Availability of Guanitoxin in Water Samples Containing Sphaerospermopsis torques-reginae Cells Submitted to Dissolution Tests
Abstract
:1. Introduction
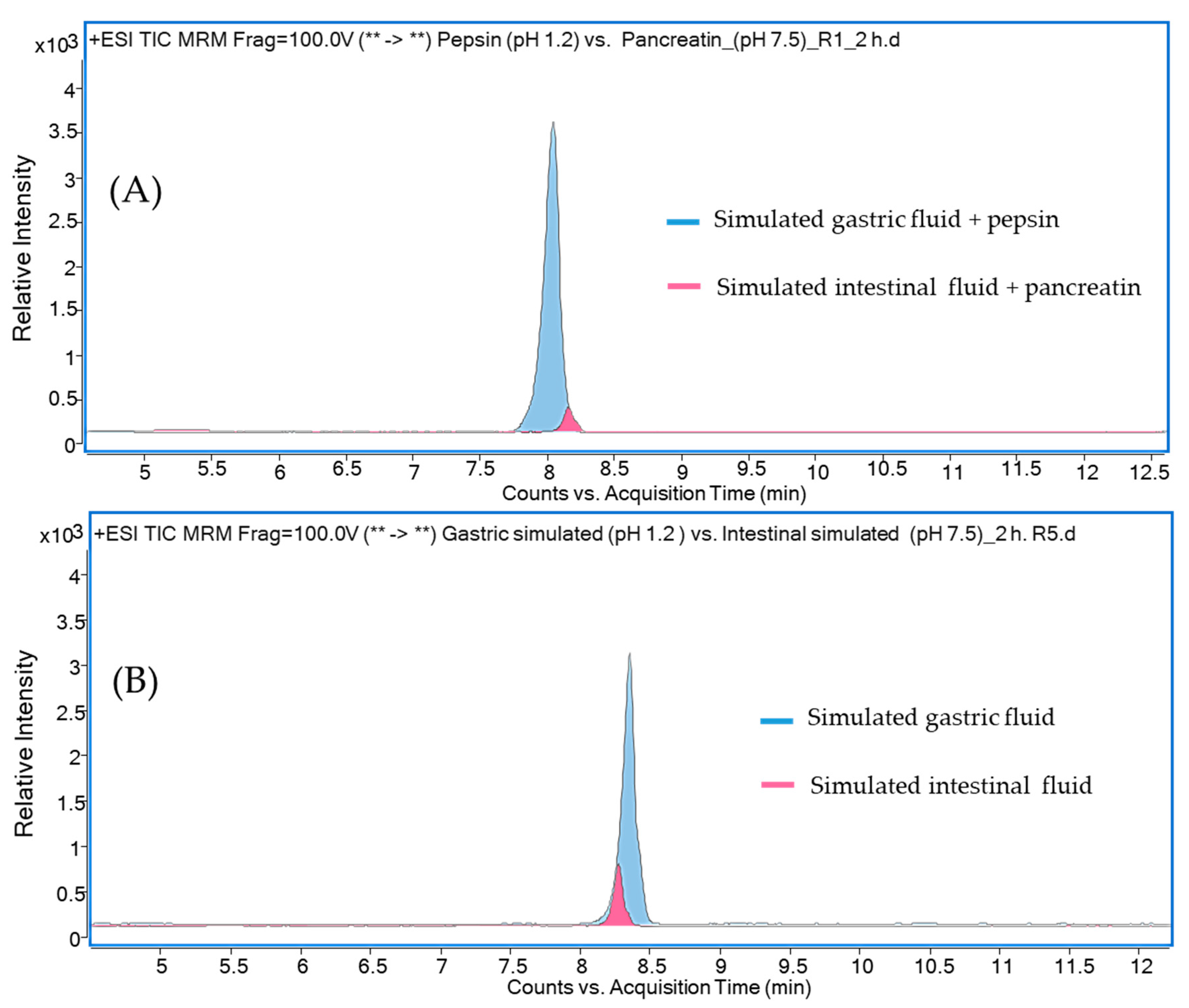
2. Results
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Cultivation of the Sphaerospermopsis torques-reginae (ITEP-24) Strain Producing Guanitoxin
4.3. Preparation of Simulated Gastrointestinal Fluid Solutions with and without Digestive Enzymes
4.4. Dissolution Test with the ITEP-24 Strain
4.5. Liquid Chromatography-Tandem Mass Spectrometry
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fiore, M.F.; de Lima, S.T.; Carmichael, W.W.; McKinnie, S.M.K.; Chekan, J.R.; Moore, B.S. Guanitoxin, re-naming a cyanobacterial organophosphate toxin. Harmful Algae 2020, 92, 101737. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, W.W.; Gorham, P.R. Anatoxins from clones of Anabaena flos-aquae isolated from lakes of western Canada: With 3 figures and 2 tables in the text. Int. Ver. für Theor. und Angew. Limnol. Mitt. 1978, 21, 285–295. [Google Scholar] [CrossRef]
- Matsunaga, S.; Moore, R.E.; Niemczura, W.P.; Carmichael, W.W. Anatoxin-a(s), a Potent Anticholinesterase from Anabaena flos-aquae. J. Am. Chem. Soc. 1989, 111, 8021–8023. [Google Scholar] [CrossRef]
- Carmichael, W.W. The Toxins of Cyanobacteria. Sci. Am. 1994, 270, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Mahmood, N.A.; Carmichael, W.W. The pharmacology of anatoxin-a(s), a neurotoxin produced by the freshwater cyanobacterium Anabaena flos-aquae NRC 525-17. Toxicon 1986, 24, 425–434. [Google Scholar] [CrossRef]
- Mahmood, N.A.; Carmichael, W.W.; Pfahler, D. Anticholinesterase poisonings in dogs from a cyanobacterial (blue-green algae) bloom dominated by Anabaena flos-aquae. Am. J. Vet. Res. 1988, 49, 500–503. [Google Scholar] [PubMed]
- Mahmood, N.A.; Carmichael, W.W. Anatoxin-a(s), an anticholinesterase from the cyanobacterium Anabaena-Flos-Aquae NRC-525-17. Toxicon 1987, 25, 1221–1227. [Google Scholar] [CrossRef]
- Cook, W.O.; Beasley, V.R.; Lovell, R.A.; Dahlem, A.M.; Hooser, S.B.; Mahmood, N.A.; Carmichael, W.W. Consistent inhibition of peripheral cholinesterases by neurotoxins from the freshwater cyanobacterium Anabaena flos-aquae: Studies of ducks, swine, mice and a steer. Environ. Toxicol. Chem. 1989, 8, 915–922. [Google Scholar] [CrossRef]
- Henriksen, P.; Carmichael, W.W.; An, J.; Moestrup, Ø. Detection of an anatoxin-a(s)-like anticholinesterase in natural blooms and cultures of cyanobacteria/blue-green algae from Danish lakes and in the stomach content of poisoned birds. Toxicon 1997, 35, 901–913. [Google Scholar] [CrossRef]
- Cook, W.O.; Dellinger, J.A.; Singh, S.S.; Dahlem, A.M.; Carmichael, W.W.; Beasley, V.R. Regional brain cholinesterase activity in rats injected intraperitoneally with anatoxin-a(s) or paraoxon. Toxicol. Lett. 1989, 49, 29–34. [Google Scholar] [CrossRef]
- Monserrat, J.M.; Yunes, J.S.; Bianchini, A. Effects of Anabaena spiroides (cyanobacteria) aqueous extracts on the acetylcholinesterase activity of aquatic species. Environ. Toxicol. Chem. 2001, 20, 1228–1235. [Google Scholar] [CrossRef]
- Freitas, E.C.; Printes, L.B.; Rocha, O. Acute effects of Anabaena spiroides extract and paraoxon-methyl on freshwater cladocerans from tropical and temperate regions: Links between the ChE activity and survival and its implications for tropical ecotoxicological studies. Aquat. Toxicol. 2014, 146, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Santos, D.S.; Rosa, M.E.; Zanatta, A.P.; Oliveira, R.S.; de Almeida, C.G.M.; Leal, A.P.; Sanz, M.; Fernandes, K.A.; de Souza, V.Q.; de Assis, D.R. Neurotoxic effects of sublethal concentrations of cyanobacterial extract containing anatoxin-a(s) on Nauphoeta cinerea cockroaches. Ecotoxicol. Environ. Saf. 2019, 171, 138–145. [Google Scholar] [CrossRef] [PubMed]
- Freitas, E.C.; Printes, L.B.; Rocha, O. Use of cholinesterase activity as an ecotoxicological marker to assess anatoxin-a(s) exposure: Responses of two cladoceran species belonging to contrasting geographical regions. Harmful Algae 2016, 55, 150–162. [Google Scholar] [CrossRef] [PubMed]
- Cook, W.O.; Iwamoto, G.A.; Schaeffer, D.J.; Beasley, V.R. Effect of anatoxin-a(s) from Anabaena flos-aquae NRC-525-17 on blood pressure, heart rate, respiratory rate, tidal volume, minute volume, and phrenic nerve activity in rats. J. Environ. Pathol. Toxicol. Oncol. Off. Organ Int. Soc. Environ. Toxicol. Cancer 1989, 9, 393–400. [Google Scholar]
- Werner, V.R.; Laughinghouse IV, H.D.; Fiore, M.F.; Sant’Anna, C.L.; Hoff, C.; de Souza Santos, K.R.; Neuhaus, E.B.; Molica, R.J.R.; Honda, R.Y.; Echenique, R.O. Morphological and molecular studies of Sphaerospermopsis torques-reginae (Cyanobacteria, Nostocales) from South American water blooms. Phycologia 2012, 51, 228–238. [Google Scholar] [CrossRef]
- Devic, E.; Li, D.; Dauta, A.; Henriksen, P.; Codd, G.A.; Marty, J.; Fournier, D. Detection of Anatoxin-a(s) in Environmental Samples of Cyanobacteria by Using a Biosensor with Engineered Acetylcholinesterases. Appl. Environ. Microbiol. 2002, 68, 4102–4106. [Google Scholar] [CrossRef] [Green Version]
- Villatte, F.; Schulze, H.; Schmid, R.; Bachmann, T. A disposable acetylcholinesterase-based electrode biosensor to detect anatoxin-a(s) in water. Anal. Bioanal. Chem. 2002, 372, 322–326. [Google Scholar] [CrossRef]
- Dörr, F.A.; Rodríguez, V.; Molica, R.; Henriksen, P.; Krock, B.; Pinto, E. Methods for detection of anatoxin-a(s) by liquid chromatography coupled to electrospray ionization-tandem mass spectrometry. Toxicon 2010, 55, 92–99. [Google Scholar] [CrossRef]
- Svirčev, Z.; Lalić, D.; Savić, G.B.; Tokodi, N.; Backović, D.D.; Chen, L.; Meriluoto, J.; Codd, G.A. Global geographical and historical overview of cyanotoxin distribution and cyanobacterial poisonings. Arch. Toxicol. 2019, 93, 2429–2481. [Google Scholar] [CrossRef]
- Metcalf, J.S.; Richer, R.; Cox, P.A.; Codd, G.A. Cyanotoxins in desert environments may present a risk to human health. Sci. Total Environ. 2012, 421–422, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Chatziefthimiou, A.D.; Richer, R.; Rowles, H.; Powell, J.T.; Metcalf, J.S. Cyanotoxins as a potential cause of dog poisonings in desert environments. Vet. Rec. 2014, 174, 484–485. [Google Scholar] [CrossRef] [PubMed]
- Ibelings, B.W.; Backer, L.C.; Kardinaal, W.E.A.; Chorus, I. Current approaches to cyanotoxin risk assessment and risk management around the globe. Harmful Algae 2014, 40, 63–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bartram, J.; Baum, R.; Coclanis, P.A.; Gute, D.M.; Kay, D.; McFadyen, S.; Pond, K.; Robertson, W.; Rouse, M.J. Routledge Handbook of Water and Health; Routledge: London, UK; New York, NY, USA, 2015; ISBN 1138910074. [Google Scholar]
- Barros, L.P.C.; Monserrat, J.M.; Yunes, J.S. Determination of optimized protocols for the extraction of anticholinesterasic compounds in environmental samples containing cyanobacteria species. Environ. Toxicol. Chem. 2004, 23, 883–889. [Google Scholar] [CrossRef] [PubMed]
- Molica, R.J.R.; Oliveira, E.J.A.; Carvalho, P.V.V.C.; Costa, A.N.S.F.; Cunha, M.C.C.; Melo, G.L.; Azevedo, S.M.F.O. Occurrence of saxitoxins and an anatoxin-a(s) -like anticholinesterase in a Brazilian drinking water supply. Harmful Algae 2005, 4, 743–753. [Google Scholar] [CrossRef]
- Metcalf, J.S.; Banack, S.A.; Powell, J.T.; Tymm, F.J.M.; Murch, S.J.; Brand, L.E.; Cox, P.A. Public health responses to toxic cyanobacterial blooms: Perspectives from the 2016 Florida event. Water Policy 2018, 20, 919–932. [Google Scholar] [CrossRef] [Green Version]
- Rockville, M.D. United Pharmacopoeia/National Formulary; USP 23/NF 18; United States Pharmacopoeil Convention, Inc.: Rockville, MD, USA, 1995; p. 1235. [Google Scholar]
- Anand, O.; Yu, L.X.; Conner, D.P.; Davit, B.M. Dissolution Testing for Generic Drugs: An FDA Perspective. AAPS J. 2011, 13, 328. [Google Scholar] [CrossRef] [Green Version]
- Qureshi, S.A.; Shabnam, J. Cause of high variability in drug dissolution testing and its impact on setting tolerances. Eur. J. Pharm. Sci. 2001, 12, 271–276. [Google Scholar] [CrossRef]
- Tsong, Y.; Shen, M.; Shah, V.P. Three-stage sequential statistical dissolution testing rules. J. Biopharm. Stat. 2004, 14, 757–779. [Google Scholar] [CrossRef]
- Kasim, N.A.; Whitehouse, M.; Ramachandran, C.; Bermejo, M.; Lennernäs, H.; Hussain, A.S.; Junginger, H.E.; Stavchansky, S.A.; Midha, K.K.; Shah, V.P. Molecular properties of WHO essential drugs and provisional biopharmaceutical classification. Mol. Pharm. 2004, 1, 85–96. [Google Scholar] [CrossRef]
- Klein, S. The Use of Biorelevant Dissolution Media to Forecast the In Vivo Performance of a Drug. AAPS J. 2010, 12, 397–406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodrigues, M.S.; Ferreira, L.S.; Converti, A.; Sato, S.; Carvalho, J.C.M. Fed-batch cultivation of Arthrospira (Spirulina) platensis: Potassium nitrate and ammonium chloride as simultaneous nitrogen sources. Bioresour. Technol. 2010, 101, 4491–4498. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.K.; Tiwari, S.P.; Rai, A.K.; Mohapatra, T.M. Cyanobacteria: An emerging source for drug discovery. J. Antibiot. (Tokyo) 2011, 64, 401–412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nowruzi, B.; Haghighat, S.; Fahimi, H.; Mohammadi, E. Nostoc cyanobacteria species: A new and rich source of novel bioactive compounds with pharmaceutical potential. J. Pharm. Heal. Serv. Res. 2018, 9, 5–12. [Google Scholar] [CrossRef]
- Marques, M.R.C. Enzymes in the dissolution testing of gelatin capsules. AAPS Pharmscitech 2014, 15, 1410–1416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stanier, R.Y.; Cohen-Bazire, G. Phototrophic prokaryotes: The cyanobacteria. Annu. Rev. Microbiol. 1977, 31, 225–274. [Google Scholar] [CrossRef]
- Palinska, K.A.; Krumbein, W.E. Perforation patterns in the peptidoglycan wall of filamentous cyanobacteria. J. Phycol. 2000, 36, 139–145. [Google Scholar] [CrossRef]
- Safi, C.; Ursu, A.V.; Laroche, C.; Zebib, B.; Merah, O.; Pontalier, P.-Y.; Vaca-Garcia, C. Aqueous extraction of proteins from microalgae: Effect of different cell disruption methods. Algal Res. 2014, 3, 61–65. [Google Scholar] [CrossRef] [Green Version]
- Murray, R.K.; Granner, D.K.; Mayes, P.A.; Rodwell, V.W. Illustrated Biochemistry; McGraw-Hill: New York, NY, USA, 2003. [Google Scholar]
- Murray, K.; Rodwell, V.; Bender, D.; Botham, K.M.; Weil, P.A.; Kennelly, P.J. Harper’s Illustrated Biochemistry, 28th ed.; McGraw-Hill: New York, NY, USA, 2009. [Google Scholar]
- Christoffersen, K. Ecological implications of cyanobacterial toxins in aquatic food webs. Phycologia 1996, 35, 42–50. [Google Scholar] [CrossRef]
- Sivonen, K.; Jones, G. Toxic Cyanobacteria in Water: A Guide to Their Public Health Consequences, Monitoring and Management; WHO: Geneva, Switzerland, 1999. [Google Scholar]
- Jaiswal, P.; Singh, P.K.; Prasanna, R. Cyanobacterial bioactive molecules—An overview of their toxic properties. Can. J. Microbiol. 2008, 54, 701–717. [Google Scholar] [CrossRef]
- Fernandes, K.; Gomes, A.; Calado, L.; Yasui, G.; Assis, D.; Henry, T.; Fonseca, A.; Pinto, E. Toxicity of Cyanopeptides from Two Microcystis Strains on Larval Development of Astyanax altiparanae. Toxins 2019, 11, 220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Azevedo, S.M.F.O.; Carmichael, W.W.; Jochimsen, E.M.; Rinehart, K.L.; Lau, S.; Shaw, G.R.; Eaglesham, G.K. Human intoxication by microcystins during renal dialysis treatment in Caruaru—Brazil. Toxicology 2002, 181, 441–446. [Google Scholar] [CrossRef]
- Rodríguez, V.; Mori, B.; Dörr, F.A.; Dal Belo, C.A.; Colepicolo, P.; Pinto, E. Effects of a cyanobacterial extract containing-anatoxin-a(s) on the cardiac rhythm of Leurolestes circunvagans. Rev. Bras. Farmacogn. 2012, 22, 775–781. [Google Scholar] [CrossRef] [Green Version]
- de Abreu, F.Q.; da Ferrão-Filho, A.S. Effects of an Anatoxin-a(s)-Producing Strain of Anabaena spiroides (Cyanobacteria) on the Survivorship and Somatic Growth of Two Daphnia similis Clones. J. Environ. Prot. (Irvine, CA) 2013, 4, 12–18. [Google Scholar] [CrossRef] [Green Version]
- Onodera, H.; Oshima, Y.; Henriksen, P.; Yasumoto, T. Confirmation of anatoxin-a(s), in the cyanobacterium Anabaena lemmermannii, as the cause of bird kills in Danish lakes. Toxicon 1997, 35, 1645–1648. [Google Scholar] [CrossRef]
- Gorham, P.R.; McLachlan, J.; Hammer, U.T.; Kim, W. Isolation and culture of toxic strains of Anabaena flos-aquae (Lyngb.) Breb. Verh. Int. Vereinigung fur Theor. und Angew. Limnol. 1964, 15, 1964. [Google Scholar] [CrossRef]
- Blakefield, M.K.; Harris, D.O. Delay of cell differentiation in Anabaena aequalis caused by UV-B radiation and the role of photoreactivation and excision repair. Photochem. Photobiol. 1994, 59, 204–208. [Google Scholar] [CrossRef]
- Moreno, I.M.; Maraver, J.; Aguete, E.C.; Leao, M.; Gago-Martínez, A.; Cameán, A.M. Decomposition of microcystin-LR, microcystin-RR, and microcystin-YR in water samples submitted to in vitro dissolution tests. J. Agric. Food Chem. 2004, 52, 5933–5938. [Google Scholar] [CrossRef]
- Team, R.C. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2016. [Google Scholar]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernandes, K.A.; Ferraz, H.G.; Vereau, F.; Pinto, E. Availability of Guanitoxin in Water Samples Containing Sphaerospermopsis torques-reginae Cells Submitted to Dissolution Tests. Pharmaceuticals 2020, 13, 402. https://doi.org/10.3390/ph13110402
Fernandes KA, Ferraz HG, Vereau F, Pinto E. Availability of Guanitoxin in Water Samples Containing Sphaerospermopsis torques-reginae Cells Submitted to Dissolution Tests. Pharmaceuticals. 2020; 13(11):402. https://doi.org/10.3390/ph13110402
Chicago/Turabian StyleFernandes, Kelly Afonsina, Humberto Gomes Ferraz, Fanny Vereau, and Ernani Pinto. 2020. "Availability of Guanitoxin in Water Samples Containing Sphaerospermopsis torques-reginae Cells Submitted to Dissolution Tests" Pharmaceuticals 13, no. 11: 402. https://doi.org/10.3390/ph13110402
APA StyleFernandes, K. A., Ferraz, H. G., Vereau, F., & Pinto, E. (2020). Availability of Guanitoxin in Water Samples Containing Sphaerospermopsis torques-reginae Cells Submitted to Dissolution Tests. Pharmaceuticals, 13(11), 402. https://doi.org/10.3390/ph13110402