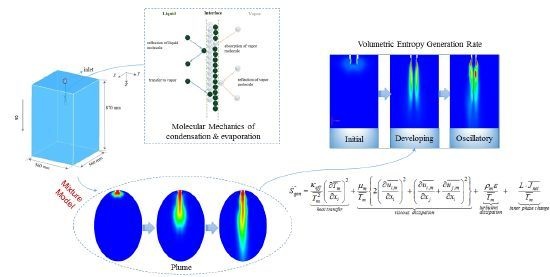
Entropy Assessment on Direct Contact Condensation of Subsonic Steam Jets in a Water Tank through Numerical Investigation
Abstract
:1. Introduction
2. Geometry Model

3. Mathematical Model
3.1. Mixture Model
3.1.1. Continuity Equation
3.1.2. Momentum Equation
3.1.3. Energy Equation
3.2. Turbulence Model
3.3. Phase Change Model
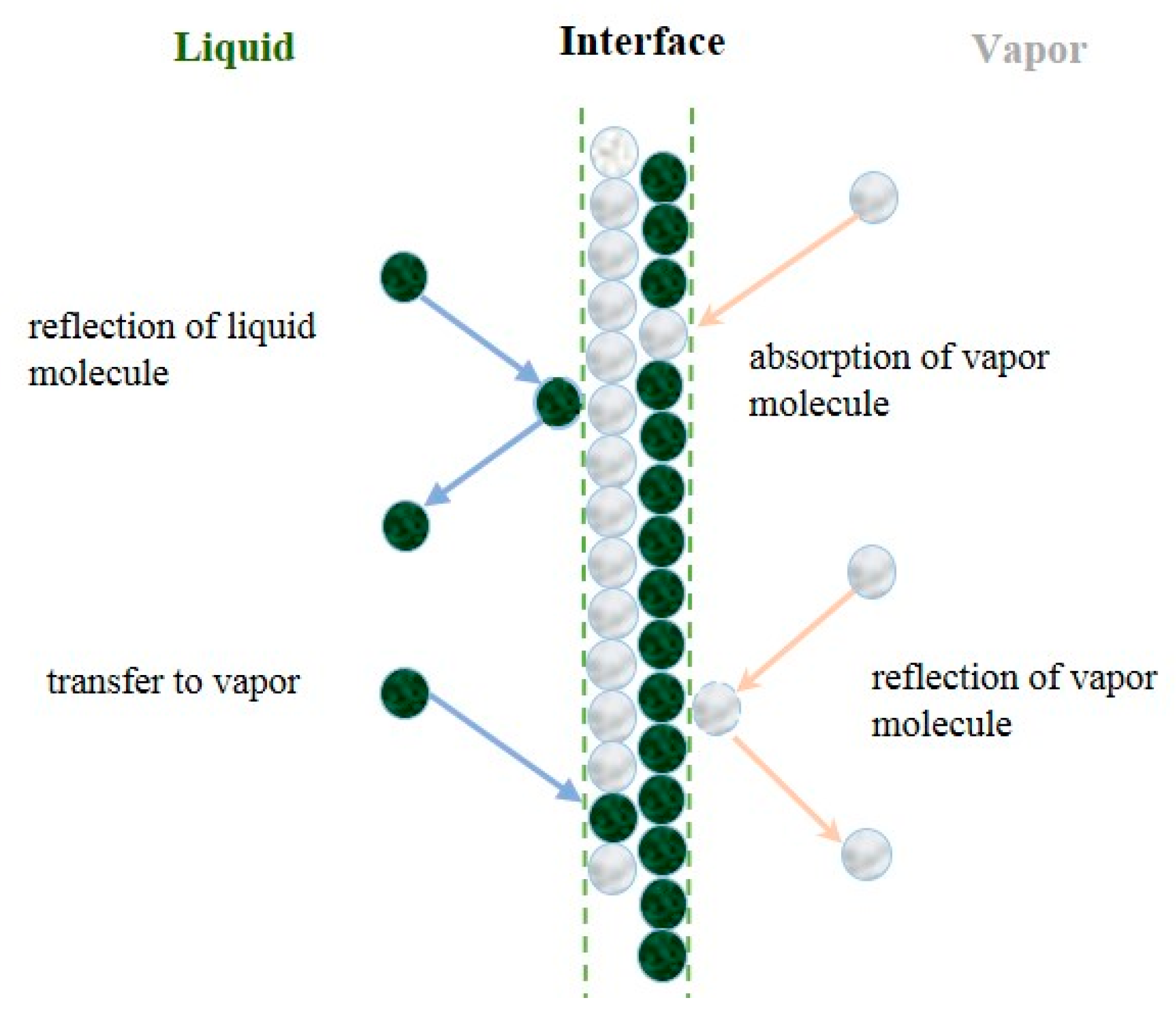
3.4 Entropy Generation Analysis Model
3.4.1. Convective Terms
3.4.2. Entropy Generation by Dissipation
3.4.3. Entropy Generation by Heat Transfer
3.4.4. Entropy Generation by Inner Phase Change
3.4.5. Time-Averaged Transport Equation for Entropy
- (1)
- The exact turbulent dissipation approximately equals to the production of density ρm and the turbulent dissipation rate ε, therefore, the entropy generation rate due to turbulent dissipation reads as:
- (2)
- Use the Boussinesque-like approach [31], and then the entropy generation due to fluctuating temperature gradients is:with a thermal diffusivity and at the turbulent thermal diffusivity. Assume that the turbulent thermal diffusivity is related as thermal diffusivity through:Then, the entropy generation due to mean temperature gradients and entropy generation due to fluctuating temperature gradients can be combined as the entropy generation due to heat transfer:From the derivation above, the local volumetric entropy generation is concluded as follows:
4. Computation Set-Up
4.1. Simulation Details
| Phases | Density (kg/m3) | Specific Heat Capacity (J/kg K) | Viscosity (Pa · s) | Thermal Conductivity (W/m K) |
|---|---|---|---|---|
| Vapor | Incompressible ideal-gas | Polynomial * | 1.34 × 10−5 | 0.0261 |
| Water | 998.2 | 4182 | 1.003 × 10−3 | 0.6 |
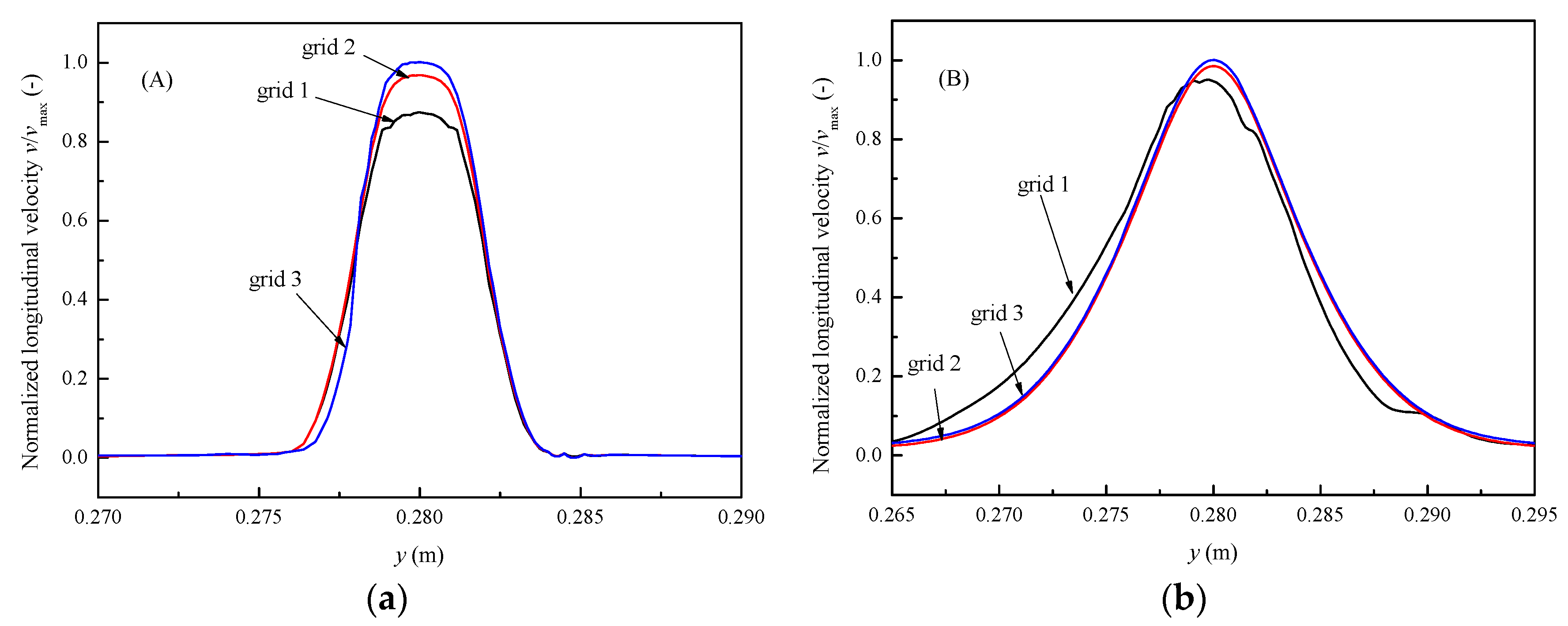
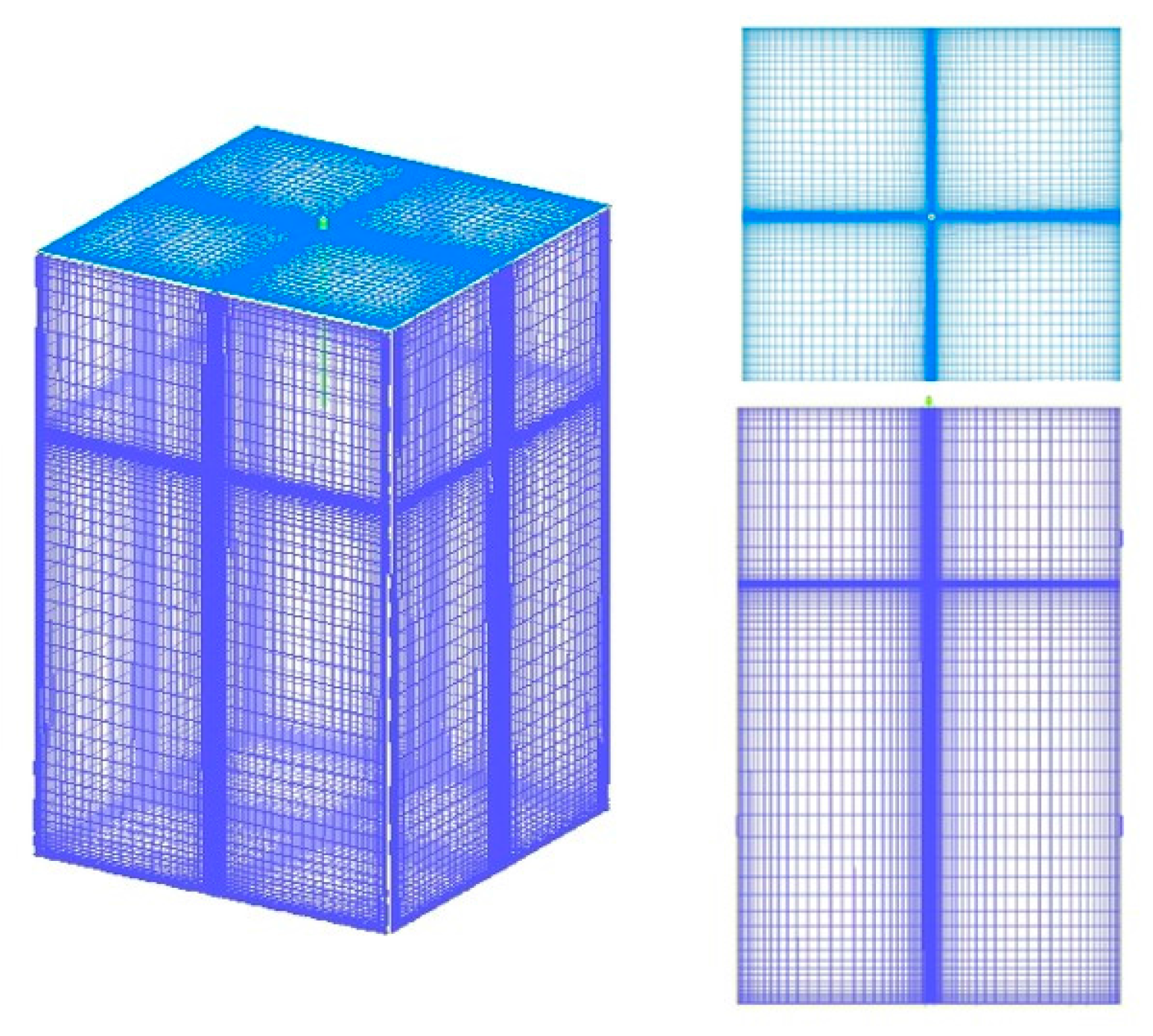
4.2. Grid Independent Verification
5. Results and Discussion
5.1. Verification & Validation

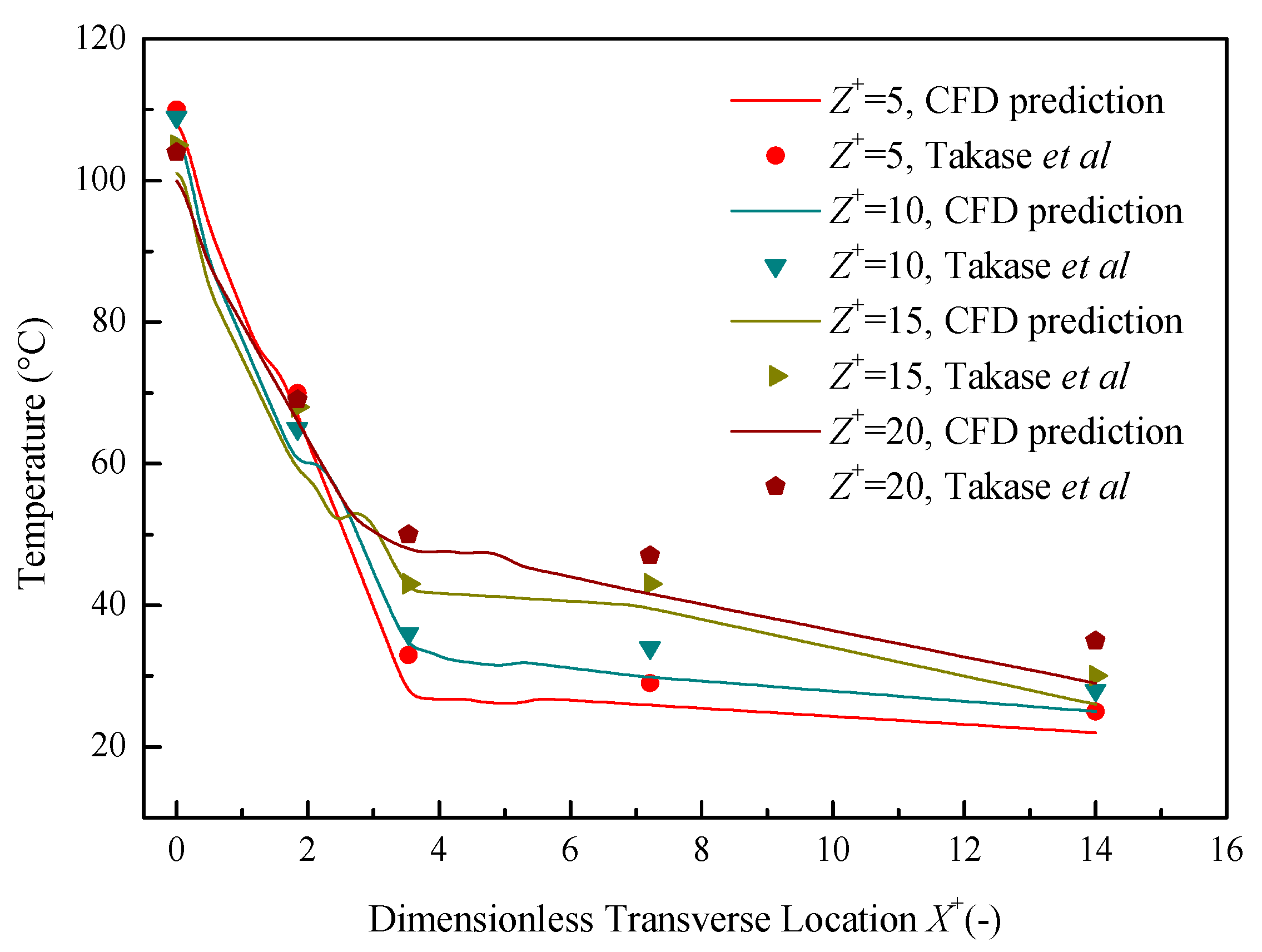
5.2. Numerical Results
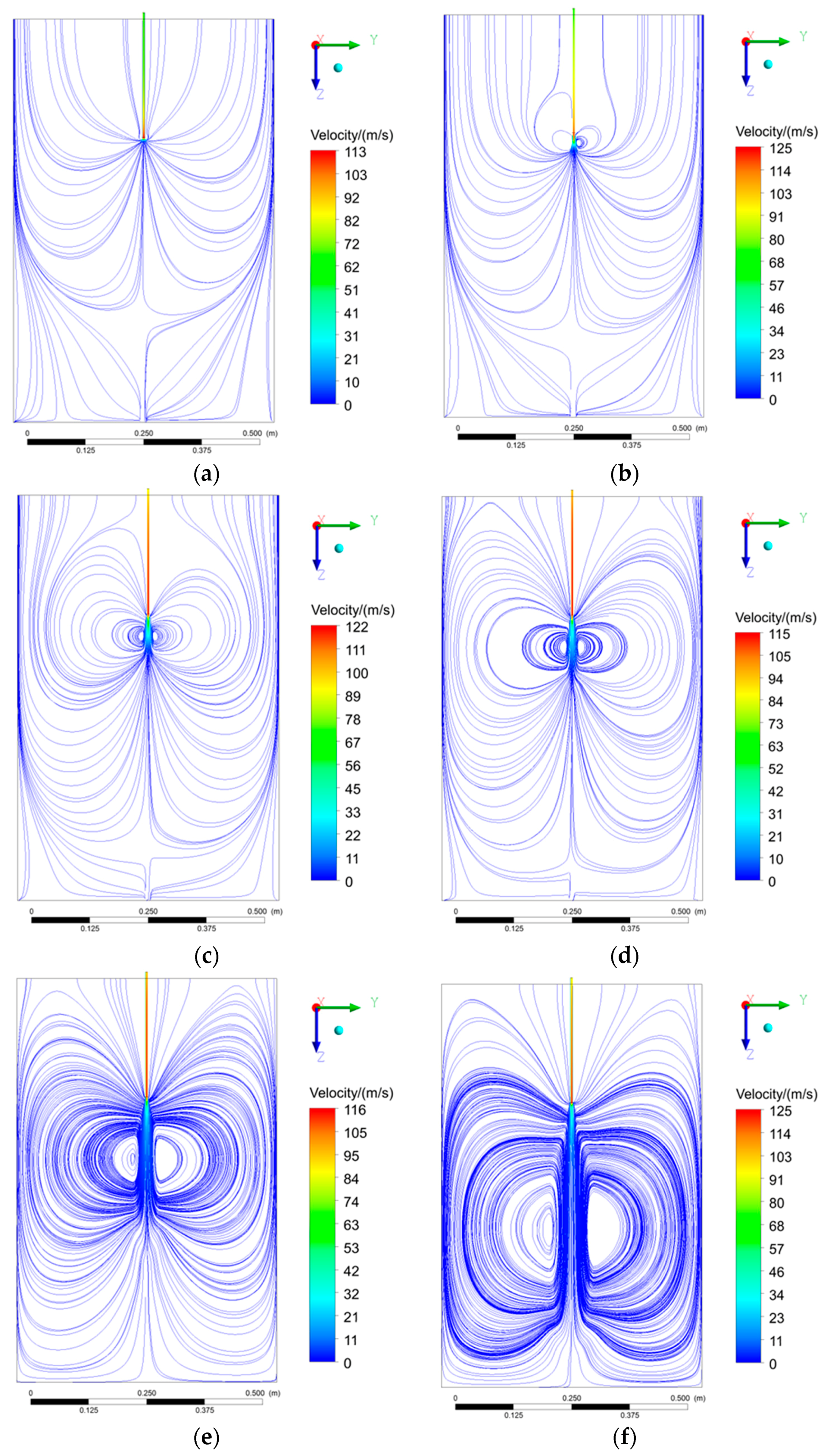
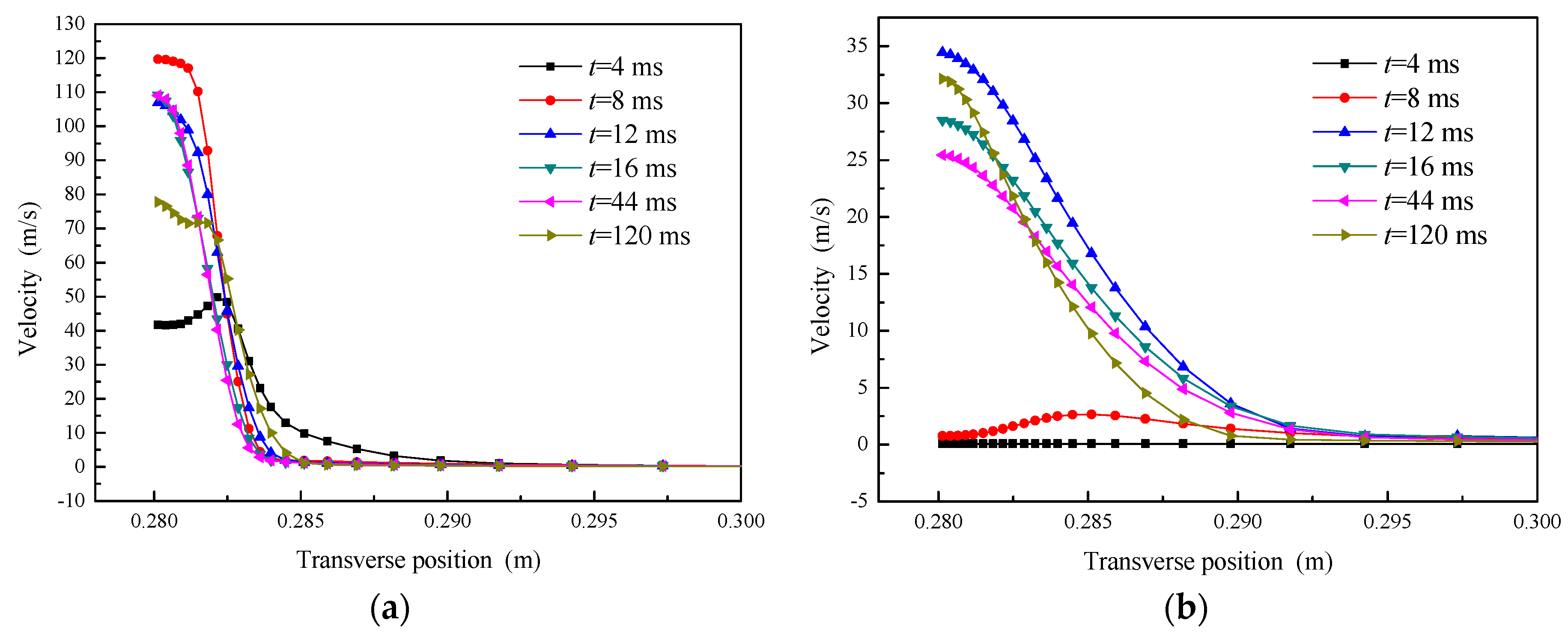
5.2.1. Velocity Profile
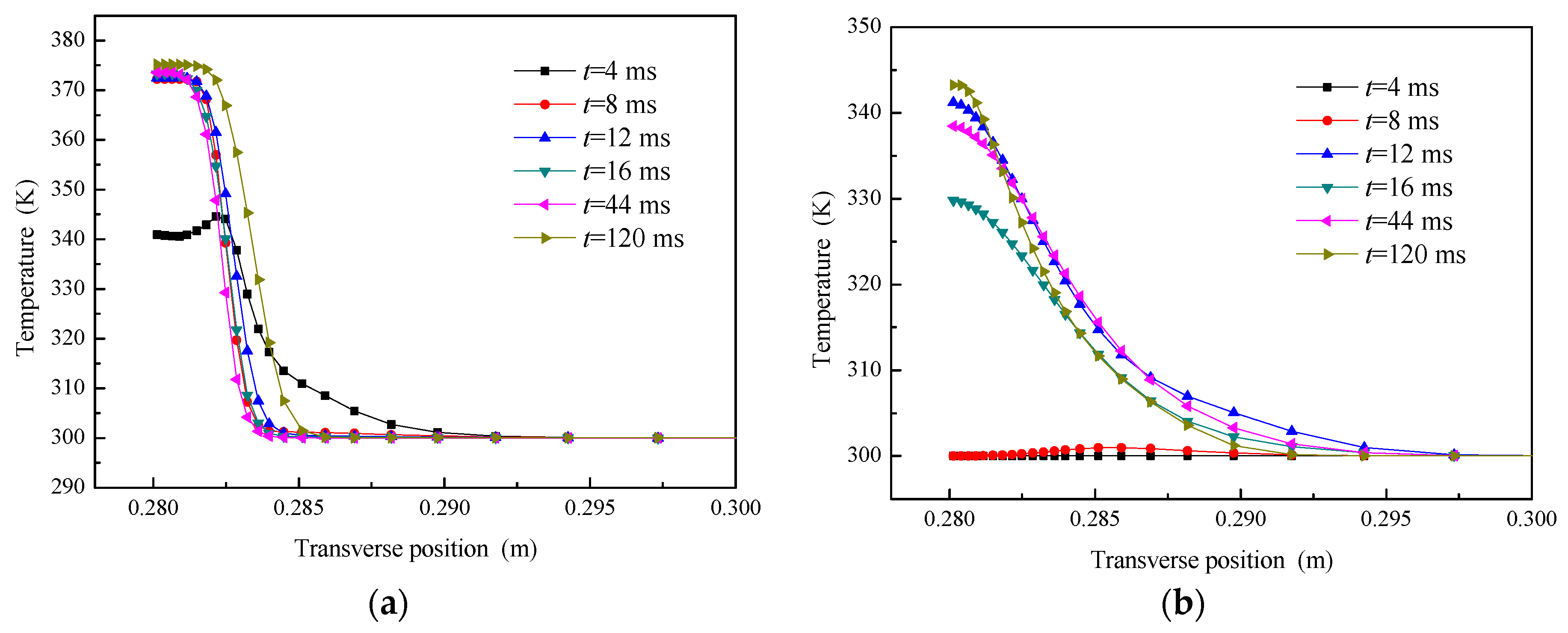
5.2.2. Temperature Field

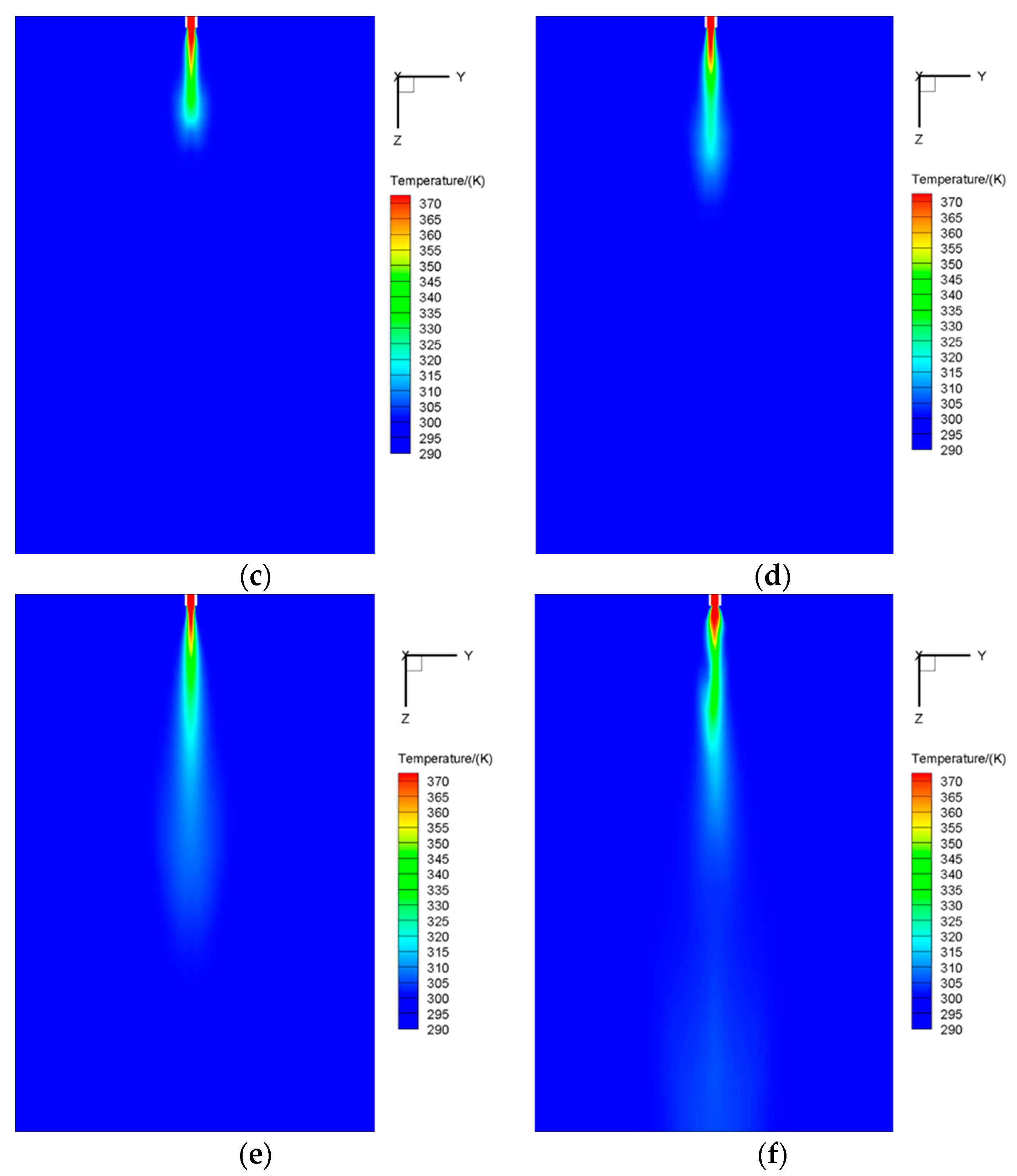
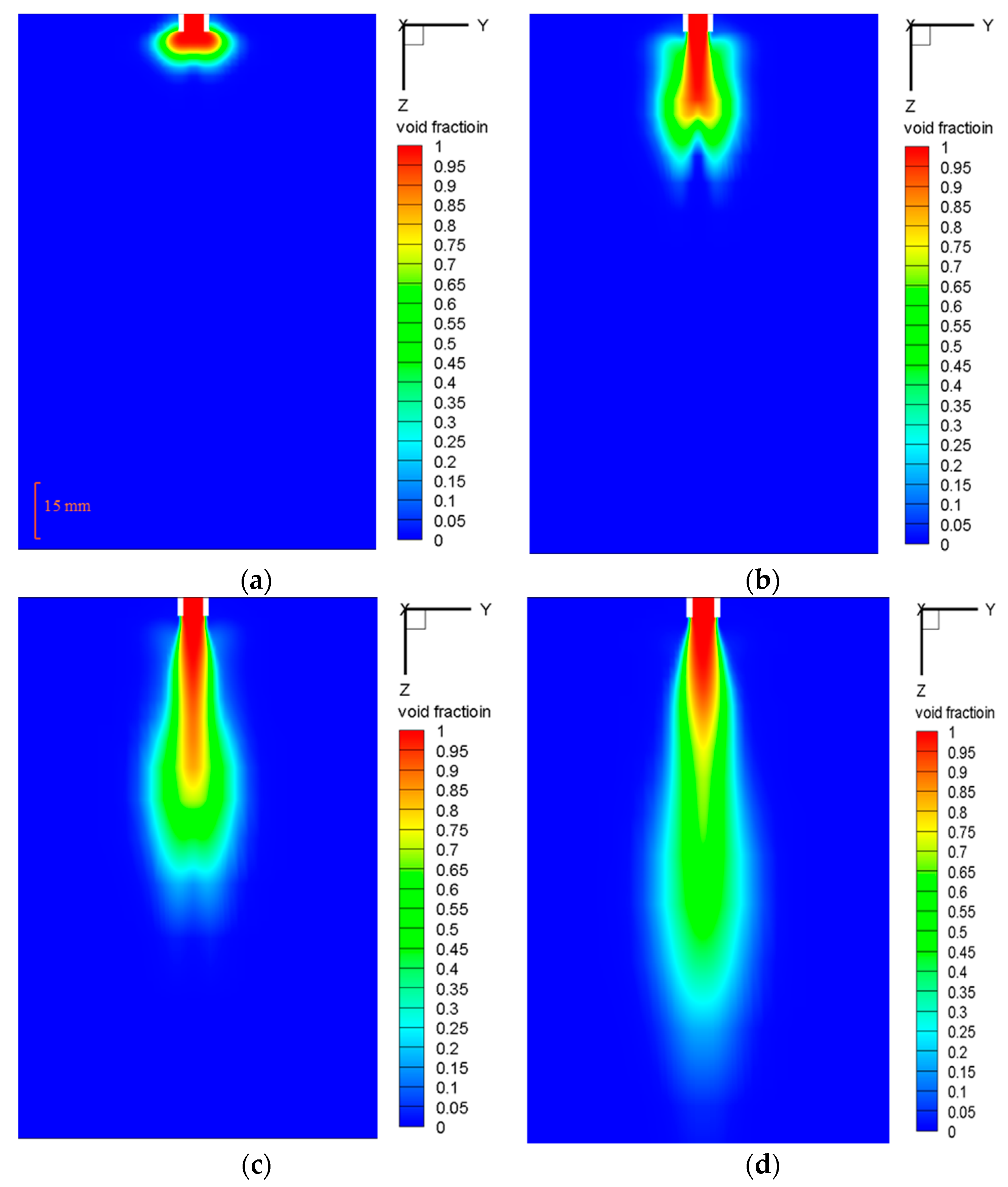
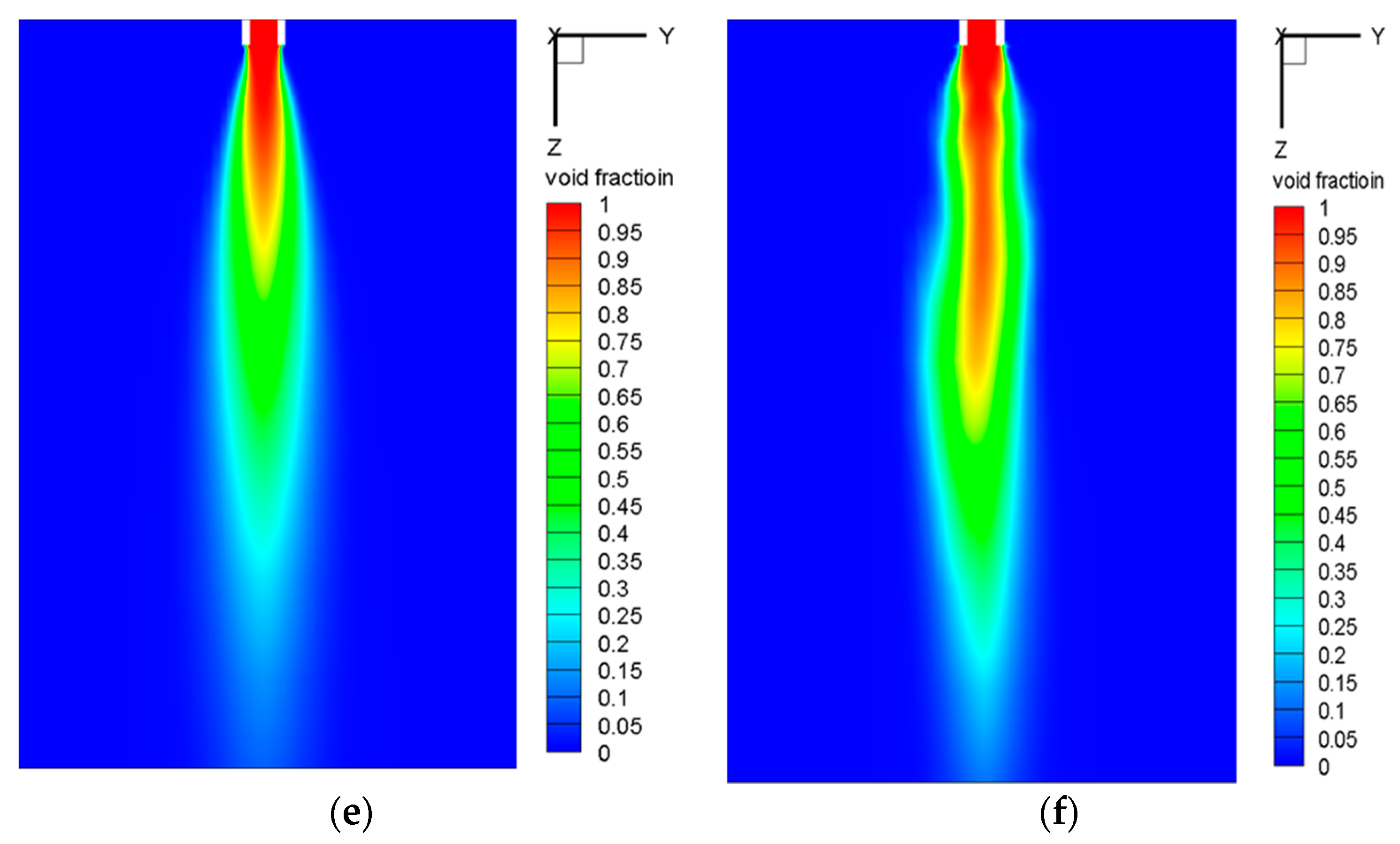
5.2.3. Plume Shape
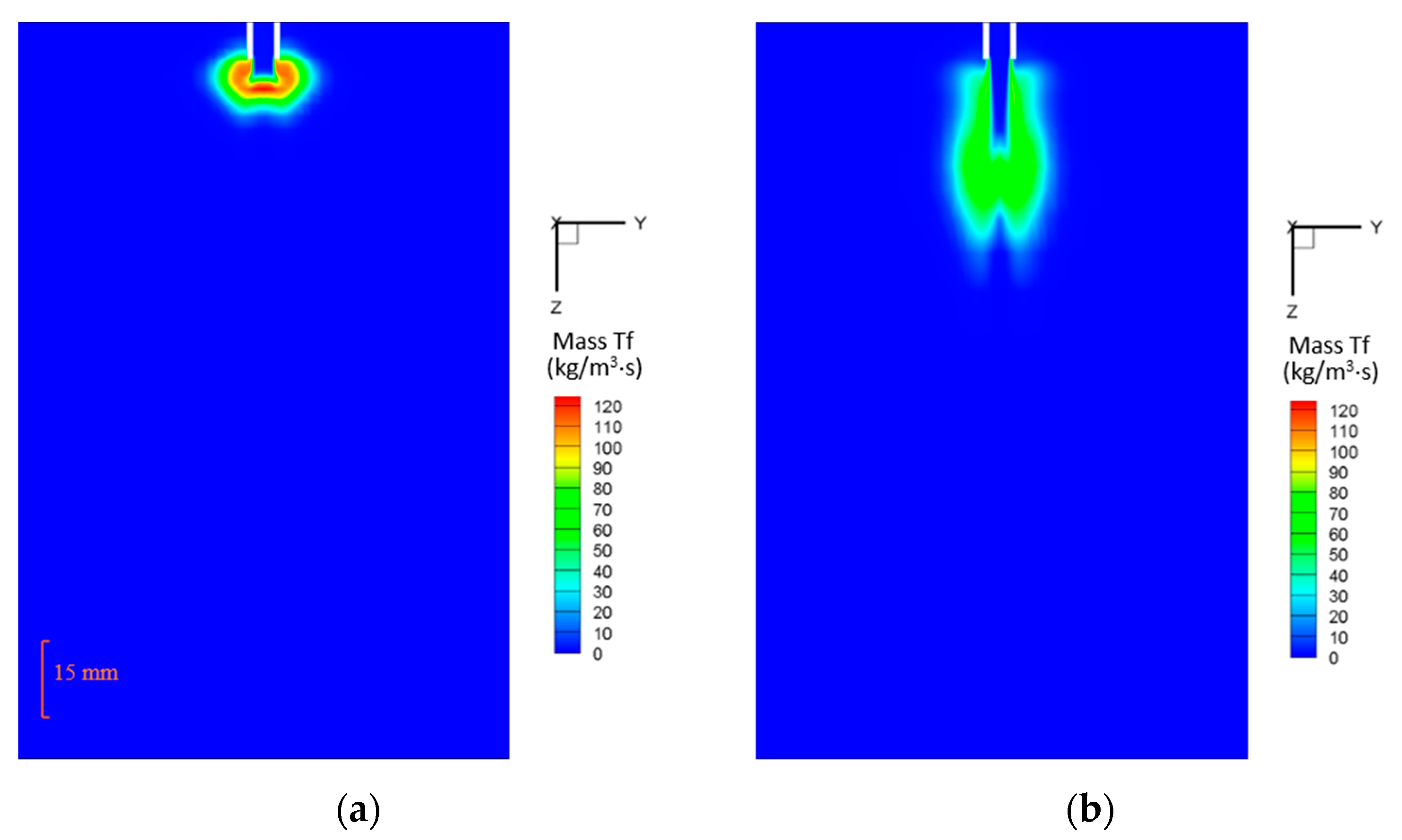
5.2.4. Mass Transfer
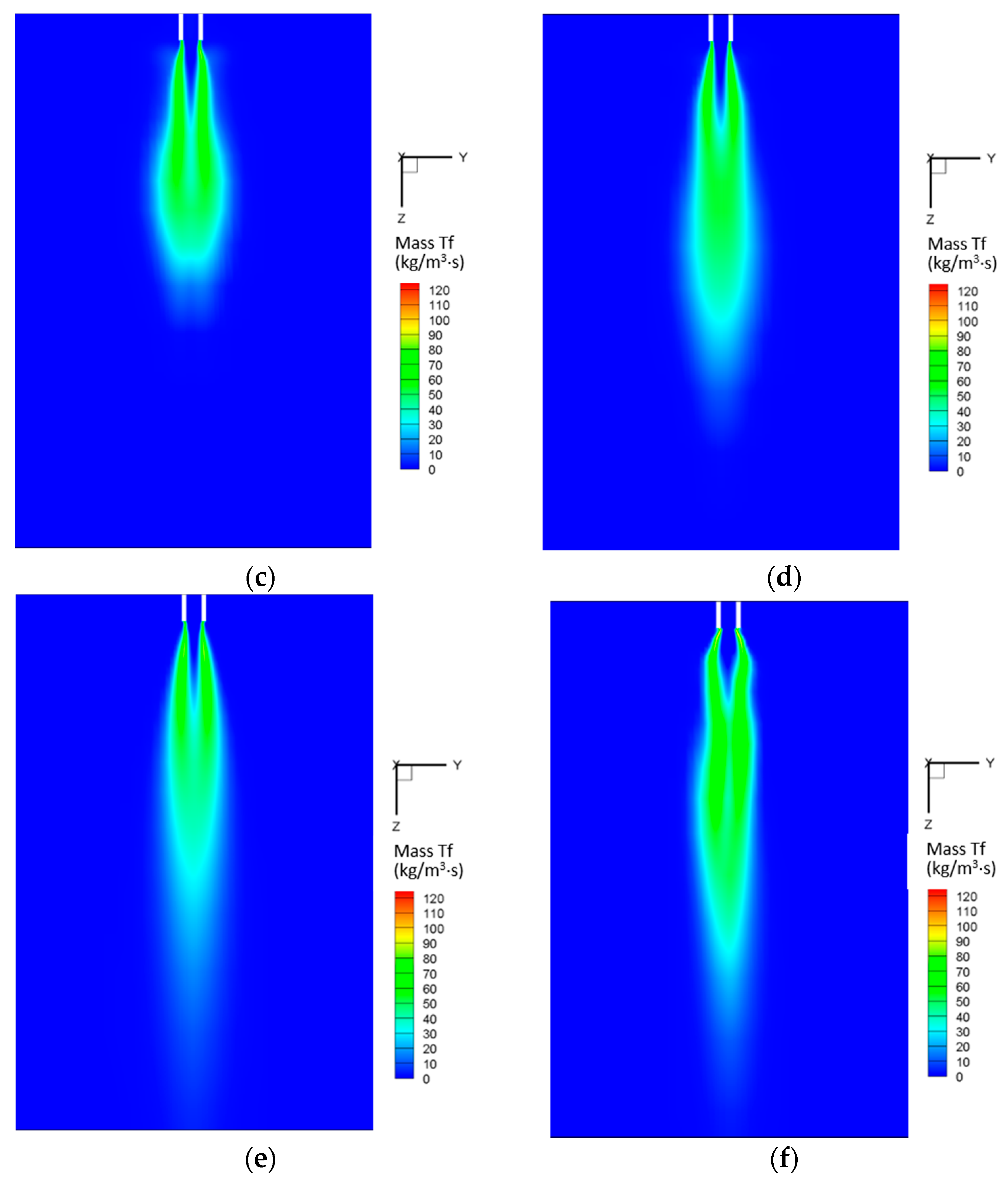
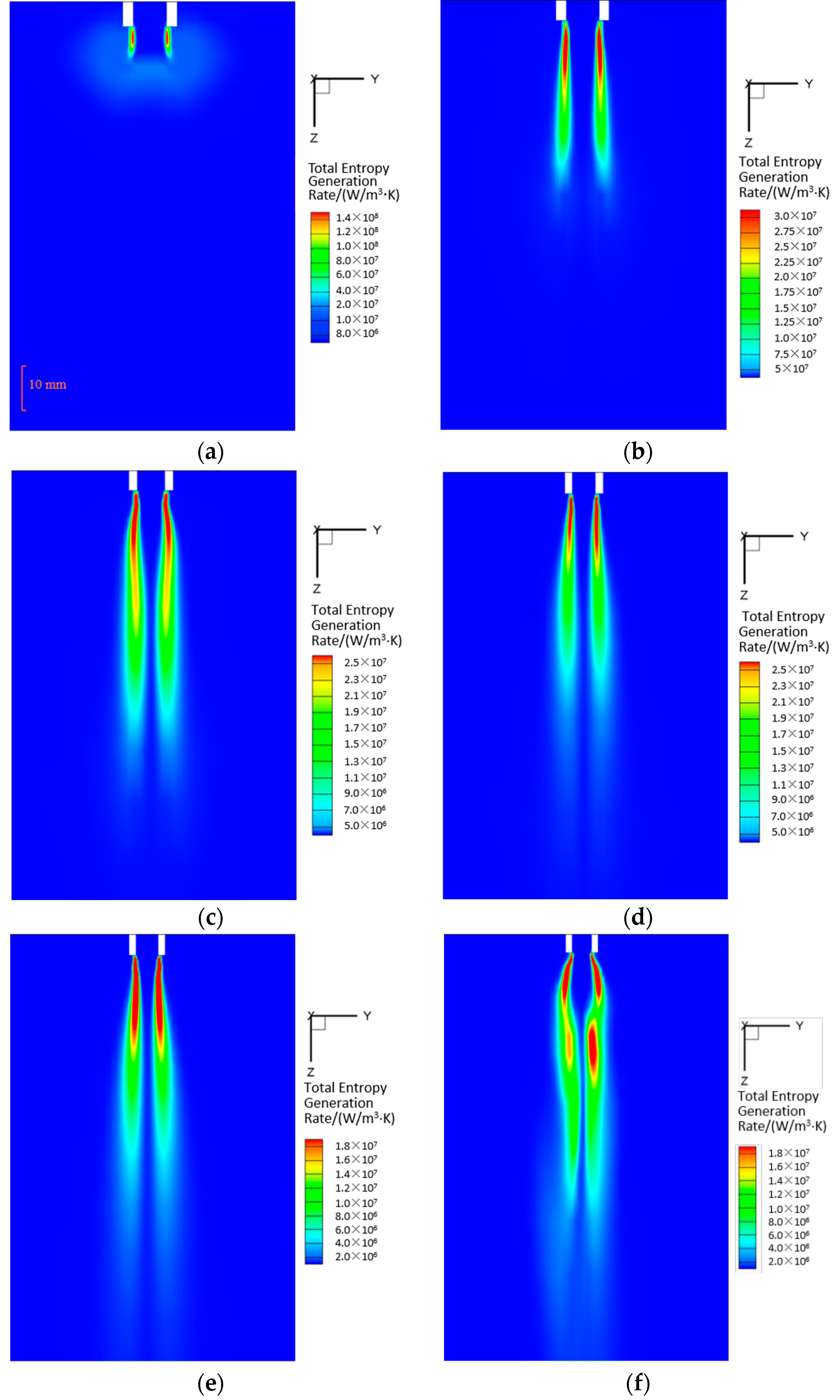
5.3. Entropy Generation
| Time/(ms) | EGR_Heat Transfer/(W/K) | EGR_Viscous/(W/K) | EGR_Turbulence/(W/K) | EGR_Inner Phase Change /(W/K) | Total EGR /(W/K) |
|---|---|---|---|---|---|
| 2 | 0.04728 | 0.00016 | 12750.01000 | 0.74853 | 12750.8080 |
| 4 | 0.52859 | 0.00031 | 1361.27000 | 2.90208 | 1364.7051 |
| 8 | 12.97281 | 0.00090 | 109.47290 | 5.96174 | 128.4084 |
| 12 | 27.41757 | 0.00099 | 28.85747 | 8.35380 | 64.6298 |
| 16 | 29.43516 | 0.00087 | 12.32487 | 9.37835 | 51.1393 |
| 20 | 31.36492 | 0.00103 | 7.30897 | 6.73753 | 45.4124 |
| 24 | 30.30458 | 0.00086 | 4.67426 | 9.81291 | 44.7926 |
| 28 | 30.89448 | 0.00086 | 3.51669 | 9.73798 | 44.1500 |
| 32 | 31.55728 | 0.00087 | 2.87975 | 9.67914 | 44.1170 |
| 36 | 32.04326 | 0.00087 | 2.49488 | 9.65655 | 44.1956 |
| 40 | 32.34219 | 0.00087 | 2.24267 | 9.65505 | 44.2471 |
| 44 | 32.49960 | 0.00087 | 2.06793 | 9.66349 | 44.2319 |
| 64 | 31.01836 | 0.00079 | 0.89096 | 9.67850 | 41.5886 |
| 140 | 32.90304 | 0.00065 | 0.53704 | 9.93461 | 43.3753 |
| 180 | 30.86176 | 0.00062 | 0.42636 | 9.48106 | 40.7698 |
6. Conclusions
- (1)
- (2)
- Three distinct stages of DCC are discriminated clearly at the present conditions, i.e., initial stage, developing stage and oscillatory stage. In the initial stage, the plume shows no fixed shape. In the developing stage, the plume begins to act as an elliptical boundary, and the size of the plume grows quickly. In the oscillatory stage, the plume shape becomes ellipsoidal shape with disturbed structure.
- (3)
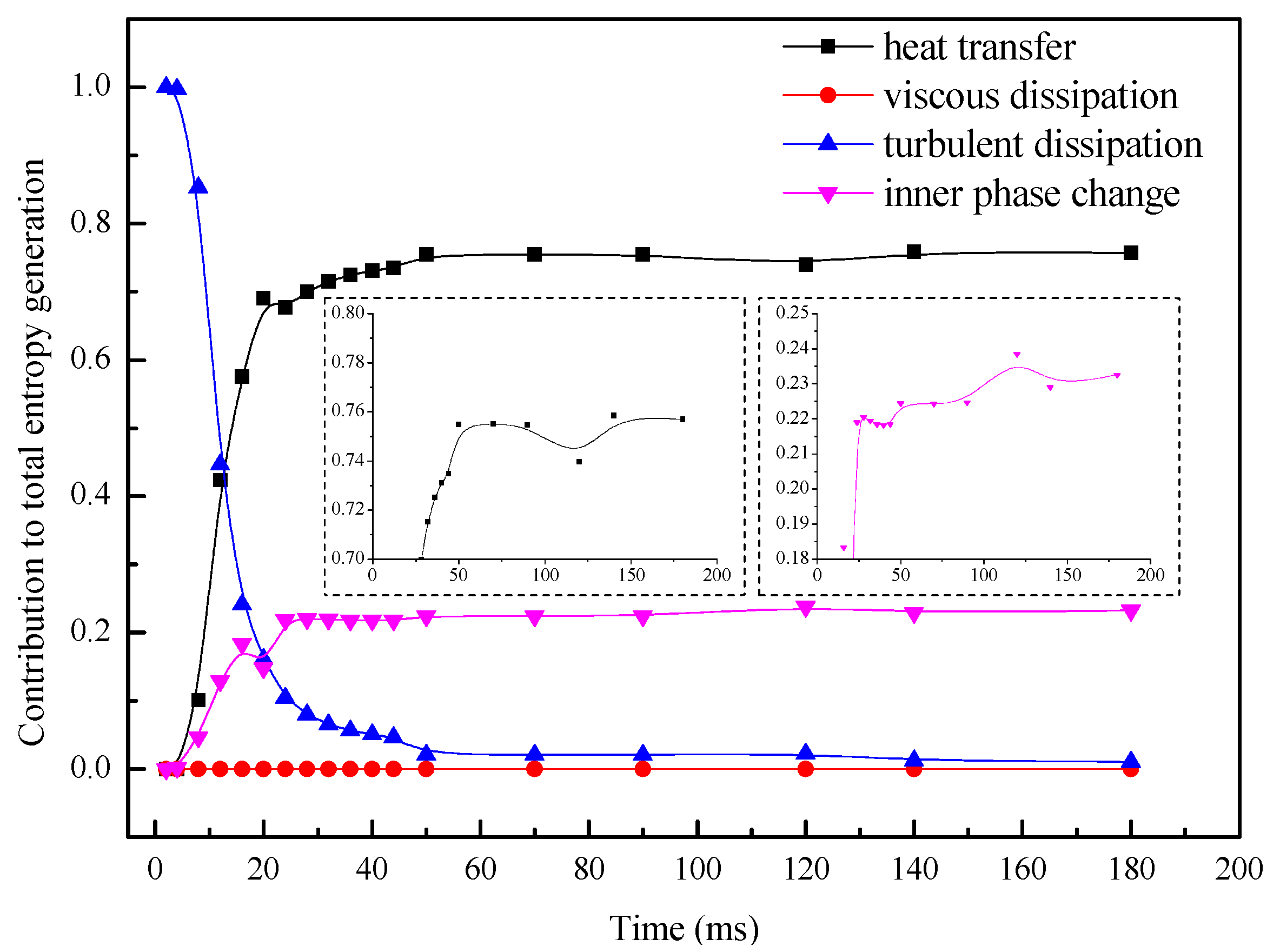
- The local volumetric EGR in the initial stage is much larger than those in other stages, but the region possessing considerable entropy generation rate is smaller than other stage. The decrease of EGR proves that the process conform to increasingly economical energy utilization.
- (4)
- The largest proportion in total EGR is occupied by turbulence fluctuation in the initial stage, and then it decreases apparently in the following time, meanwhile, the contributions of heat transfer irreversibility and inner phase change irreversibility to the local entropy generation increase, which makes DCC process become heat dominant in the developing and the oscillatory stage. The variation of EGR can be used to characterize the the dissipation and proceeding of DCC process.
Acknowledgments
Author Contributions
Conflicts of Interest
Nomenclature
| A | interfacial area per unit volume, m2/m3 |
| Cμ | parameter in the turbulent model |
| C1ε | parameter in the turbulent model |
| C2ε | parameter in the turbulent model |
| dp | diameter of dispersed phase, m |
| E | total energy, J |
| Fdr | interaction force between phases, N/m3 |
| g | gravitational acceleration vector, m/s2 |
| j | evaporation-condensation flux, kg/m2·s |
| J | volumetric phase change rate, kg/m3·s |
| k | turbulent kinetic energy, m2/s2 |
| L | latent heat, J/kg |
| M | molar mass, kg/mol |
| p | pressure, Pa |
| T | temperature, K |
| v | specific volume, m3/kg |
| v | mean velocity, m/s |
Greek Letters
| α | volume fraction |
| γ | factor characterizing intensity of evaporation and condensation, m3/s |
| ε | turbulent energy dissipated per unit mass, m2/s3 |
| κeff | effective thermal conductivity, W/m·K |
| μ | viscosity, kg/m·s |
| ρ | density, kg/m3 |
Subscripts and Superscripts
| c | condensation |
| e | evaporation |
| g | vapor |
| l | liquid |
| m | mixture |
| q | qth phase |
| sat | saturated state |
| T | transpose matrix |
| + | condensation process |
| - | evaporation process |
Abbreviations
| CFD | computational fluid dynamics |
| DCC | direct contact condensation |
| EGR | entropy generation rate |
| HTC | heat transfer coefficient |
References
- Kerney, P.J.; Faeth, G.M.; Olson, D.R. Penetration characteristics of a submerged steam jet. AIChE J. 1972, 18, 548–553. [Google Scholar] [CrossRef]
- Weimer, J.C.; Faeth, G.M.; Olson, D.R. Penetration of vapor jets submerged in subcooled liquids. AIChE J. 1973, 19, 552–558. [Google Scholar] [CrossRef]
- Aya, I.; Nariai, H. Evaluation of heat-transfer coefficient at direct-contact condensation of cold water and steam. Nucl. Eng. Des. 1991, 131, 17–24. [Google Scholar] [CrossRef]
- Chan, C.K.; Lee, C.K.B. A regime map for direct contact condensation. Int. J. Multiph. Flow 1982, 8, 11–20. [Google Scholar] [CrossRef]
- Gulawani, S.S.; Joshi, J.; Shah, M.; RamaPrasad, C.S.; Shukla, D.S. CFD analysis of flow pattern and heat transfer in direct contact steam condensation. Chem. Eng. Sci. 2006, 61, 5204–5220. [Google Scholar] [CrossRef]
- Gulawani, S.S.; Dahikar, S.K.; Mathpati, C.S.; Joshi, J.B.; Shah, M.S.; RamaPrasad, C.S.; Shukla, D.S. Analysis of flow pattern and heat transfer in direct contact condensation. Chem. Eng. Sci. 2009, 64, 1719–1738. [Google Scholar] [CrossRef]
- Dahikar, S.K.; Sathe, M.J.; Joshi, J.B. Investigation of flow and temperature patterns in direct contact condensation using PIV, PLIF and CFD. Chem. Eng. Sci. 2010, 65, 4606–4620. [Google Scholar] [CrossRef]
- Shah, A.; Chughtai, I.R.; Inayat, M.H. Numerical simulation of direct-contact condensation from a supersonic steam jet in subcooled water. Chin. J. Chem. Eng. 2010, 18, 577–587. [Google Scholar] [CrossRef]
- Li, S.Q.; Wang, P.; Lu, T. Numerical simulation of direct contact condensation of subsonic steam injected in a water pool using VOF method and LES turbulence model. Prog. Nucl. Energy 2015, 78, 201–215. [Google Scholar] [CrossRef]
- Torabi, M.; Zhang, K. Classical entropy generation analysis in cooled homogenous and functionally graded material slabs with variation of internal heat generation with temperature, and convective-radiative boundary conditions. Energy 2014, 65, 387–397. [Google Scholar] [CrossRef]
- Hassan, M.; Sadri, R.; Ahmadi, G.; Ahmadi, G.; Dahari, M.B.; Kazi, S.N.; Safaei, M.R.; Sadeghinezhad, E. Numerical study of entropy generation in a flowing nanofluid used in micro-and minichannels. Entropy 2013, 15, 144–155. [Google Scholar] [CrossRef]
- Makinde, O.D.; Eegunjobi, A.S. Entropy generation in a couple stress fluid flow through a vertical channel filled with saturated porous media. Entropy 2013, 15, 4589–4606. [Google Scholar] [CrossRef]
- Seo, J.W.; Kim, Y.H.; Kim, D.; Choi, Y.D.; Lee, K.J. Heat Transfer and Pressure Drop Characteristics in Straight Microchannel of Printed Circuit Heat Exchangers. Entropy 2015, 17, 3438–3457. [Google Scholar] [CrossRef]
- Cho, C.C.; Yau, H.T.; Chiu, C.H.; Chiu, K.C. Numerical Investigation into Natural Convection and Entropy Generation in a Nanofluid-Filled U-Shaped Cavity. Entropy 2015, 17, 5980–5994. [Google Scholar] [CrossRef]
- Bejan, A. Entropy Generation Minimization: The Method of Thermodynamic Optimization oF Finite Size System and Finite-Time Processes; CRC Press: Boca Raton, FL, USA, 1995. [Google Scholar]
- Bejan, A. Entropy Generation Through Heat and Fluid Flow; Wiley: New York, NY, USA, 1982. [Google Scholar]
- Vargas, J.V.C.; Bejan, A. Thermodynamic optimization of the match between two streams with phase change. Energy 2000, 25, 15–33. [Google Scholar] [CrossRef]
- Goudarzi, N.; Talebi, S. An approach to stability analysis and entropy generation minimization in the single-phase natural circulation loops. Energy 2015, 80, 213–226. [Google Scholar] [CrossRef]
- Lior, N.; Sarmiento, D.W.; Al-Sharqawi, H.S. The exergy fields in transport processes: Their calculation and use. Energy 2006, 31, 553–578. [Google Scholar] [CrossRef]
- Adeyinka, O.B.; Naterer, G.F. Optimization correlation for entropy production and energy availability in film condensation. Int. Commun. Heat Mass Transf. 2004, 31, 513–524. [Google Scholar] [CrossRef]
- Thiel, G.P.; Lienhard, J.H. Entropy generation in condensation in the presence of high concentrations of noncondensable gases. Int. J. Heat Mass Transf. 2012, 55, 5133–5147. [Google Scholar] [CrossRef]
- Revellin, R.; Lips, S.; Khandekar, S.; Bonjour, J. Local entropy generation for saturated two-phase flow. Energy 2009, 34, 1113–1121. [Google Scholar] [CrossRef]
- Herwig, H.; Schmandt, B. How to Determine Losses in a Flow Field: A Paradigm Shift towards the Second Law Analysis. Entropy 2014, 16, 2959–2989. [Google Scholar] [CrossRef]
- Wenterodt, T.; Herwig, H. The Entropic Potential Concept: A New Way to Look at Energy Transfer Operations. Entropy 2014, 16, 2071–2084. [Google Scholar] [CrossRef]
- Fluent Inc. Fluent 14.0 Theory Guide; Fluent Inc.: Pittsburgh, PA, USA, 2012. [Google Scholar]
- Marek, R.; Straub, J. Analysis of the evaporation coefficient and the condensation coefficient of water. Int. J. Heat Mass Transf. 2001, 44, 39–53. [Google Scholar] [CrossRef]
- Zhang, X.B.; Li, J.F.; Zhu, J.K.; Qiu, L.M. Computational fluid dynamics study on liquefied natural gas dispersion with phase change of water. Int. J. Heat Mass Transf. 2015, 91, 347–354. [Google Scholar] [CrossRef]
- Knudsen, M. Die maximale verdampfungsgeschwindigkeit des quecksilbers. Ann. Phys. 1915, 352, 697–708. [Google Scholar] [CrossRef]
- Pru, W. Verdampfungsvorgang. Forsch. Gebiete Ing. 1941, 12, 258–260. [Google Scholar]
- Jou, D.; Casas-Vázquez, J.; Lebon, G. Extended Irreversible Thermodynamics; Springer: Berlin/Heidelberg, Germany, 1996. [Google Scholar]
- Kock, F.; Herwig, H. Local entropy production in turbulent shear flows: A high-Reynolds number model with wall functions. Int. J. Heat Mass Transf. 2004, 47, 2205–2215. [Google Scholar] [CrossRef]
- Patankar, S.V. Numerical Heat Transfer and Fluid Flow; McGraw-Hill: New York, NY, USA, 1981. [Google Scholar]
- Narusawa, U.; Springer, G.S. Measurement of evaporation rates of water. J. Colloid Interface Sci. 1975, 50, 392–395. [Google Scholar] [CrossRef]
- Nabavian, K.; Bromley, L.A. Condensation coefficient of water. Chem. Eng. Sci. 1963, 18, 651–660. [Google Scholar] [CrossRef]
- Takase, K.; Ose, Y.; Kunugi, T. Numerical study on direct-contact condensation of vapor in cold water. Fusion Eng. Des. 2002, 63, 424–428. [Google Scholar] [CrossRef]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ji, Y.; Zhang, H.-C.; Tong, J.-F.; Wang, X.-W.; Wang, H.; Zhang, Y.-N. Entropy Assessment on Direct Contact Condensation of Subsonic Steam Jets in a Water Tank through Numerical Investigation. Entropy 2016, 18, 21. https://doi.org/10.3390/e18010021
Ji Y, Zhang H-C, Tong J-F, Wang X-W, Wang H, Zhang Y-N. Entropy Assessment on Direct Contact Condensation of Subsonic Steam Jets in a Water Tank through Numerical Investigation. Entropy. 2016; 18(1):21. https://doi.org/10.3390/e18010021
Chicago/Turabian StyleJi, Yu, Hao-Chun Zhang, Jian-Fei Tong, Xu-Wei Wang, Han Wang, and Yi-Ning Zhang. 2016. "Entropy Assessment on Direct Contact Condensation of Subsonic Steam Jets in a Water Tank through Numerical Investigation" Entropy 18, no. 1: 21. https://doi.org/10.3390/e18010021
APA StyleJi, Y., Zhang, H.-C., Tong, J.-F., Wang, X.-W., Wang, H., & Zhang, Y.-N. (2016). Entropy Assessment on Direct Contact Condensation of Subsonic Steam Jets in a Water Tank through Numerical Investigation. Entropy, 18(1), 21. https://doi.org/10.3390/e18010021