Characterizing Normal and Pathological Gait through Permutation Entropy
Abstract
:1. Introduction
2. Results
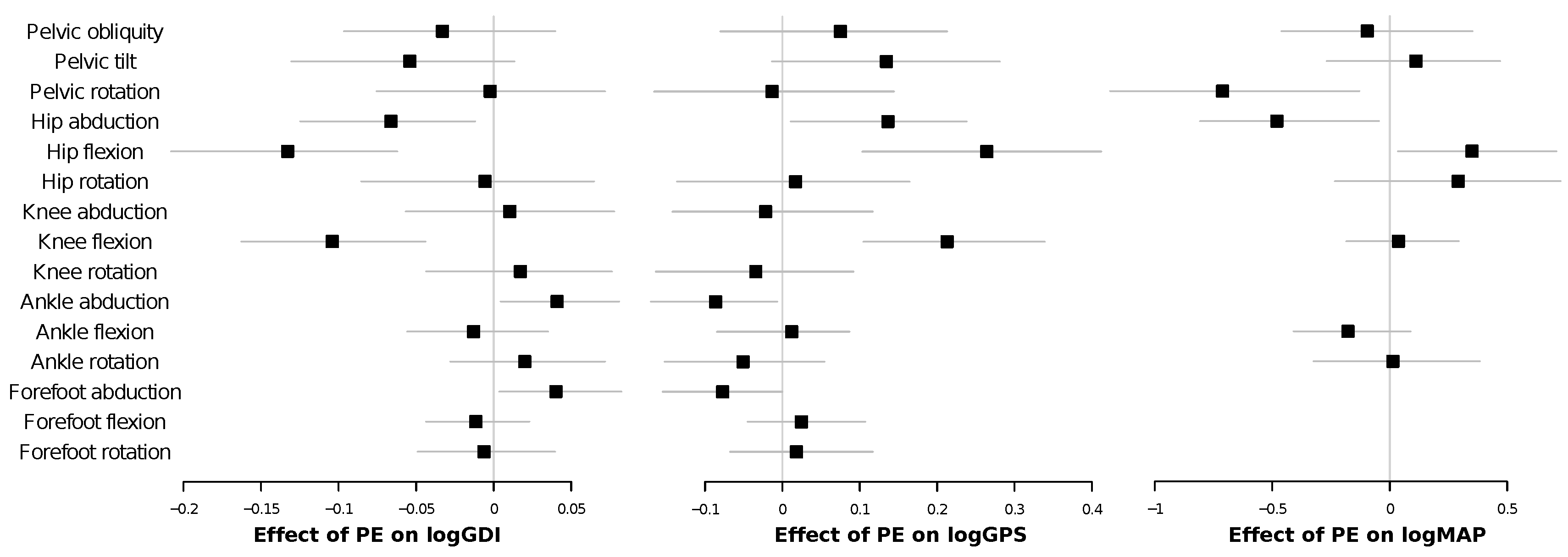
2.1. Gait Permutation Entropy: Single-Scale
2.2. Gait Permutation Entropy: Multi-Scale
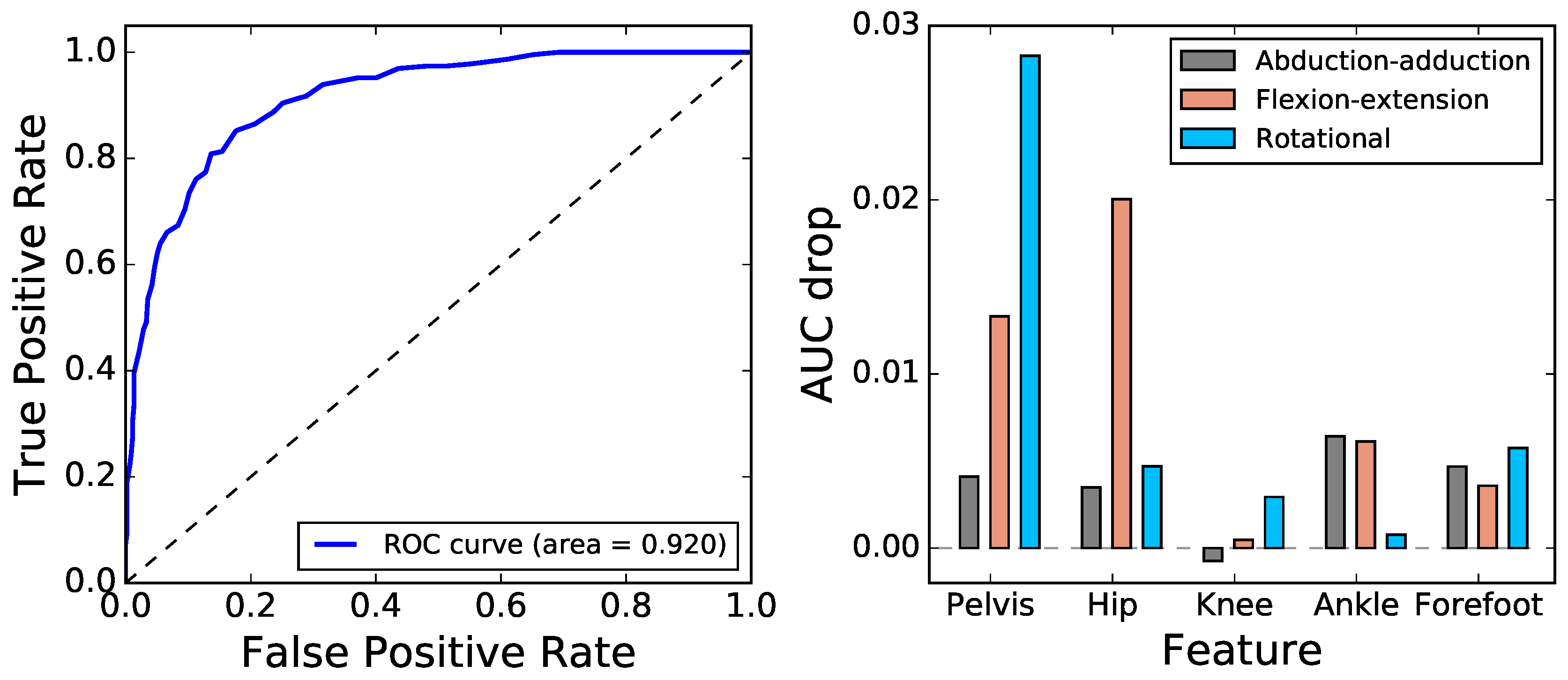
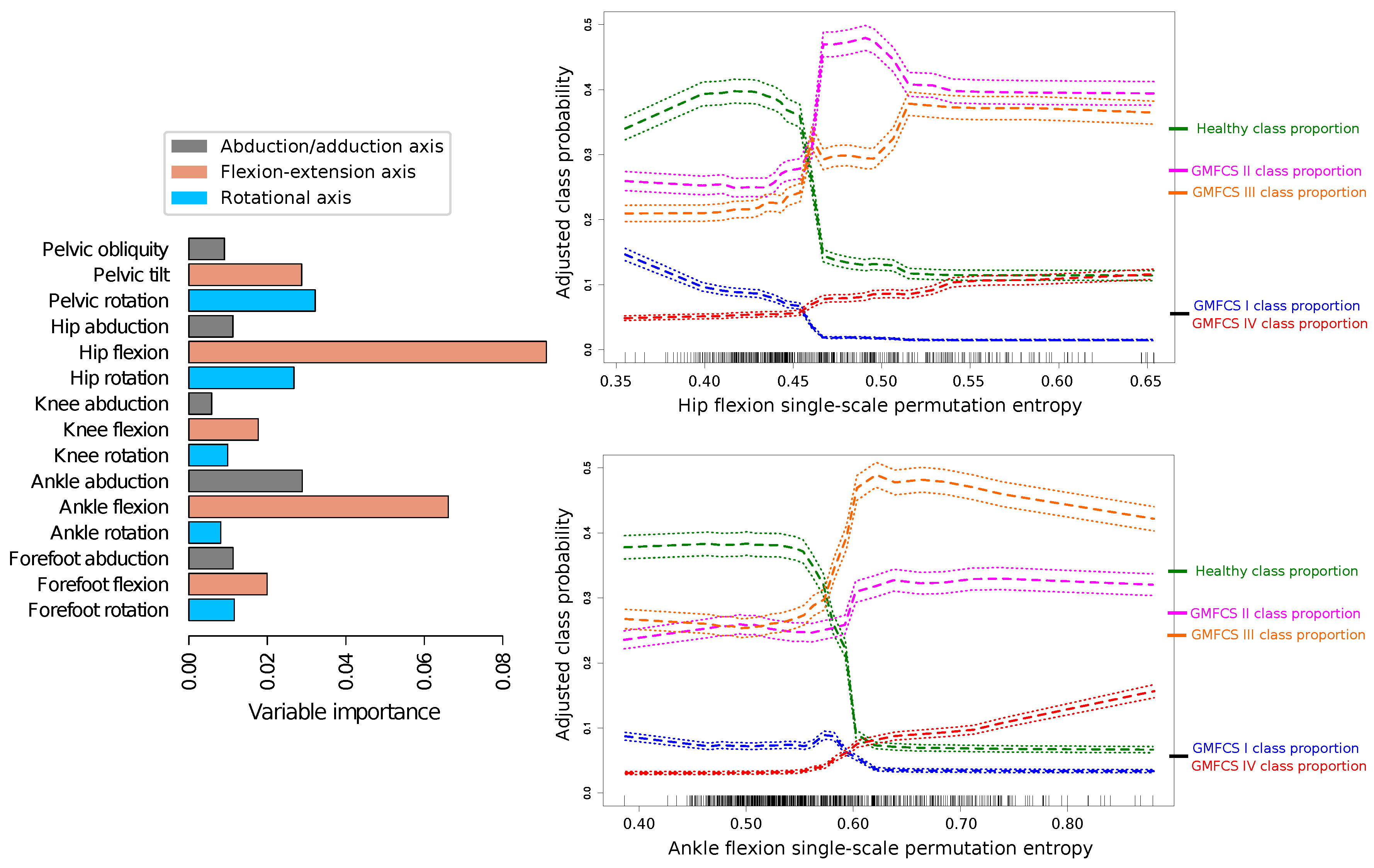
2.3. Gait Entropy in Classification Tasks
3. Discussion and Conclusions
4. Materials and Methods
4.1. The Gait Dataset
4.1.1. Participants
4.1.2. Clinical and 3D-Gait Analysis
4.2. Permutation Entropy Analysis
4.2.1. Single-Scale Entropy
4.2.2. Multi-Scale Entropy
4.3. Linear Mixed Models
4.4. Classification Task
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CFCS | Communication Function Classification System, scale used to classify the effectiveness of everyday communication of an impaired individual, and specifically of cerebral palsy patients [36]. |
| CP | Cerebral Palsy. |
| GDI | Gait Deviation Index [27]. |
| GMFCS | Gross Motor Function Classification System, scale describing the impairment of patients based on everyday movements such as sitting and walking [18]. |
| GPS | Gait Profile Score [28]. |
| IGA | Instrumental Gait Analysis. |
| MACS | Manual Ability Classification System, scale assessing the ability of CP children to handle objects in everyday activities [37]. |
| MAP | Movement Analysis Profile [28]. |
| PE | Permutation Entropy [12,13]. |
| Tanner | Scale of physical development in children and adolescents [25]. |
References
- Colver, A.; Fairhurst, C.; Pharoah, P.O. Cerebral palsy. Lancet 2014, 383, 1240–1249. [Google Scholar] [CrossRef]
- Richards, C.L.; Malouin, F. Cerebral palsy: Definition, assessment and rehabilitation. Handb. Clin. Neurol. 2013, 111, 183–195. [Google Scholar] [PubMed]
- Shumway-Cook, A.; Woollacott, M.H. Motor Control: Translating Research into Clinical Practice; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2007. [Google Scholar]
- Marret, S.; Vanhulle, C.; Laquerriere, A. Pathophysiology of cerebral palsy. Handb. Clin. Neurol. 2013, 111, 169–176. [Google Scholar] [PubMed]
- Gage, J.R.; Schwartz, M.H.; Koop, S.E.; Novacheck, T.F. The Identification and Treatment of Gait Problems in Cerebral Palsy; John Wiley & Sons: Hoboken, NJ, USA, 2009; Volume 4. [Google Scholar]
- Riley, M.A.; Turvey, M.T. Variability and determinism in motor behavior. J. Motor Behav. 2002, 34, 99–125. [Google Scholar] [CrossRef] [PubMed]
- Stergiou, N.; Decker, L.M. Human movement variability, nonlinear dynamics, and pathology: Is there a connection? Hum. Mov. Sci. 2011, 30, 869–888. [Google Scholar] [CrossRef] [PubMed]
- Gavrishchaka, V.; Senyukova, O.; Davis, K. Multi-complexity ensemble measures for gait time series analysis: Application to diagnostics, monitoring and biometrics. In Signal and Image Analysis for Biomedical and Life Sciences; Springer: Berlin, Germany, 2015; pp. 107–126. [Google Scholar]
- Harbourne, R.T.; Stergiou, N. Movement variability and the use of nonlinear tools: Principles to guide physical therapist practice. Phys. Ther. 2009, 89, 267–282. [Google Scholar] [CrossRef] [PubMed]
- Arpin, D.J.; Stuberg, W.; Stergiou, N.; Kurz, M.J. Motor control of the lower extremity musculature in children with cerebral palsy. Res. Dev. Disabil. 2013, 34, 1134–1143. [Google Scholar]
- Tao, W.; Zhang, X.; Chen, X.; Wu, D.; Zhou, P. Multi-scale complexity analysis of muscle coactivation during gait in children with cerebral palsy. Front. Hum. Neurosc. 2015, 9, 367. [Google Scholar] [CrossRef] [PubMed]
- Bandt, C.; Pompe, B. Permutation entropy: A natural complexity measure for time series. Phys. Rev. Lett. 2002, 88, 174102. [Google Scholar] [CrossRef] [PubMed]
- Zanin, M.; Zunino, L.; Rosso, O.A.; Papo, D. Permutation entropy and its main biomedical and econophysics applications: A review. Entropy 2012, 14, 1553–1577. [Google Scholar] [CrossRef]
- Khandoker, A.H.; Palaniswami, M.; Begg, R.K. A comparative study on approximate entropy measure and poincaré plot indexes of minimum foot clearance variability in the elderly during walking. J. Neuroeng. Rehabilit. 2008, 5, 4. [Google Scholar] [CrossRef] [PubMed]
- Kurz, M.J.; Hou, J.G. Levodopa influences the regularity of the ankle joint kinematics in individuals with Parkinson’s disease. J. Comput. Neurosci. 2010, 28, 131–136. [Google Scholar] [CrossRef] [PubMed]
- Decker, L.M.; Cignetti, F.; Stergiou, N. Wearing a safety harness during treadmill walking influences lower extremity kinematics mainly through changes in ankle regularity and local stability. J. Neuroeng. Rehabilit. 2012, 9, 8. [Google Scholar] [CrossRef] [PubMed]
- Hillen, B.K.; Yamaguchi, G.T.; Abbas, J.J.; Jung, R. Joint-specific changes in locomotor complexity in the absence of muscle atrophy following incomplete spinal cord injury. J. Neuroeng. Rehabilit. 2013, 10, 97. [Google Scholar] [CrossRef] [PubMed]
- Palisano, R.; Rosenbaum, P.; Walter, S.; Russell, D.; Wood, E.; Galuppi, B. Development and reliability of a system to classify gross motor function in children with cerebral palsy. Dev. Med. Child Neurol. 1997, 39, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Hintze, J.L.; Nelson, R.D. Violin Plots: A Box Plot-Density Trace Synergism. Am. Stat. 1998, 52, 181–184. [Google Scholar]
- Šidák, Z. Rectangular confidence regions for the means of multivariate normal distributions. J. Am. Stat. Assoc. 1967, 62, 626–633. [Google Scholar]
- Amigó, J.M.; Zambrano, S.; Sanjuán, M.A. True and false forbidden patterns in deterministic and random dynamics. EPL Europhys. Lett. 2007, 79, 50001. [Google Scholar] [CrossRef]
- Zanin, M.; Papo, D.; Sousa, P.A.; Menasalvas, E.; Nicchi, A.; Kubik, E.; Boccaletti, S. Combining complex networks and data mining: Why and how. Phys. Rep. 2016, 635, 1–44. [Google Scholar] [CrossRef]
- Schwartz, M.H.; Rozumalski, A.; Steele, K.M. Dynamic motor control is associated with treatment outcomes for children with cerebral palsy. Dev. Med. Child Neurol. 2016, 58, 1139–1145. [Google Scholar] [CrossRef] [PubMed]
- Gracies, J.M. Pathophysiology of spastic paresis. II: Emergence of muscle overactivity. Muscle Nerve 2005, 31, 552–571. [Google Scholar] [PubMed]
- Tanner, J.M. Growth at Adolescence; Blackwell Scientific Publications: Oxford, UK, 1962. [Google Scholar]
- Pulido-Valdeolivas, I.; Gómez-Andrés, D.; Martín-Gonzalo, J.; López-López, J.; Gómez-Barrena, E.; Hernández, J.S.; Rausell, E. Gait parameters in a reference sample of healthy Spanish schoolchildren: Multivariate descriptive statistics and asymmetries observed in left and right cycles. Neurología 2013, 28, 145–152. [Google Scholar] [PubMed]
- Schwartz, M.H.; Rozumalski, A. The gait deviation index: A new comprehensive index of gait pathology. Gait Posture 2008, 28, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Baker, R.; McGinley, J.L.; Schwartz, M.H.; Beynon, S.; Rozumalski, A.; Graham, H.K.; Tirosh, O. The Gait Profile Score and Movement Analysis Profile. Gait Posture 2009, 30, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.; Goldberger, A.L.; Peng, C.K. Multiscale entropy analysis of complex physiologic time series. Phys. Rev. Lett. 2002, 89, 068102. [Google Scholar]
- Li, D.; Li, X.; Liang, Z.; Voss, L.J.; Sleigh, J.W. Multiscale permutation entropy analysis of EEG recordings during sevoflurane anesthesia. J. Neural Eng. 2010, 7, 046010. [Google Scholar] [CrossRef] [PubMed]
- Ho, T.K. Random decision forests. In Proceedings of the Third International Conference on Document Analysis and Recognition, Quebec, QC, USA, 14–16 August 1995; Volume 1, pp. 278–282. [Google Scholar]
- Ho, T.K. The random subspace method for constructing decision forests. IEEE Trans. Pattern Anal. Mach. Intell. 1998, 20, 832–844. [Google Scholar]
- Pedregosa, F.; Varoquaux, G.; Gramfort, A.; Michel, V.; Thirion, B.; Grisel, O.; Blondel, M.; Prettenhofer, P.; Weiss, R.; Dubourg, V.; et al. Scikit-learn: Machine Learning in Python. J. Mach. Learn. Res. 2011, 12, 2825–2830. [Google Scholar]
- Ishwaran, H.; Kogalur, U. Random Forests for Survival, Regression, and Classification (RF-SRC), R Package Version 2.5.1. Available online: https://cran.r-project.org/package=randomForestSRC (accessed on 18 January 2018).
- Kohavi, R. A study of cross-validation and bootstrap for accuracy estimation and model selection. In Proceedings of the 14th international joint conference on Artificial intelligence, Montreal, QC, Canada, 20–25 August 1995; Volume 14, pp. 1137–1145. [Google Scholar]
- Hidecker, M.J.C.; Paneth, N.; Rosenbaum, P.L.; Kent, R.D.; Lillie, J.; Eulenberg, J.B.; CHESTER, J.; Johnson, B.; Michalsen, L.; Evatt, M.; et al. Developing and validating the Communication Function Classification System for individuals with cerebral palsy. Dev. Med. Child Neurol. 2011, 53, 704–710. [Google Scholar] [CrossRef] [PubMed]
- Eliasson, A.C.; Krumlinde-Sundholm, L.; Rösblad, B.; Beckung, E.; Arner, M.; Öhrvall, A.M.; Rosenbaum, P. The Manual Ability Classification System (MACS) for children with cerebral palsy: Scale development and evidence of validity and reliability. Dev. Med. Child Neurol. 2006, 48, 549–554. [Google Scholar] [CrossRef] [PubMed]
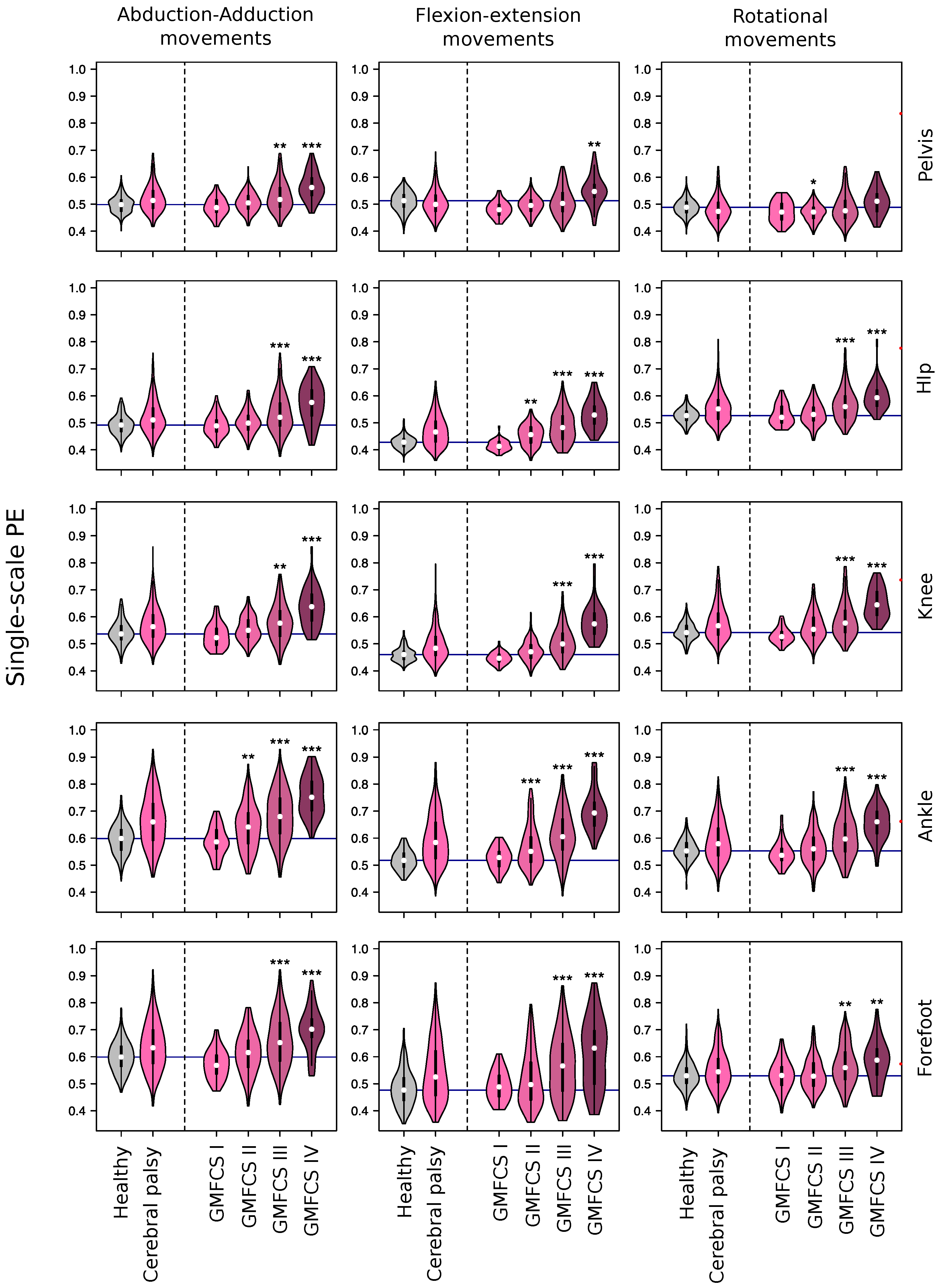
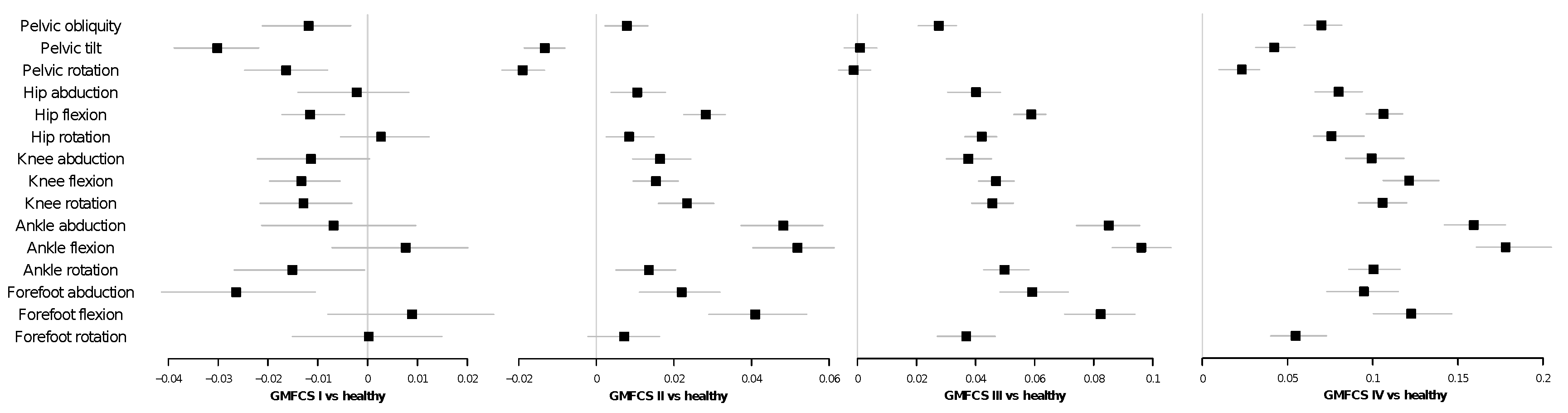
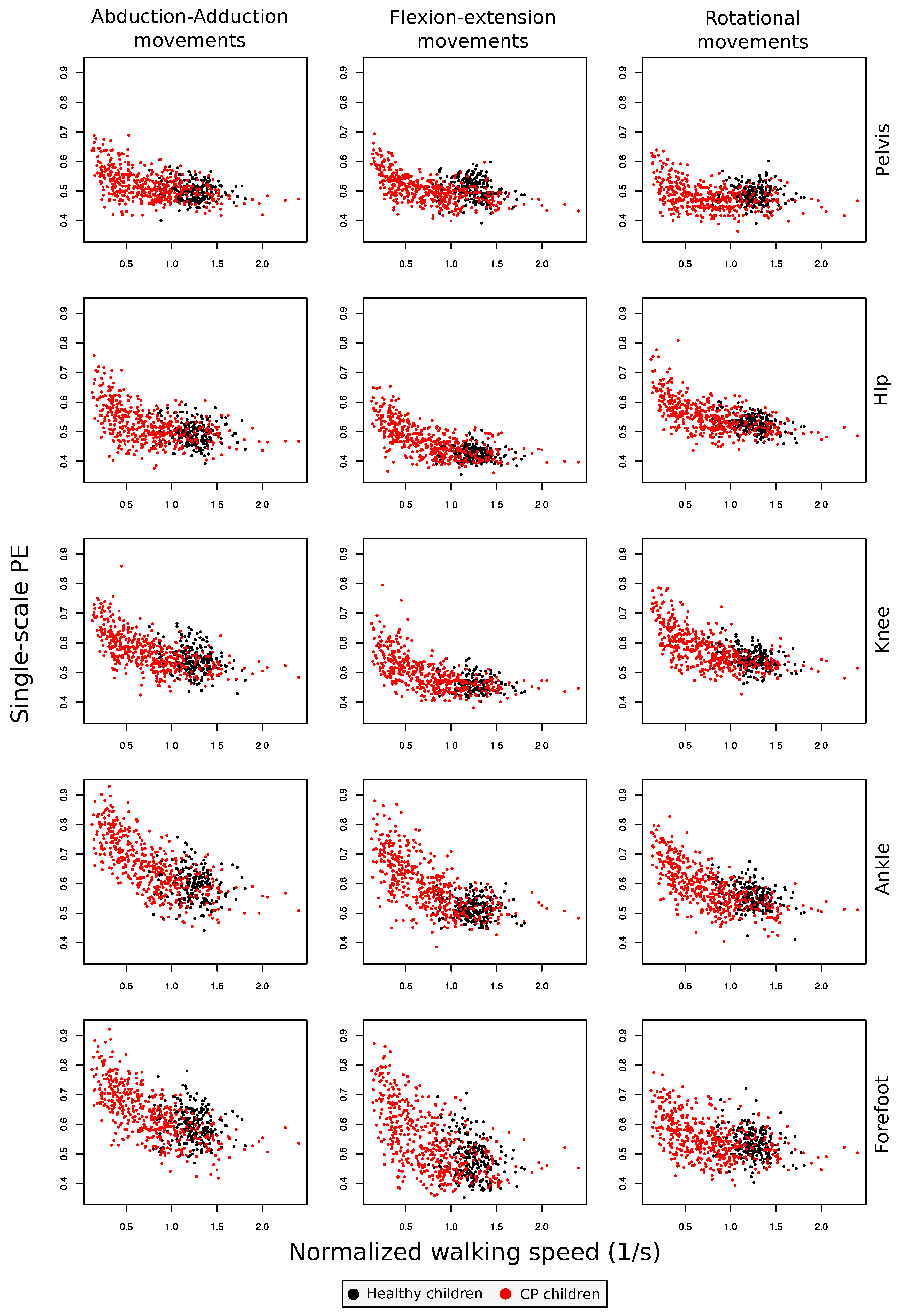
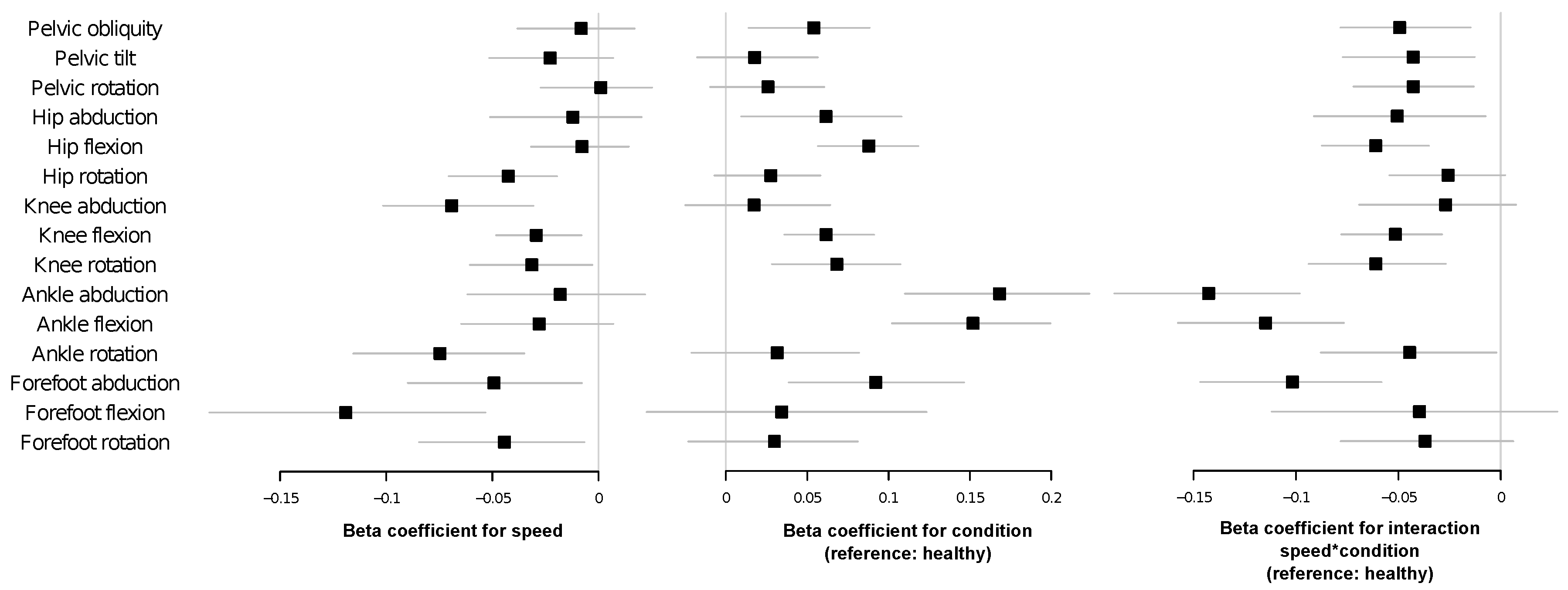
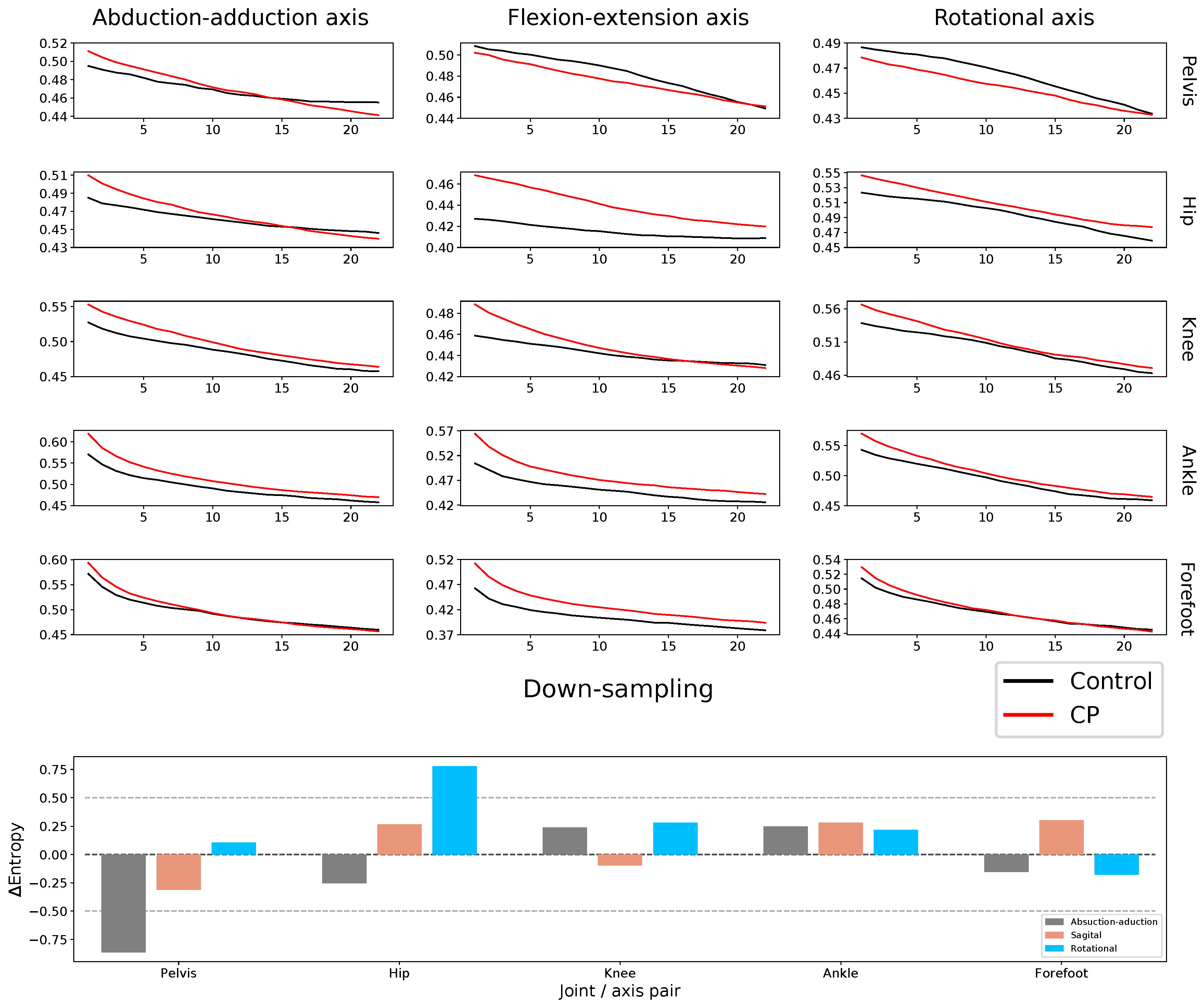
| Abduction-Adduction Axis | Sagittal Axis | Rotational Axis | |
|---|---|---|---|
| Pelvis | |||
| Hip | |||
| Knee | |||
| Ankle | |||
| Forefoot |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zanin, M.; Gómez-Andrés, D.; Pulido-Valdeolivas, I.; Martín-Gonzalo, J.A.; López-López, J.; Pascual-Pascual, S.I.; Rausell, E. Characterizing Normal and Pathological Gait through Permutation Entropy. Entropy 2018, 20, 77. https://doi.org/10.3390/e20010077
Zanin M, Gómez-Andrés D, Pulido-Valdeolivas I, Martín-Gonzalo JA, López-López J, Pascual-Pascual SI, Rausell E. Characterizing Normal and Pathological Gait through Permutation Entropy. Entropy. 2018; 20(1):77. https://doi.org/10.3390/e20010077
Chicago/Turabian StyleZanin, Massimiliano, David Gómez-Andrés, Irene Pulido-Valdeolivas, Juan Andrés Martín-Gonzalo, Javier López-López, Samuel Ignacio Pascual-Pascual, and Estrella Rausell. 2018. "Characterizing Normal and Pathological Gait through Permutation Entropy" Entropy 20, no. 1: 77. https://doi.org/10.3390/e20010077
APA StyleZanin, M., Gómez-Andrés, D., Pulido-Valdeolivas, I., Martín-Gonzalo, J. A., López-López, J., Pascual-Pascual, S. I., & Rausell, E. (2018). Characterizing Normal and Pathological Gait through Permutation Entropy. Entropy, 20(1), 77. https://doi.org/10.3390/e20010077







