Abstract
Pimpinella brachycarpa Nakai, known as cham-na-mul in Korea, is a popular edible herb and vegetable. Phenolic compounds are recognized as a vital group of plant secondary metabolites that provide innumerable, valuable therapeutic properties. Elicitors are biofactors or chemicals from diverse sources that can trigger morphological and physiological responses in the target organism. This study examined the effect of methyl jasmonate (MeJA), salicylic acid (SA), and chitosan treatment on the accretion of phenolic compounds in P. brachycarpa Nakai. This plant was harvested under different concentration of elicitor treatment for time course. Eight phenolic compounds including were detected in response to elicitor using HPLC. While the untreated controls showed the lowest phenolic content, treatment with 0.3% chitosan, 0.1 mM SA, and 0.1 mM MeJA resulted in 1.43-, 1.39-, and 1.35-fold increase in the phenolic content, respectively. The highest content of most of the individual phenolic compounds followed a similar trend according to treatment type, with chitosan treatment showing the highest content, followed by SA and then MeJA treatments. Thus, we demonstrate that the treatment with optimal concentrations of these elicitors for an optimal period of time increases the production of phenolic compounds in P. brachycarpa Nakai.
1. Introduction
Pimpinella brachycarpa Nakai, which belongs to the Apiaceae family, is widely distributed in Asia, Africa, and Europe [1]. This plant, known as cham-na-mul in Korea, is a popular edible herb and vegetable [2]. In addition, this plant has been used as folk medicine in Korea for the treatment of persistent cough, bronchial asthma, gastrointestinal disturbances, and insomnia [3]. In previous phytochemical studies, essential oil components, terpenes, and flavonoids were isolated from P. brachycarpa Nakai [3,4,5]. An EtOH extract of P. brachycarpa Nakai exhibited a positive effect on the lipid metabolism of rats with high cholesterol and chronic hepatotoxicity in rat liver induced by ethanol [3,6]. Moreover, a MeOH extract of P. brachycarpa Nakai showed positive effects on antioxidant, antifungal, antithrombotic, and antibacterial activities [7,8]. In addition, many previous studies have reported the content of inorganic components, tannin, fiber, general components, and vitamins in the stem and leaves of P. brachycarpa Nakai [7,8,9,10].
Phenolic compounds, which are commonly found in the plant kingdom, are secondary metabolites arising biogenetically from either the shikimate or the phenylpropanoid pathway [11]. They play a key role as defense compounds against biotic and abiotic stresses, such as pathogen infection, insects, low temperature, and high light, and perform many physiological functions such as the mediation of plant-pollinator interactions [12,13]. Previous studies have reported that phenolic compounds affect antioxidant [14], anticarcinogenic [15], anti-inflammatory [16], anticancer, [17] and cardioprotective [18] activities. Phenolic compounds are classified into different groups according to their basic skeleton: C6 (simple phenol, benzoquinones), C6—C1 (phenolic acid), C6—C2 (acetophenone, phenylacetic acid), C6—C3 (hydroxycinnamic acid, coumarin, phenylpropanes, chromones), C6—C4 (naphthoquinones), C6—C1—C6 (xanthones), C6—C2—C6 (stilbenes, anthraquinones), and C6—C3—C6 (flavonoids, isoflavonoids, neoflavonoids) [11]. The biosynthesis of phenylpropanoids and flavonoids as well as the various catalyzing enzymes involved in the process are shown in Figure 1 [19].
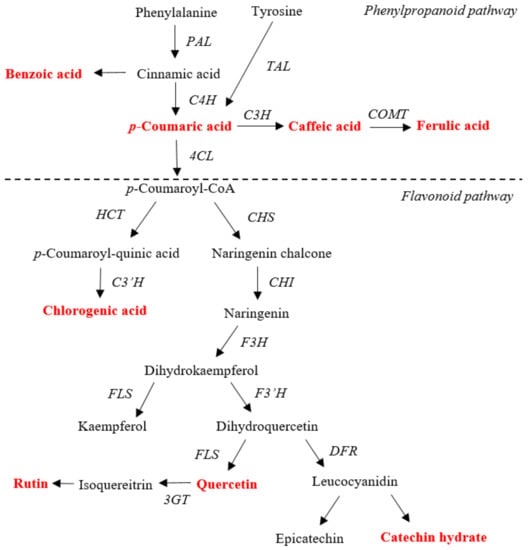
Figure 1.
Schematic representation of phenylpropanoid and flavonoid biosynthetic pathways. Red bold color indicates the phenylpropanoids and flavonoids measured in this study. PAL, phenylalanine ammonia lyase; TAL, tyrosine ammonia lyase; C4H, cinnamate 4-hydroxylase; C3H, coumarate 3-hydroxylase; COMT, caffeic acid 3-O-methyltransferase; 4CL, 4-coumaroyl CoA ligase; HCT, hydroxycinnamoyl-coA shilimate/quinate hydroxycinnamoil transferase; C3′H, p-coumaroyl ester 3′-hydroxilase; CHS, chalcone synthase; CHI, chalcone isomerase; F3H, flavanone 3-hydroxylase; F3′H, flavonoid 3′-hydroxylase; FLS, flavonol synthase; DFR, dihydroflavonol-4 reductase; 3GT, flavonoid 3-O-glucosyltransferase [19].
Elicitors are biofactors or chemicals from diverse sources that can trigger morphological and physiological responses in the target organism [20]. Elicitors are classified as either biotic or abiotic. Biotic elicitors may include viruses, bacteria, fungi, or herbivores, and abiotic elicitors may include inorganic compounds and metal ions. In addition, they can include chemicals that are generated for defense against herbivore and pathogen attack [20]. The production of secondary metabolites in plants can be activated by signal molecules, abiotic stresses, and elicitors. In particular, the accumulation of secondary metabolites can be stimulated by elicitors such as jasmonic acid (JA), salicylic acid (SA), chitosan, and ultraviolet-A or B (UV-A or B) radiation [21]. Previous studies have reported that JA from the cells of Hypericum perforatum L. (St. John’s wort) cause an increase in the total phenolic content [22]. Furthermore, the phenolic content of chitosan-elicited Cocos nucifera (coconut) cells was shown to be increased [23]. In tomato seedlings, UV-A treatment stimulated phenylalanine ammonia lyase (PAL), which is an important enzyme in the phenylpropanoid biosynthesis and increases the accumulation of anthocyanin in the hypocotyls and fruits [24].
Likewise, the production of hypericin and hyperforin in H. perforatum was shown to be increased upon exposure to UV-B [25].
Studies on the phenolic content of P. brachycarpa Nakai are insufficient and the effect of elicitor treatment has not been investigated. Therefore, this study examined the production of phenolic compounds in P. brachycarpa Nakai upon treatment with the elicitor methyl jasmonate (MeJA), SA, and chitosan.
2. Materials and Methods
2.1. Plant Materials and Elicitor Treatment
P. brachycarpa Nakai seedlings were planted at intervals of 15 to 20 cm in a shading-shielded greenhouse and cultivated for 60 days. Then, 0.1, 0.3, or 0.5 mM MeJA (Sigma-Aldrich Co., St. Louis, MO, USA) in aqueous solution containing 0.05% Tween 20 was sprayed on the leaves and stems of P. brachycarpa Nakai. Treated samples were harvested on days 3, 6, and 9 post-treatment for comparison of their phenylpropanoid content. SA (Sigma-Aldrich Co., St. Louis, MO, USA) and chitosan (Ja Kwang Co. Ltd., Anseong, Korea) treatments were performed using the same method. Treatment concentrations of SA were 0.1, 0.3, and 0.5 mM. The treatment concentrations of chitosan were 0.1, 0.3, and 0.5%. For each elicitor treatment, three plants were harvested. As a control, 0.05% Tween 20 in distilled water was used.
2.2. Extraction Procedure of Phenolic Compounds
For phenolic compound extraction, the harvested P. brachycarpa Nakai samples were deep-frozen in liquid nitrogen and freeze-dried at −70 °C for 72 h. Then, the samples were pulverized to fine powder using a pestle until 100 mg of each sample was obtained. For phenylpropanoid extraction, 2 mL of 80% methanol was added to the samples, followed by vortexing for 2 min and sonication at 37 °C for 1 h. Then, the sonicated samples were centrifuged at 12,000 rpm for 12 min at 4 °C and the supernatants were filtered through 0.45 µm filter papers. The solution was then transferred to vials for high-performance liquid chromatography (HPLC) analysis. The content of each phenolic compound was calculated based on the calibration curve and the peak area of the standard compounds for each sample.
2.3. HPLC Analysis of Phenolic Compounds
The HPLC conditions are presented in Table 1. For peak identification and quantification, spike tests were performed, and the content of phenolic compounds was quantified using standard calibration curves. The calibration curves of the standard compounds were as follows: catechin hydrate, y = 28025.39 x − 2223.87 (R2 = 0.9997); chlorogenic acid, y = 52135.70 x − 57351.97 (R2 = 0.9991); caffeic acid, y = 101427.93 x − 19999.04 (R2 = 0.9990); p-coumaric acid, y = 172376.91 x − 2288.50 (R2 = 0.9991); ferulic acid, y = 115029.24 x − 16713.48 (R2 = 0.9995); benzoic acid, y = 16424.67 x + 1917.49 (R2 = 0.9996); rutin, y = 25662.41 x − 3352.89 (R2 = 0.9990); quercetin, y = 40520.94 x + 12547.82 (R2 = 0.9994). All standards used in this work were purchased from Sigma-Aldrich (USA).

Table 1.
The HPLC conditions for phenylpropanoid analysis.
2.4. Statistical Analysis
The content of the different phenylpropanoid compounds is presented as the mean ± standard deviation of three replicates. The data were analyzed using IBM SPSS statistics 26 software (IBM Corp., Armonk, NY, USA). Statistical significance was evaluated with analysis of variance (ANOVA) using Duncan’s multiple range test (DMRT) with a significance level of p ≤ 0.05.
3. Results
3.1. Effect of MeJA Treatment on The Phenolic Content (mg/g dry wt.) of P. brachycarpa Nakai Samples
HPLC analysis was performed to examine and compare the phenolic content of P. brachycarpa Nakai sprouts taken at different harvest times (3, 6, and 9 days) after MeJA treatment. Eight phenolic compounds (catechin hydrate, chlorogenic acid, caffeic acid, p-coumaric acid, ferulic acid, benzoic acid, rutin, and quercetin) were detected in different amounts at different sampling times (Figure 2). MeJA treatment significantly influenced the production of all phenolic compounds except quercetin. The total phenolic content ranged between 10.20 and 13.78 mg/g dry wt., depending on the harvesting day. The highest total phenolic content (13.78 mg/g dry wt.) was detected 9 days after treatment with 0.1 mM MeJA, which was 1.35 times higher than the lowest total phenolic content detected in a control sample. The second highest total phenolic content (13.71 mg/g dry wt.) was detected 6 days after treatment with 0.1 mM MeJA. The content of catechin hydrate MeJA-treated samples ranged between 0.19 and 0.37 mg/g dry wt., depending on the harvesting time, with the highest content (0.37 mg/g dry wt.) detected 3 days after treatment with 0.3 mM MeJA, followed by that found 6 days after treatment with 0.1 mM MeJA (0.37 mg/g dry wt.). After 6 days from MeJA application, the catechin hydrate content tended to decline. It is noteworthy that the catechin hydrate content in any of the MeJA-treated samples was higher than that in any of the control samples. Among all detected phenolic compounds, chlorogenic acid showed the highest content, ranging between 8.14. and 11.38 mg/g dry wt. The highest chlorogenic acid content (11.38 mg/g dry wt.) was detected 9 days after exposure to 0.1 mM MeJA, which was 1.4 times higher than the lowest chlorogenic acid content found in a control sample. The second highest chlorogenic acid content was observed 6 days after exposure, to 0.3 mM MeJA (11.29 mg/g dry wt.), followed by that found 3 days after exposure to 0.1 mM MeJA (10.92 mg/g dry wt.). Remarkably, the chlorogenic acid content found in any of the MeJA-treated samples was higher than that found in any of the control samples. The levels of caffeic acid, p-coumaric acid, and ferulic acid were lower and their values were almost similar irrespective of the treatment used. The range of caffeic acid content in MeJA-treated samples at different harvesting times was 0.09–0.16 mg/g dry wt. The highest content of caffeic acid at any harvesting time was detected after treatment with 0.1 mM MeJA, with the highest amount (0.16 mg/g dry wt.) found 9 days after treatment, which was 1.78 times higher than the lowest content found in a control sample. The range of p-coumaric acid content in MeJA-treated samples at different harvesting times was 0.04–0.09 mg/g dry wt. The highest content of p-coumaric acid at any harvesting time was detected after treatment with 0.3 mM MeJA, with the highest amount (0.09 mg/g dry wt.) found 3 days after treatment, which was 2.25 times higher than the lowest content found in a control sample. The range of ferulic acid content in MeJA-treated samples at different harvesting times was 0.04–0.05 mg/g dry wt., showing hardly any variation between samples. The trend in caffeic acid, benzoic acid, rutin, and total phenolic content was the same, with the highest content of these compounds at any harvesting time detected after treatment with 0.1 mM MeJA. The range of benzoic acid content in MeJA-treated samples at different harvesting times was 0.49–0.71 mg/g dry wt. The highest benzoic acid content (0.71 mg/g dry wt.) was found 9 days after treatment with 0.1 mM MeJA, which was 1.45 times higher than that found 9 days after exposure to 0.5 mM MeJA, corresponding to the lowest benzoic acid content found. The rutin content in MeJA-treated samples at different harvesting times ranged between 0.60. and 0.93 mg/g dry wt. The highest rutin content (0.93 mg/g dry wt.) was found 3 days after treatment with 0.1 mM, which was 1.55 times higher than that found 9 days after exposure to 0.5 mM MeJA, corresponding to the lowest rutin content found. The quercetin content in MeJA-treated samples at different harvesting times ranged between 0.28 and 0.51 mg/g dry wt., and the highest quercetin content was observed in the control samples. This indicated that MeJA treatment did not influence quercetin content.
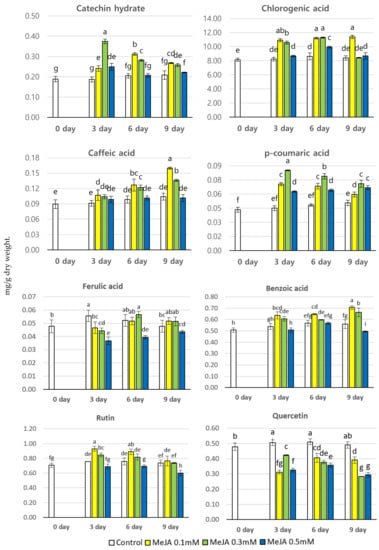
Figure 2.
The content of phenolic compounds (mg/g dry wt.) in P. brachycarpa Nakai samples after treatment with different concentrations of methyl jasmonate (MeJA). The values marked above the bars with different letters (a–i) indicate a statistically significant difference between treatments, as determined using DMRT (p < 0.05).
3.2. Effect of SA Treatment on The Phenolic Content (mg/g dry wt.) of P. brachycarpa Nakai Samples
The levels of phenolic compounds in P. brachycarpa Nakai sprouts were investigated at different harvesting times (3, 6, and 9 days) after treatment with different concentrations of SA using HPLC. Eight different phenolic compounds were observed in different amounts at different sampling times (Figure 3). Treatment with SA increased the content of most phenolic compounds, except for benzoic acid, rutin, and quercetin. Here, the content of benzoic acid, rutin, and quercetin was lower in SA-treated samples than in the control samples. The content of the different phenolic compounds followed different trends depending on the SA concentration used and the harvesting time. SA treatments had a significant influence on the total phenolic content, ranging between 10.20 and 4.14 mg/g dry wt. at different harvesting times. The highest total phenolic content (14.14 mg/g dry wt.) was detected 9 days after exposure to 0.1 mM SA, which was 1.39 times higher than the lowest content found in a control sample. The second highest total phenolic content (13.47 mg/g dry wt.) was detected 6 days after treatment with 0.1 mM SA, followed by that found 3 days after exposure to 0.3 mM SA (13.5 mg/g dry wt.). The content of chlorogenic acid was much higher than that of any other phenolic compound, irrespective of the treatment type. The chlorogenic acid content in SA-treated samples ranged between 8.14. and 12.34 mg/g dry wt. The highest content (12.34 mg/g dry wt.) was detected 9 days after treatment with 0.1 mM SA, which was 1.52 times higher than the lowest content found in a control sample. The second highest chlorogenic acid content was observed 6 days after treatment with 0.1 mM SA (11.61 mg/g dry wt.), followed by that found 3 days after exposure to 0.3 mM SA (11.60 mg/g dry wt.). The chlorogenic acid content was much higher in any of the SA-treated samples than in any control sample. The content of catechin hydrate in SA-treated samples ranged between 0.19. and 0.32 mg/g dry wt. The highest content (0.32 mg/g dry wt.) was detected 3 days after treatment with 0.1 mM SA. The catechin hydrate content of the samples treated with either 0.1 mM or 0.3 mM SA was very similar at all sampling dates. The catechin hydrate content was higher in any of the SA-treated samples than that in the control samples. In comparison, the content of p-coumaric acid, caffeic acid, and ferulic acid was much lower irrespective of the treatment type. The caffeic acid content in SA-treated samples ranged between 0.09 and 0.12 mg/g dry wt. The highest caffeic acid content was detected 3 days after treatment with 0.1 mM SA (0.12 mg/g dry wt.). On the other hand, the content of p-coumaric acid in SA-treated samples ranged between 0.04 and 0.06 mg/g dry wt., and was almost the same in all treated samples, indicating that SA-treatment did not influence the content of p-coumaric acid. The content of ferulic acid in SA-treated samples ranged between 0.05. and 0.08 mg/g dry wt., and the values were almost the same among the different treated samples.
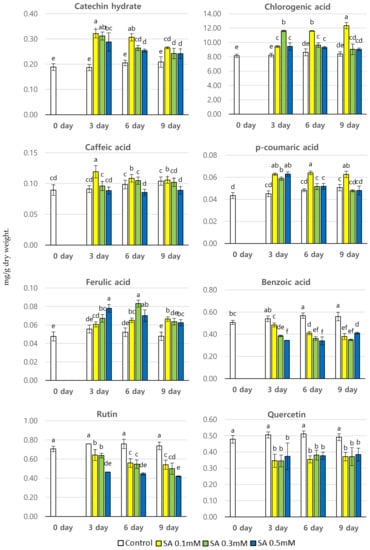
Figure 3.
The content of phenolic compounds (mg/g dry wt.) in P. brachycarpa Nakai samples after treatment with different concentrations of salicylic acid (SA). The values marked above the bars with different letters (a–f) indicate a statistically significant difference between treatments, as determined using DMRT (p < 0.05).
3.3. Effect of Chitosan Treatment on The Phenolic Content (mg/g dry wt.) of P. brachycarpa Nakai Samples
P. brachycarpa Nakai samples treated with different concentrations of chitosan were harvested on days 3, 6, and 9 post-treatment to examine the content of different phenolic compounds using HPLC analysis. Eight different phenolic compounds were detected at varying amounts on different sampling dates (Figure 4). Chitosan treatment increased the content of all detected phenolic compounds except for rutin. Here, the content of rutin was lower in the chitosan-treated samples than in the control samples. The different phenolic compounds followed different trends depending on the chitosan concentration and the harvesting times. The total phenolic content of chitosan-treated samples ranged between 10.20 and 14.63 mg/g dry wt. The highest total phenolic content (14.63 mg/g dry wt.) was detected 9 days after treatment with 0.3% chitosan, which was 1.43 times higher than the lowest total phenolic content of a control sample. The second highest total phenolic content (14.47 mg/g dry wt.) was detected 6 days after treatment with 0.5% chitosan. The content of chlorogenic acid was much higher than that of any other phenolic compounds irrespective of the treatment type. The range of chlorogenic acid content in chitosan-treated samples was 8.14–11.87 mg/g dry wt., with the highest content (11.874 mg/g dry wt.) detected 9 days after treatment with 0.3% chitosan. The second highest chlorogenic acid content (11.74 mg/g dry wt.) was found 6 days after treatment with 0.5% chitosan. The catechin hydrate content in chitosan-treated samples ranged between 0.19 and 0.47 mg/g dry wt. The highest catechin hydrate content (0.47 mg/g dry wt.) was detected 9 days after treatment with 0.5% chitosan. The levels of p-coumaric acid, caffeic acid, and ferulic acid were much lower, and the values were almost identical irrespective of the treatment type. The caffeic acid content in chitosan-treated samples ranged between 0.09 and 0.13 mg/g dry wt. The highest caffeic acid content at any harvesting time was detected after treatment with the highest concentration of chitosan (0.5%). The range of p-coumaric acid content in chitosan-treated samples was 0.04–0.12 mg/g dry wt. The highest p-coumaric acid content (0.12 mg/g dry wt.) was detected 9 days after treatment with 0.5% chitosan. The ferulic acid content in chitosan-treated samples ranged between 0.05 and 0.11 mg/g dry wt. The highest ferulic acid content (0.11 mg/g dry wt.) was detected 6 and 9 days after treatment with 0.5% chitosan. The benzoic acid content in chitosan-treated samples was 0.51–0.79 mg/g dry wt. The highest benzoic acid content (0.79 mg/g dry wt.) was found 3 days after treatment with 0.5% chitosan, which was 1.55 times higher than the lowest content found in a control sample. The content of quercetin in chitosan-treated samples ranged between 0.48 and 0.73 mg/g dry wt. The highest quercetin content (0.73 mg/g dry wt.) was found 6 days after treatment with 0.5% chitosan, which was 1.52 times higher than the lowest content detected in a control sample.
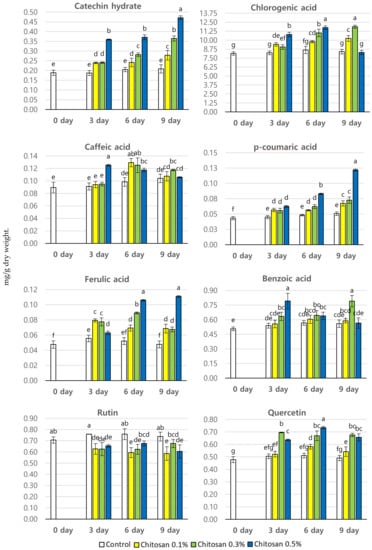
Figure 4.
The content of phenolic compounds (mg/g dry wt.) in P. brachycarpa Nakai samples taken at different harvesting days after treatment with different chitosan concentrations. The values marked above the bars with different letters (a–f) indicate statistically significant differences between treatments, as determined using DMRT (p < 0.05).
3.4. Comparison of The Effect of MeJA, SA, and Chitosan Treatments on The Phenolic Content (mg/g dry wt.) of P. brachycarpa Nakai Samples
The highest total phenolic content (14.63 mg/g dry wt.) was detected 9 days after treatment with 0.3% chitosan, followed by that found 9 days after treatment with 0.1 mM SA (14.14 mg/g dry wt.) and then by that found 9 days after treatment with 0.1 mM MeJA (13.78 mg/g dry wt.) (Table 2). The phenolic content 9 days after treatment with 0.3% chitosan, 0.1 mM SA, or 0.1 mM MeJA was 1.43, 1.39, and 1.35 times higher than the lowest content found in a control sample (10.20 mg/g dry wt.), respectively. The highest chlorogenic acid content (12.34 mg/g dry wt.) was detected 9 days after treatment with 0.1 mM SA, followed by that found 9 days after treatment with 0.3% chitosan (11.87 mg/g dry wt.) and then by that found 9 days after treatment with 0.1 mM MeJA (11.38 mg/g dry wt.).

Table 2.
The content of total phenolic compounds (mg/g dry wt.) in P. brachycarpa Nakai under different elicitor treatment.
The chlorogenic acid content 9 days after treatment with 0.1 mM SA, 0.3% chitosan, or 0.1 mM MeJA was 1.52, 1.46, and 1.40 times higher than the lowest content found in a control sample (8.14 mg/g dry wt.), respectively. The highest catechin hydrate content (0.47 mg/g dry wt.) was detected 9 days after treatment with 0.5% chitosan, followed by that found 3 days after 0.3 mM MeJA treatment (0.37 mg/g dry wt.), and then by that found 3 days after 0.1 mM SA treatment (0.32 mg/g dry wt.). The catechin hydrate content in these three samples was 2.47, 1.95, and 1.68 times higher than the lowest catechin hydrate content found in a control sample (0.19 mg/g dry wt.), respectively. The highest caffeic acid content (0.16 mg/g dry wt.) was detected 9 days after treatment with 0.1 mM MeJA, followed by that found 3 days after treatment with 0.5% chitosan (0.13 mg/g dry wt.), and then that found 3 days after treatment with 0.1 mM SA (0.12 mg/g dry wt.). These treatments resulted in 1.78, 1.44, and 1.33 times higher caffeic acid content than the lowest content found in a control sample (0.09 mg/g dry wt.), respectively. The highest p-coumaric acid content (0.12 mg/g dry wt.) was detected 9 days after treatment with 0.5% chitosan, followed by those found 3 days after treatment with 0.3 mM MeJA (0.0.9 mg/g dry wt.) and 0.1 mM SA (0.06 mg/g dry wt.). These three treatments resulted in 3.0, 2.25, and 1.5 times higher p-coumaric acid content than the lowest content found in a control sample (0.09 mg/g dry wt.), respectively. The highest ferulic acid content (0.11 mg/g dry wt.) was detected 9 days after treatment with 0.5% chitosan, followed by that found 3 days after treatment with 0.5 mM SA (0.0.9 mg/g dry wt.). These treatments resulted in 2.2 and 1.6 times higher ferulic acid content, respectively, than the lowest content found in a control sample (0.05 mg/g dry wt.). The highest benzoic acid content (0.79 mg/g dry wt.) was detected 9 days after treatment with 0.3% chitosan, followed by that found 9 days after treatment with 0.1 mM MeJA (0.71 mg/g dry wt.). These treatments resulted in 1.55 and 1.39 times higher benzoic acid content, respectively, than the lowest content found in a control sample (0.51 mg/g dry wt.). A slightly higher rutin content was only observed after 0.1 mM MeJA treatment. SA and chitosan treatments did not influence the rutin content. Moreover, the quercetin content was increased slightly after treatment with 0.5% chitosan, whereas MeJA and SA treatments did not show any effect on quercetin content.
4. Discussion
Elicitation is recognized as one of the most important techniques for the stimulation of secondary metabolites. The accretion of any metabolites from either part of parent or transformed plants is largely reliant on the source; however, it may be influenced by treatments as well as environmental factors. Elicitors trigger an increase in the content of phenolic compounds, pigments, flavones, phytoalexins, and other defense-related compounds when in contact with the cells of higher plants [26,27,28,29,30,31,32].
The levels of phenolic compounds in P. brachycarpa Nakai were investigated in response to different elicitor (MeJA, SA, and chitosan) treatments at different harvesting times (3, 6, and 9 days) post-treatment. In this study, eight phenolic compounds (catechin hydrate, chlorogenic acid, caffeic acid, p-coumaric acid, ferulic acid, benzoic acid, rutin, and quercetin) were detected in different amounts at different sampling times. All the elicitors significantly increased the content of most of the phenolic compounds compared to the untreated controls. These results are consistent with previous studies reporting the identification of these types of compounds in common buckwheat [33,34], tartary buckwheat [35], hairy root cultures of tartary buckwheat [36], Agastache rugosa [37], sorghum [38], Scutellaria baicalensis [39], and Kohlrabi [40].
Our time course analysis revealed that MeJA, SA, and chitosan treatment progressively enhanced the production of phenolic compounds in the studied species. This might be due to an increase in the expression levels of phenylpropanoid-related genes following treatment with MeJA, SA, or chitosan, since previous studies proclaimed that different types of elicitors increase the expression levels of phenlypropanoid-related genes and enhance the accumulation of phenolic compounds. Some of these findings are relevant to our study and are discussed here. MeJA treatment was shown to increase the expression levels of phenlypropanoid-related genes and enhance the accumulation of phenolic compounds in radish sprouts [41] and A. rugosa Kuntze [37]. Light treatment with 100 μM methyl jasmonate of radish seedlings increased the anthocyanin content in radish [42]. The rosmarinic acid content in A. rugosa Kuntze was 2.1-, 4.7-, and 3.9-fold higher after exposure to 10, 50, and 100 mM MeJA, respectively, than that in untreated controls [37]. The content of phenolic acids was increased in Exacum affine shoot cultures by adding the precursor (L-phenylalanine), the elicitor (MeJA), and by increasing the sucrose concentration [43]. At least a 6–8-fold increase in PAL and CHI activities was observed in JA-elicited cells, confirming a strong activation of the phenylpropanoid pathway. JA elicitation increased the production of phenylpropanoids and naphtodianthrones in a Hypericum perforatum cell suspension without differentiation of dark glands under a 16 h photoperiod [22]. Most of the above-mentioned findings coincide with our study, in which MeJA treatment significantly increased the content of all detected phenolic compounds, except quercetin.
The rutin content has been shown to increase following treatment with SA in tartary buckwheat [44]. After 72 h from the application of SA, the levels of 4-hydroxybenzoic acid, 4-coumaric acid, and ferulic acid were shown to be 3.5-, 2.9-, and 3.8-fold higher than those in the control samples, respectively, in eggplant roots [45]. At 50 µM, SA enhanced the production of hypericin (7.98-fold) and pseudohypericin (13.58-fold) in H. hirsutum, and at 200 µM, enhanced the production of hypericin (2.2-fold) and pseudohypericin (3.94-fold) in H. maculatum [46]. Thus, our results are consistent with those from others, as SA treatment increased the content of most phenolic compounds, except benzoic acid, rutin, and quercetin.
Eight different phenolic compounds were detected in different amounts at different sampling times in response to different concentrations of chitosan in P. brachycarpa Nakai. Treatment with chitosan increased the content of all detected phenolic compounds except rutin. Our results on phenolic content upon chitosan treatment are supported by previous studies. In soybean leaves, the total phenolic content increased following chitosan and chitin oligomer treatments [47]. Treatment with 0.1% chitosan and 150 µM JA resulted in the highest levels of phenolic compounds compared to those of the control, whereas SA treatment did not affect the production of phenolic compounds in sprouted buckwheat. A sample of buckwheat taken 72 h after 0.1% chitosan treatment showed higher levels of phenolic compounds than those in all control samples [48]. The treatment of Cocos nucifera (coconut) endosperm cell suspension cultures with chitosan led to the rapid formation of phenylpropanoid derivatives [23]. Moreover, 48 h post-elicitation with chitosan, the content of 4-hydroxybenzoic acid, 4-coumaric acid, and ferulic acid in the cell wall of eggplants was 2.6-, 2.8-, and 3.0-fold higher than that in the control sample, respectively [45].
Qi et al. [49] revealed that exogenous MeJA treatment up-regulated many photosynthesis and defense-related proteins, and down-regulated a number of ribosomal proteins. They also discovered that MeJA treatment can maintain a better reactive oxygen species (ROS) scavenging capability, by decreasing Hydrogen peroxide, peroxidase. And they suggested that up-regulated proteins stress-related, such as DHAR1, ATCOR47, PDE334, ATHM1 can affect antioxidant activity. The previous study suggested that salicylic acid was synthesized in cells can move freely in and out of the cells, tissues, and organs, and this movement is finely regulated by ROS and Ca2+ [50]. The treatment with a moderate concentration of SA increased the activity of the enzyme, finally increased growth and bio-productivity. However, the treatment with higher concentrations of SA decreased the activity of the enzyme by reducing the photosynthesis-related enzyme activity such as ribulose-1,5-biphosphate carboxylase/oxygenase (RuBPCO) [50]. In a previous study revealed that chitosan-treated plants up-regulated PAL, peroxidase, glucanase, polyphenol oxidase, and defense-responsive enzymes [51]. Farag et al. [52] investigated elicitor-specific biosynthesis of isoflavonoids in response to yeast elicitor (YE) and MeJA in Medicago truncatula Cell Cultures. The response to YE increased isoflavonoid biosynthesis, including the shikimic acid pathway and flavonoid biosynthesis. MeJA treatment resulted in contrast to that of YE, increased in afromosin and medicarpin and, but many pathway precursors were not changed. Based on the abundant precursor pathways, an increased medicarpine of isoflavonoid for YE is achieved from de novo biosynthesis; in contrast, MeJA-induced medicarpine accumulation occurs through remigration of the vacuole pool of formononetin glucoside and hydrolysis. Kubes et al. [53] investigated the elicitation effect of two vanadium compounds, which could make a positive effect on the isoflavones production in Trifolium pratense. The content of isoflavones in T. pratense suspension culture was increased after 24 h by vanadium compounds, NH4VO3. However, increased isoflavones since been suppressed by transport-mechanisms inhibitors. This suppress process was mostly facilitated by vesicle transport and ATP-binding cassette protein. These findings suggest that elicitors can have a positive effect on plants through various mechanisms and physiological activities.
A previous study revealed that studies on the biological activities of rare phenylpropanoids of the genus Pimpinella, such as anoltiglate, isoeugenolisobutyrate, epoxy-pseudoisoeugenoltiglate [54]. Delazar et al. [55] investigated a chemical profile of the essential oils of Pimpinella aurea that was significantly different from the previously published reports, β-Bisabolene (39.559%) and geranyl 2-methylbutanoate (25.334%) were the two most abundant components. Additionally, both phenylpropanoid derivatives, erythro-1′-(4-methoxyphenyl)-propan-1′,2′-diol, and erythro-1′-[4-(sec-butyl)-phenyl]-propan-1′,2′-diol, were identified by using vacuum liquid chromatography (VLC) and preparative thin-layer chromatography (PTLC). And these were exhibited low levels of antioxidant activity in the DPPH assay. In this study, we investigate the accumulation of phenolic compounds in P. brachycarpa Nakai upon treatment with the elicitor MeJA, SA, and chitosan. The samples were taken 9 days after treatment with 0.3% chitosan, 0.1 mM SA, and 0.1 mM MeJA showed 1.43-, 1.39-, and 1.35-fold increases in total phenolic content, respectively, compared to the lowest total phenolic content found in a control sample. Therefore, chitosan, SA, and MeJA treatment augment the production of phenolic compounds in P. brachycarpa Nakai.
5. Conclusions
This study shows that chitosan, SA, and MeJA enhance the production of different phenolic compounds. Eight different phenolic compounds, including chlorogenic acid, catechin hydrate, caffeic acid, p-coumaric acid, ferulic acid, benzoic acid, rutin, and quercetin were detected in P. brachycarpa Nakai. All the elicitors tested increased the content of most of the detected phenolic compounds. Particularly, 1.43-, 1.39-, and 1.35-fold increases in total phenolic content were achieved 9 days after treatment with 0.3% chitosan, 0.1 mM SA, and 0.1 mM MeJA, respectively, compared to the lowest total phenolic content detected in a control sample. These results might help to create robust strategies for the improvement of phenolic production by P. brachycarpa Nakai, which will positively impact its use as an edible herb and folk medicine in Korea and elsewhere.
Author Contributions
H.W.P. and S.U.P. designed the experiments and analyzed the data. N.S.K., D.H.J., C.R.J. and K.S.J. performed the experiments and analyzed the data. N.S.K., H.W.P. and S.U.P. wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by the National Institute of Forest Science, Korea Forest Service, Korea (Project No. FG0502-2017-01).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Lee, C. Coloured Flora of Korean Medicinal Herbs; Koy-Hak Publishing, Co.: Seoul, Korea, 1998; pp. 430–436. [Google Scholar]
- Ahn, S.-M.; Kim, M.-S.; Jung, I.-C.; Sohn, H.-Y. Antibacterial, antioxidative and anti-proliferative activity against human colorectal cell of Pimpinella brachycarpa. Korean J. Food Preserv. 2011, 18, 590–596. [Google Scholar]
- Lee, J.-J.; Choo, M.-H.; Lee, M.-Y. Physicochemical compositions of Pimpinella brachycarpa. J. Korean Soc. Food Sci. Nutr. 2007, 36, 327–331. [Google Scholar]
- Oh, S.-S.; Lee, J.-M.; Choi, N.-S. Change of biologically functional compounds of Pimpinella brachycarpa (Chamnamul) by blanching conditions. J. Korean Soc. Food Cult. 2001, 16, 388–397. [Google Scholar]
- Lee, S.Y.; Shin, Y.J.; Lee, K.R. Two new sesquiterpenes from the aerial parts of Pimpinella brachycarpa Nakai. Notes 2013, 34, 2215. [Google Scholar] [CrossRef]
- Lee, J.-J.; Choo, M.-H.; Lee, M.-Y. Effect of Pimpinella brachycarpa extract on lipid metabolism in rats fed high cholesterol diet. J. Korean Soc. Food Sci. Nutr. 2006, 35, 1151–1158. [Google Scholar]
- Chang, K. Cookery study of Pimpinella brachycarpa for Practical Utilization. Ph.D. Thesis, Duksung Women’s University, Seoul, Korea, 2003. [Google Scholar]
- Kwon, C.; Kwon, Y.; Kim, Y.; Kwon, G.; Jin, I.; Ryu, G.; Sohn, H. Inhibitory activities of edible and medicinal herbs against human thrombin. J. Life Sci. 2004, 14, 509–513. [Google Scholar]
- Choi, M.-H.; Kim, G.-H. A study on quality characteristics of Pimpinella brachycarpa Kimchi during storage at different temperatures. J. Korean Soc. Food Sci. Nutr. 2002, 31, 45–49. [Google Scholar]
- Park, S.; Jeong, G. The Use and Efficacy of Korean Folk Vegetables; Seowon Publishing, Co.: Daegu, Korea, 1995; pp. 204–205. [Google Scholar]
- Lattanzio, V. Phenolic Compounds: Introduction 50. Nat. Prod. 2013, 1543–1580. [Google Scholar] [CrossRef]
- Tossi, V.; Amenta, M.; Lamattina, L.; Cassia, R. Retracted: Nitric oxide enhances plant ultraviolet-B protection up-regulating gene expression of the phenylpropanoid biosynthetic pathway. Plant Cell Environ. 2011, 34, 909–921. [Google Scholar]
- Singh, A.; Sarma, B.K.; Upadhyay, R.S.; Singh, H.B. Compatible rhizosphere microbes mediated alleviation of biotic stress in chickpea through enhanced antioxidant and phenylpropanoid activities. Microbiol. Res. 2013, 168, 33–40. [Google Scholar]
- Critchfield, J.W.; Butera, S.T.; Folks, T.M. Inhibition of HIV activation in latently infected cells by flavonoid compounds. AIDS Res. Hum. Retrovir. 1996, 12, 39–46. [Google Scholar] [CrossRef] [PubMed]
- Ferrazzano, G.F.; Amato, I.; Ingenito, A.; De Natale, A.; Pollio, A. Anti-cariogenic effects of polyphenols from plant stimulant beverages (cocoa, coffee, tea). Fitoterapia 2009, 80, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Fang, S.-C.; Hsu, C.-L.; Yen, G.-C. Anti-inflammatory effects of phenolic compounds isolated from the fruits of Artocarpus heterophyllus. J. Agric. Food Chem. 2008, 56, 4463–4468. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Luo, Q.; Sun, M.; Corke, H. Antioxidant activity and phenolic compounds of 112 traditional Chinese medicinal plants associated with anticancer. Life Sci. 2004, 74, 2157–2184. [Google Scholar] [CrossRef]
- Cook, N.C.; Samman, S. Flavonoids—chemistry, metabolism, cardioprotective effects, and dietary sources. J. Nutr. Biochem. 1996, 7, 66–76. [Google Scholar] [CrossRef]
- Cuong, D.M.; Kwon, S.J.; Jeon, J.; Park, Y.J.; Park, J.S.; Park, S.U. Identification and characterization of phenylpropanoid biosynthetic genes and their accumulation in bitter melon (Momordica charantia). Molecules 2018, 23, 469. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Davis, L.C.; Verpoorte, R. Elicitor signal transduction leading to production of plant secondary metabolites. Biotechnol. Adv. 2005, 23, 283–333. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.R.; Ravishankar, G. Plant cell cultures: Chemical factories of secondary metabolites. Biotechnol. Adv. 2002, 20, 101–153. [Google Scholar]
- Gadzovska, S.; Maury, S.; Delaunay, A.; Spasenoski, M.; Joseph, C.; Hagege, D. Jasmonic acid elicitation of Hypericum perforatum L. cell suspensions and effects on the production of phenylpropanoids and naphtodianthrones. Plant Cell Tissue Organ Cult. 2007, 89, 1–13. [Google Scholar] [CrossRef]
- Chakraborty, M.; Karun, A.; Mitra, A. Accumulation of phenylpropanoid derivatives in chitosan-induced cell suspension culture of Cocos nucifera. J. Plant Physiol. 2009, 166, 63–71. [Google Scholar] [CrossRef]
- Guo, J.; Wang, M.-H. Ultraviolet A-specific induction of anthocyanin biosynthesis and PAL expression in tomato (Solanum lycopersicum L.). Plant Growth Regul. 2010, 62, 1–8. [Google Scholar] [CrossRef]
- Brechner, M.L.; Albright, L.D.; Weston, L.A. Effects of UV-B on secondary metabolites of St. John’s Wort (Hypericum perforatum L.) grown in controlled environments. Photochem. Photobiol. 2011, 87, 680–684. [Google Scholar] [CrossRef]
- Flores, H.E.; Curtis, W.R. Approaches to Understanding and Manipulating the Biosynthetic Potential of Plant Roots a. Ann. N. Y. Acad. Sci. USA 1992, 665, 188–209. [Google Scholar] [CrossRef] [PubMed]
- Sim, S.J.; Chang, H.N.; Liu, J.R.; Jung, K.H. Production and secretion of indole alkaloids in hairy root cultures of Catharanthus roseus: Effects of in situ adsorption, fungal elicitation and permeabilization. J. Ferment. Bioeng. 1994, 78, 229–234. [Google Scholar]
- Bhagyalakshmi, N.; Bopanna, K. Elicitation and immobilization of cell cultures for enhanced synthesis of pharmaceutical compounds. In Role of Biotechnology in Medicinal and Aromatic Plants; Ukaaz Publications: Hyderabad, India, 1998; Volume 1, pp. 305–325. [Google Scholar]
- Singh, G. Elicitation—Manipulating and enhancing secondary metabolite production. In Plant Cell and Tissue Culture for the Production of Food Ingredients; Springer: Berlin/Heidelberg, Germany, 1999; pp. 101–111. [Google Scholar]
- Uddin, M.R.; Park, K.W.; Kim, Y.K.; Park, S.U.; Pyon, J.Y. Enhancing sorgoleone levels in grain sorghum root exudates. J. Chem. Ecol. 2010, 36, 914–922. [Google Scholar] [CrossRef] [PubMed]
- Uddin, M.R.; Park, W.T.; Kim, Y.K.; Pyon, J.Y.; Park, S.-U. Effects of auxins on sorgoleone accumulation and genes for sorgoleone biosynthesis in sorghum roots. J. Agric. Food Chem. 2011, 59, 12948–12953. [Google Scholar] [CrossRef]
- Uddin, M.R.; Thwe, A.A.; Kim, Y.B.; Park, W.T.; Chae, S.C.; Park, S.U. Effects of jasmonates on sorgoleone accumulation and expression of genes for sorgoleone biosynthesis in sorghum roots. J. Chem. Ecol. 2013, 39, 712–722. [Google Scholar] [CrossRef]
- Li, X.; Park, N.I.; Xu, H.; Woo, S.-H.; Park, C.H.; Park, S.U. Differential expression of flavonoid biosynthesis genes and accumulation of phenolic compounds in common buckwheat (Fagopyrum esculentum). J. Agric. Food Chem. 2010, 58, 12176–12181. [Google Scholar] [CrossRef]
- Uddin, M.R.; Li, X.; Park, W.T.; Kim, Y.B.; Kim, S.J.; Kim, Y.S.; Lee, M.Y.; Park, C.H.; Park, S.U. Phenolic compound content in different organs of Korean common buckwheat cultivars. Asian J. Chem. 2013, 25, 424. [Google Scholar] [CrossRef]
- Uddin, M.R.; Li, X.; Kim, Y.B.; Chae, S.C.; Kim, S.-J.; Park, S.U. Phenolic compounds in different organs of tartary buckwheat (Fagopyrum tataricum Gaertn.) cultivars. Aust. J. Crop Sci. 2013, 7, 1861–1865. [Google Scholar]
- Kim, Y.K.; Li, X.; Xu, H.; Park, N.I.; Uddin, M.R.; Pyon, J.Y.; Park, S.U. Production of phenolic compounds in hairy root culture of tartary buckwheat (Fagopyrum tataricum Gaertn). J. Crop Sci. Biotechnol. 2009, 12, 53–57. [Google Scholar]
- Kim, Y.B.; Kim, J.K.; Uddin, M.R.; Xu, H.; Park, W.T.; Tuan, P.A.; Li, X.; Chung, E.; Lee, J.-H.; Park, S.U. Metabolomics analysis and biosynthesis of rosmarinic acid in Agastache rugosa Kuntze treated with methyl jasmonate. PLoS ONE 2013, 8, e64199. [Google Scholar] [CrossRef]
- Won, O.; Uddin, M.; Park, K.; Pyon, J.; Park, S. Phenolic compounds in sorghum leaf extracts and their effects on weed control. Allelopath. J. 2013, 31, 147–156. [Google Scholar]
- Im, A.; Kim, Y.-H.; Uddin, M.; Lee, H.W.; Chae, S.W.; Kim, Y.H.; Jung, W.S.; Kang, B.J.; Mun, C.S.; Lee, M.-Y. Scutellaria baicalensis extracts and flavonoids protect rat L6 cells from antimycin A-induced mitochondrial dysfunction. Evid. Based Complementary Altern. Med. 2012, 2012, 517965. [Google Scholar] [CrossRef]
- Park, W.T.; Kim, J.K.; Park, S.; Lee, S.-W.; Li, X.; Kim, Y.B.; Uddin, M.R.; Park, N.I.; Kim, S.-J.; Park, S.U. Metabolic profiling of glucosinolates, anthocyanins, carotenoids, and other secondary metabolites in kohlrabi (Brassica oleracea var. gongylodes). J. Agric. Food Chem. 2012, 60, 8111–8116. [Google Scholar]
- Chen, H.; Seguin, P.; Archambault, A.; Constan, L.; Jabaji, S. Gene expression and isoflavone concentrations in soybean sprouts treated with chitosan. Crop Sci. 2009, 49, 224–236. [Google Scholar]
- Park, W.T.; Kim, Y.B.; Seo, J.M.; Kim, S.-J.; Chung, E.; Lee, J.-H.; Park, S.U. Accumulation of anthocyanin and associated gene expression in radish sprouts exposed to light and methyl jasmonate. J. Agric. Food Chem. 2013, 61, 4127–4132. [Google Scholar] [CrossRef]
- Skrzypczak-Pietraszek, E.; Słota, J.; Pietraszek, J. The influence of L-phenylalanine, methyl jasmonate and sucrose concentration on the accumulation of phenolic acids in Exacum affine Balf. f. ex Regel shoot culture. Acta Biochim. Pol. 2014, 61, 61. [Google Scholar] [CrossRef]
- Sun, Z.; Hou, S.; Yang, W.; Han, Y. Exogenous application of salicylic acid enhanced the rutin accumulation and influenced the expression patterns of rutin biosynthesis related genes in Fagopyrum tartaricum Gaertn leaves. Plant Growth Regul. 2012, 68, 9–15. [Google Scholar] [CrossRef]
- Mandal, S.; Gupta, C. Inducing cell wall-bound phenolic compounds by elicitors in eggplant (Solanum melongena). Appl. Biochem. Microbiol. 2016, 52, 650–656. [Google Scholar] [CrossRef]
- Coste, A.; Vlase, L.; Halmagyi, A.; Deliu, C.; Coldea, G. Effects of plant growth regulators and elicitors on production of secondary metabolites in shoot cultures of Hypericum hirsutum and Hypericum maculatum. Plant Cell Tissue Organ Cult. 2011, 106, 279–288. [Google Scholar] [CrossRef]
- Khan, W.; Prithiviraj, B.; Smith, D.L. Chitosan and chitin oligomers increase phenylalanine ammonia-lyase and tyrosine ammonia-lyase activities in soybean leaves. J. Plant Physiol. 2003, 160, 859–863. [Google Scholar] [CrossRef] [PubMed]
- Park, C.H.; Yeo, H.J.; Park, Y.E.; Chun, S.W.; Chung, Y.S.; Lee, S.Y.; Park, S.U. Influence of chitosan, salicylic acid and jasmonic acid on phenylpropanoid accumulation in germinated buckwheat (Fagopyrum esculentum Moench). Foods 2019, 8, 153. [Google Scholar] [CrossRef] [PubMed]
- Qi, J.; Zhao, X.; Li, Z. iTRAQ-Based Quantitative Proteomic Analysis of the Arabidopsis Mutant opr3-1 in Response to Exogenous MeJA. Int. J. Mol. Sci. 2020, 21, 571. [Google Scholar] [CrossRef] [PubMed]
- Hayat, Q.; Hayat, S.; Irfan, M.; Ahmad, A. Effect of exogenous salicylic acid under changing environment: A review. Environ. Exp. Bot. 2010, 68, 14–25. [Google Scholar] [CrossRef]
- Jogaiah, S.; Satapute, P.; De Britto, S.; Konappa, N.; Udayashankar, C. Exogenous priming of chitosan induces upregulation of phytohormones and resistance against cucumber powdery mildew disease is correlated with localized biosynthesis of defense enzymes. Int. J. Biol. Macromol. 2020, 162, 1825–1838. [Google Scholar] [CrossRef]
- Farag, M.A.; Huhman, D.V.; Dixon, R.A.; Sumner, L.W. Metabolomics reveals novel pathways and differential mechanistic and elicitor-specific responses in phenylpropanoid and isoflavonoid biosynthesis in Medicago truncatula cell cultures. Plant Physiol. 2008, 146, 387–402. [Google Scholar] [CrossRef]
- Kubes, J.; Skalicky, M.; Tumova, L.; Martin, J.; Hejnak, V.; Martinkova, J. Vanadium elicitation of Trifolium pratense L. cell culture and possible pathways of produced isoflavones transport across the plasma membrane. Plant Cell Rep. 2019, 38, 657–671. [Google Scholar] [CrossRef]
- Reichling, J.; Merkel, B.; Hofmeister, P. Studies on the Biological Activites of Rare Phenylpropanoids of the Genus Pimpinella. J. Nat. Prod. 1991, 54, 1416–1418. [Google Scholar] [CrossRef]
- Delazar, A.; Biglari, F.; Esnaashari, S.; Nazemiyeh, H.; Talebpour, H.; Nahar, L.; Sarker, S.D. GC-MS analysis of the essential oils, and the isolation of phenylpropanoid derivatives from the aerial parts of Pimpinella aurea. Phytochemistry 2006, 67, 2176–2181. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).