Multiscale Entropy Analysis of Surface Electromyographic Signals from the Urethral Sphincter as a Prognostic Indicator for Surgical Candidates with Primary Bladder Neck Obstruction
Abstract
:1. Introduction
2. Methods
2.1. Study Population and Grouping
2.2. Study Protocol and Parameters
2.3. Definitions
2.4. Equipment for Videourodynamic Testing and Procedures
2.5. Study Method
- (1)
- Define the data series x(n) with length N and the two parameters of m and r (where m = Embedded dimension of the vector; r = tolerance).
- (2)
- Define N – m + 1 vectors, each of size m, composed as follows:
- (3)
- Define d[um(i), um(j)] as the maximum value: d[um(i), um(j)] = max{|xi + k − xj + k|: 0 ≤ k ≤ m − 1} (i ≠ j). Calculate the number of d[um(i), um(j)] within distance r and calculate the ratio of the number to the total N – m for each value of i ≤ N − m + 1 and an average to all points is defined as:
- (4)
- Increase the embedded dimension to m + 1, gives:
- (5)
- Therefore, sample entropy (SE) is defined as:
2.6. Statistical Analysis
3. Results
3.1. Age and Urodynamic Parameters of Testing Subjects
| Group 1 (n = 14) | Group 2 (n = 8) | Group 3 (Negative Control) (n = 7) | Group 4 (Positive Control) (n = 12) | |
|---|---|---|---|---|
| Age (years) | 66.29 ± 6.67 | 72.75 ± 7.31 ## | 63.86 ± 15.40 | 38.25 ± 13.85 |
| Pdet (cmH2O) | 46.69 ± 26.97 | 34.13 ± 12.06 # | 36.14 ± 8.18 | 57.25 ± 17.86 |
| Qmax (mL/sec) | 6.50 ± 3.22 | 8.50 ± 3.91 | 11.71 ± 4.71 * | 8.75 ± 9.22 |
| Vol (mL) | 225.07 ± 74.65 | 219.75 ± 109.86 # | 195.86 ± 86.60 | 86.33 ± 49.60 |
| PVR (mL) | 48.46 ± 54.47 | 67.50 ± 96.53 | 21.43 ± 19.59 | 82.92 ± 81.76 |
| SD of pre-void EMG (mV) | 2.94 ± 1.61 | 5.99 ± 5.82 | 3.04 ± 0.79 | 6.13 ± 5.26 |
| SD of EMG during voiding (mV) | 2.18 ± 1.63 | 4.81 ± 3.60 | 3.16 ± 1.27 † | 14.07 ± 13.86 |
3.2. Multiscale Entropy (MSE) Analysis of Electromyographic (EMG) Signals
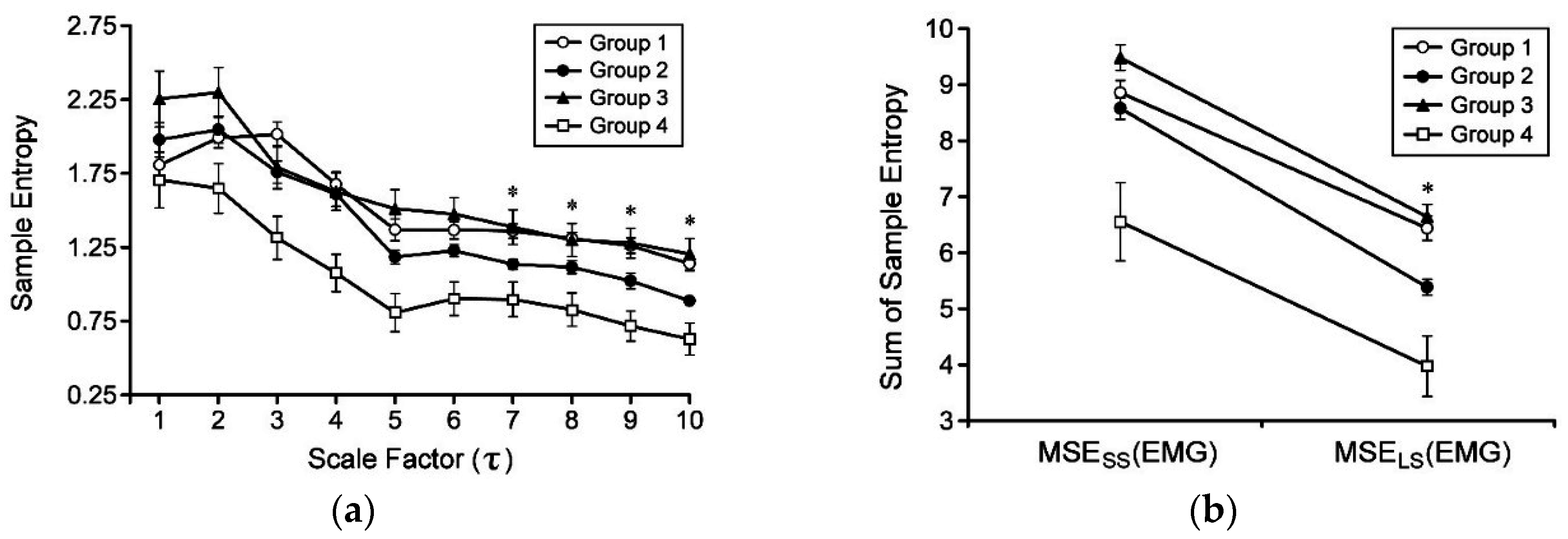
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wein, A.J. Re: Urodynamics: Examining the Current Role of UDS Testing. What is the Role of Urodynamic Testing in Light of Recent AUA Urodynamics and Overactive Bladder Guidelines and the VALUE Study? J. Urol. 2015, 193, 1311–1334. [Google Scholar] [CrossRef] [PubMed]
- Danforth, T.L.; Ginsberg, D.A. Neurogenic lower urinary tract dysfunction: How, when, and with which patients do we use urodynamics? Urol. Clin. N. Am. 2014, 41, 445–452. [Google Scholar] [CrossRef] [PubMed]
- Padmanabhan, P.; Nitti, V.W. Primary bladder neck obstruction in men, women, and children. Curr. Urol. Rep. 2007, 8, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Nitti, V.W.; Lefkowitz, G.; Ficazzola, M.; Dixon, C.M. Lower urinary tract symptoms in young men: Videourodynamic findings and correlation with noninvasive measures. J. Urol. 2002, 168, 135–138. [Google Scholar] [CrossRef]
- Nitti, V.W. Primary bladder neck obstruction in men and women. Rev. Urol. 2005, 7, S12–S17. [Google Scholar] [PubMed]
- Sacco, E.; Tienforti, D.; Bientinesi, R.; D’Addessi, A.; Racioppi, M.; Pinto, F.; Totaro, A.; Vittori, M.; D’Agostino, D.; Bassi, P. OnabotulinumtoxinA injection therapy in men with LUTS due to primary bladder-neck dysfunction: Objective and patient-reported outcomes. Neurourol. Urodyn. 2014, 33, 142–146. [Google Scholar] [CrossRef] [PubMed]
- Trockman, B.A.; Gerspach, J.; Dmochowski, R.; Haab, F.; Zimmern, P.E.; Leach, G.E. Primary bladder neck obstruction: Urodynamic findings and treatment results in 36 men. J. Urol. 1996, 156, 1418–1420. [Google Scholar] [CrossRef]
- Van Batavia, J.P.; Combs, A.J.; Fast, A.M.; Glassberg, K.I. Use of non-invasive uroflowmetry with simultaneous electromyography to monitor patient response to treatment for lower urinary tract conditions. J. Pediatr. Urol. 2014, 10, 532–537. [Google Scholar] [CrossRef] [PubMed]
- Istenic, R.; Kaplanis, P.A.; Pattichis, C.S.; Zazula, D. Multiscale entropy-based approach to automated surface EMG classification of neuromuscular disorders. Med. Biol. Eng. Comput. 2010, 48, 773–781. [Google Scholar] [CrossRef] [PubMed]
- Cashaback, J.G.; Cluff, T.; Potvin, J.R. Muscle fatigue and contraction intensity modulates the complexity of surface electromyography. J. Electromyogr. Kinesiol. 2013, 23, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Abrams, P.; Cardozo, L.; Fall, M.; Griffiths, D.; Rosier, P.; Ulmsten, U.; van Kerrebroeck, P.; Victor, A.; Wein, A. The standardisation of terminology of lower urinary tract function: Report from the Standardisation Sub-committee of the International Continence Society. Neurourol. Urodyn. 2002, 21, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Chapple, C.R.; Araño, P.; Bosch, J.L.; de Ridder, D.; Kramer, A.E.; Ridder, A.M. Solifenacin appears effective and well tolerated in patients with symptomatic idiopathic detrusor overactivity in aplacebo- and tolterodine-controlled phase 2 dose-finding study. BJU Int. 2004, 93, 71–77. [Google Scholar] [CrossRef] [PubMed]
- De, E.J.; Patel, C.Y.; Tharian, B.; Westney, O.L.; Graves, D.E.; Hairston, J.C. Diagnostic discordance of electromyography (EMG) versus voiding cystourethrogram (VCUG) for detrusor-external sphincter dyssynergy (DESD). Neurourol. Urodyn. 2005, 24, 616–621. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.; Goldberger, A.L.; Peng, C.K. Multiscale entropy to distinguish physiologic and synthetic RR time series. Comput. Cardiol. 2002, 29, 137–140. [Google Scholar] [PubMed]
- Costa, M.; Goldberger, A.L.; Peng, C.K. Multiscale entropy analysis of complex physiologic time series. Phys. Rev. Lett. 2002, 89, 068102. [Google Scholar] [CrossRef] [PubMed]
- Costa, M.; Peng, C.K.; Goldberger, A.L.; Jeffrey, M.H. Multiscale entropy analysis of human gait dynamics. Physica A 2003, 330, 53–60. [Google Scholar] [CrossRef]
- Wu, H.T.; Hsu, P.C.; Lin, C.F.; Wang, H.J.; Sun, C.K.; Liu, A.B.; Lo, M.T.; Tang, C.J. Multiscale entropy analysis of pulse wave velocity for assessing atherosclerosis in the aged and diabetic. IEEE Trans. Biomed. Eng. 2011, 58, 2978–2981. [Google Scholar] [PubMed]
- Richman, J.S.; Moorman, J.R. Physiological time-series analysis using approximate entropy and sample entropy. Am. J. Phys. Heart Circ. Physiol. 2000, 278, H2039–H2049. [Google Scholar]
- Costa, M.; Goldberger, A.L.; Peng, C.K. Multiscale entropy analysis of biological signals. Phys. Rev. E 2005, 71, 021906. [Google Scholar] [CrossRef] [PubMed]
- Baumert, M.; Javorka, M.; Seeck, A.; Faber, R.; Sanders, P.; Voss, A. Multiscale entropy and detrended fluctuation analysis of QT interval and heart rate variability during normal pregnancy. Comput. Biol. Med. 2012, 42, 347–352. [Google Scholar] [CrossRef] [PubMed]
- Javorka, M.; Trunkvalterova, Z.; Tonhajzerova, I.; Javorkova, J.; Javorka, K.; Baumert, M. Short-term heart rate complexity is reduced in patients with type 1 diabetes mellitus. Clin. Neurophysiol. 2008, 119, 1071–1081. [Google Scholar] [CrossRef] [PubMed]
- Turianikova, Z.; Javorka, K.; Baumert, M.; Calkovska, A.; Javorka, M. The effect of orthostatic stress on multiscale entropy of heart rate and blood pressure. Physiol. Meas. 2011, 32, 1425–1437. [Google Scholar] [CrossRef] [PubMed]
- Huang, N.E.; Zheng, S.; Steven, R.L.; Wu, M.C.; Shih, H.H.; Zheng, Q.; Yen, N.C.; Tung, C.C.; Liu, H.H. The empirical mode decomposition and the Hilbert spectrum for nonlinear and non-stationary time series analysis. Proc. R. Soc. A Math. Phys. Eng. Sci. 1998, 454, 903–995. [Google Scholar] [CrossRef]
- Wu, Z.; Huang, N.E. Ensemble empirical mode decomposition: A noise-assisted data analysis method. Adv. Adapt. Data Anal. 2009, 1, 1–41. [Google Scholar] [CrossRef]
- Lo, M.T.; Tsai, P.H.; Lin, P.F.; Lin, C.; Hsin, Y.L. The nonlinear and nonstationary properties in EEG signals: Probing the complex fluctuations by Hilbert–Huang transform. Adv. Adapt. Data Anal. 2009, 1, 461–482. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, X.; Barkhaus, P.E.; Zhou, P. Multiscale entropy analysis of different spontaneous motor unit discharge patterns. IEEE J. Biomed. Health Inform. 2013, 17, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Roehrborn, C.G.; Oesterling, J.E.; Auerbach, S.; Kaplan, S.A.; Lloyd, L.K.; Milam, D.E.; Padley, R.J. The Hytrin Community Assessment Trial study: A one-year study of terazosin versus placebo in the treatment of men with symptomatic benign prostatic hyperplasia. Urology 1996, 2, 159–168. [Google Scholar] [CrossRef]
- Nitti, V.W.; Carlson, K.V.; Blaivas, J.G.; Dmochowski, R.R. Early results of pubovaginal sling lysis by midline sling incision. Urology 2002, 59, 47–51. [Google Scholar] [CrossRef]
- Kochakarn, W.; Lertsithichai, P. Unilateral transurethral incision for primary bladder neck obstruction: Symptom relief and fertility preservation. World J. Urol. 2003, 21, 159–162. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, H.-T.; Jiang, Y.-H.; Liu, A.-B.; Liu, C.-W.; Ou, Y.-N.; Kuo, H.-C.; Sun, C.-K. Multiscale Entropy Analysis of Surface Electromyographic Signals from the Urethral Sphincter as a Prognostic Indicator for Surgical Candidates with Primary Bladder Neck Obstruction. Entropy 2015, 17, 8089-8098. https://doi.org/10.3390/e17127863
Wu H-T, Jiang Y-H, Liu A-B, Liu C-W, Ou Y-N, Kuo H-C, Sun C-K. Multiscale Entropy Analysis of Surface Electromyographic Signals from the Urethral Sphincter as a Prognostic Indicator for Surgical Candidates with Primary Bladder Neck Obstruction. Entropy. 2015; 17(12):8089-8098. https://doi.org/10.3390/e17127863
Chicago/Turabian StyleWu, Hsien-Tsai, Yuan-Hong Jiang, An-Bang Liu, Chun-Wei Liu, Yu-Nian Ou, Hann-Chorng Kuo, and Cheuk-Kwan Sun. 2015. "Multiscale Entropy Analysis of Surface Electromyographic Signals from the Urethral Sphincter as a Prognostic Indicator for Surgical Candidates with Primary Bladder Neck Obstruction" Entropy 17, no. 12: 8089-8098. https://doi.org/10.3390/e17127863
APA StyleWu, H.-T., Jiang, Y.-H., Liu, A.-B., Liu, C.-W., Ou, Y.-N., Kuo, H.-C., & Sun, C.-K. (2015). Multiscale Entropy Analysis of Surface Electromyographic Signals from the Urethral Sphincter as a Prognostic Indicator for Surgical Candidates with Primary Bladder Neck Obstruction. Entropy, 17(12), 8089-8098. https://doi.org/10.3390/e17127863