Estimating the Lower Limit of the Impact of Amines on Nucleation in the Earth’s Atmosphere
Abstract
:1. Introduction
2. Methods
3. Results and Discussion
3.1. Equilibrium Geometries and Cluster Properties
3.2. New Thermochemical Data and Their Analysis
3.2.1. Hydration
3.2.2. Affinity of H2SO4 to Nucleating (H2SO4)m(CH3NH2)n(H2O)k Clusters
3.2.3. Affinity of CH3NH2 to Nucleating (H2SO4)m(CH3NH2)n(H2O)k Clusters
3.3. Impacts of the MA on Formation of Nucleating Clusters under Atmospheric Conditions
4. Conclusions
- (H2SO4)m(CH3NH2)n(H2O)k clusters are strongly hydrated under typical atmospheric conditions and, thus, (H2SO4)m(CH3NH2)n(H2O)k formation rates are RH-dependent. The effect of the MA depends strongly on the RH and the ambient temperature. Low RH and low ambient temperatures are clearly the favorable conditions, at which the strongest effect of MA on the formation of stable binary H2SO4-H2O clusters is achieved. While the enhancing effect is very strong at low RH, it decreases quickly as RH is growing;
- At the identical concentrations of MA and DMA, the enhancement in the production of stable (H2SO4)m(H2O)k due to MA is close to that due to DMA;
- The MA ternary nucleation begins to dominate over ternary nucleation of sulfuric acid, water and ammonia at [MA]/[NH3] > ~10−3.
Supplement
Acknowledgments
Author Contributions
References
- Zhang, R. Getting to the Critical Nucleus of Aerosol Formation. Science 2010, 328, 1366–1367. [Google Scholar]
- Yu, F.; Luo, G. Simulation of Particle Size Distribution with a Global Aerosol Model: Contribution of Nucleation to Aerosol and CCN Number Concentrations. Atmos. Chem. Phys. 2009, 9, 7691–7710. [Google Scholar]
- Paasonen, P.; Asmi, A.; Petäjä, T.; Kajos, M.K.; Äijälä, M.; Junninen, H.; Holst, T.; Abbatt, J.P.D.; Arneth, A.; Birmili, W.; et al. Warming-induced increase in aerosol number concentration likely to moderate climate change. Nature Geosci. 2013, 6, 438–442. [Google Scholar]
- Yu, F.; Hallar, G. Difference in Particle Formation at a Mountain-Top Location during the Spring and Summer: Implications for the Role of Sulfuric Acid and Organics in Nucleation. J. Geophys. Res. 2014, 119, 12–246. [Google Scholar]
- Knaapen, M.; Borm, P.; Albrecht, C.; Schins, R.P.F. Inhaled Particles and Lung Cancer. Part A: Mechanisms. Int. J. Cancer. 2004, 109, 799–809. [Google Scholar]
- Saxon, D.; Diaz-Sanchez, D. Air pollution and allergy: You are what you breathe. Nature Immunol. 2005, 6, 223–226. [Google Scholar]
- Zhang, R.; Khalizov, A.F.; Wang, L.; Hu, M.; Wen, X. Nucleation and Growth of Nanoparticles in the Atmosphere. Chem. Rev. 2012, 112. [Google Scholar] [CrossRef]
- Flood, H. Tröpfchenbildung in übersättigten Äthylalkohol-wasserdampfgemischen. Z Phys Chem A 1934, 170, 286–295. [Google Scholar]
- Reiss, H. The Kinetics of Phase Transitions in Binary Systems. J. Chem. Phys. 1950, 18, 840–849. [Google Scholar]
- Kulmala, M.; Pirjola, L.; Makela, J.M. Stable Sulphate Clusters as a Source of New Atmospheric Particles. Nature 2000, 404, 66–69. [Google Scholar]
- Yu, F.; Turco, R.P. Ultrafine Aerosol Formation via Ion-Mediated Nucleation. Geophys. Res. Lett. 2000, 27, 883–886. [Google Scholar]
- Yu, F.; Luo, G. Effect of Solar Variations on Particle Formation and Cloud Condensation Nuclei. Environ. Res. Lett. 2014, 9, 045004. [Google Scholar]
- Kirkby, J.; Curtius, J.; Almeida, J.; Dunne, E.; Duplissy, J.; Ehrhart, S.; Franchin, A.; Gagné, S.; Ickes, L.; Kürten, A.; et al. Role of Sulphuric Acid, Ammonia and Galactic Cosmic Rays in Atmospheric Aerosol Nucleation. Nature 2011, 476, 429–433. [Google Scholar]
- Napari, I.; Noppel, M.; Vehkamaki, H.; Kulmala, M. An Improved Model for Ternary Nucleation of Sulfuric Acid-ammonia-water. J. Chem. Phys. 2002, 116. [Google Scholar] [CrossRef]
- Anttila, T.; Vehkamaki, H.; Napari, I.; Kulmala, M. Effect of Ammonium Bisulphate Formation on Atmospheric Water-sulphuric Acid-ammonia Nucleation. Boreal Environ. Res. 2005, 10, 511–523. [Google Scholar]
- Yu, F. Effect of Ammonia on New Particle Formation: A Kinetic H2SO4-H2O-NH3 Nucleation Model Constrained by Laboratory Measurements. J. Geophys. Res. 2006, 111. [Google Scholar] [CrossRef]
- Ball, S.M.; Hanson, D.R.; Eisele, F.L.; McMurry, P.H. Laboratory Studies of Particle Nucleation: Initial Results for H2SO4, H2O, and NH3 Vapors. J. Geophys. Res. 1999, 104, 23709–23718. [Google Scholar]
- Benson, D.R.; Erupe, M.E.; Lee, S.-H. Laboratory-measured H2SO4-H2O-NH3 Ternary Homogeneous Nucleation Rates: Initial Observations. Geophys. Res. Lett. 2009, 36, L15818. [Google Scholar]
- Loukonen, V.; Kurten, T.; Ortega, I.K.; Vehkamaki, H.; Padua, A.A.H.; Sellegri, K.; Kulmala, M. Enhancing Effect of Dimethylamine in Sulfuric Acidnucleation in the Presence of Water—a Computational Study. Atmos. Chem. Phys. 2010, 10, 4961–4974. [Google Scholar]
- Nadykto, A.B.; Yu, F.; Jakovleva, M.; Herb, J.; Xu, Y. Amines in the Earth’s Atmosphere: A Density Functional Theory Study of the Thermochemistry of Pre-Nucleation Clusters. Entropy 2011, 13, 554–569. [Google Scholar]
- Ortega, I.K.; Kupiainen, O.; Kurten, T.; Olenius, T.; Wilkman, O.; McGrath, M.J.; Loukonen, V.; Vehkamaki, H. From quantum chemical formation free energies to evaporation rates. Atmos. Chem. Phys. 2012, 12, 225–235. [Google Scholar] [CrossRef]
- Nadykto, A.B.; Herb, J.; Yu, F.; Xu, Y. Enhancement in the Production of Nucleating Clusters due to Dimethylamine and Large Uncertainties in the Thermochemistry of Amine-enhanced Nucleation. Chem. Phys. Lett. 2014, 609, 42–49. [Google Scholar]
- Almeida, J.; Schobesberger, S.; Kürten, A.; Ortega, I.K.; Kupiainen-Määttä, O.; Praplan, A.P.; Adamov, A.; Amorim, A.; Bianchi, F.; Breitenlechner, M.; et al. Molecular Understanding of Sulphuric Acid-amine Particle Nucleation in the Atmosphere. Nature 2013, 502, 359–363. [Google Scholar]
- Zhang, R.Y.; Suh, I.; Zhao, J.; Zhang, D.; Fortner, E.C.; Tie, X.X.; Molina, L.T.; Molina, M.J. Atmospheric New Particle Formation Enhanced by Organic acids. Science 2004, 304, 1487–1490. [Google Scholar]
- Nadykto, A.B.; Yu, F. Strong Hydrogen Bonding between Atmospheric Nucleation Precursors and Common Organics. Chem. Phys. Lett. 2007, 435. [Google Scholar] [CrossRef]
- Nadykto, A.B.; Al Natsheh, A.; Yu, F.; Mikkelsen, K.V.; Herb, J. Computational Quantum Chemistry: A New Approach to Atmospheric Nucleation. Adv. Quant. Chem. 2008, 55, 449–478. [Google Scholar]
- Xu, Y.-S.; Nadykto, A.B.; Yu, F.; Jiang, L.; Wang, L. Formation and Properties of Hydrogen-bonded Complexes of Common Organic Oxalic Acid with Atmospheric Nucleation Precursors. J. Mol. Struct. Theochem. 2010, 951, 28–33. [Google Scholar]
- Xu, Y.-S.; Nadykto, A.B.; Yu, F.; Jiang, L.; Wang, L. Interaction between Common Organic Acids and Trace Nucleation Species in the Earth’s Atmosphere. J. Phys. Chem. A 2010, 114. [Google Scholar] [CrossRef]
- Zhao, J.; Khalizov, A.; Zhang, R.; McGraw, R. Hydrogen-bonding Interaction in Molecular Complexes and Clusters of Aerosol Nucleation Precursors. J. Phys. Chem. A 2009, 113, 680–689. [Google Scholar]
- Xu, W.; Zhang, R. A Theoretical Study of Hydrated Molecular Clusters of Amines and Dicarboxylic Acids. J. Chem. Phys. 2013, 139, 064312. [Google Scholar]
- Elm, J.; Kurtén, T.; Bilde, M.; Mikkelsen, K.V. Molecular Interaction of Pinic Acid with Sulfuric Acid: Exploring the Thermodynamic Landscape of Cluster Growth. J. Phys. Chem. A 2014, 118, 7892–7900. [Google Scholar]
- Elm, J.; Jørgensen, S.; Bilde, M.; Mikkelsen, K.V. Ambient Reaction Kinetics of Atmospheric Oxygenated Organics with the OH Radical: A Computational Methodology Study. Phys. Chem. Chem. Phys. 2013, 15, 9636–9645. [Google Scholar]
- Ge, X.; Wexler, A.S.; Clegg, S.L. Atmospheric Amines—Part II. Thermodynamic Properties and Gas/Particle Partitioning. Atmos. Environ. 2010, 45, 561–577. [Google Scholar]
- You, Y.; Kanawade, V.P.; de Gouw, J.A.; Guenther, A.B.; Madronich, S.; Sierra-Hernández, M.R.; Lawler, M.; Smith, J.N.; Takahama, S.; Ruggeri, G.; et al. Atmospheric amines and ammonia measured with a chemical ionization mass spectrometer (CIMS). Atmos. Chem. Phys. 2014, 14, 12181–12194. [Google Scholar]
- Hertel, O.; Reis, S.; Skjøth, C.A.; Bleeker, A.; Harrison, R.; Cape, J.N.; Fowler, D.; Skiba, U.; Simpson, D.; Jickells, T.; Baker, A.; Kulmala, M.; et al. Nitrogen Processes in the Atmosphere. Philos. Trans. R. Soc. 2013, 368, 177–207. [Google Scholar]
- Lee, D.; Wexler, A.S. Atmospheric amines—Part III: Photochemistry and Toxicity. Atmos. Environ. 2013, 71, 95–103. [Google Scholar]
- Ge, X.; Wexler, A.S.; Clegg, S.L. Atmospheric Amines—Part I. a Review. Atmos. Environ. 2010, 45, 524–546. [Google Scholar]
- Finlayson-Pitts, B.J.; Pitts, J.N. Chemistry of the Upper and Lower Atmosphere: Theory, Experiments, and Applications; Academic Press: San Diego, CA, USA, 2000. [Google Scholar]
- Nielsen, C.J.; D’Anna, B.; Karl, M.; Aursnes, M.; Boreave, A.; Bossi, R.; Bunkan, A.J.C.; Glasius, M.; Hallquist, M.; Hansen, A.M.K.; et al. Summary Report: Photo-Oxidation of Methylamine; Dimethylamine and Trimethylamine CLIMIT Project no. 201604; Norwegian Institute for Air Research: Kjeller, Norway, 2011. [Google Scholar]
- Akyüz, M. Simultaneous Determination of Aliphatic and Aromatic Amines in Indoor and Outdoor Air Samples by Gas Chromatography-Mass Spectrometry. Talanta 2007, 71, 486–492. [Google Scholar]
- Dawson, M.L.; Perraud, V.; Gomez, A.; Arquero, K.D.; Ezell, M.J.; Finlayson-Pitts, B.J. Measurement of Gas-phase Ammonia and Amines in Air by Collection onto an Ion Exchange Resin and Analysis by Ion Chromatography. Atmos. Meas. Tech. Discuss. 2014, 7, 1573–1602. [Google Scholar]
- Yu, H.; Lee, S.-H. A Chemical Ionization Mass Spectrometer for the Detection of Atmospheric Amines. Environ. Chem. 2012, 9, 190–201. [Google Scholar]
- Benson, D.R.; Markovich, A.; Al-Refai, M.; Lee, S.-H. A Chemical Ionization Mass Spectrometer for Ambient Measurements of Ammonia. Atmos. Meas. Tech. 2010, 3, 1075–1087. [Google Scholar]
- Erupe, M.E.; Benson, D.R.; Li, J.; Young, L.-H.; Verheggen, B.; Al-Refai, M.; Tahboub, O.; Cunningham, V.; Frimpong, F.; Viggiano, A.A.; Lee, S.-H. Correlation of Aerosol Nucleation Rate with Sulfuric Acid and Ammonia in Kent Ohio: an Atmospheric Observation. J. Geophys. Res. 2010, 115, D23216. [Google Scholar]
- Nowak, J.B.; Neuman, J.A.; Bahreini, R.; Brock, C.A.; Middlebrook, A.M.; Wollny, A.G.; Holloway, J.S.; Peischl, J.; Ryerson, T.B.; Fehsenfeld, F.C. Airborne Observations of Ammonia and Ammonium Nitrate Formation over Houston, Texas. J. Geophys. Res. 2010, 115. [Google Scholar] [CrossRef]
- Yu, H.; McGraw, R.; Lee, S.-H. Effects of Amines on Formation of Sub-3nm Particles and their Subsequent Growth. Geophys. Res. Lett. 2012, 39, L02807. [Google Scholar]
- Erupe, M.E.; Viggiano, A.A.; Lee, S.-H. The Effect of Trimethylamine on Atmospheric Nucleation Involving H2SO4. Atmos. Chem. Phys. 2011, 11, 4767–4775. [Google Scholar]
- Berndt, T.; Sipilä, M.; Stratmann, F.; Petäjä, T.; Vanhanen, J.; Mikkilä, J.; Patokoski, J.; Taipale, R.; Mauldin, R.L.; Kulmala, M. Enhancement of Atmospheric H2SO4/H2O Nucleation: Organic Oxidation Products versus Amines. Atmos. Chem. Phys. 2014, 14, 751–764. [Google Scholar]
- Zollner, J.H.; Glasoe, W.A.; Panta, B.; Carlson, K.K.; McMurry, P.H.; Hanson, D.R. Sulfuric Acid Nucleation: Power Dependencies, Variation with Relative Humidity, and Effect of Bases. Atmos. Chem. Phys. 2012, 12, 4399–4411. [Google Scholar]
- Jen, C.N.; McMurry, P.H.; Hanson, D.R. Stabilization of Sulfuric Acid Dimers by Ammonia, Methylamine, Dimethylamine, and Trimethylamine. J. Geophys. Res. Atmos. 2014, 119, 7502–7514. [Google Scholar]
- Qiu, C.; Zhang, R. Multiphase Chemistry of Atmospheric Amines. Phys. Chem. Chem. Phys. 2013, 15, 5738–5752. [Google Scholar]
- Al Natsheh, A.; Nadykto, A.B.; Mikkelsen, K.V.; Yu, F.; Ruuskanen, J. Quantum Nature of the Sign Preference in Ion-Induced Nucleation. J. Phys. Rev. Lett. 2006, 96, 125701. [Google Scholar]
- Nadykto, A.B.; Yu, F.; Herb, J. Towards Understanding the Sign Preference in Binary Atmospheric Nucleation. Phys. Chem. Chem. Phys. 2008, 10, 7073–7078. [Google Scholar]
- Du, H.; Nadykto, A.B.; Yu, F. Quantum-mechanical Solution to Fundamental Problems of Classical Theory of Water Vapor Nucleation. Phys. Rev. E 2009, 79, 021604. [Google Scholar]
- Hanson, D.R.; Lovejoy, E.R. Measurement of the Thermodynamics of the Hydrated Dimer and Trimer of Sulfuric Acid. J. Phys. Chem. A 2006, 110, 9525–9528. [Google Scholar]
- Froyd, K.D.; Lovejoy, E.R. Experimental Thermodynamics of Cluster Ions Composed of H2SO4 and H2O Positive Ions. J. Phys. Chem. A 2003, 107, 9812–9824. [Google Scholar]
- Temelso, B.; Ngoc Phan, T.; Shields, G.C. Computational Study of the Hydration of Sulfuric Acid Dimers: Implications for Acid Dissociation and Aerosol Formation. J. Phys. Chem. A 2012, 116, 9745–9758. [Google Scholar]
- Bustos, D.J.; Temelso, B.; Shields, G.C. Hydration of the Sulfuric Acid-Methylamine Complex and Implications for Aerosol Formation. J. Phys. Chem. A 2014, 118, 7430–7441. [Google Scholar]
- Husar, D.E.; Temelso, B.; Ashworth, A.L.; Shields, G.C. Hydration of the Bisulfate ion: Atmospheric Implications. J. Phys. Chem. A 2012, 116, 5151–5163. [Google Scholar]
- Gaussian 09; Revision D.01; Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Mennucci, B.; Petersson, G.A.; Nakatsuji, H.; Caricato, M.; Li, X.; Hratchian, H.P.; Izmaylov, A.F.; Bloino, J.; Zheng, G.; Sonnenberg, J.L.; Hada, M.; Ehara, M.; Toyota, K.; Fukuda, R.; Hasegawa, J.; Ishida, M.; Nakajima, T.; Honda, Y.; Kitao, O.; Nakai, H.; Vreven, T.; Montgomery, J.A., Jr.; Peralta, J.E.; Ogliaro, F.; Bearpark, M.; Heyd, J.J.; Brothers, E.; Kudin, K.N.; Staroverov, V.N.; Kobayashi, R.; Normand, J.; Raghavachari, K.; Rendell, A.; Burant, J.C.; Iyengar, S.S.; Tomasi, J.; Cossi, M.; Rega, N.; Millam, J.M.; Klene, M.; Knox, J.E.; Cross, J.B.; Bakken, V.; Adamo, C.; Jaramillo, J.; Gomperts, R.; Stratmann, R.E.; Yazyev, O.; Austin, A.J.; Cammi, R.; Pomelli, C.; Ochterski, J.W.; Martin, R.L.; Morokuma, K.; Zakrzewski, V.G.; Voth, G.A.; Salvador, P.; Dannenberg, J.J.; Dapprich, S.; Daniels, A.D.; Farkas, Ö.; Foresman, J.B.; Ortiz, J.V.; Cioslowski, J.; Fox, D.J. Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Herb, J.; Nadykto, A.B.; Yu, F. Large Ternary Hydrogen-bonded Pre-nucleation Clusters in the Earth’s Atmosphere. Chem. Phys. Lett. 2011, 518, 7–14. [Google Scholar]
- Paasonen, P.; Olenius, T.; Kupiainen, O.; Kurten, T.; Petaja, T.; Birmili, W.; Hamed, A.; Hu, M.; Huey, L.G.; Plass-Duelmer, C.; et al. On the Formation of Sulphuric Acid—Amine Clusters in Varying Atmospheric Conditions and its Influence on Atmospheric new Particle Formation. Atmos. Chem. Phys. 2012, 12, 9113–9133. [Google Scholar]
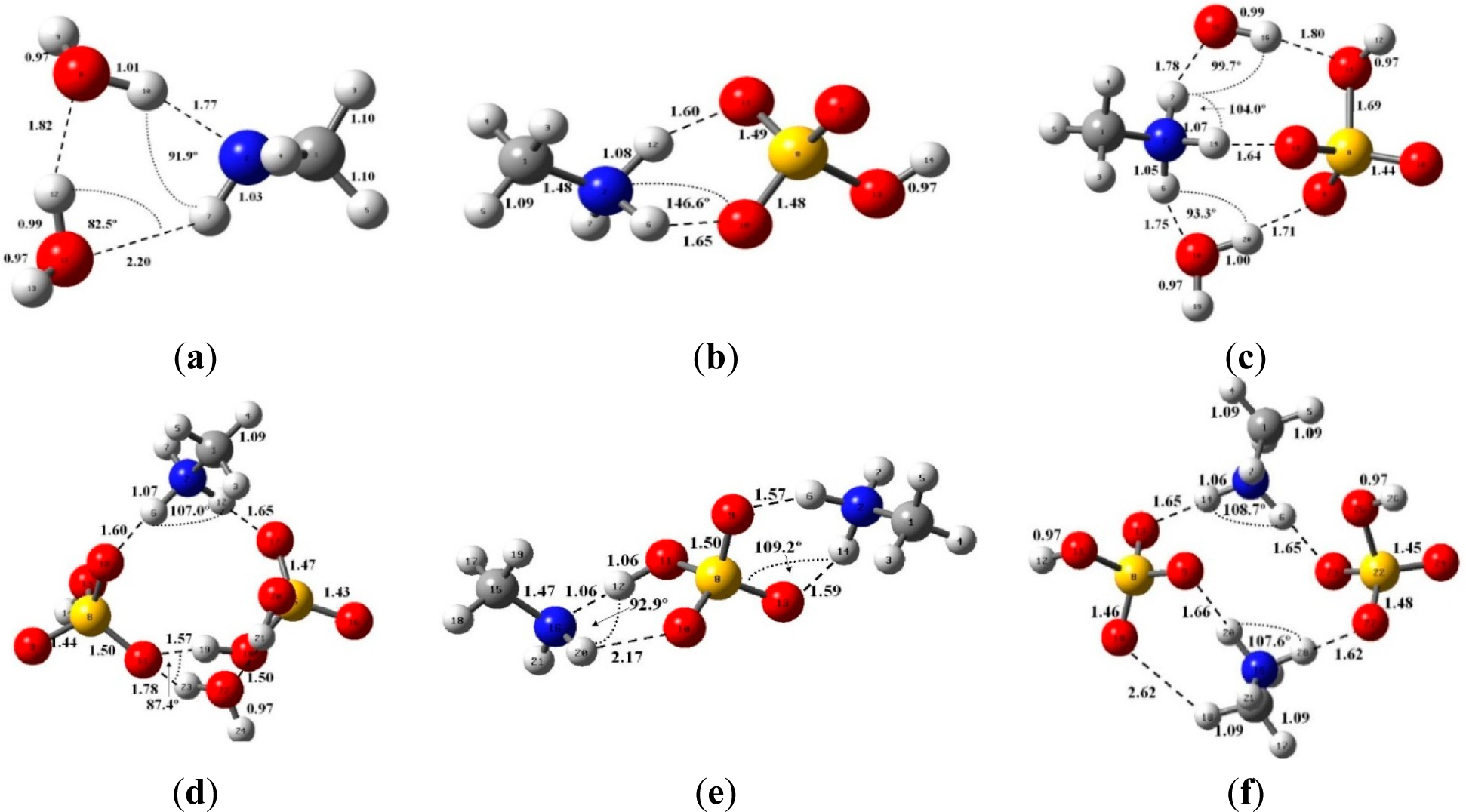
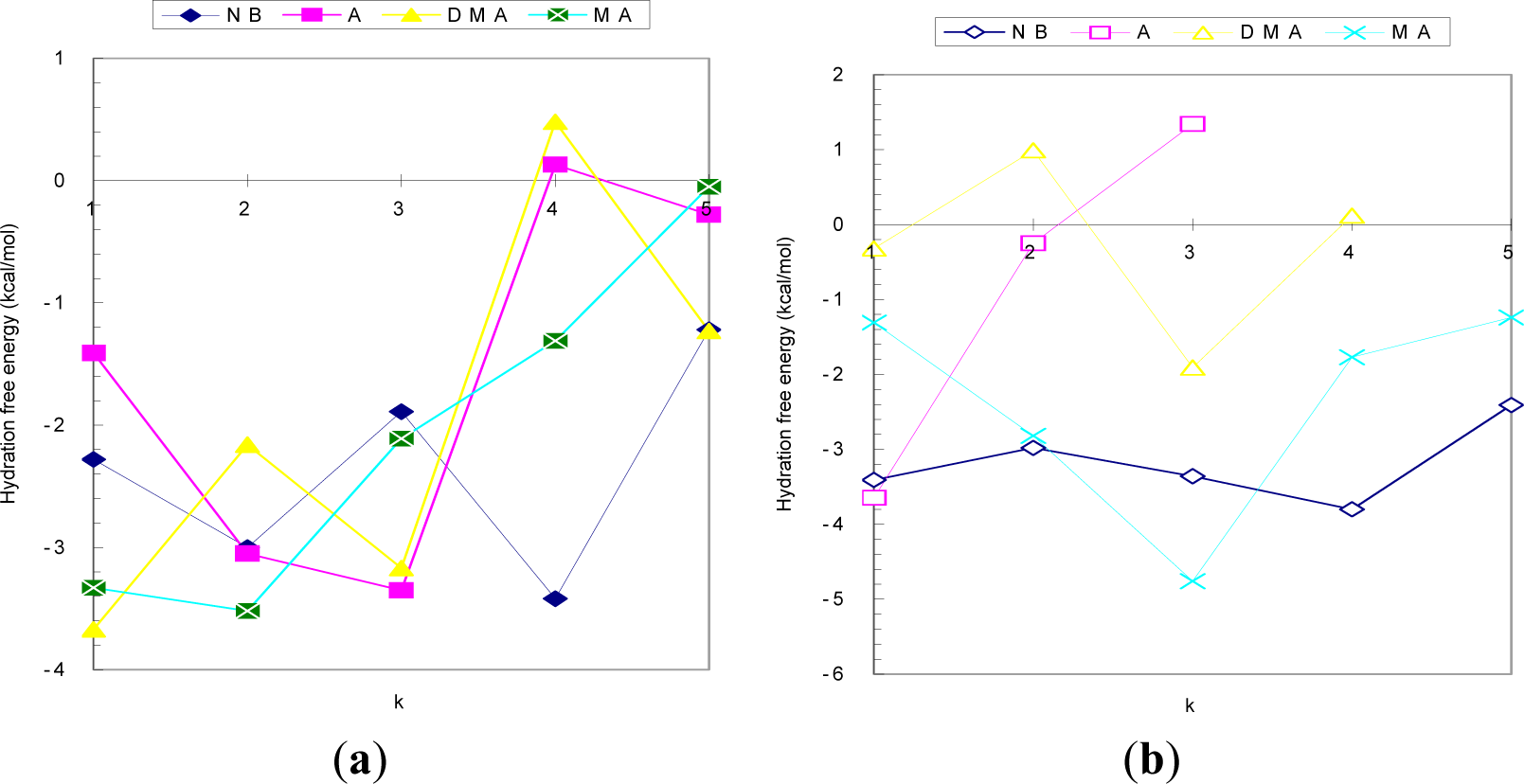
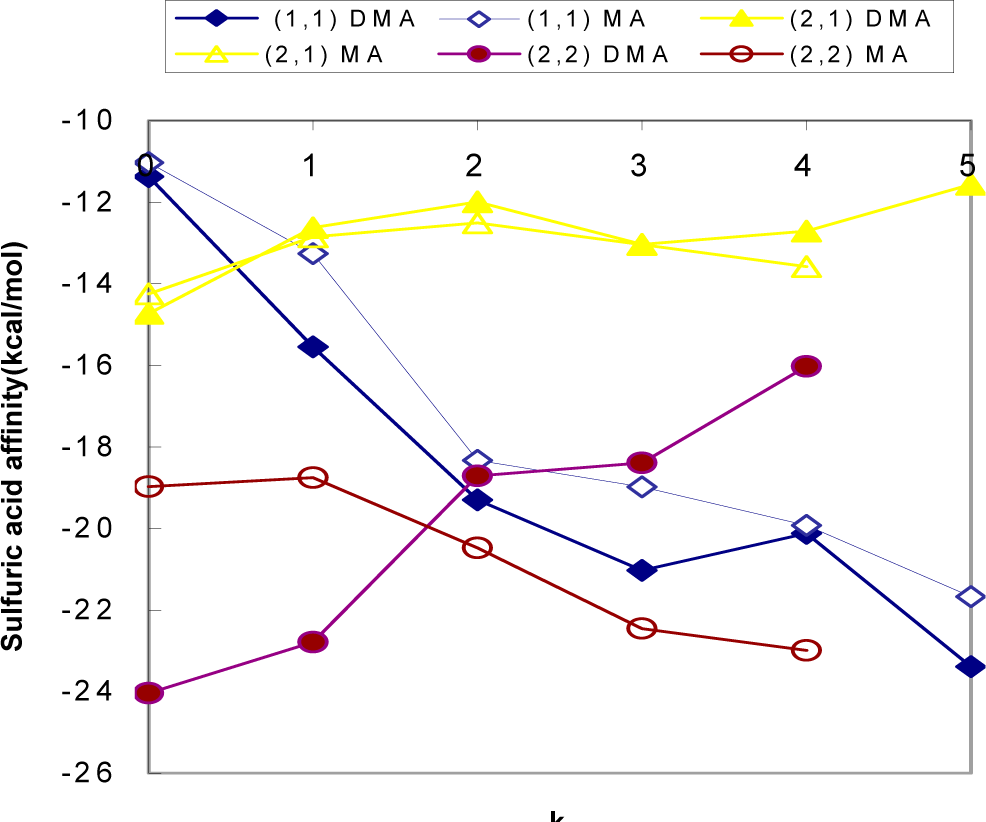

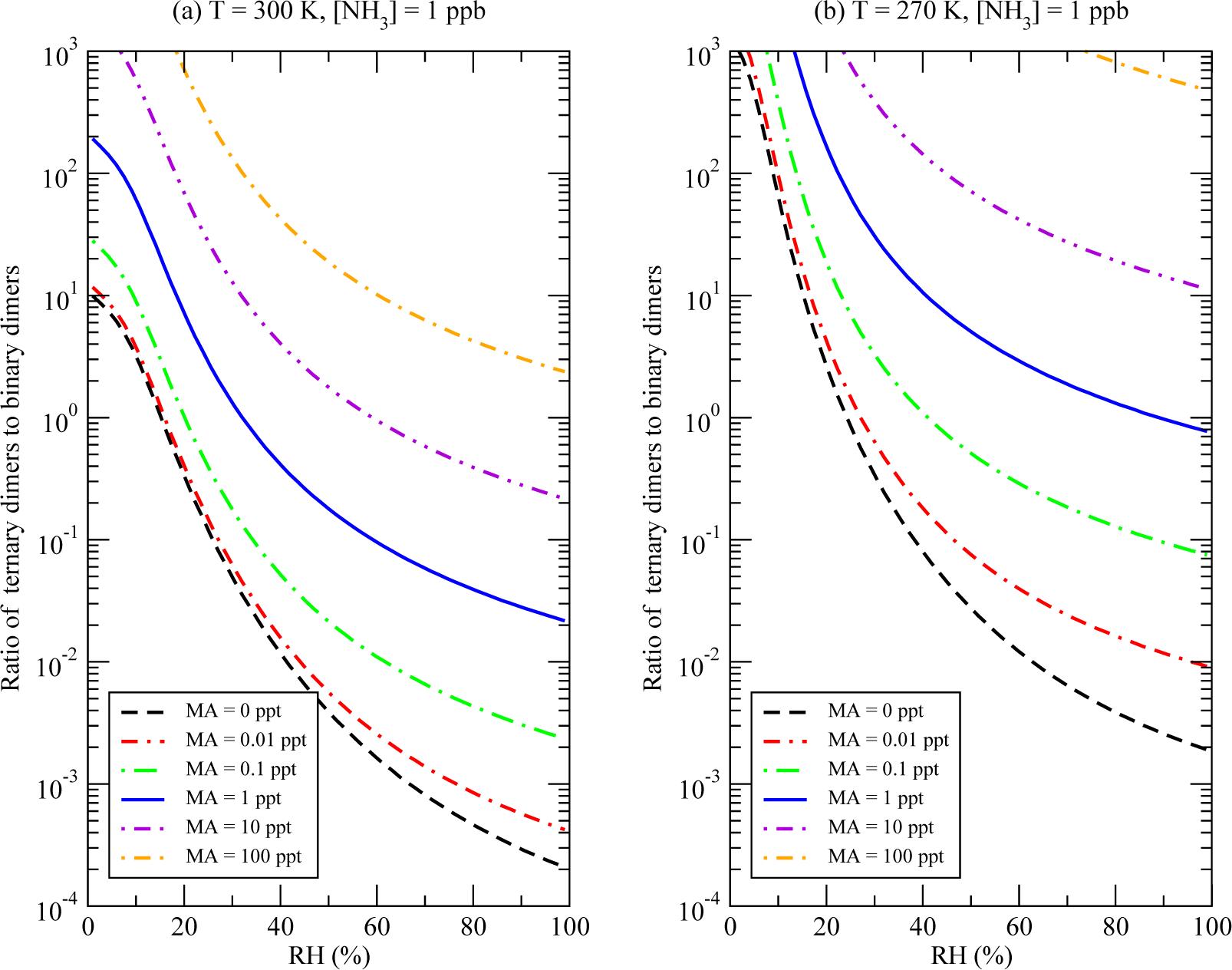
| Reaction | Δ H | Δ S | Δ G | Δ GBA |
|---|---|---|---|---|
| (CH3NH2)+(H2O) <=> (CH3NH2)(H2O) | −7.73 | −22.25 | −1.10 | |
| (CH3NH2)(H2O)+(H2O) <=> (CH3NH2)(H2O)2 | −8.70 | −34.40 | 1.55 | |
| (CH3NH2)(H2O)2+(H2O) <=> (CH3NH2)(H2O)3 | −11.21 | −32.72 | −1.46 | |
| (CH3NH)(H2O)3+(H2O) <=> (CH3NH)(H2O)4 | −9.10 | −29.31 | −0.36 | |
| (CH3NH2)(H2O)4+(H2O) <=> (CH3NH2)(H2O)5 | −7.36 | −30.37 | 1.70 | |
| (H2SO4)(CH3NH2)+(H2O) <=> (H2SO4)(CH3NH2)(H2O) | −13.02 | −32.49 | −3.33 | −3.53 |
| (H2SO4)(CH3NH2)(H2O)+(H2O) <=> (H2SO4)(CH3NH2)(H2O)2 | −13.85 | −34.66 | −3.52 | −3.95 |
| (H2SO4)(CH3NH2)(H2O)2+(H2O) <=> (H2SO4 H2SO4)(CH3NH2)(H2O)3 | −10.97 | −29.73 | −2.11 | |
| (H2SO4)(CH3NH2)(H2O)3+(H2O) <=> (H2SO4)(CH3NH2)(H2O)4 | −10.61 | −31.21 | −1.31 | |
| (H2SO4)(CH3NH2)(H2O)4+(H2O) <=> (H2SO4)(CH3NH2)(H2O)5 | −11.84 | −39.55 | −0.05 | |
| (H2SO4)(CH3NH2)2+(H2O) <=> (H2SO4)(CH3NH2)2(H2O) | −12.26 | −36.04 | −1.51 | |
| (H2SO4)(CH3NH2)2(H2O)+(H2O) <=> (H2SO4)(CH3NH2)2(H2O)2 | −11.73 | −35.65 | −1.10 | |
| (H2SO4)(CH3NH2)2(H2O)2+(H2O) <=> (H2SO4)(CH3NH2)2(H2O)3 | −13.30 | −35.31 | −2.77 | |
| (H2SO4)(CH3NH2)2(H2O)3+(H2O) <=> (H2SO4)(CH3NH2)2(H2O)4 | −11.30 | −33.74 | −1.24 | |
| (H2SO4)2(CH3NH2)+(H2O) <=> (H2SO4)2(CH3NH2)(H2O) | −11.26 | −31.27 | −1.94 | |
| (H2SO4)2(CH3NH2)(H2O)+(H2O) <=> (H2SO4)2(CH3NH2)(H2O)2 | −13.49 | −34.61 | −3.17 | |
| (H2SO4)2(CH3NH2)(H2O)2+(H2O) <=> (H2SO4)2(CH3NH2)(H2O)3 | −11.99 | −31.32 | −2.65 | |
| (H2SO4)2(CH3NH2)(H2O)3+(H2O) <=> (H2SO4)2(CH3NH2)(H2O)4 | −12.33 | −35.18 | −1.84 | |
| (H2SO4)2(CH3NH2)(H2O)4+(H2O) <=> (H2SO4)2(CH3NH2)(H2O)5 | −10.82 | −30.46 | −1.74 | |
| (H2SO4)2(CH3NH2)2+(H2O) <=> (H2SO4)2(CH3NH2)2(H2O) | −12.07 | −36.10 | −1.31 | |
| (H2SO4)2(CH3NH2)2(H2O)+(H2O) <=> (H2SO4)2(CH3NH2)2 (H2O)2 | −11.62 | −29.53 | −2.82 | |
| (H2SO4)2(CH3NH2)2(H2O)2+(H2O) <=> (H2SO4)2(CH3NH2)2(H2O)3 | −14.55 | −32.83 | −4.76 | |
| (H2SO4)2(CH3NH2)2 (H2O)3+(H2O) <=> (H2SO4)2(CH3NH2)2 (H2O)4 | −11.09 | −31.28 | −1.77 | |
| (H2SO4)2(CH3NH2)2 (H2O)4+(H2O) <=> (H2SO4)2(CH3NH2)2 (H2O)5 | −11.25 | −33.56 | −1.24 |
| Reaction | Δ H | Δ S | Δ G |
|---|---|---|---|
| (CH3NH2)+(H2SO4) <=> (H2SO4)(CH3NH2) | −20.40 | −31.42 | −11.03 (−11.61)BA |
| (CH3NH2)(H2O)+(H2SO4) <=> (H2SO4)(CH3NH2)(H2O) | −25.69 | −41.67 | −13.26 |
| (CH3NH2)(H2O)2+(H2SO4) <=> (H2SO4)(CH3NH2)(H2O)2 | −30.83 | −41.93 | −18.33 |
| (CH3NH2)(H2O)3+(H2SO4) <=> (H2SO4)(CH3NH2)(H2O)3 | −30.59 | −38.95 | −18.98 |
| (CH3NH2)(H2O)4+(H2SO4) <=> (H2SO4)(CH3NH2)(H2O)4 | −32.11 | −40.85 | −19.93 |
| (CH3NH2)(H2O)5+(H2SO4) <=> (H2SO4)(CH3NH2)(H2O)5 | −36.60 | −50.04 | −21.68 |
| (H2SO4)(CH3NH2)+(H2SO4) <=> (H2SO4)2(CH3NH2) | −27.42 | −44.17 | −14.25 |
| (H2SO4)(CH3NH2)(H2O)+(H2SO4) <=> (H2SO4)2(CH3NH2)(H2O) | −25.66 | −42.94 | −12.86 |
| (H2SO4)(CH3NH2)(H2O)2+(H2SO4) <=> (H2SO4)2(CH3NH2)(H2O)2 | −25.30 | −42.90 | −12.51 |
| (H2SO4)(CH3NH2)(H2O)3+(H2SO4) <=> (H2SO4)2(CH3NH2)(H2O)3 | −26.31 | −44.49 | −13.05 |
| (H2SO4)(CH3NH2)(H2O)4+(H2SO4) <=> (H2SO4)2(CH3NH2)(H2O)4 | −28.03 | −48.46 | −13.58 |
| (CH3NH2)2+(H2SO4) <=> (H2SO4)(CH3NH2)2 | −31.93 | −42.75 | −19.18 |
| (CH3NH2)2(H2O)+(H2SO4) <=> (H2SO4)(CH3NH2)2(H2O) | −33.74 | −45.36 | −20.22 |
| (CH3NH2)2(H2O)2+(H2SO4) <=> (H2SO4)(CH3NH2)2(H2O)2 | −33.81 | −49.43 | −19.07 |
| (CH3NH2)2(H2O)3+(H2SO4) <=> (H2SO4)(CH3NH2)2(H2O)3 | −38.21 | −54.86 | −21.85 |
| (CH3NH2)2(H2O)4+(H2SO4) <=> (H2SO4)(CH3NH2)2(H2O)4 | −42.26 | −60.50 | −24.22 |
| (H2SO4)(CH3NH2)2+(H2SO4) <=> (H2SO4)2(CH3NH2)2 | −31.08 | −40.62 | −18.97 |
| (H2SO4)(CH3NH2)2(H2O)+(H2SO4) <=> (H2SO4)2(CH3NH2)2(H2O) | −30.89 | −40.68 | −18.76 |
| (H2SO4)(CH3NH2)2(H2O)2+(H2SO4) <=> (H2SO4)2(CH3NH2)2(H2O)2 | −30.78 | −34.57 | −20.48 |
| (H2SO4)(CH3NH2)2(H2O)3+(H2SO4) <=> (H2SO4)2(CH3NH2)2(H2O)3 | −32.03 | −32.09 | −22.46 |
| (H2SO4)(CH3NH2)2(H2O)4+(H2SO4) <=> (H2SO4)2(CH3NH2)2(H2O)4 | −31.83 | −29.62 | −22.99 |
| Reaction | Δ H | Δ S | Δ G |
|---|---|---|---|
| (CH3NH2) +(CH3NH2) <=> (CH3NH2)2 | −4.93 | −21.07 | 1.35 |
| (CH3NH2)2 +(CH3NH2) <=> (CH3NH2)3 | −3.17 | −24.5 | 4.13 |
| (H2SO4) +(CH3NH2) <=> (H2SO4)(CH3NH2) | −20.4 | −31.42 | −11.03 |
| (H2SO4)(H2O)+(CH3NH2) <=> (H2SO4)(CH3NH2)(H2O) | −21.65 | −32.11 | −12.08 |
| (H2SO4)(H2O)2+(CH3NH2) <=> (H2SO4)(CH3NH2)(H2O)2 | −22.94 | −34.69 | −12.59 |
| (H2SO4)(H2O)3+(CH3NH2) <=> (H2SO4)(CH3NH2)(H2O)3 | −21.96 | −32.17 | −12.37 |
| (H2SO4)(H2O)4+(CH3NH2) <=> (H2SO4)(CH3NH2)(H2O)4 | −18.76 | −28.24 | −10.34 |
| (H2SO4)(H2O)5+(CH3NH2) <=> (H2SO4)(CH3NH2)(H2O)5 | −20.54 | −36.92 | −9.53 |
| (H2SO4)(CH3NH2) +(CH3NH2) <=> (H2SO4)(CH3NH2)2 | −16.47 | −32.40 | −6.80 |
| (H2SO4) (CH3NH2)(H2O)+(CH3NH2) <=> (H2SO4)(CH3NH2)2(H2O) | −15.70 | −35.95 | −4.98 |
| (H2SO4)(CH3NH2)(H2O)2+(CH3NH2) <=> (H2SO4)(CH3NH2)2(H2O)2 | −13.58 | −36.94 | −2.57 |
| (H2SO4)(CH3NH2)(H2O)3+(CH3NH2) <=> (H2SO4)(CH3NH2)2(H2O)3 | −15.91 | −42.52 | −3.24 |
| (H2SO4)(CH3NH2)(H2O)4+(CH3NH2) <=> (H2SO4)(CH3NH2)2(H2O)4 | −16.60 | −45.06 | −3.17 |
| (H2SO4)2+(CH3NH2) <=> (H2SO4)2(CH3NH2) | −31.65 | −40.13 | −19.69 |
| (H2SO4)2(H2O)+(CH3NH2) <=> (H2SO4)2(CH3NH2)(H2O) | −28.47 | −34.41 | −18.21 |
| (H2SO4)2(H2O)2+(CH3NH2) <=> (H2SO4)2(CH3NH2)(H2O)2 | −29.11 | −35.91 | −18.41 |
| (H2SO4)2(H2O)3+(CH3NH2) <=> (H2SO4)2(CH3NH2)(H2O)3 | −26.53 | −29.64 | −17.70 |
| (H2SO4)2(H2O)4+(CH3NH2) <=> (H2SO4)2(CH3NH2)(H2O)4 | −24.05 | −27.87 | −15.74 |
| (H2SO4)2(CH3NH2)+(CH3NH2) <=> (H2SO4)2(CH3NH2)2 | −20.13 | −28.86 | −11.52 |
| (H2SO4)2(CH3NH2)(H2O)+(CH3NH2) <=> (H2SO4)2(CH3NH2)2(H2O) | −20.94 | −33.69 | −10.89 |
| (H2SO4)2(CH3NH2)(H2O)2+(CH3NH2) <=> (H2SO4)2(CH3NH2)2(H2O)2 | −19.07 | −28.61 | −10.54 |
| (H2SO4)2(CH3NH2)(H2O)3+(CH3NH2) <=> (H2SO4)2(CH3NH2)2(H2O)3 | −21.63 | −30.12 | −12.65 |
| (H2SO4)2(CH3NH2)(H2O)4+(CH3NH2) <=> (H2SO4)2(CH3NH2)2(H2O)4 | −20.4 | −26.21 | −12.58 |
© 2015 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nadykto, A.B.; Herb, J.; Yu, F.; Xu, Y.; Nazarenko, E.S. Estimating the Lower Limit of the Impact of Amines on Nucleation in the Earth’s Atmosphere. Entropy 2015, 17, 2764-2780. https://doi.org/10.3390/e17052764
Nadykto AB, Herb J, Yu F, Xu Y, Nazarenko ES. Estimating the Lower Limit of the Impact of Amines on Nucleation in the Earth’s Atmosphere. Entropy. 2015; 17(5):2764-2780. https://doi.org/10.3390/e17052764
Chicago/Turabian StyleNadykto, Alexey B., Jason Herb, Fangqun Yu, Yisheng Xu, and Ekaterina S. Nazarenko. 2015. "Estimating the Lower Limit of the Impact of Amines on Nucleation in the Earth’s Atmosphere" Entropy 17, no. 5: 2764-2780. https://doi.org/10.3390/e17052764
APA StyleNadykto, A. B., Herb, J., Yu, F., Xu, Y., & Nazarenko, E. S. (2015). Estimating the Lower Limit of the Impact of Amines on Nucleation in the Earth’s Atmosphere. Entropy, 17(5), 2764-2780. https://doi.org/10.3390/e17052764





