Non-Equilibrium Thermodynamic Analysis of Double Diffusive, Nanofluid Forced Convection in Catalytic Microreactors with Radiation Effects
Abstract
:1. Introduction
2. Theoretical methods
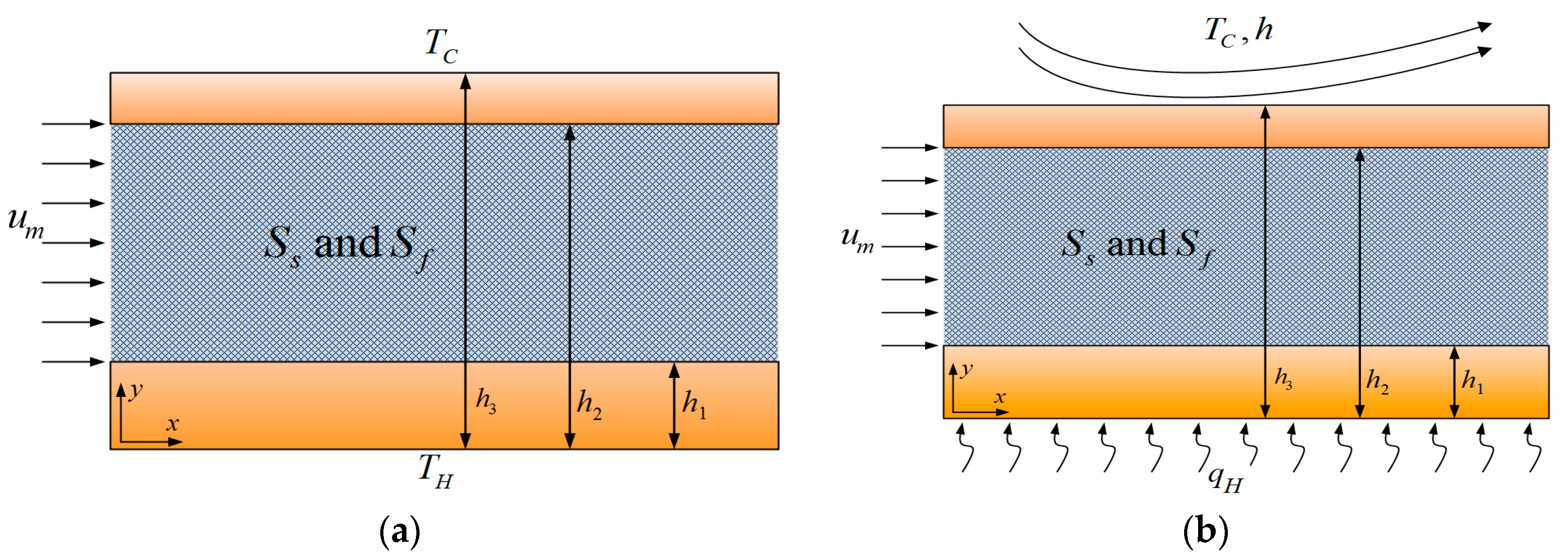
2.1. Problem Configuration and Assumptions
- The porous medium is homogenous and isotropic, fluid saturated and includes uniform and steady internal heat generation representing heat of reaction and/or absorption of electromagnetic waves.
- The fluid flow is laminar, steady and incompressible, with uniform heat generation.
- A local thermal non-equilibrium condition has been considered within the porous section of the microreactor.
- Fully developed conditions hold within the microreactor.
- It is assumed that the temperature of the solid phase of the porous medium is high enough to include the effect of radiation on the temperature distribution [32].
2.2. Governing Equations
- Case one:
- Case two:
3. Dimensionless Parameters and Non-Dimensionalised Equations
- Case one:
- Case two:
4. Solution of Momentum, Energy and Dispersion Equations
5. Results and Discussion
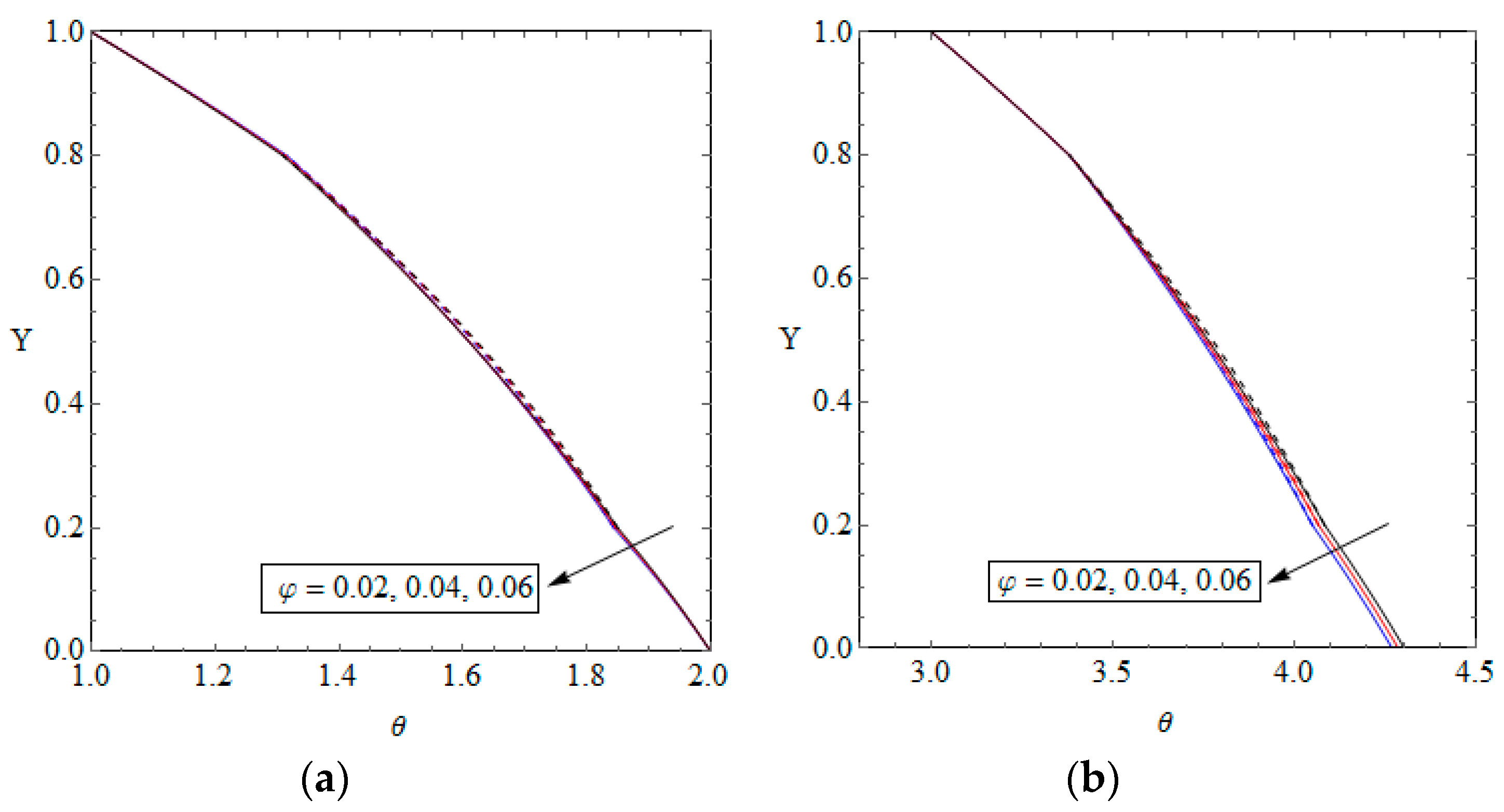
5.1. Validation
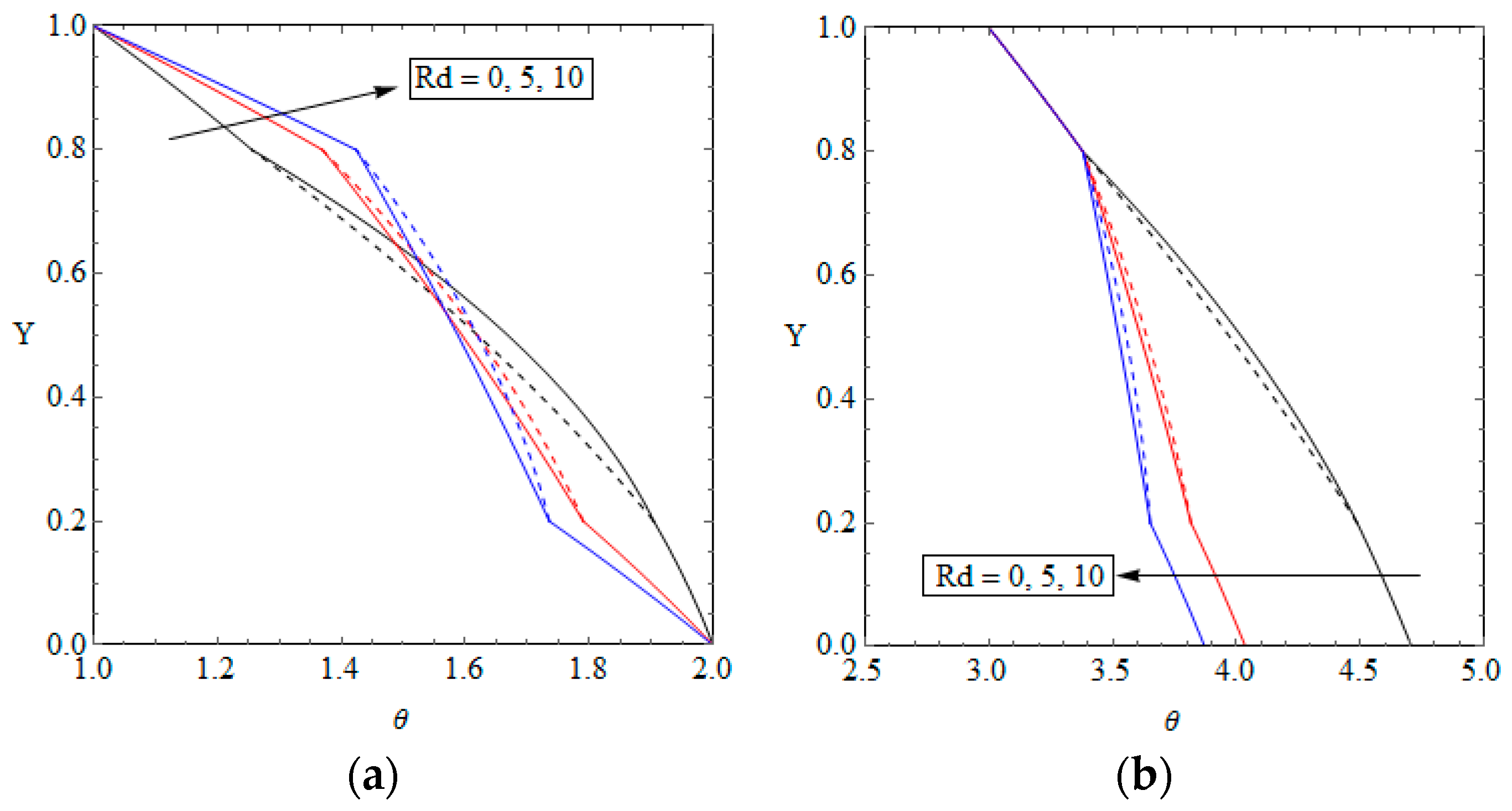
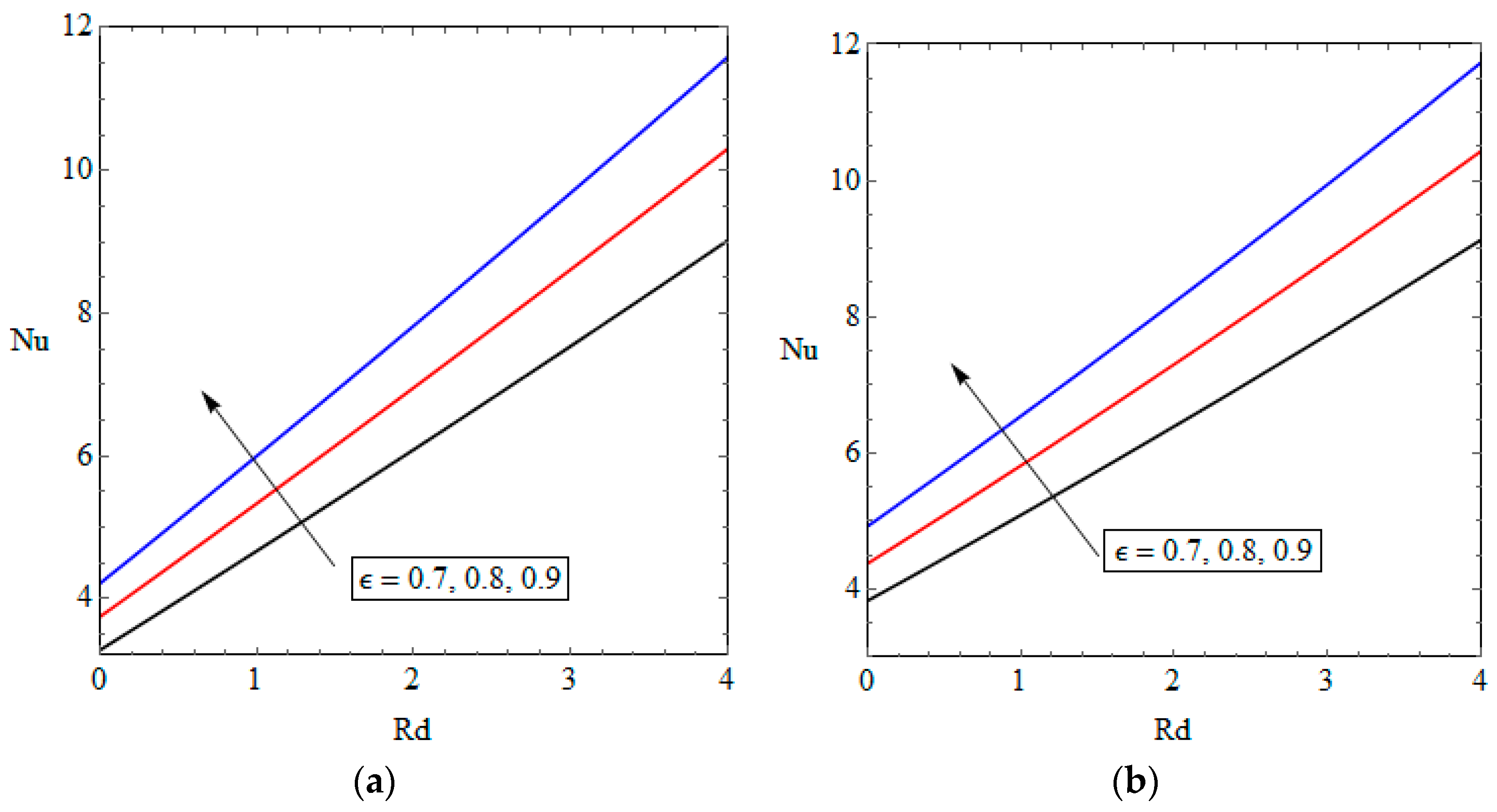
5.2. Temperature Distribution and Nusselt Number
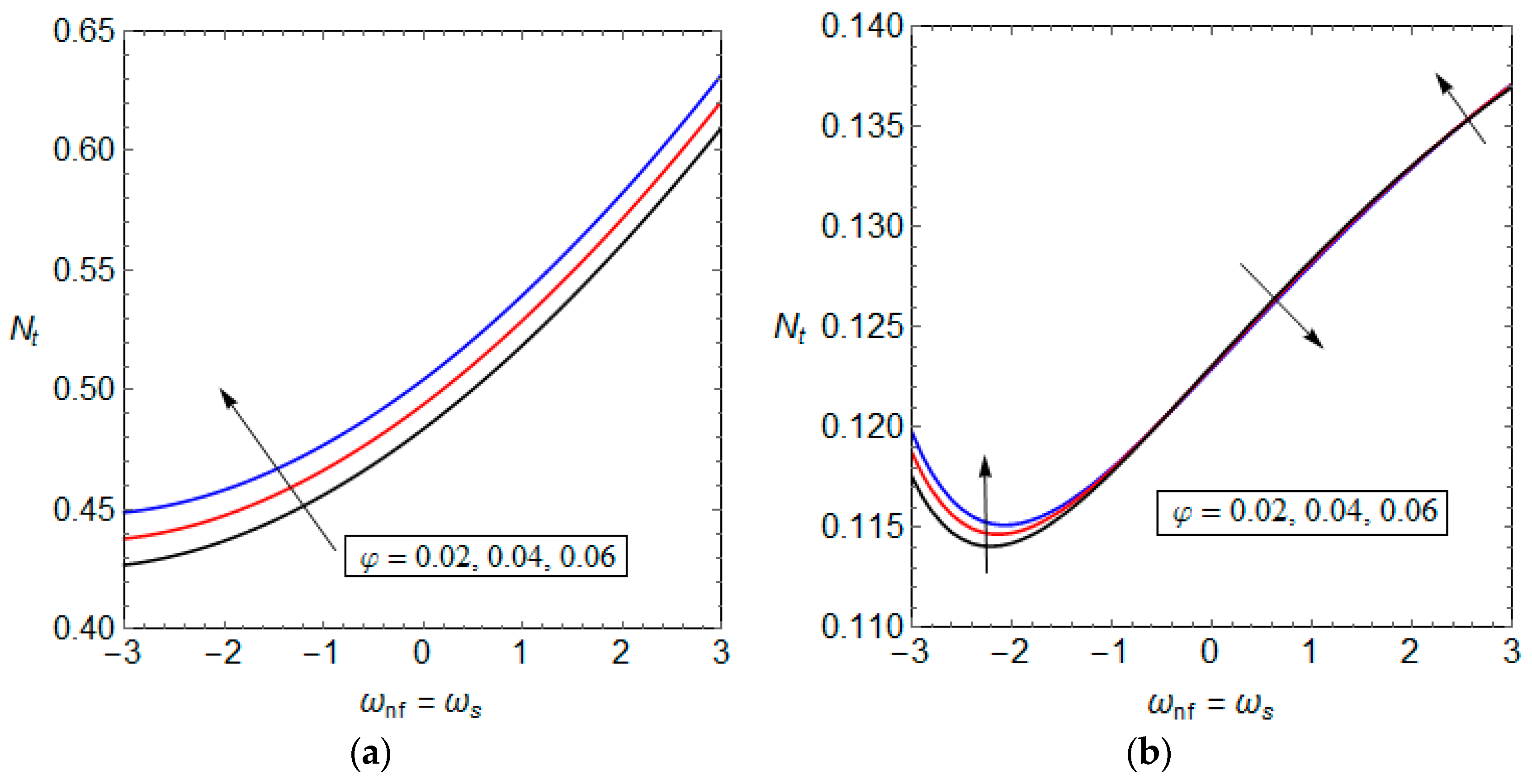
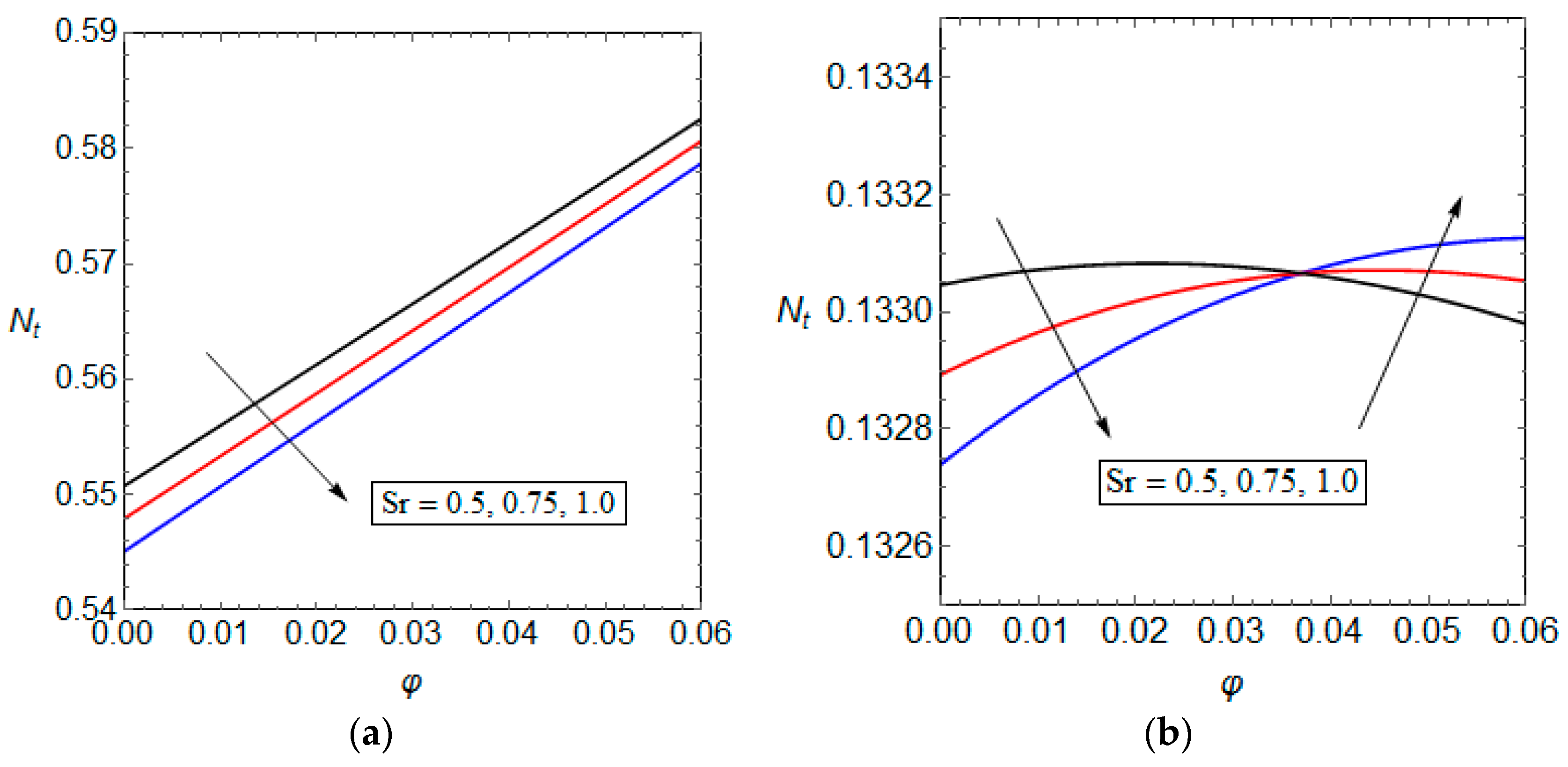
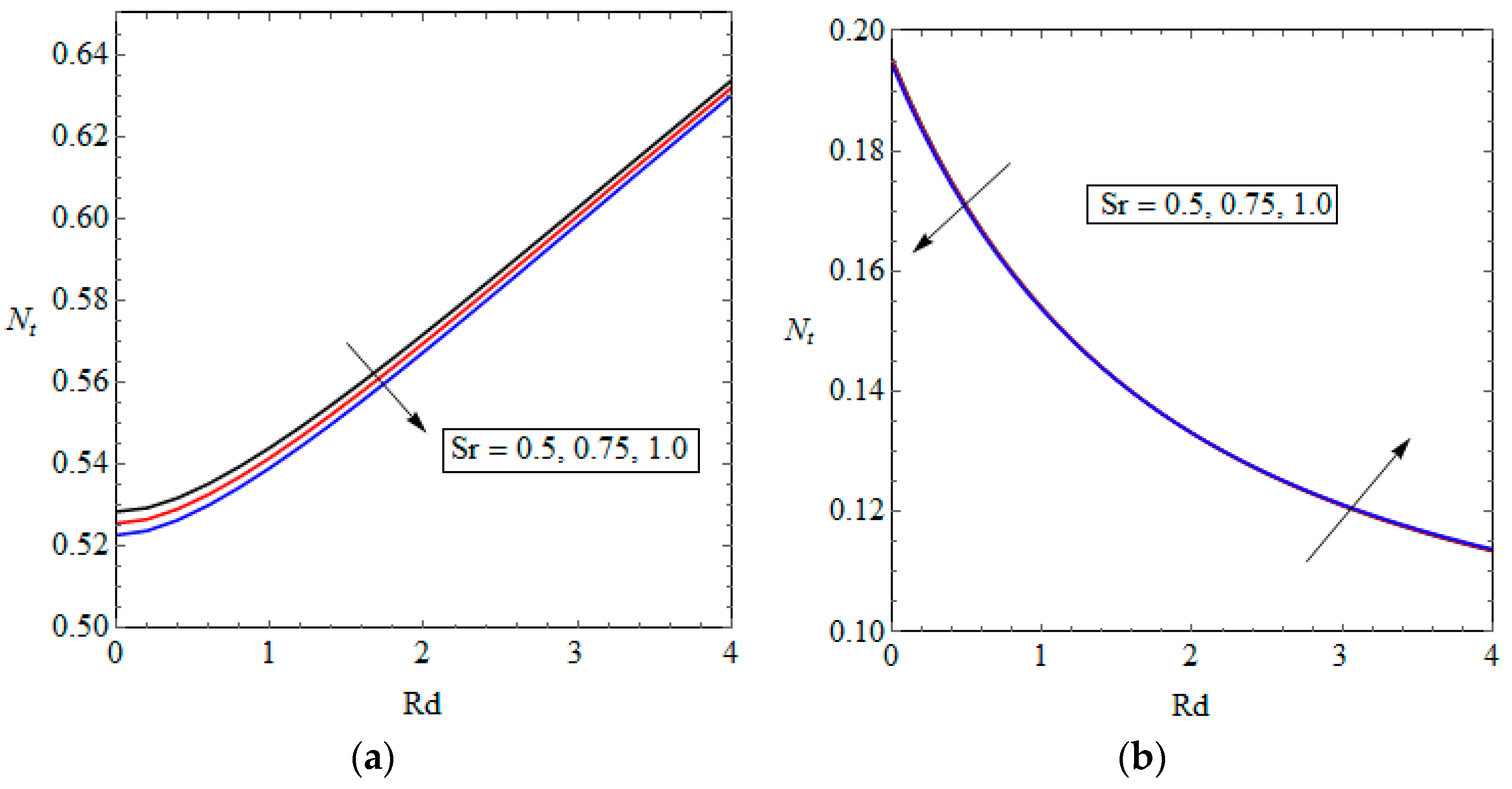
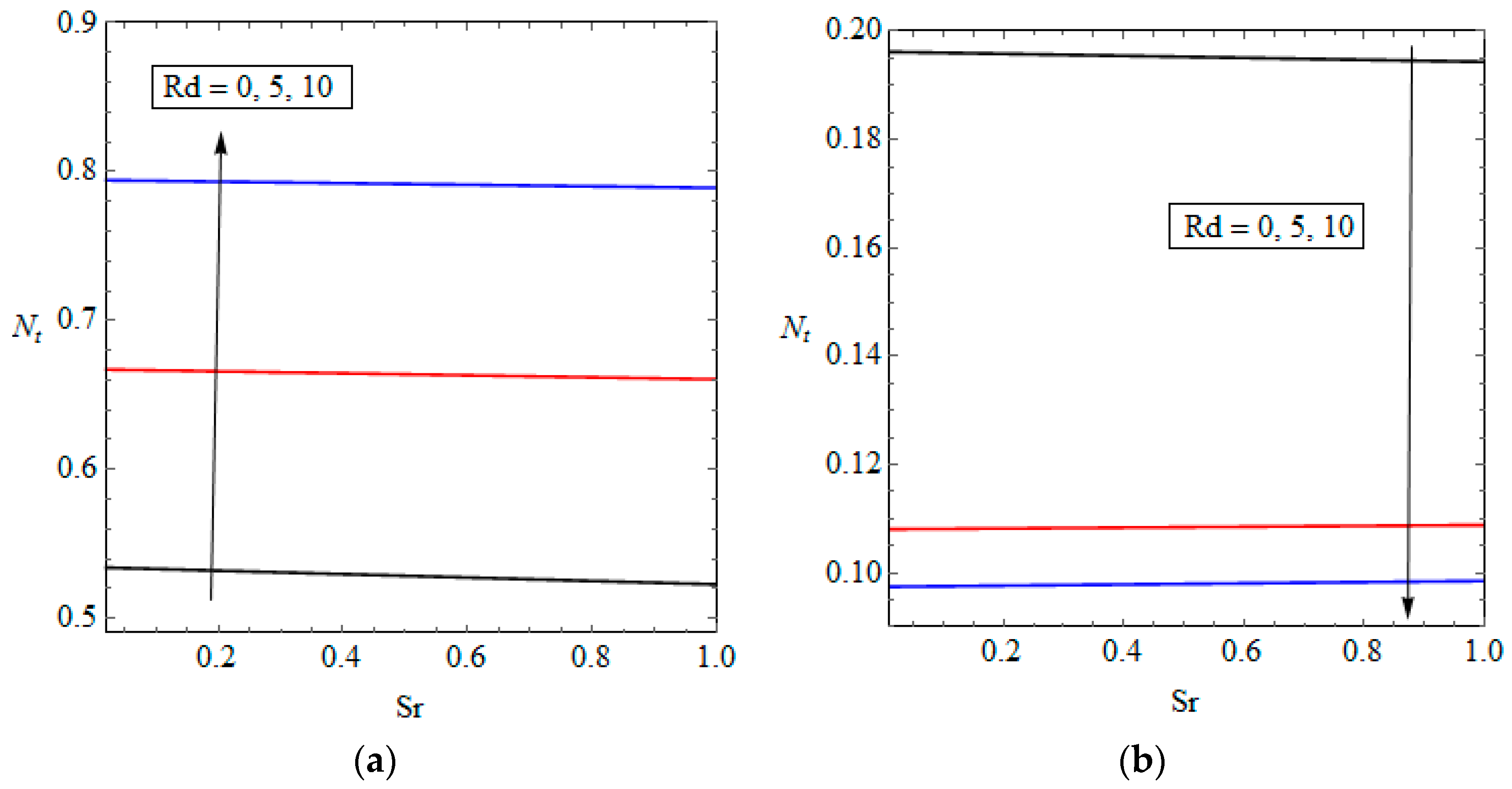
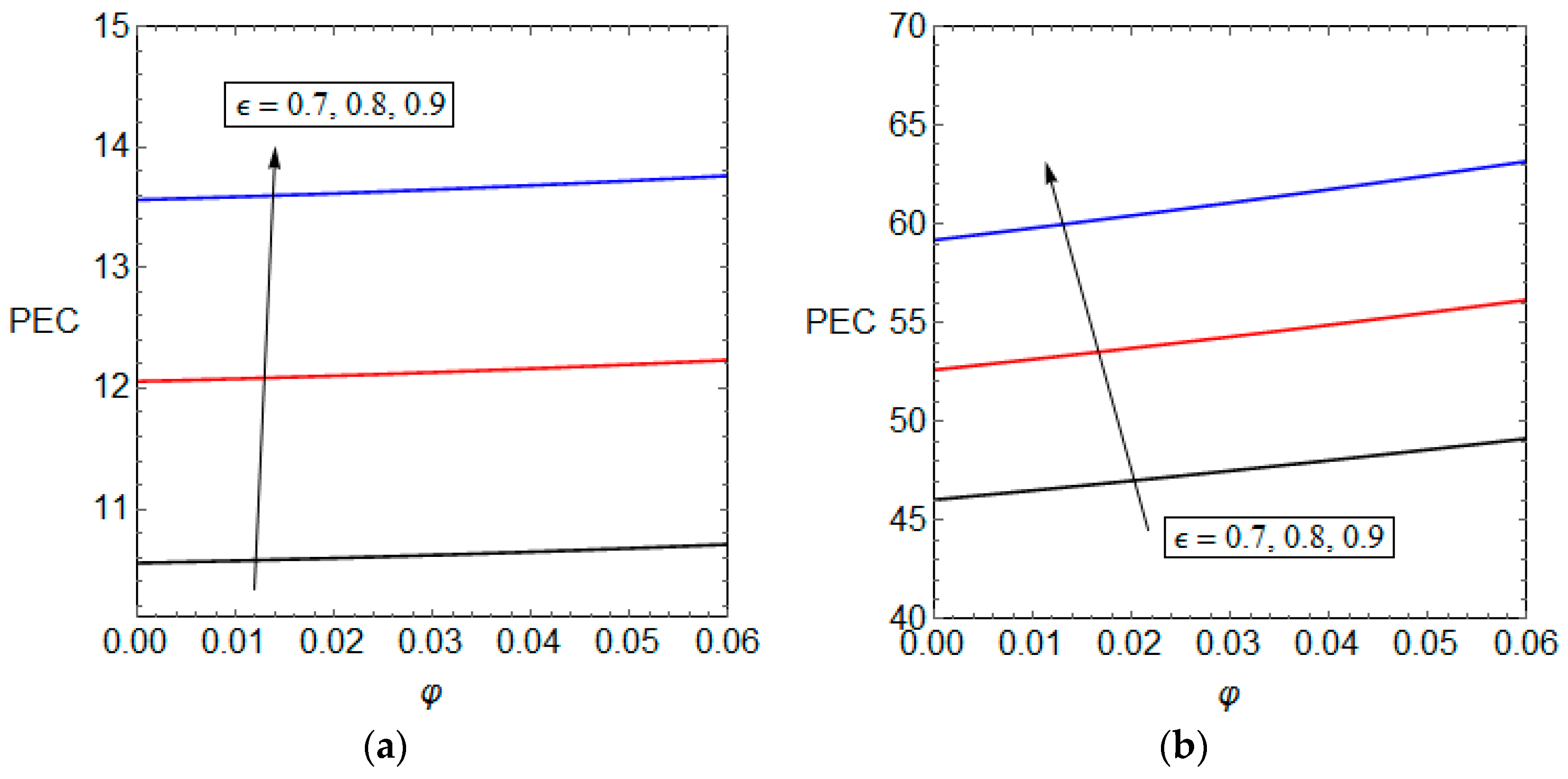
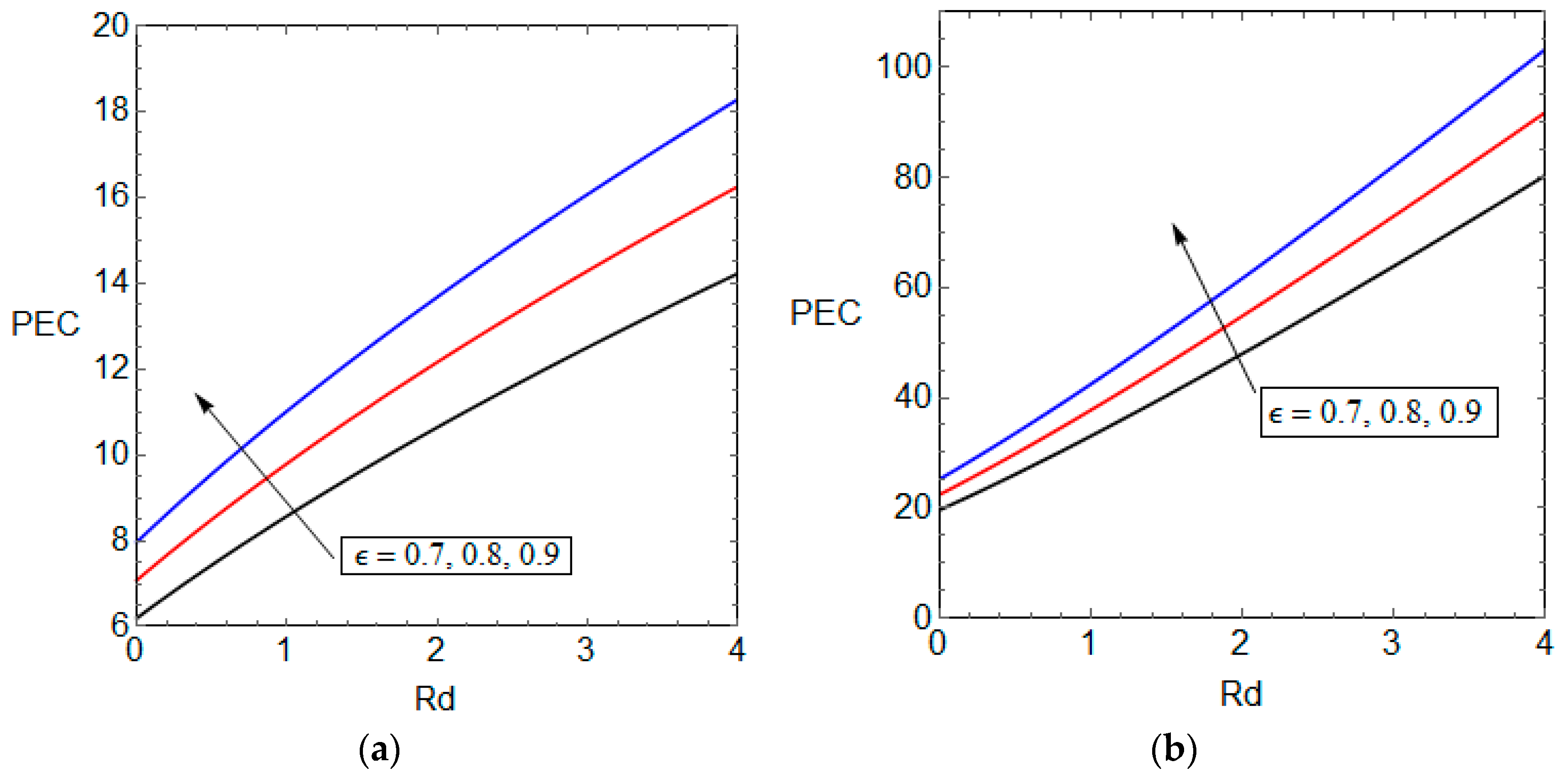
5.3. Entropy Generation and Performance Evaluation Criterion
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Nomenclature
| interfacial area per unit volume of porous media, | |
| Biot number | |
| Concentration of the chemical products per unit volume, | |
| Specific heat of the fluid phase of the porous medium, | |
| Diffusion coefficient, | |
| Darcy number | |
| Thermodiffusion coefficient, | |
| h1 | Height of the lower wall, |
| h2 | Height of the lower boundary of the upper wall, |
| h3 | Height of the upper boundary of the upper wall, |
| h | External heat convection coefficient, |
| hsf | Internal heat convection coefficient, |
| k | Solid to fluid effective thermal conductivity ratio |
| k1 | Reference thermal conductivity for lower solid material, |
| k2 | Reference thermal conductivity for upper solid material, |
| ke1 | Ratio of the fluid to lower solid material thermal conductivities |
| ke2 | Ratio of the fluid to upper solid material thermal conductivities |
| ke,nf | Effective thermal conductivity of the nanofluid phase of the porous medium, |
| kes | Effective thermal conductivity of the solid phase of the porous medium, |
| kf | Thermal conductivity of the base fluid, |
| knf | Thermal conductivity of the nanofluid, |
| kp | Thermal conductivity of the nanoparticles, |
| kR | Kinetic constant, |
| ks | Thermal conductivity of the solid phase of the porous medium, |
| Nc | dimensionless convection heat transfer (Case two) |
| Nu | Nusselt Number |
| Ns | Dimensionless local volumetric entropy generation rate |
| Nt | Dimensionless total entropy generation rate |
| p | Pressure, Pa |
| Dimensionless volumetric internal heat generation rate for the lower solid material | |
| Dimensionless volumetric internal heat generation rate for the upper solid material | |
| Dimensionless heat flux boundary condition (Case two) | |
| Volumetric internal heat generation rate for the lower solid material, | |
| Volumetric internal heat generation rate for the upper solid material, | |
| Heat flux boundary condition (Case two), | |
| Radiation heat flux, | |
| Rd | Dimensionless radiation parameter |
| Dimensionless volumetric internal heat generation rate for the solid phase of the porous medium | |
| Sr | Soret Number |
| Dimensionless volumetric internal heat generation rate for the nanofluid phase of the porous medium | |
| Temperature, | |
| Temperature of the lower solid material, | |
| Temperature of the upper solid material, | |
| Outer temperature of the upper solid material, | |
| Outer temperature of the lower solid material, | |
| Temperature of the fluid phase of the porous medium, | |
| Temperature of the solid phase of the porous medium, | |
| Um | Average dimensionless velocity |
| Velocity of the fluid in porous medium, | |
| Dimensionless velocity | |
| Y1 | Dimensionless height of the lower wall |
| Y2 | Dimensionless height of the upper wall lower boundary |
| Greek symbols | |
| Damköhler number | |
| Porosity | |
| Dimensionless temperature | |
| Dimensionless temperature of the lower solid material | |
| Dimensionless temperature of the upper solid material | |
| Dimensionless temperature of the fluid phase of the porous medium | |
| Dimensionless average temperature of the fluid phase of the porous medium | |
| Dimensionless temperature of the solid phase of the porous medium | |
| Dimensionless temperature at outer side of the lower wall | |
| Permeability, | |
| Rosseland mean absorption coefficient | |
| Dynamic viscosity of porous medium, | |
| Dynamic viscosity of the base fluid, | |
| Dynamic viscosity of the nanofluid, | |
| Dimensionless volumetric internal heat generation rate for the solid phase of the porous medium | |
| Dimensionless volumetric internal heat generation rate for the fluid phase of the porous medium | |
| density of the fluid phase, | |
| Stefan–Boltzmann constant, | |
| Dimensionless concentration | |
| Constant defined in entropy generation formulation | |
References
- Mahian, O.; Kianifar, A.; Sahin, A.Z.; Wongwises, S. Heat transfer, pressure drop, and entropy generation in a solar collector using SiO 2/water nanofluids: Effects of nanoparticle size and pH. J. Heat Transf. 2015, 137, 61011. [Google Scholar] [CrossRef]
- Hepbasli, A. Exergetic modeling and assessment of solar assisted domestic hot water tank integrated ground-source heat pump systems for residences. Energy Build. 2007, 39, 1211–1217. [Google Scholar] [CrossRef]
- Kuravi, S.; Trahan, J.; Goswami, D.Y.; Rahman, M.M.; Stefanakos, E.K. Thermal energy storage technologies and systems for concentrating solar power plants. Prog. Energy Combust. Sci. 2013, 39, 285–319. [Google Scholar] [CrossRef]
- Shamoushaki, M.; Ehyaei, M.A.; Ghanatir, F. Exergy, economic and environmental analysis and multi-objective optimization of a SOFC-GT power plant. Energy 2017, 134, 515–531. [Google Scholar] [CrossRef]
- Chen, Q.; Ja, M.K.; Li, Y.; Chua, K.J. On the second law analysis of a multi-stage spray-assisted low-temperature desalination system. Energy Convers. Manag. 2017, 148, 1306–1316. [Google Scholar] [CrossRef]
- Mistry, K.H.; Lienhard, J.H.; Zubair, S.M. Effect of entropy generation on the performance of humidification- dehumidification desalination cycles. Int. J. Therm. Sci. 2010, 49, 1837–1847. [Google Scholar] [CrossRef]
- Yao, X.; Zhang, Y.; Du, L.; Liu, J.; Yao, J. Review of the applications of microreactors. Renew. Sustain. Energy Rev. 2015, 47, 519–539. [Google Scholar] [CrossRef]
- Roberge, D.M.; Ducry, L.; Bieler, N.; Cretton, P.; Zimmermann, B. Microreactor technology: A revolution for the fine chemical and pharmaceutical industries? Chem. Eng. Technol. 2005, 28, 318–323. [Google Scholar] [CrossRef]
- Kolb, G.; Hessel, V. Micro-structured reactors for gas phase reactions. Chem. Eng. J. 2004, 98, 1–38. [Google Scholar] [CrossRef]
- Ju, Y.; Maruta, K. Microscale combustion: Technology development and fundamental research. Prog. Energy Combust. Sci. 2011, 37, 669–715. [Google Scholar] [CrossRef]
- Li, J.; Chou, S.K.; Li, Z.W.; Yang, W.M. Experimental investigation of porous media combustion in a planar micro-combustor. Fuel 2010, 89, 708–715. [Google Scholar] [CrossRef]
- Torabi, M.; Karimi, N.; Peterson, G.P.; Yee, S. Challenges and progress on the modeling of entropy generation in porous media: A review. Int. J. Heat Mass Transf. 2017, 114, 31–46. [Google Scholar] [CrossRef]
- Torabi, M.; Torabi, M.; Peterson, G.P. Entropy generation of double diffusive forced convection in porous channels with thick walls and Soret effect. Entropy 2017, 19, 171. [Google Scholar] [CrossRef]
- Hunt, G.; Karimi, N.; Torabi, M. Analytical investigation of heat transfer and classical entropy generation in microreactors—The influences of exothermicity and asymmetry. Appl. Therm. Eng. 2017, 119, 403–424. [Google Scholar] [CrossRef]
- Elliott, A.; Torabi, M.; Karimi, N. Thermodynamics analyses of porous microchannels with asymmetric thick walls and exothermicity: An entropic model of microreactors. J. Therm. Sci. Eng. Appl. 2017, 9, 41013. [Google Scholar] [CrossRef]
- Delsman, E.R.; de Croon, M.H.J.M.; Elzinga, G.D.; Cobden, P.D.; Kramer, G.J.; Schouten, J.C. The influence of differences between microchannels on microreactor performance. Chem. Eng. Technol. 2005, 28, 367–375. [Google Scholar] [CrossRef]
- Ting, T.W.; Hung, Y.M.; Guo, N. Entropy generation of viscous dissipative nanofluid convection in asymmetrically heated porous microchannels with solid-phase heat generation. Energy Convers. Manag. 2015, 105, 731–745. [Google Scholar] [CrossRef]
- Ting, T.W.; Hung, Y.M.; Guo, N. Entropy generation of viscous dissipative nanofluid flow in thermal non-equilibrium porous media embedded in microchannels. Int. J. Heat Mass Transf. 2015, 81, 862–877. [Google Scholar] [CrossRef]
- Torabi, M.; Zhang, K. Temperature distribution, local and total entropy generation analyses in MHD porous channels with thick walls. Energy 2015, 87, 540–554. [Google Scholar] [CrossRef]
- Wang, W.; Zuo, Z.; Liu, J.; Yang, W. Entropy generation analysis of fuel premixed CH4/H2/air flames using multistep kinetics. Int. J. Hydrog. Energy 2016, 41, 20744–20752. [Google Scholar] [CrossRef]
- Tang, A.; Xu, Y.; Pan, J.; Yang, W.; Jiang, D.; Lu, Q. Combustion characteristics and performance evaluation of premixed methane/air with hydrogen addition in a micro-planar combustor. Chem. Eng. Sci. 2015, 131, 235–242. [Google Scholar] [CrossRef]
- Sahu, K.C. Double diffusive effects on pressure-driven miscible channel flow: Influence of variable diffusivity. Int. J. Multiph. Flow 2013, 55, 24–31. [Google Scholar] [CrossRef]
- Bhagat, K.D.; Tripathi, M.K.; Sahu, K.C. Instability due to double-diffusive phenomenon in pressure-driven displacement flow of one fluid by another in an axisymmetric pipe. Eur. J. Mech. B Fluids 2016, 55, 63–70. [Google Scholar] [CrossRef]
- Karimi, N.; Agbo, D.; Talat Khan, A.; Younger, P.L. On the effects of exothermicity and endothermicity upon the temperature fields in a partially-filled porous channel. Int. J. Therm. Sci. 2015, 96, 128–148. [Google Scholar] [CrossRef]
- Yang, K.; Vafai, K. Analysis of temperature gradient bifurcation in porous media—An exact solution. Int. J. Heat Mass Transf. 2010, 53, 4316–4325. [Google Scholar] [CrossRef]
- Chen, W.H.; Cheng, T.C.; Hung, C.I. Modeling and simulation of microwave double absorption on methanol steam reforming for hydrogen production. Int. J. Hydrog. Energy 2011, 36, 333–344. [Google Scholar] [CrossRef]
- Chen, W.H.; Cheng, T.C.; Hung, C.I. Numerical predictions on thermal characteristic and performance of methanol steam reforming with microwave-assisted heating. Int. J. Hydrog. Energy 2011, 36, 8279–8791. [Google Scholar] [CrossRef]
- Civan, F. Porous Media Transport Phenomena; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Torabi, M.; Peterson, G.P.; Torabi, M.; Karimi, N. A thermodynamic analysis of forced convection through porous media using pore scale modeling. Int. J. Heat Mass Transf. 2016, 99, 303–316. [Google Scholar] [CrossRef]
- Torabi, M.; Torabi, M.; Peterson, G.P. Heat transfer and entropy generation analyses of forced convection through porous media using pore scale modeling. J. Heat Transf. 2017, 139, 12601. [Google Scholar] [CrossRef]
- Chen, J.; Song, W.; Gao, X.; Xu, D. Hetero-/homogeneous combustion and flame stability of fuel-lean propane–air mixtures over platinum in catalytic micro-combustors. Appl. Therm. Eng. 2016, 100, 932–943. [Google Scholar] [CrossRef]
- Wang, P.; Vafai, K.; Liu, D.Y. Analysis of radiative effect under local thermal non-equilibrium conditions in porous media-application to a solar air receiver. Numer. Heat Transf. Part A 2014, 65, 931–948. [Google Scholar] [CrossRef]
- Torabi, M.; Dickson, C.; Karimi, N. Theoretical investigation of entropy generation and heat transfer by forced convection of copper–water nanofluid in a porous channel—Local thermal non-equilibrium and partial filling effects. Powder Technol. 2016, 301, 234–254. [Google Scholar] [CrossRef]
- Modest, M.F. Radiative Heat Transfer; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Elliott, A.; Torabi, M.; Karimi, N.; Cunningham, S. On the effects of internal heat sources upon forced convection in porous channels with asymmetric thick walls. Int. Commun. Heat Mass Transf. 2016, 73, 100–110. [Google Scholar] [CrossRef]
- Torabi, M.; Peterson, G.P. Effects of velocity slip and temperature jump on the heat transfer and entropy generation in micro porous channels under magnetic field. Int. J. Heat Mass Transf. 2016, 102, 585–595. [Google Scholar] [CrossRef]
- Ibáñez, G.; López, A.; Pantoja, J.; Moreira, J. Combined effects of uniform heat flux boundary conditions and hydrodynamic slip on entropy generation in a microchannel. Int. J. Heat Mass Transf. 2014, 73, 201–206. [Google Scholar] [CrossRef]
- Ibáñez, G. Entropy generation in MHD porous channel with hydrodynamic slip and convective boundary conditions. Int. J. Heat Mass Transf. 2015, 80, 274–280. [Google Scholar] [CrossRef]
- Ibáñez, G.; López, A.; Pantoja, J.; Moreira, J. Entropy generation analysis of a nanofluid flow in MHD porous microchannel with hydrodynamic slip and thermal radiation. Int. J. Heat Mass Transf. 2016, 100, 89–97. [Google Scholar] [CrossRef]
- Kaisare, N.S.; Vlachos, D.G. A review on microcombustion: Fundamentals, devices and applications. Prog. Energy Combust. Sci. 2012, 38, 321–359. [Google Scholar] [CrossRef]
- Ellahi, R.; Hassan, M.; Zeeshan, A. Shape effects of nanosize particles in Cu-H2O nanofluid on entropy generation. Int. J. Heat Mass Transf. 2015, 81, 449–456. [Google Scholar] [CrossRef]
- Torabi, M.; Karimi, N.; Zhang, K. Heat transfer and second law analyses of forced convection in a channel partially filled by porous media and featuring internal heat sources. Energy 2015, 93, 106–127. [Google Scholar] [CrossRef]
- Torabi, M.; Karimi, N.; Zhang, K.; Peterson, G.P. Generation of entropy and forced convection of heat in a conduit partially filled with porous media—Local thermal non-equilibrium and exothermicity effects. Appl. Therm. Eng. 2016, 106, 518–536. [Google Scholar] [CrossRef]
- Torabi, M.; Zhang, K.; Karimi, N.; Peterson, G.P. Entropy generation in thermal systems with solid structures—A concise review. Int. J. Heat Mass Transf. 2016, 97, 917–931. [Google Scholar] [CrossRef]
- Dickson, C.; Torabi, M.; Karimi, N. First and second law analyses of nanofluid forced convection in a partially-filled porous channel—The effects of local thermal non-equilibrium and internal heat sources. Appl. Therm. Eng. 2016, 103, 459–480. [Google Scholar] [CrossRef]
- Mehta, S.; Chauhan, K.P.; Kanagaraj, S. Modeling of thermal conductivity of nanofluids by modifying Maxwell’s equation using cell model approach. J. Nanopart. Res. 2011, 13, 2791–2798. [Google Scholar] [CrossRef]
- Mishra, P.C.; Mukherjee, S.; Nayak, S.K.; Panda, A. A brief review on viscosity of nanofluids. Int. Nano Lett. 2014, 4, 109–120. [Google Scholar] [CrossRef]
- Miroshnichenko, I.V.; Sheremet, M.A.; Oztop, H.F.; Al-Salem, K. MHD natural convection in a partially open trapezoidal cavity filled with a nanofluid. Int. J. Mech. Sci. 2016, 119, 294–302. [Google Scholar] [CrossRef]
- Sheremet, M.A.; Oztop, H.F.; Pop, I.; Abu-Hamdeh, N. Analysis of entropy generation in natural convection of nanofluid inside a square cavity having hot solid block: Tiwari and das’ model. Entropy 2016, 18, 9. [Google Scholar] [CrossRef]
- Sheremet, M.A.; Cimpean, D.S.; Pop, I. Free convection in a partially heated wavy porous cavity filled with a nanofluid under the effects of Brownian diffusion and thermophoresis. Appl. Therm. Eng. 2017, 113, 413–418. [Google Scholar] [CrossRef]
- Amani, M.; Amani, P.; Kasaeian, A.; Mahian, O.; Yan, W.M. Two-phase mixture model for nanofluid turbulent flow and heat transfer: Effect of heterogeneous distribution of nanoparticles. Chem. Eng. Sci. 2017, 167, 135–144. [Google Scholar] [CrossRef]
- Torabi, M.; Torabi, M.; Ghiaasiaan, S.M.; Peterson, G.P. The effect of Al2O3-water nanofluid on the heat transfer and entropy generation of laminar forced convection through isotropic porous media. Int. J. Heat Mass Transf. 2017, 111, 804–816. [Google Scholar] [CrossRef]

© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Govone, L.; Torabi, M.; Hunt, G.; Karimi, N. Non-Equilibrium Thermodynamic Analysis of Double Diffusive, Nanofluid Forced Convection in Catalytic Microreactors with Radiation Effects. Entropy 2017, 19, 690. https://doi.org/10.3390/e19120690
Govone L, Torabi M, Hunt G, Karimi N. Non-Equilibrium Thermodynamic Analysis of Double Diffusive, Nanofluid Forced Convection in Catalytic Microreactors with Radiation Effects. Entropy. 2017; 19(12):690. https://doi.org/10.3390/e19120690
Chicago/Turabian StyleGovone, Lilian, Mohsen Torabi, Graeme Hunt, and Nader Karimi. 2017. "Non-Equilibrium Thermodynamic Analysis of Double Diffusive, Nanofluid Forced Convection in Catalytic Microreactors with Radiation Effects" Entropy 19, no. 12: 690. https://doi.org/10.3390/e19120690
APA StyleGovone, L., Torabi, M., Hunt, G., & Karimi, N. (2017). Non-Equilibrium Thermodynamic Analysis of Double Diffusive, Nanofluid Forced Convection in Catalytic Microreactors with Radiation Effects. Entropy, 19(12), 690. https://doi.org/10.3390/e19120690




