Enhanced Heparin Adsorption from Porcine Mucosa Using Beta Zeolites: Optimization and Kinetic Analysis
Abstract
1. Introduction
2. Materials and Methods
Experimental
3. Results and Discussion
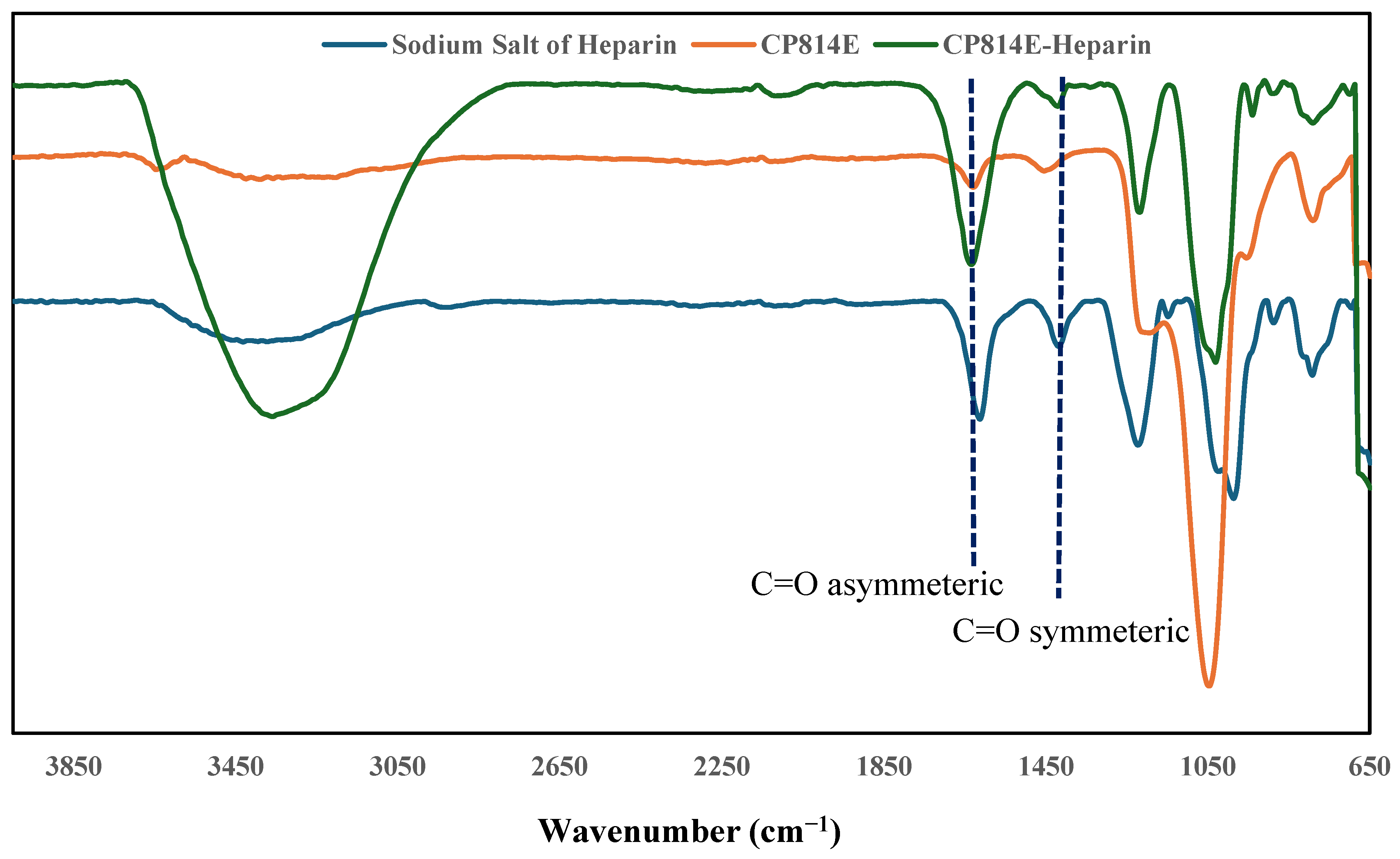
3.1. Adsorption Studies of Heparin
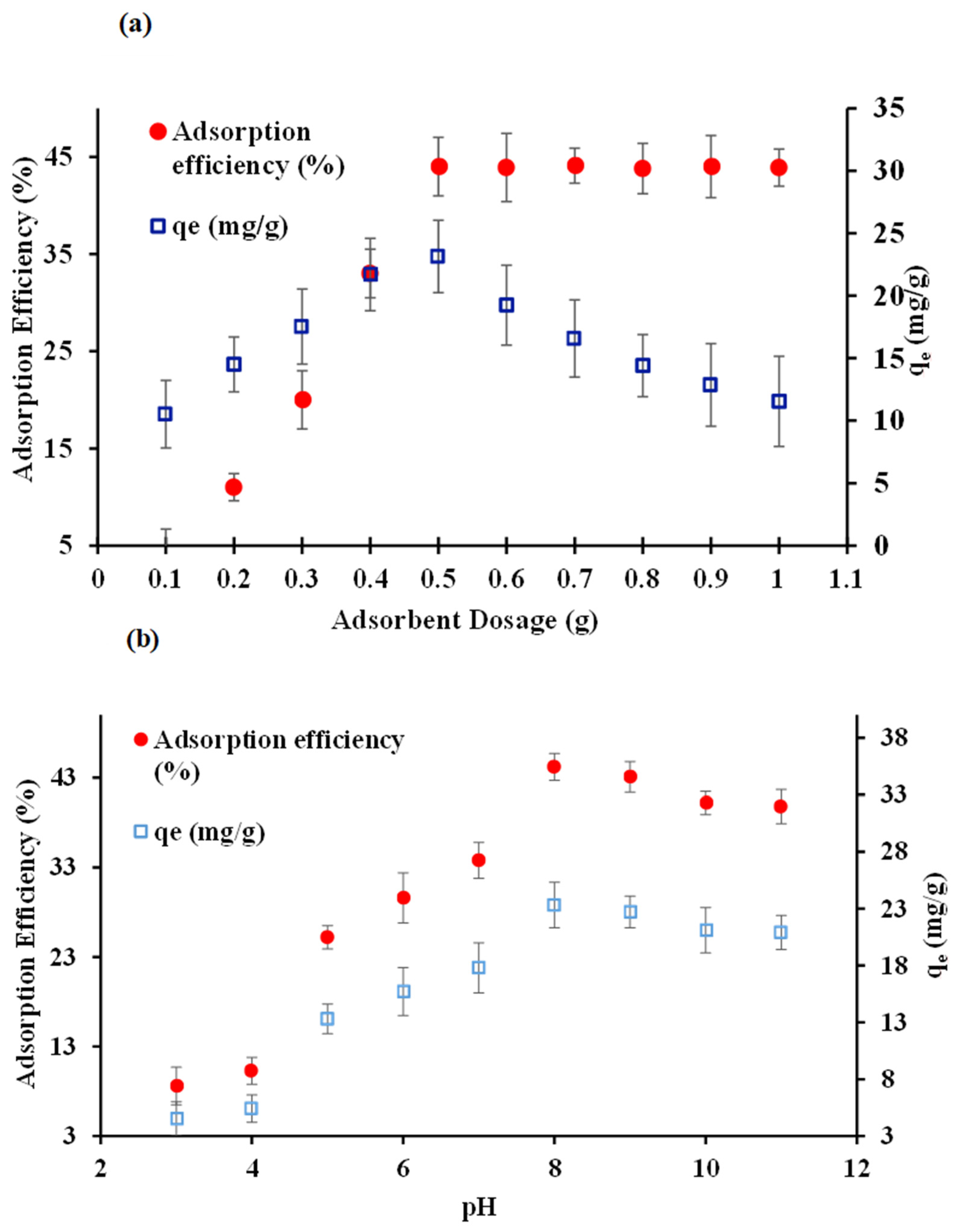
3.1.1. Adsorption Dosage Optimization
3.1.2. pH Optimization
3.1.3. Effect of Temperature and Duration on Heparin Uptake in Real Sample
3.1.4. Adsorption Kinetics
3.1.5. Thermodynamics of Heparin Adsorption
- -
- Shift from monolayer to multilayer adsorption;
- -
- Changes in adsorbent porosity at elevated temperatures;
- -
- Altered solubility of the adsorbate at higher temperatures.
- -
- Decreasing heparin concentration in the medium;
- -
- Reduced molecular collisions;
- -
- Established adsorption equilibrium.
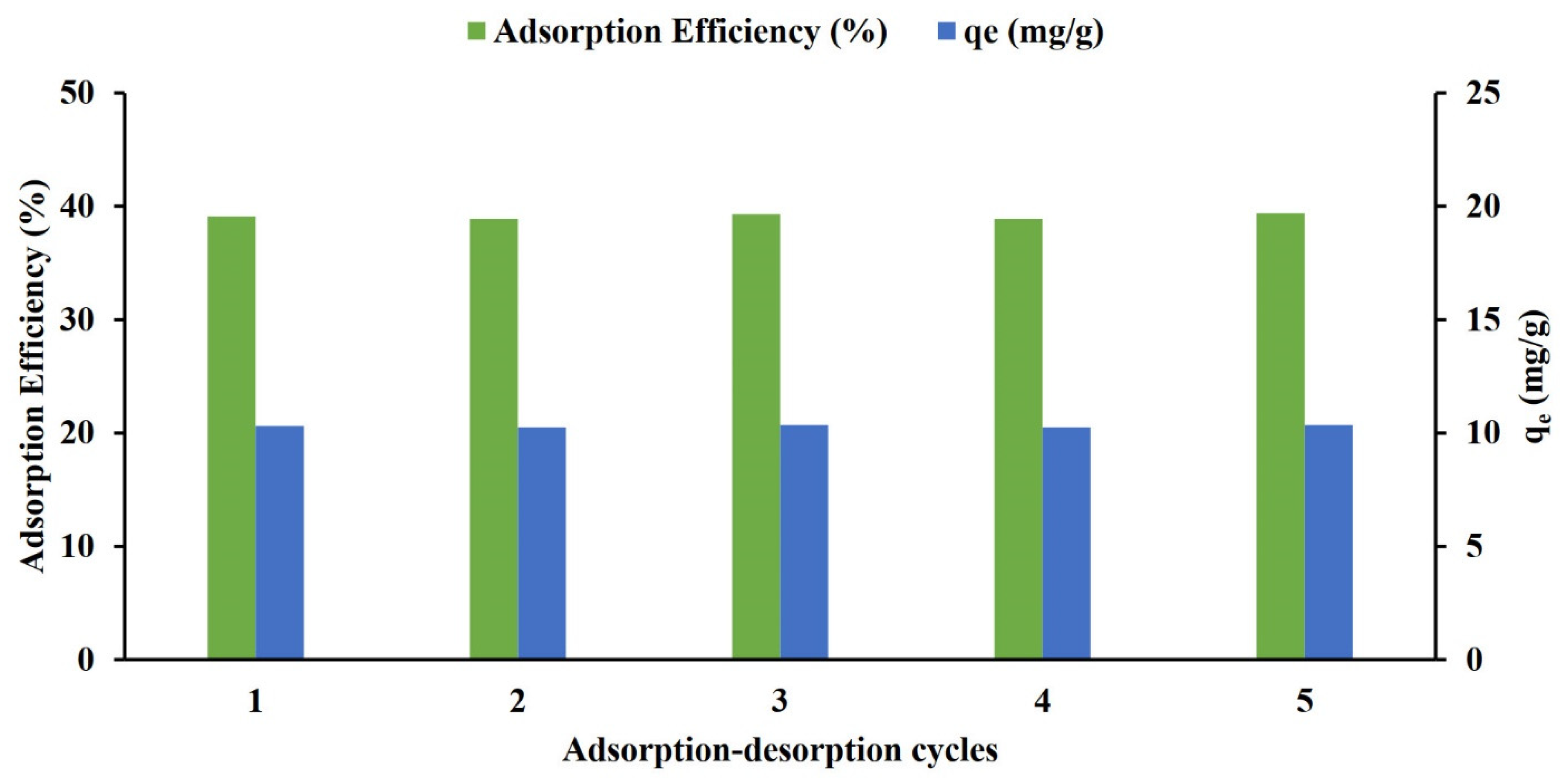
3.1.6. Sorbet Reusability
3.1.7. Molecular Weight of the Extracted Heparin
3.2. Sheep Plasma Clotting Assay
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Reid, K.R.; Tabibi, A.; Adami, P.; Karimi, B. Optimized heparin adsorption using macroporous zeolites: A comprehensive study. Heliyon 2025, 11, e41398. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.S.; Kong, J. Heparin: Physiology, Pharmacology, and Clinical Application. RCM 2015, 16, 189–199. [Google Scholar] [CrossRef] [PubMed]
- Oduah, E.I.; Linhardt, R.J.; Sharfstein, S.T. Heparin: Past, Present, and Future. Pharmaceuticals 2016, 9, 38. [Google Scholar] [CrossRef]
- Atallah, J.; Khachfe, H.H.; Berro, J.; Assi, H.I. The Use of Heparin and Heparin-like Molecules in Cancer Treatment: A Review. Cancer Treat. Res. Commun. 2020, 24, 100192. [Google Scholar] [CrossRef] [PubMed]
- Scott, L.J.; Perry, C.M. Tramadol: A review of its use in perioperative pain. Drugs 2000, 60, 139–176. [Google Scholar] [CrossRef]
- Urbinati, C.; Milanesi, M.; Lauro, N.; Bertelli, C.; David, G.; D’Ursi, P.; Rusnati, M.; Chiodelli, P. HIV-1 tat and heparan sulfate proteoglycans orchestrate the setup of in cis and in trans cell-surface interactions functional to lymphocyte trans-endothelial migration. Molecules 2021, 26, 7488. [Google Scholar] [CrossRef]
- Barrowcliffe, T. History of heparin. Heparin-A Century Prog. 2012, 207, 3–22. [Google Scholar]
- Anderson, J.; Saenko, E. Editorial I: Heparin Resistance; Oxford University Press: Oxford, UK, 2002; Volume 88, pp. 467–469. [Google Scholar]
- Flengsrud, R.; Larsen, M.L.; Ødegaard, O.R. Purification, characterization and in vivo studies of salmon heparin. Thromb. Res. 2010, 126, e409–e417. [Google Scholar] [CrossRef]
- Vreeburg, J.; Baauw, A. Method for Preparation of Heparin from Mucosa. Patent No. WO2010/110654 A, 26 February 2010. [Google Scholar]
- Karimi Abdolmaleki, M.; Das, A.; Khambhati, D.P.; Shafiee, A.; Dimas, K.; Velazquez, C.A.; Davachi, S.M.; Choubtarash Abardeh, S. Efficient and economic heparin recovery from porcine intestinal mucosa using quaternary ammonium-functionalized silica gel. Bioengineering 2022, 9, 606. [Google Scholar] [CrossRef]
- Das, A.; Khambhati, D.P.; Longoria, N.D.; Tabibi, A.; Davachi, S.M.; Dimas, K.; Laurencin, Y.; Carmona, L.; Avalos, P.Z.; Karimi Abdolmaleki, M. Modified diatomaceous earth in heparin recovery from porcine intestinal mucosa. Molecules 2023, 28, 7982. [Google Scholar] [CrossRef]
- Karimi Abdolmaleki, M.; Ganta, D.; Shafiee, A.; Velazquez, C.A.; Khambhati, D.P. Efficient heparin recovery from porcine intestinal mucosa using zeolite imidazolate framework-8. Molecules 2022, 27, 1670. [Google Scholar] [CrossRef] [PubMed]
- Van der Meer, J.-Y.; Kellenbach, E.; Van den Bos, L.J. From farm to pharma: An overview of industrial heparin manufacturing methods. Molecules 2017, 22, 1025. [Google Scholar] [CrossRef]
- Hoke, D.E.; Carson, D.D.; Höök, M. A heparin binding synthetic peptide from human HIP/RPL29 fails to specifically differentiate between anticoagulantly active and inactive species of heparin. J. Negat. Results Biomed. 2003, 2, 1–10. [Google Scholar] [CrossRef]
- Linhardt, R.J.; Ampofo, S.A.; Fareed, J.; Hoppensteadt, D.; Folkman, J.; Mulliken, J.B. Isolation and characterization of human heparin. Biochem. J. 1992, 31, 12441–12445. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wang, Q.; Zhang, A.; Shao, X.; Liu, T.; Tang, B.; Fang, G. Strategies for sustained release of heparin: A review. Carbohydr. Polym. 2022, 294, 119793. [Google Scholar] [CrossRef]
- Ryu, U.; Jee, S.; Rao, P.C.; Shin, J.; Ko, C.; Yoon, M.; Park, K.S.; Choi, K.M. Recent advances in process engineering and upcoming applications of metal–organic frameworks. Coord. Chem. Rev. 2021, 426, 213544. [Google Scholar] [CrossRef]
- Haley, R.A.; Ringo, J.M.; Hopgood, H.; Denlinger, K.L.; Das, A.; Waddell, D.C. Graduate student designed and delivered: An upper-level online course for undergraduates in green chemistry and sustainability. J. Chem. Educ. 2018, 95, 560–569. [Google Scholar] [CrossRef]
- Eskandarloo, H.; Godec, M.; Arshadi, M.; Padilla-Zakour, O.I.; Abbaspourrad, A. Multi-porous quaternized chitosan/polystyrene microbeads for scalable, efficient heparin recovery. J. Chem. Eng. 2018, 348, 399–408. [Google Scholar] [CrossRef]
- Gu, H.; Liu, Y.; Wang, L.; Zhang, B.; Yin, D.; Zhang, Q. Monolithic macroporous hydrogels prepared from oil-in-water high internal phase emulsions for high-efficiency purification of Enterovirus 71. J. Chem. Eng. 2020, 401, 126051. [Google Scholar] [CrossRef]
- Welzel, P.B.; Grimmer, M.; Renneberg, C.; Naujox, L.; Zschoche, S.; Freudenberg, U.; Werner, C. Macroporous starPEG-heparin cryogels. Biomacromolecules 2012, 13, 2349–2358. [Google Scholar] [CrossRef] [PubMed]
- Aw, M.S.; Simovic, S.; Yu, Y.; Addai-Mensah, J.; Losic, D. Porous silica microshells from diatoms as biocarrier for drug delivery applications. Powder Technol. 2012, 223, 52–58. [Google Scholar] [CrossRef]
- Serati-Nouri, H.; Jafari, A.; Roshangar, L.; Dadashpour, M.; Pilehvar-Soltanahmadi, Y.; Zarghami, N. Biomedical applications of zeolite-based materials: A review. Mater. Sci. Eng. C. 2020, 116, 111225. [Google Scholar] [CrossRef]
- Bacakova, L.; Vandrovcova, M.; Kopova, I.; Jirka, I. Applications of zeolites in biotechnology and medicine–a review. Biomater. Sci. 2018, 6, 974–989. [Google Scholar] [CrossRef]
- Rubio-Martinez, M.; Avci-Camur, C.; Thornton, A.W.; Imaz, I.; Maspoch, D.; Hill, M.R. New synthetic routes towards MOF production at scale. Chem. Soc. Rev. 2017, 46, 3453–3480. [Google Scholar] [CrossRef] [PubMed]
- Boer, D.G.; Langerak, J.; Pescarmona, P.P. Zeolites as selective adsorbents for CO2 separation. ACS Appl. Energy Mater. 2023, 6, 2634–2656. [Google Scholar] [CrossRef]
- Efe, C.; van der Wielen, L.A.; Straathof, A.J. High silica zeolites as an alternative to weak base adsorbents in succinic acid recovery. Ind. Eng. Chem. Res. 2010, 49, 1837–1843. [Google Scholar] [CrossRef]
- Wang, L.; Ozawa, K.; Komatsu, T.; Ikeda, T. Ca2+-exchanged ferrierite: Quasi one-dimensional zeolite for highly selective and stable formation of light alkenes in catalytic cracking of n-octane. Appl. Catal. A Gen. 2011, 407, 127–133. [Google Scholar] [CrossRef]
- Kirdeciler, S.K.; Soy, E.; Öztürk, S.; Kucherenko, I.; Soldatkin, O.; Dzyadevych, S.; Akata, B. A novel urea conductometric biosensor based on zeolite immobilized urease. Talanta 2011, 85, 1435–1441. [Google Scholar] [CrossRef]
- Ninan, N.; Muthiah, M.; Park, I.-K.; Elain, A.; Wong, T.W.; Thomas, S.; Grohens, Y. Faujasites incorporated tissue engineering scaffolds for wound healing: In vitro and in vivo analysis. ACS Appl. Mater. Interfaces 2013, 5, 11194–11206. [Google Scholar] [CrossRef]
- Moshoeshoe, M.; Nadiye-Tabbiruka, M.S.; Obuseng, V. A review of the chemistry, structure, properties and applications of zeolites. Am. J. Mater. Sci. 2017, 7, 196–221. [Google Scholar]
- Matsui, M.; Kiyozumi, Y.; Mizushina, Y.; Sakaguchi, K.; Mizukami, F. Adsorption and desorption behavior of basic proteins on zeolites. Sep. Purif. Technol. 2015, 149, 103–109. [Google Scholar] [CrossRef]
- Halasz, I.; Kim, S.; Marcus, B. Uncommon adsorption isotherm of methanol on a hydrophobic Y-zeolite. J. Phys. Chem. B 2001, 105, 10788–10796. [Google Scholar] [CrossRef]
- Zhang, X.; Sun, J.; Liu, J.; Xu, H.; Dong, B.; Sun, X.; Zhang, T.; Xu, S.; Xu, L.; Bai, X. Label-free electrochemical immunosensor based on conductive Ag contained EMT-style nano-zeolites and the application for α-fetoprotein detection. Sens. Actuators B Chem. 2018, 255, 2919–2926. [Google Scholar] [CrossRef]
- Vasconcelos, A.A.; Len, T.; de Oliveira, A.d.N.; Costa, A.A.F.d.; Souza, A.R.d.S.; Costa, C.E.F.d.; Luque, R.; Rocha Filho, G.N.d.; Noronha, R.C.R.; Nascimento, L.A.S.d. Zeolites: A theoretical and practical approach with uses in (bio) chemical processes. Appl. Sci. 2023, 13, 1897. [Google Scholar] [CrossRef]
- Souza, I.M.; García-Villén, F.; Viseras, C.; Pergher, S.B. Zeolites as ingredients of medicinal products. Pharmaceutics 2023, 15, 1352. [Google Scholar] [CrossRef] [PubMed]
- Nsengiyumva, E.M.; Heitz, M.P.; Alexandridis, P. Carboxymethyl hydroxypropyl guar gum physicochemical properties in dilute aqueous media. Int. J. Biol. Macromol. 2024, 262, 129775. [Google Scholar] [CrossRef] [PubMed]
- Rauschkolb, J.C.; Ribeiro, B.C.; Feiden, T.; Fischer, B.; Weschenfelder, T.A.; Cansian, R.L.; Junges, A. Parameter Estimation of Mark-Houwink Equation of Polyethylene Glycol (PEG) Using Molecular Mass and Intrinsic Viscosity in Water. Biointerface Res. Appl. Chem. 2021, 12, 1778–1790. [Google Scholar] [CrossRef]
- Rahmani, S.; Zeynizadeh, B.; Karami, S. Removal of cationic methylene blue dye using magnetic and anionic-cationic modified montmorillonite: Kinetic, isotherm and thermodynamic studies. Appl. Clay Sci. 2020, 184, 105391. [Google Scholar] [CrossRef]
- Jia, S.-X.; Chi, Q.-N.; Zhang, Y.; Liu, T.; Kou, X.; Wang, F.; Qi, Y.-K.; Du, S.-S.; Xing, X.-H. Binding ability of methylene blue with heparin dependent on its sulfate level rather than its sulfation location or basic saccharide structure. Glycoconj. J. 2021, 38, 551–560. [Google Scholar] [CrossRef]
- Bergua, J.F.; Alvarez-Diduk, R.; Idili, A.; Parolo, C.; Maymó, M.; Hu, L.; Merkoçi, A. Low-cost, user-friendly, all-integrated smartphone-based microplate reader for optical-based biological and chemical analyses. Anal. Chem. 2022, 94, 1271–1285. [Google Scholar] [CrossRef]
- Jiang, N.; Shang, R.; Heijman, S.G.; Rietveld, L.C. Adsorption of triclosan, trichlorophenol and phenol by high-silica zeolites: Adsorption efficiencies and mechanisms. Sep. Purif. Technol. 2020, 235, 116152. [Google Scholar] [CrossRef]
- Kim, K.-J.; Ahn, H.-G. The effect of pore structure of zeolite on the adsorption of VOCs and their desorption properties by microwave heating. Micropor. Mesopor. Mater. 2012, 152, 78–83. [Google Scholar] [CrossRef]
- Dinda, A.K.; Tripathy, D.R.; Das, A.; Dasgupta, S. Comparison of the ribonucleolytic activity of the dityrosine cross-linked Ribonuclease A dimer with its monomer in the presence of inhibitors. Int. J. Biol. Macromol. 2014, 63, 107–113. [Google Scholar] [CrossRef]
- Grant, D.; Long, W.; Moffat, C.F.; Williamson, F.B. Infrared spectroscopy of chemically modified heparins. Biochem. J. 1989, 261, 1035–1038. [Google Scholar] [CrossRef]
- Das, A.; Lao, E.A.; Gudmundsdottir, A.D. Photoenolization of o-Methylvalerophenone Ester Derivative. Photochem. Photobiol. 2016, 92, 388–398. [Google Scholar] [CrossRef]
- Pandey, A.; Dalal, S.; Dutta, S.; Dixit, A. Structural characterization of polycrystalline thin films by X-ray diffraction techniques. J. Mater. Sci. Mater. Electron. 2021, 32, 1341–1368. [Google Scholar] [CrossRef]
- Mongkolsuttirat, K.; Buajarern, J. Uncertainty evaluation of crystallite size measurements of nanoparticle using X-Ray Diffraction analysis (XRD). J. Phys. Conf. Ser. 2021, 1719, 012054. [Google Scholar] [CrossRef]
- Li, J.; Gao, M.; Yan, W.; Yu, J. Regulation of the Si/Al ratios and Al distributions of zeolites and their impact on properties. Chem. Sci. 2023, 14, 1935–1959. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Meng, B.; Wang, X.; Liu, Y.; Zeng, J.; Weng, G.; Xu, J.; Xiang, J.; Song, Y.; Jin, L. Quaternized Fe3O4@ chitosan nanoparticles for efficient and selective isolation of heparin. Int. J. Biol. Macromol. 2025, 293, 139368. [Google Scholar] [CrossRef]
- Aumeier, B.M.; Augustin, A.; Thönes, M.; Sablotny, J.; Wintgens, T.; Wessling, M. Linking the effect of temperature on adsorption from aqueous solution with solute dissociation. J. Hazard. Mater. 2022, 429, 128291. [Google Scholar] [CrossRef]
- Wang, J.; Guo, X. Adsorption kinetics and isotherm models of heavy metals by various adsorbents: An overview. Crit. Rev. Environ. Sci. Technol. 2023, 53, 1837–1865. [Google Scholar] [CrossRef]
- Ho, Y.-S.; McKay, G. Pseudo-second order model for sorption processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Simonin, J.-P. On the comparison of pseudo-first order and pseudo-second order rate laws in the modeling of adsorption kinetics. Chem. Eng. J. 2016, 300, 254–263. [Google Scholar] [CrossRef]
- Das, A.; Thomas, S.S.; Garofoli, A.A.; Chavez, K.A.; Krause, J.A.; Bohne, C.; Gudmundsdottir, A.D. Steric Demand and Rate-determining Step for Photoenolization of Di-ortho-substituted Acetophenone Derivatives. Photochem. Photobiol. 2019, 95, 154–162. [Google Scholar] [CrossRef]
- Azizi, S.; Mahdavi Shahri, M.; Mohamad, R. Green synthesis of zinc oxide nanoparticles for enhanced adsorption of lead ions from aqueous solutions: Equilibrium, kinetic and thermodynamic studies. Molecules 2017, 22, 831. [Google Scholar] [CrossRef]
- Sarkar, S.K.; Gatlin, D.M.; Das, A.; Loftin, B.; Krause, J.A.; Abe, M.; Gudmundsdottir, A.D. Laser flash photolysis of nanocrystalline α-azido-p-methoxy-acetophenone. Org. Biomol. Chem. 2017, 15, 7380–7386. [Google Scholar] [CrossRef] [PubMed]
- Wu, F.-C.; Tseng, R.-L.; Juang, R.-S. Initial behavior of intraparticle diffusion model used in the description of adsorption kinetics. J. Chem. Eng. 2009, 153, 1–8. [Google Scholar] [CrossRef]
- Musah, M.; Azeh, Y.; Mathew, J.T.; Umar, M.T.; Abdulhamid, Z.; Muhammad, A.I. Adsorption kinetics and isotherm models: A review. Caliphate J. Sci. Technol. 2022, 4, 20–26. [Google Scholar] [CrossRef]


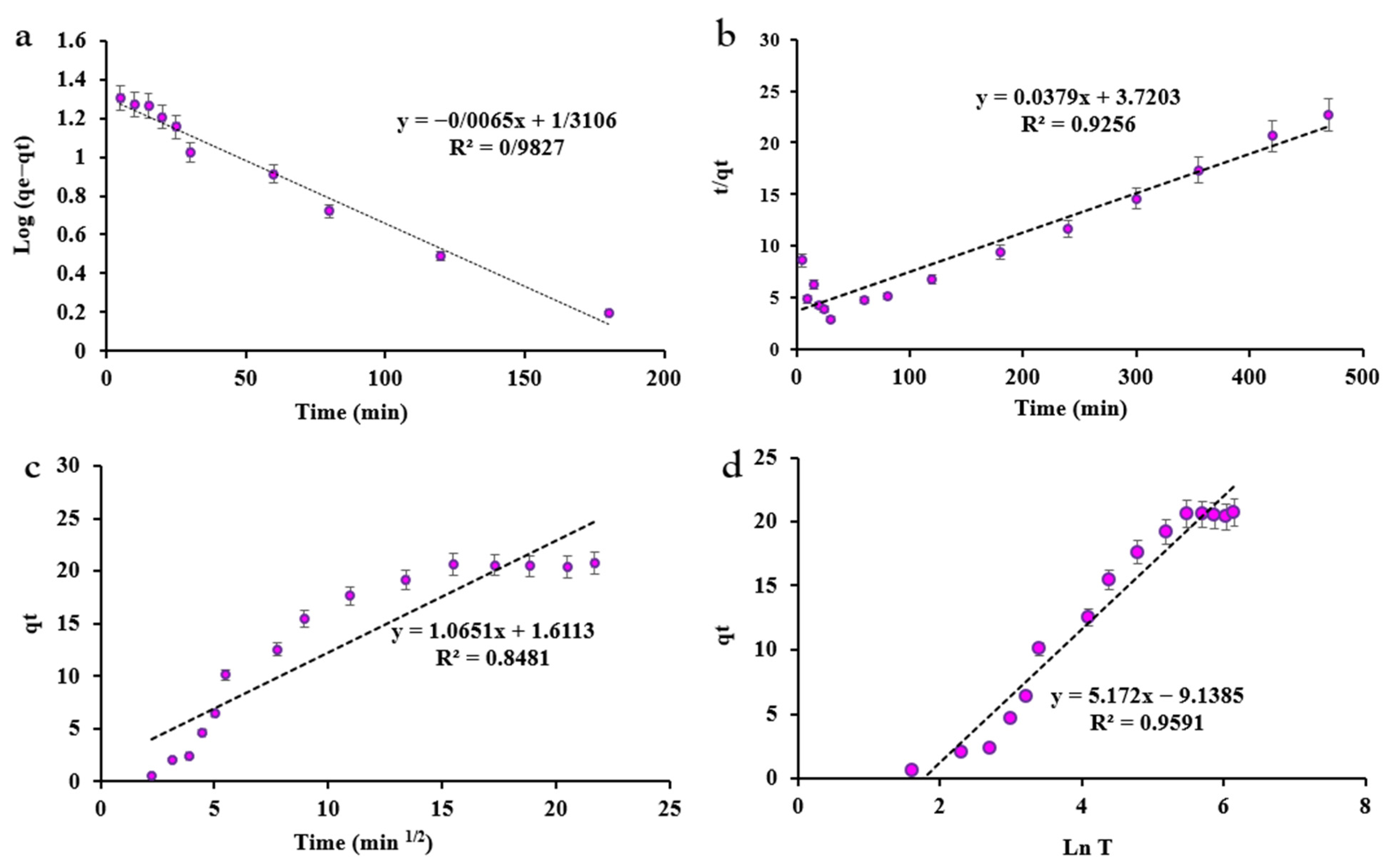

| Model | Parameter | Value |
|---|---|---|
| Pseudo-first-order | K1 | 0.0149 |
| qe (cal) | 20.4456 | |
| R2 | 0.9827 | |
| Pseudo-second-order | K2 | 0.0004 |
| qe (cal) | 26.3852 | |
| R2 | 0.9256 | |
| Intraparticle Diffusion | Kdiff | 1.0651 |
| C | 1.6113 | |
| R2 | 0.8481 | |
| Elovich | β | 0.1933 |
| α | 0.8837 | |
| R2 | 0.9591 |
| Temperature (K) | ||||||||
|---|---|---|---|---|---|---|---|---|
| 298.15 | 303.15 | 318.15 | 328.15 | 333.15 | 338.15 | 343.15 | 348.15 | |
| ∆H° (kJ/mol) | 53.8430 | −50.6528 | ||||||
| ∆S° (kJ/mol. K) | 0.1918 | −0.1272 | ||||||
| Ea (kJ/mol) | 11.1055 | −14.2084 | ||||||
| S* | 0.0105 | 112.3480 | ||||||
| ∆G (KJ/mol) | ||||||||
| −3.3273 | −4.2860 | −7.1623 | −8.9023 | −8.2661 | −7.6299 | −6.9938 | −6.3577 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Butt, L.; Das, A.; Tabibi, A.; Rehmani, M.; Karimi, B. Enhanced Heparin Adsorption from Porcine Mucosa Using Beta Zeolites: Optimization and Kinetic Analysis. Purification 2025, 1, 6. https://doi.org/10.3390/purification1020006
Butt L, Das A, Tabibi A, Rehmani M, Karimi B. Enhanced Heparin Adsorption from Porcine Mucosa Using Beta Zeolites: Optimization and Kinetic Analysis. Purification. 2025; 1(2):6. https://doi.org/10.3390/purification1020006
Chicago/Turabian StyleButt, Laiba, Anushree Das, Alireza Tabibi, Mousab Rehmani, and Benson Karimi. 2025. "Enhanced Heparin Adsorption from Porcine Mucosa Using Beta Zeolites: Optimization and Kinetic Analysis" Purification 1, no. 2: 6. https://doi.org/10.3390/purification1020006
APA StyleButt, L., Das, A., Tabibi, A., Rehmani, M., & Karimi, B. (2025). Enhanced Heparin Adsorption from Porcine Mucosa Using Beta Zeolites: Optimization and Kinetic Analysis. Purification, 1(2), 6. https://doi.org/10.3390/purification1020006






