scholar] [CrossRef]
- Fernandez, A.; Hornero, R.; Gomez, C.; Turrero, A.; Gil-Gregorio, P.; Matias-Santos, J.; Ortiz, T. Complexity analysis of spontaneous brain activity in Alzheimer disease and mild cognitive impairment: An MEG study. Alzheimer Dis. Assoc. Disord. 2010, 24, 182–189. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Tong, S.; Liu, D.; Gai, Y.; Wang, X.; Wang, J.; Qiu, Y.; Zhu, Y. Abnormal EEG complexity in patients with schizophrenia and depression. Clin. Neurophysiol. 2008, 119, 1232–1241. [Google Scholar] [CrossRef] [PubMed]
- Mallat, S.; Zhang, Z. Matching pursuits with time-frequency dictionaries. IEEE Trans. Signal Proc. 1993, 41, 3397–3415. [Google Scholar] [CrossRef]
- Franaszczuk, P.; Bergey, G.; Durka, P.; Eisenberg, H. Time frequency analysis using the matching pursuit algorithm applied to seizures originating from the mesial temporal lobe. Electroencephalogr. Clin. Neurophysiol. 1998, 106, 51–521. [Google Scholar] [CrossRef]
- Durka, P.; Zygierewicz, J.; Klekowicz, H.; Ginter, J.; Blinowska, K. On the statistical significance of event-related EEG desynchronization and synchronization in the time-frequency plane. IEEE Trans. Biomed. Eng. 2004, 51, 1167–1175. [Google Scholar] [CrossRef] [PubMed]
- Durka, P. Adaptive time-frequency parametrization of epileptic spikes. Phys. Rev. E 2004, 69, 051914. [Google Scholar] [CrossRef] [PubMed]
- Jouny, C.; Franaszczuk, P.; Bergey, G. Characterization of epileptic seizure dynamics using Gabor atom density. Clin. Neurophysiol. 2003, 114, 426–437. [Google Scholar] [CrossRef]
- Z-Flores, E.; Trujillo, L.; Sotelo, A.; Legrand, P.; Coria, L. Regularity and matching pursuit feature extraction for the detection of epileptic seizures. J. Neurosci. Methods 2016, 266, 107–125. [Google Scholar] [CrossRef] [PubMed]
- Jouny, C.; Bergey, G.; Franaszczuk, P. Partial seizures are associated with early increases in signal complexity. Clin. Neurophysiol. 2010, 121, 7–13. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jouny, C.; Bergey, G. Characterization of early partial seizure onset: Frequency, complexity and entropy. Clin. Neurophysiol. 2012, 123, 658–669. [Google Scholar] [CrossRef] [PubMed]
- Schnwald, S.; de Santa-Helena, E.; Rossatto, R.; Chaves, M.; Gerhardt, G. Benchmarking matching pursuit to find sleep spindles. J. Neurosci. Methods 2006, 156, 314–321. [Google Scholar] [CrossRef] [PubMed]
- Gribonval, R.; Bacry, E. Harmonic decomposition of audio signals with matching pursuit. IEEE Trans. Signal Proc. 2003, 51, 101–111. [Google Scholar] [CrossRef]
- Tropp, J.; Gilbert, A.; Strauss, M. Algorithms for simultaneous sparse approximation. Part I: Greedy Purs. Signal Proc. 2006, 86, 572–588. [Google Scholar]
- Barthelemy, Q.; Gouy-Pailler, C.; Isaac, Y.; Souloumiac, A.; Larue, A.; Mars, J. Multivariate temporal dictionary learning for EEG. J. Neurosci. Methods 2013, 215, 19–28. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Benar, C.; Papadopoulo, T.; Torrsani, B.; Clerc, M. Consensus matching pursuit for multi-trial EEG signals. J. Neurosci. Methods 2009, 180, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Durka, P.; Matysiak, A.; Montes, E.; Sosa, P.; Blinowska, K. Multichannel matching pursuit and EEG inverse solutions. J. Neurosci. Methods 2005, 148, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Sieluzycki, C.; Konig, R.; Matysiak, A.; Kus, R.; Ircha, D.; Durka, P. Single-trial evoked brain responses modeled by multivariate matching pursuit. IEEE Trans. Biomed. Eng. 2009, 56, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Lelic, D.; Gratkowski, M.; Hennings, K.; Drewes, A. Multichannel matching pursuit validation and clustering: A simulation and empirical study. J. Neurosci. Methods 2011, 196, 190–200. [Google Scholar] [CrossRef] [PubMed]
- Fisher, R.; Boas, W.; Blume, W.; Elger, C.; Genton, P.; Lee, P.; Engel, J. Epileptic seizures and epilepsy: Definitions proposed by the international league against epilepsy (ILAE) and the international bureau for epilepsy (IBE). Epilepsia 2005, 46, 470–472. [Google Scholar] [CrossRef] [PubMed]
- Sackellares, J.; Iasemidis, L.; Gilmore, R.; Roper, S. Epilepsy When Chaos Fails; World Scientific: Singapore, 2000. [Google Scholar]
- Iasemidis, L.; Shiau, D.S.; Sackellares, J.; Pardalos, P.; Prasad, A. Dynamical resetting of the human brain at epileptic seizures: Application of nonlinear dynamics and global optimization techniques. IEEE Trans. Biomed. Eng. 2004, 51, 493–506. [Google Scholar] [CrossRef] [PubMed]
- Sabesan, S.; Chakravarthy, N.; Tsakalis, K.; Pardalos, P.; Iasemidis, L. Measuring resetting of brain dynamics at epileptic seizures: Application of global optimization and spatial synchronization techniques. J. Comb. Optim. 2009, 17, 74–97. [Google Scholar] [CrossRef] [PubMed]
- Iasemidis, L. Seizure prediction and its applications. Neurosurg. Clin. N. Am. 2011, 22, 489–506. [Google Scholar] [CrossRef] [PubMed]
- Mormann, F.; Kreuz, T.; Andrzejak, R.; David, P.; Lehnertz, K.; Elger, C. Epileptic seizures are preceded by a decrease in synchronization. Epilepsy Res. 2003, 53, 173–185. [Google Scholar] [CrossRef]
- Le van Quyen, M.; Soss, J.; Navarro, V.; Robertson, R.; Chavez, M.; Baulac, M.; Martinerie, J. Preictal state identification by synchronization changes in long-term intracranial EEG recordings. Clin. Neurophysiol. 2005, 116, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Jiruska, P.; de Curtis, M.; Jeerys, J.; Schevon, C.; Schi, S.; Schindler, K. Synchronization and desynchronization in epilepsy: Controversies and hypotheses. J. Physiol. 2013, 591, 787–797. [Google Scholar] [CrossRef] [PubMed]
- Jouny, C.; Franaszczuk, P.; Bergey, G. Signal complexity and synchrony of epileptic seizures: Is there an identifiable preictal period? Clin. Neurophysiol. 2005, 116, 552–558. [Google Scholar] [CrossRef] [PubMed]
- Cantero, J.L.; Atienza, M.; Cruz-Vadell, A.; Suarez-Gonzalez, A.; Gil-Neciga, E. Increased synchronization and decreased neural complexity underlie thalamocortical oscillatory dynamics in mild cognitive impairment. NeuroImage 2009, 46, 938–948. [Google Scholar] [CrossRef] [PubMed]
- Martinerie, J.; Adam, C.; le van Quyen, M.; Baulac, M.; Clemenceau, S.; Renault, B.; Varela, F. Epileptic seizures can be anticipated by non-linear analysis. Nat. Med. 1998, 4, 1173–1176. [Google Scholar] [CrossRef] [PubMed]
- Jia, W.; Kong, N.; Li, F.; Gao, X.; Gao, S.; Zhang, G.; Wang, Y.; Yang, F. An epileptic seizure prediction algorithm based on second-order complexity measure. Physiol. Meas. 2005, 26, 609. [Google Scholar] [CrossRef] [PubMed]
- Spustek, T.; Jedrzejczak, W.; Blinowska, K. Matching pursuit with asymmetric functions for signal decomposition and parameterization. PLoS ONE 2015, 10, e0131007. [Google Scholar] [CrossRef] [PubMed]
- Lucena, F.; Cavalcante, A.; Takeuchi, Y.; Barros, A.; Ohnishi, N. Wavelet Entropy Measure Based on Matching Pursuit Decomposition and Its Analysis to Heartbeat Intervals; Springer: Berlin/Heidelberg, Germany, 2010; pp. 503–511. [Google Scholar]
- Liu, R.; Vlachos, I.; Karumuri, B.; Adkinson, J.; Iasemidis, L. Normalized Gabor entropy analysis of iEEG for prediction of epileptic seizures. In Proceedings of the 2016 32nd Southern Biomedical Engineering Conference (SBEC), Shreveport, LA, USA, 11–13 March 2016; pp. 15–16. [Google Scholar]
- Inouye, T.; Shinosaki, K.; Sakamoto, H.; Toi, S.; Ukai, S.; Iyama, A.; Katsuda, Y.; Hirano, M. Quantification of EEG irregularity by use of the entropy of the power spectrum. Electroencephalogr. Clin. Neurophysiol. 1991, 79, 204–210. [Google Scholar] [CrossRef]
- Rosso, O.; Blanco, S.; Yordanova, J.; Kolev, V.; Figliola, A.; Schrmann, M.; Baar, E. Wavelet entropy: A new tool for analysis of short duration brain electrical signals. J. Neurosci. Methods 2001, 105, 65–75. [Google Scholar] [CrossRef]
- Lorenz, E. Deterministic nonperiodic flow. J. Atmos. Sci. 1963, 20, 130–141. [Google Scholar] [CrossRef]
- Wolf, A.; Swift, J.; Swinney, H.; Vastano, J. Determining Lyapunov exponents from a time series. Phys. D Nonlinear Phenom. 1985, 16, 285–317. [Google Scholar] [CrossRef]
- Kaplan, J.; Yorke, J. Chaotic Behavior of Multidimensional Difference Equations; Springer: Berlin/Heidelberg, Germany, 1979; pp. 204–227. [Google Scholar]
- Moghtadaei, M.; Golpayegani, M. Complex dynamic behaviors of the complex Lorenz system. Sci. Iran. 2012, 19, 733–738. [Google Scholar] [CrossRef]
- Sprott, J.C. Chaos and Time-Series Analysis; Oxford University Press: Oxford, UK, 2003. [Google Scholar]
- Abarbanel, H. Analysis of Observed Chaotic Data, Institute for Nonlinear Science; Springer: New York, NY, USA, 1997. [Google Scholar]
- Ramgopal, S.; Thome-Souza, S.; Jackson, M.; Kadish, N.; Fernndez, I.; Klehm, J.; Bosl, W.; Reinsberger, C.; Schachter, S.; Loddenkemper, T. Seizure detection, seizure prediction, and closed-loop warning systems in epilepsy. Epilepsy Behav. 2007, 37, 291–307. [Google Scholar] [CrossRef] [PubMed]
- Lilliefors, H. On the Kolmogorov-Smirnov test for normality with mean and variance unknown. J. Am. Stat. Assoc. 1967, 62, 399–402. [Google Scholar] [CrossRef]
- Hollander, M.; Wolfe, D.; Chicken, E. Nonparametric Statistical Methods; Wiley Series in Probability and Statistics–Applied Probability and Statistics Section; Wiley: Hoboken, NJ, USA, 2015. [Google Scholar]
- Benjamini, Y.H.Y. Controlling the false discovery rate: A practical and powerful approach to multiple testing. J. R. Stat. Soc. Ser. B 1995, 57, 289–300. [Google Scholar]
- Yekutieli, D.; Benjamini, Y. Resampling-based false discovery rate controlling multiple test procedures for correlated test statistics. J. Stat. Plan. Inference 1999, 82, 171–196. [Google Scholar] [CrossRef]
- Chaovalitwongse, W.; Iasemidis, L.; Pardalos, P.; Carney, P.; Shiau, D.; Sackellares, J. Performance of a seizure warning algorithm based on the dynamics of intracranial EEG. Epilepsy Res. 2005, 64, 93–113. [Google Scholar] [CrossRef] [PubMed]
- Iasemidis, L.; Shiau, D.; Chaovalitwongse, W.; Sackellares, J.; Pardalos, P.; Principe, J.; Carney, P.; Prasad, A.; Veeramani, B.; Tsakalis, K. Adaptive epileptic seizure prediction system. IEEE Trans. Biomed. Eng. 2003, 50, 616–627. [Google Scholar] [CrossRef] [PubMed]
- Iasemidis, L. Epileptic seizure prediction and control. IEEE Trans. Biomed. Eng. 2003, 50, 549–558. [Google Scholar] [CrossRef] [PubMed]
- Iasemidis, L.; Pardalos, P.; Shiau, D.; Chaovalitwongse, W.; Narayanan, K.; Kumar, S.; Carney, P.; Sackellares, J. Prediction of human epileptic seizures based on optimization and phase changes of brain electrical activity. Optim. Methods Softw. 2003, 18, 81–104. [Google Scholar] [CrossRef]
- Gadhoumi, K.; Lina, J.-M.; Mormann, F.; Gotman, J. Seizure prediction for therapeutic devices: A review. J. Neurosci. Methods 2016, 260, 270–282. [Google Scholar] [CrossRef] [PubMed]
- Acharya, U.R.; Fujita, H.; Sudarshan, V.K.; Bhat, S.; Koh, J.E.W. Application of entropies for automated diagnosis of epilepsy using EEG. Knowl.-Based Syst. 2015, 88, 85–96. [Google Scholar] [CrossRef]

| Patient | Gender | # Recording Electrodes | Available iEEG Duration (hours) | Number of Isolated Clinical Seizures |
|---|---|---|---|---|
| 1 | F | 40 | 34.67 | 4 |
| 2 | M | 28 | 281.68 | 6 |
| 3 | F | 28 | 86.3 | 14 |
| 4 | M | 28 | 334.62 | 7 |
| 5 | M | 28 | 85.02 | 3 |
| 6 | M | 28 | 156.22 | 2 |
| 7 | M | 28 | 145.77 | 3 |
| 8 | F | 28 | 18.77 | 3 |
| Measure | Model | FDR Adjusted p-Value | |
|---|---|---|---|
| GAD | m3 | 0.0092 | 1 |
| GMF | m3 | 0.0032 | 1 |
| GEn | m3 | 1 | |
| GE | m3 | 1 | |
| NGE | m3 | 1 |
| Complexity Measure | GAD | GMF | GEn | GE | NGE |
|---|---|---|---|---|---|
| Statistic of Measure | Optimized Model for Trend Identification of Statistic across Epochs | ||||
| m1 | m3 | m1 | m1 | m1 | |
| m1 | m3 | m2 | m1 | m2 | |
| Patient | P1 (α1) | P2 (α2) | P3 (α3) | P4 (α4) | P5 (α5) | P6 (α6) | P7 (α7) | P8 (α8) | FDR Adjusted p-Value for Significance | |
|---|---|---|---|---|---|---|---|---|---|---|
| Statistic (Measure) | ||||||||||
| (GAD) | 0.0242 | −0.0102 | 0.479 | −0.0173 | −0.0097 | −0.0213 | −0.0215 | 0.1185 | 0.0138 | 0.7266 |
| (GAD) | 0.031 | 0.0241 | 0.0248 | 0.0424 | 0.0501 | 0.0467 | 0.0237 | 0.0588 | 0.0316 | 0.0206 |
| (GMF) | 0.0177 | 0.0089 | 0.0209 | −0.0031 | −0.0047 | −0.0017 | −0.0079 | 0.052 | 0.0103 | 0.2359 |
| (GMF) | 0.0076 | 0.0042 | 0.0025 | 0.003 | 0.0036 | 0.0013 | 0.0026 | 0.0243 | 0.0061 | 0.0195 |
| (GEn) | −0.0009 | 0.0063 | −0.0035 | 0.0088 | 0.0038 | 0.0023 | 0.0126 | −0.0018 | 0.0034 | 0.1773 |
| (GEn) | 0.0013 | 0.0034 | 0.0014 | 0.002 | 0.0024 | 0.0005 | 0.0062 | 0.0055 | 0.0028 | 0.0195 |
| (GE) | −0.0018 | −0.0082 | 0.0141 | −0.0147 | −0.0035 | −0.019 | −0.0162 | 0.0252 | −0.003 | 0.6645 |
| (GE) | 0.0471 | 0.0201 | 0.021 | 0.0777 | 0.0371 | 0.0652 | 0.06 | 0.1004 | 0.0536 | 0.0097 |
| (NGE) | −0.0077 | −0.0074 | −0.001 | −0.0091 | 0 | −0.0117 | −0.0098 | −0.0176 | −0.008 | 0.0195 |
| (NGE) | 0.0017 | 0.0014 | −0.0004 | 0.0027 | 0.0024 | 0.0039 | 0.004 | −0.001 | 0.0018 | 0.0409 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, R.; Karumuri, B.; Adkinson, J.; Hutson, T.N.; Vlachos, I.; Iasemidis, L. Multivariate Matching Pursuit Decomposition and Normalized Gabor Entropy for Quantification of Preictal Trends in Epilepsy. Entropy 2018, 20, 419. https://doi.org/10.3390/e20060419
Liu R, Karumuri B, Adkinson J, Hutson TN, Vlachos I, Iasemidis L. Multivariate Matching Pursuit Decomposition and Normalized Gabor Entropy for Quantification of Preictal Trends in Epilepsy. Entropy. 2018; 20(6):419. https://doi.org/10.3390/e20060419
Chicago/Turabian StyleLiu, Rui, Bharat Karumuri, Joshua Adkinson, Timothy Noah Hutson, Ioannis Vlachos, and Leon Iasemidis. 2018. "Multivariate Matching Pursuit Decomposition and Normalized Gabor Entropy for Quantification of Preictal Trends in Epilepsy" Entropy 20, no. 6: 419. https://doi.org/10.3390/e20060419
APA StyleLiu, R., Karumuri, B., Adkinson, J., Hutson, T. N., Vlachos, I., & Iasemidis, L. (2018). Multivariate Matching Pursuit Decomposition and Normalized Gabor Entropy for Quantification of Preictal Trends in Epilepsy. Entropy, 20(6), 419. https://doi.org/10.3390/e20060419






