Effects of Resistance Training on the Redox Status of Skeletal Muscle in Older Adults
Abstract
:1. Introduction
2. Materials and Methods
2.1. Ethical Approval
2.2. Participants
2.3. Study Design
2.4. Testing Sessions
2.5. Muscle Biopsies
2.6. Resistance Training (RT) Program
2.7. mRNA Analysis
2.8. Western Blotting
2.9. Enzymatic Activities
2.10. Statistics
3. Results
3.1. Participant Characteristics and Training Adaptations
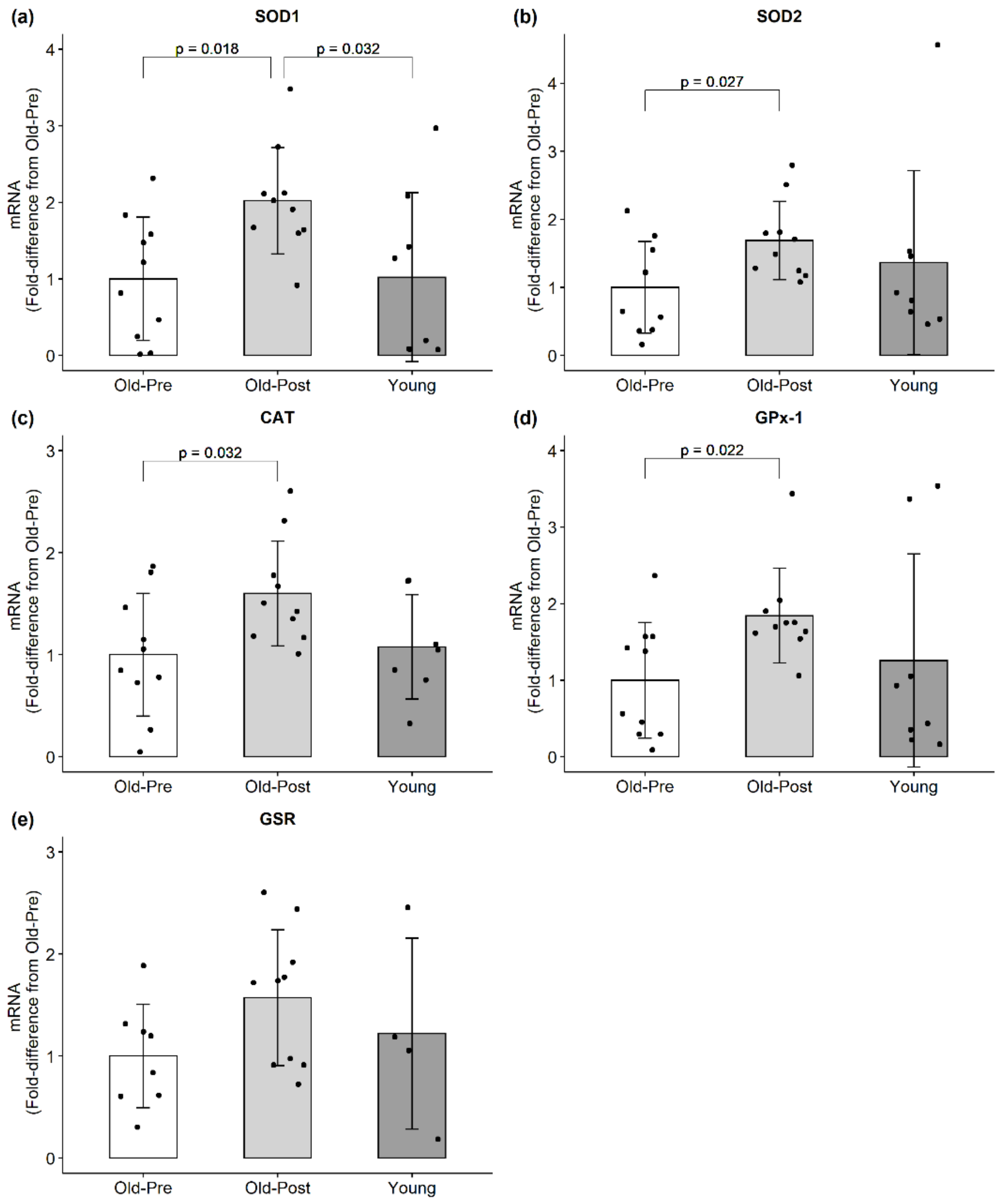
3.2. mRNA Expression
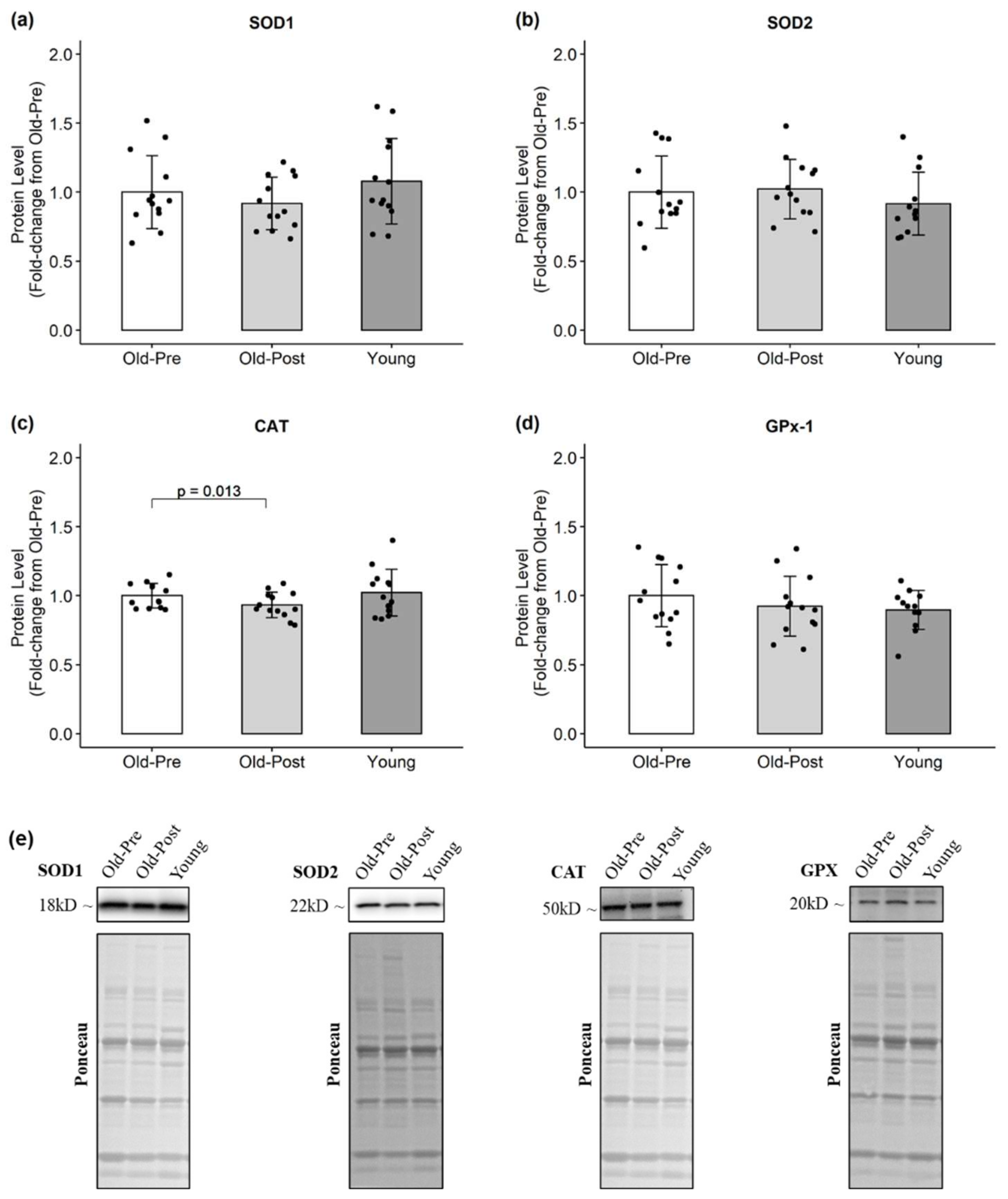
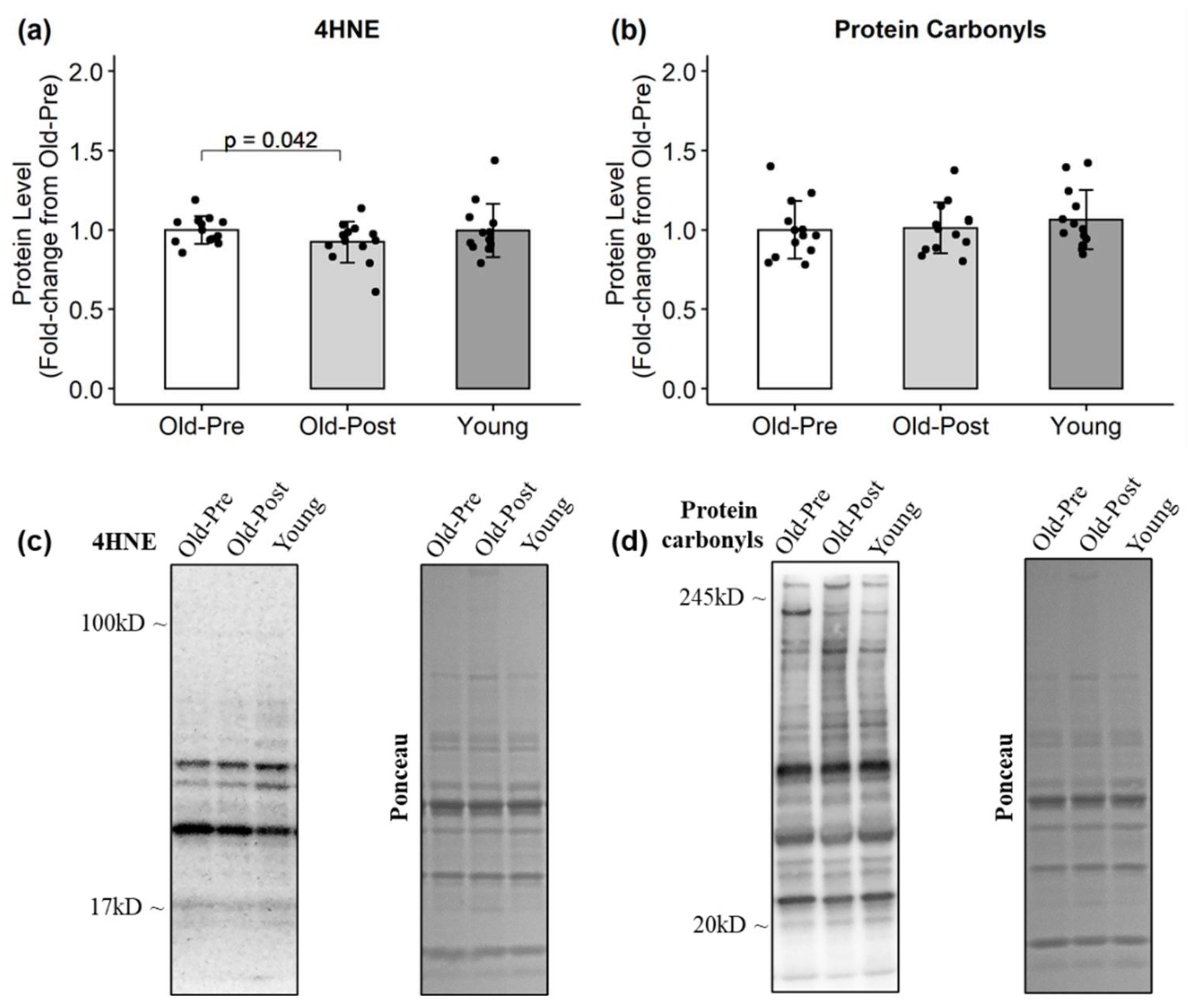
3.3. Protein Levels
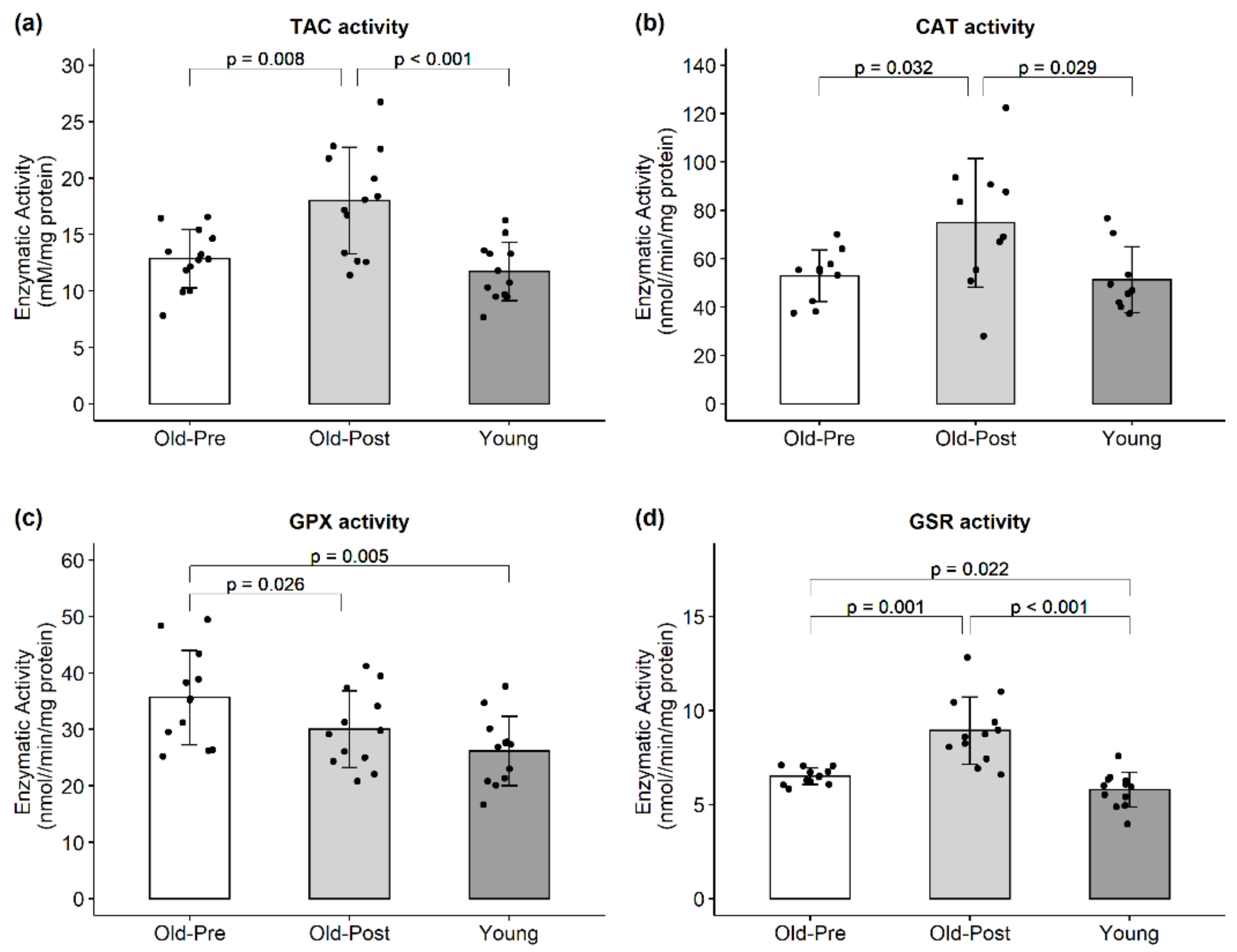
3.4. Enzymatic Activity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Berger, M.J.; Doherty, T.J. Sarcopenia: Prevalence, mechanisms, and functional consequences. Interdiscip. Top. Gerontol. 2010, 37, 94–114. [Google Scholar] [CrossRef] [PubMed]
- Niccoli, T.; Partridge, L. Ageing as a risk factor for disease. Curr. Biol. 2012, 22, R741–R752. [Google Scholar] [CrossRef] [Green Version]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dai, D.-F.; Chiao, Y.; Marcinek, D.J.; Szeto, H.H.; Rabinovitch, P.S. Mitochondrial oxidative stress in aging and healthspan. Longev. Healthspan 2014, 3, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harman, D. Aging: A theory based on free radical and radiation chemistry. J. Gerontol. 1956, 11, 298–300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jang, Y.C.; Van Remmen, H. Age-associated alterations of the neuromuscular junction. Exp. Gerontol. 2011, 46, 193–198. [Google Scholar] [CrossRef] [Green Version]
- Salmon, A.B.; Richardson, A.; Pérez, V.I. Update on the oxidative stress theory of aging: Does oxidative stress play a role in aging or healthy aging? Free Radic. Biol. Med. 2010, 48, 642–655. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gianni, P.; Jan, K.J.; Douglas, M.J.; Stuart, P.M.; Tarnopolsky, M.A. Oxidative stress and the mitochondrial theory of aging in human skeletal muscle. Exp. Gerontol. 2004, 39, 1391–1400. [Google Scholar] [CrossRef]
- Pansarasa, O.; Castagna, L.; Colombi, B.; Vecchiet, J.; Felzani, G.; Marzatico, F. Age and sex differences in human skeletal muscle: Role of reactive oxygen species. Free Radic. Res. 2000, 33, 287–293. [Google Scholar] [CrossRef]
- Ryan, M.J.; Dudash, H.J.; Docherty, M.; Geronilla, K.B.; Baker, B.A.; Haff, G.G.; Cutlip, R.G.; Alway, S.E. Aging-dependent regulation of antioxidant enzymes and redox status in chronically loaded rat dorsiflexor muscles. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2008, 63, 1015–1026. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Powers, S.K.; Duarte, J.; Kavazis, A.N.; Talbert, E.E. Reactive oxygen species are signalling molecules for skeletal muscle adaptation. Exp. Physiol. 2010, 95, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Powers, S.K.; Ji, L.L.; Kavazis, A.N.; Jackson, M.J. Reactive oxygen species: Impact on skeletal muscle. Compr. Physiol. 2011, 1, 941–969. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dimauro, I.; Mercatelli, N.; Caporossi, D. Exercise-induced ROS in heat shock proteins response. Free Radic. Biol. Med. 2016, 98, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Morton, J.P.; Kayani, A.C.; McArdle, A.; Drust, B. The exercise-induced stress response of skeletal muscle, with specific emphasis on humans. Sports Med. 2009, 39, 643–662. [Google Scholar] [CrossRef]
- Cobley, J.N.; Moult, P.R.; Burniston, J.G.; Morton, J.P.; Close, G.L. Exercise improves mitochondrial and redox-regulated stress responses in the elderly: Better late than never! Biogerontology 2015, 16, 249–264. [Google Scholar] [CrossRef] [PubMed]
- De Sousa, C.V.; Sales, M.M.; Rosa, T.S.; Lewis, J.E.; de Andrade, R.V.; Simões, H.G. The antioxidant effect of exercise: A systematic review and meta-analysis. Sports Med. 2017, 47, 277–293. [Google Scholar] [CrossRef]
- Cuyul-Vásquez, I.; Berríos-Contreras, L.; Soto-Fuentes, S.; Hunter-Echeverría, K.; Marzuca-Nassr, G.N. Effects of resistance exercise training on redox homeostasis in older adults. A systematic review and meta-analysis. Exp. Gerontol. 2020, 138, 111012. [Google Scholar] [CrossRef]
- Parker, L.; Trewin, A.; Levinger, I.; Shaw, C.S.; Stepto, N.K. Exercise-intensity dependent alterations in plasma redox status do not reflect skeletal muscle redox-sensitive protein signaling. J. Sci. Med. Sport 2018, 21, 416–421. [Google Scholar] [CrossRef] [Green Version]
- Cobley, J.N.; Close, G.L.; Bailey, D.M.; Davison, G.W. Exercise redox biochemistry: Conceptual, methodological and technical recommendations. Redox Biol. 2017, 12, 540–548. [Google Scholar] [CrossRef] [PubMed]
- Lamb, D.A.; Moore, J.H.; Smith, M.A.; Vann, C.G.; Osburn, S.C.; Ruple, B.A.; Fox, C.D.; Smith, K.S.; Altonji, O.M.; Power, Z.M.; et al. The effects of resistance training with or without peanut protein supplementation on skeletal muscle and strength adaptations in older individuals. J. Int. Soc. Sports Nutr. 2020, 17, 66. [Google Scholar] [CrossRef]
- Kephart, W.C.; Mobley, C.B.; Fox, C.D.; Pascoe, D.D.; Sefton, J.M.; Wilson, T.J.; Goodlett, M.D.; Kavazis, A.N.; Roberts, M.D.; Martin, J.S. A single bout of whole-leg, peristaltic pulse external pneumatic compression upregulates PGC-1α mRNA and endothelial nitric oxide sythase protein in human skeletal muscle tissue. Exp. Physiol. 2015, 100, 852–864. [Google Scholar] [CrossRef] [PubMed]
- Parry, H.A.; Brooks Mobley, C.; Mumford, P.W.; Romero, M.A.; Haun, C.T.; Zhang, Y.; Roberson, P.A.; Zempleni, J.; Ferrando, A.A.; Vechetti, I.J.; et al. Bovine milk extracellular vesicles (EVs) modification elicits skeletal muscle growth in rats. Front. Physiol. 2019, 10, 436. [Google Scholar] [CrossRef]
- Wang, L.; Gao, J.; Liu, J.; Siedlak, S.L.; Torres, S.; Fujioka, H.; Huntley, M.L.; Jiang, Y.; Ji, H.; Yan, T.; et al. Mitofusin 2 regulates axonal transport of calpastatin to prevent neuromuscular synaptic elimination in skeletal muscles. Cell Metab. 2018, 28, 400–414.e8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Apostolopoulos, A.; Nakamura, A.; Yokoyama, S.; Aoshima, M.; Fujimoto, R.; Nakamura, K.; Ito, R.; Goto, K. Nuclear accumulation of HSP70 in mouse skeletal muscles in response to heat stress, aging, and unloading with or without reloading. Front. Genet. 2018, 9, 617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Basso, E.; Rigotto, G.; Zucchetti, A.E.; Pozzan, T. Slow activation of fast mitochondrial Ca2+ uptake by cytosolic Ca2+. J. Biol. Chem. 2018, 293, 17081–17094. [Google Scholar] [CrossRef] [Green Version]
- Tarpey, M.D.; Valencia, A.P.; Jackson, K.C.; Amorese, A.J.; Balestrieri, N.P.; Renegar, R.H.; Pratt, S.J.P.; Ryan, T.E.; McClung, J.M.; Lovering, R.M.; et al. Induced in vivo knockdown of the Brca1 gene in skeletal muscle results in skeletal muscle weakness. J. Physiol. 2019, 597, 869–887. [Google Scholar] [CrossRef]
- Lamb, D.A.; Moore, J.H.; Mesquita, P.H.C.; Smith, M.A.; Vann, C.G.; Osburn, S.C.; Fox, C.D.; Lopez, H.L.; Ziegenfuss, T.N.; Huggins, K.W.; et al. Resistance training increases muscle NAD+ and NADH concentrations as well as NAMPT protein levels and global sirtuin activity in middle-aged, overweight, untrained individuals. Aging 2020, 12. [Google Scholar] [CrossRef] [PubMed]
- Mesquita, P.H.C.; Lamb, D.A.; Parry, H.A.; Moore, J.H.; Smith, M.A.; Vann, C.G.; Osburn, S.C.; Fox, C.D.; Ruple, B.A.; Huggins, K.W.; et al. Acute and chronic effects of resistance training on skeletal muscle markers of mitochondrial remodeling in older adults. Physiol. Rep. 2020, 8, e14526. [Google Scholar] [CrossRef]
- Ji, L.L. Antioxidant signaling in skeletal muscle: A brief review. Exp. Gerontol. 2007, 42, 582–593. [Google Scholar] [CrossRef]
- Ji, L.L. Modulation of skeletal muscle antioxidant defense by exercise: Role of redox signaling. Free Radic. Biol. Med. 2008, 44, 142–152. [Google Scholar] [CrossRef]
- Ji, L.L. Redox signaling in skeletal muscle: Role of aging and exercise. Adv. Physiol. Educ. 2015, 39, 352–359. [Google Scholar] [CrossRef]
- Oh-ishi, S.; Kizaki, T.; Yamashita, H.; Nagata, N.; Suzuki, K.; Taniguchi, N.; Ohno, H. Alterations of superoxide dismutase iso-enzyme activity, content, and mRNA expression with aging in rat skeletal muscle. Mech. Ageing Dev. 1995, 84, 65–76. [Google Scholar] [CrossRef]
- Ryan, M.J.; Dudash, H.J.; Docherty, M.; Geronilla, K.B.; Baker, B.A.; Haff, G.G.; Cutlip, R.G.; Alway, S.E. Vitamin E and C supplementation reduces oxidative stress, improves antioxidant enzymes and positive muscle work in chronically loaded muscles of aged rats. Exp. Gerontol. 2010, 45, 882–895. [Google Scholar] [CrossRef] [Green Version]
- García-López, D.; Häkkinen, K.; Cuevas, M.J.; Lima, E.; Kauhanen, A.; Mattila, M.; Sillanpää, E.; Ahtiainen, J.P.; Karavirta, L.; Almar, M.; et al. Effects of strength and endurance training on antioxidant enzyme gene expression and activity in middle-aged men. Scand. J. Med. Sci. Sports 2007, 17, 595–604. [Google Scholar] [CrossRef]
- Lambertucci, R.H.; Levada-Pires, A.C.; Rossoni, L.V.; Curi, R.; Pithon-Curi, T.C. Effects of aerobic exercise training on antioxidant enzyme activities and mRNA levels in soleus muscle from young and aged rats. Mech. Ageing Dev. 2007, 128, 267–275. [Google Scholar] [CrossRef]
- Parise, G.; Brose, A.N.; Tarnopolsky, M.A. Resistance exercise training decreases oxidative damage to DNA and increases cytochrome oxidase activity in older adults. Exp. Gerontol. 2005, 40, 173–180. [Google Scholar] [CrossRef] [PubMed]
- Murlasits, Z.; Cutlip, R.G.; Geronilla, K.B.; Rao, K.M.K.; Wonderlin, W.F.; Alway, S.E. Resistance training increases heat shock protein levels in skeletal muscle of young and old rats. Exp. Gerontol. 2006, 41, 398–406. [Google Scholar] [CrossRef]
- Holloway, G.P.; Holwerda, A.M.; Miotto, P.M.; Dirks, M.L.; Verdijk, L.B.; van Loon, L.J.C. Age-associated impairments in mitochondrial ADP sensitivity contribute to redox stress in senescent human skeletal muscle. Cell Rep. 2018, 22, 2837–2848. [Google Scholar] [CrossRef] [Green Version]
- Mecocci, P.; Fanó, G.; Fulle, S.; MacGarvey, U.; Shinobu, L.; Polidori, M.C.; Cherubini, A.; Vecchiet, J.; Senin, U.; Beal, M.F. Age-dependent increases in oxidative damage to DNA, lipids, and proteins in human skeletal muscle. Free Radic. Biol. Med. 1999, 26, 303–308. [Google Scholar] [CrossRef]
- Parise, G.; Phillips, S.M.; Kaczor, J.J.; Tarnopolsky, M.A. Antioxidant enzyme activity is up-regulated after unilateral resistance exercise training in older adults. Free Radic. Biol. Med. 2005, 39, 289–295. [Google Scholar] [CrossRef]
- Azizbeigi, K.; Stannard, S.R.; Atashak, S.; Mosalman, M. Antioxidant enzymes and oxidative stress adaptation to exercise training: Comparison of endurance, resistance, and concurrent training in untrained males. J. Exerc. Sci. Fit. 2014, 12, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Cobley, J.N.; Sakellariou, G.K.; Owens, D.J.; Murray, S.; Waldron, S.; Gregson, W.; Fraser, W.D.; Burniston, J.G.; Iwanejko, L.A.; McArdle, A.; et al. Lifelong training preserves some redox-regulated adaptive responses after an acute exercise stimulus in aged human skeletal muscle. Free Radic. Biol. Med. 2014, 70, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Gjøvaag, T.F.; Dahl, H.A. Effect of training and detraining on the expression of heat shock proteins in m. triceps brachii of untrained males and females. Eur. J. Appl. Physiol. 2006, 98, 310–322. [Google Scholar] [CrossRef]
- Paulsen, G.; Hanssen, K.E.; Rønnestad, B.R.; Kvamme, N.H.; Ugelstad, I.; Kadi, F.; Raastad, T. Strength training elevates HSP27, HSP70 and αB-crystallin levels in musculi vastus lateralis and trapezius. Eur. J. Appl. Physiol. 2012, 112, 1773–1782. [Google Scholar] [CrossRef] [PubMed]
- Bloomer, R.J.; Goldfarb, A.H.; Wideman, L.; McKenzie, M.J.; Consitt, L.A. Effects of acute aerobic and anaerobic exercise on blood markers of oxidative stress. J. Strength Cond. Res. 2005, 19, 276–285. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bloomer, R.J.; Fry, A.C.; Falvo, M.J.; Moore, C.A. Protein carbonyls are acutely elevated following single set anaerobic exercise in resistance trained men. J. Sci. 2007, 10, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Çakır-Atabek, H.; Özdemir, F.; Çolak, R. Oxidative stress and antioxidant responses to progressive resistance exercise intensity in trained and untrained males. Biol. Sport 2015, 32, 321–328. [Google Scholar] [CrossRef]
- Güzel, N.A.; Hazar, S.; Erbas, D. Effects of different resistance exercise protocols on nitric oxide, lipid peroxidation and creatine kinase activity in sedentary males. J. Sports Sci. Med. 2007, 6, 417–422. [Google Scholar] [PubMed]
- Parry, H.A.; Roberts, M.D.; Kavazis, A.N. Human skeletal muscle mitochondrial adaptations following resistance exercise training. Int. J. Sports Med. 2020. [Google Scholar] [CrossRef]
- Roberts, M.D.; Haun, C.T.; Vann, C.G.; Osburn, S.C.; Young, K.C. Sarcoplasmic hypertrophy in skeletal muscle: A scientific “unicorn” or resistance training adaptation? Front. Physiol. 2020, 11, 816. [Google Scholar] [CrossRef]

| Gene | Primer Sequences | Amplicon Length | Position | NCBI Ref. Seq. |
|---|---|---|---|---|
| SOD1 | FP (5′ → 3′): TGTGGCCGATGTGTCTATTGAA RP (5′ → 3′): CACCTTTGCCCAAGTCATCTG | 109 bp | FP: 430–451 RP: 519–539 | NM_000454 |
| SOD2 | FP (5′ → 3′): GTTGGGGTTGGCTTGGTTTC RP (5′ → 3′): GCCTGTTGTTCCTTGCAGTG | 89 bp | FP: 511–530 RP: 580–599 | NM_000636 |
| CAT | FP (5′ → 3′): CTGACTACGGGAGCCACATC RP (5′ → 3′): AGATCCGGACTGCACAAAGG | 92 bp | FP: 1540–1559 RP: 1612–1631 | NM_001752 |
| GPx-1 | FP (5′ → 3′): ACGAGGGAGGAACACCTGAT RP (5′ → 3′): TCTGGCAGAGACTGGGATCA | 80 bp | FP: 775–794 RP: 835–854 | NM_000581 |
| GSR | FP (5′ → 3′): AAAAAGTACACCGCCCCACA RP (5′ → 3′): ATCTGGCTCTCATGAGGGGT | 71 bp | FP: 576–595 RP: 1612–1631 | NM_000637 |
| GAPDH | FP (5′ → 3′): AACCTGCCAAATATGATGAC RP (5′ → 3′): TCATACCAGGAAATGAGCTT | 193 bp | FP: 828–847 RP: 1001–1020 | NM_002046 |
| Old-Pre | Old-Post | Young | |
|---|---|---|---|
| Body mass (kg) | 89.8 ± 11.7 | 90.1 ± 11.5 | 89.7 ± 11.7 |
| FFM (kg) | 59.2 ± 6.1 | 59.9 ± 6.4 * | 69.3 ± 8.0 *# |
| FM (kg) | 27.4 ± 7.5 | 27.0 ± 7.2 | 17.2 ± 7.2 *# |
| VL thickness (cm) | 2.18 ± 0.4 | 2.31 ± 0.3 * | 2.8 ± 0.3 *# |
| mCSA (cm2) | 148.9 ± 21.4 | 151.1 ± 21.7 | 201.3 ± 28.9 *# |
| Knee ext—60°/s (N.m) | 154 ± 55 | 173 ± 41 * | 229 ± 46 *# |
| Knee ext—120°/s (N.m) | 125 ± 41 | 142 ± 34 * | 191 ± 36 *# |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mesquita, P.H.C.; Lamb, D.A.; Godwin, J.S.; Osburn, S.C.; Ruple, B.A.; Moore, J.H.; Vann, C.G.; Huggins, K.W.; Fruge, A.D.; Young, K.C.; et al. Effects of Resistance Training on the Redox Status of Skeletal Muscle in Older Adults. Antioxidants 2021, 10, 350. https://doi.org/10.3390/antiox10030350
Mesquita PHC, Lamb DA, Godwin JS, Osburn SC, Ruple BA, Moore JH, Vann CG, Huggins KW, Fruge AD, Young KC, et al. Effects of Resistance Training on the Redox Status of Skeletal Muscle in Older Adults. Antioxidants. 2021; 10(3):350. https://doi.org/10.3390/antiox10030350
Chicago/Turabian StyleMesquita, Paulo H. C., Donald A. Lamb, Joshua S. Godwin, Shelby C. Osburn, Bradley A. Ruple, Johnathon H. Moore, Christopher G. Vann, Kevin W. Huggins, Andrew D. Fruge, Kaelin C. Young, and et al. 2021. "Effects of Resistance Training on the Redox Status of Skeletal Muscle in Older Adults" Antioxidants 10, no. 3: 350. https://doi.org/10.3390/antiox10030350








