First Identification of P230L and H134R Mutations Conferring SDHIs Resistance in Stemphylium vesicarium Isolated from an Italian Experimental Pear Orchard
Abstract
1. Introduction
2. Materials and Methods
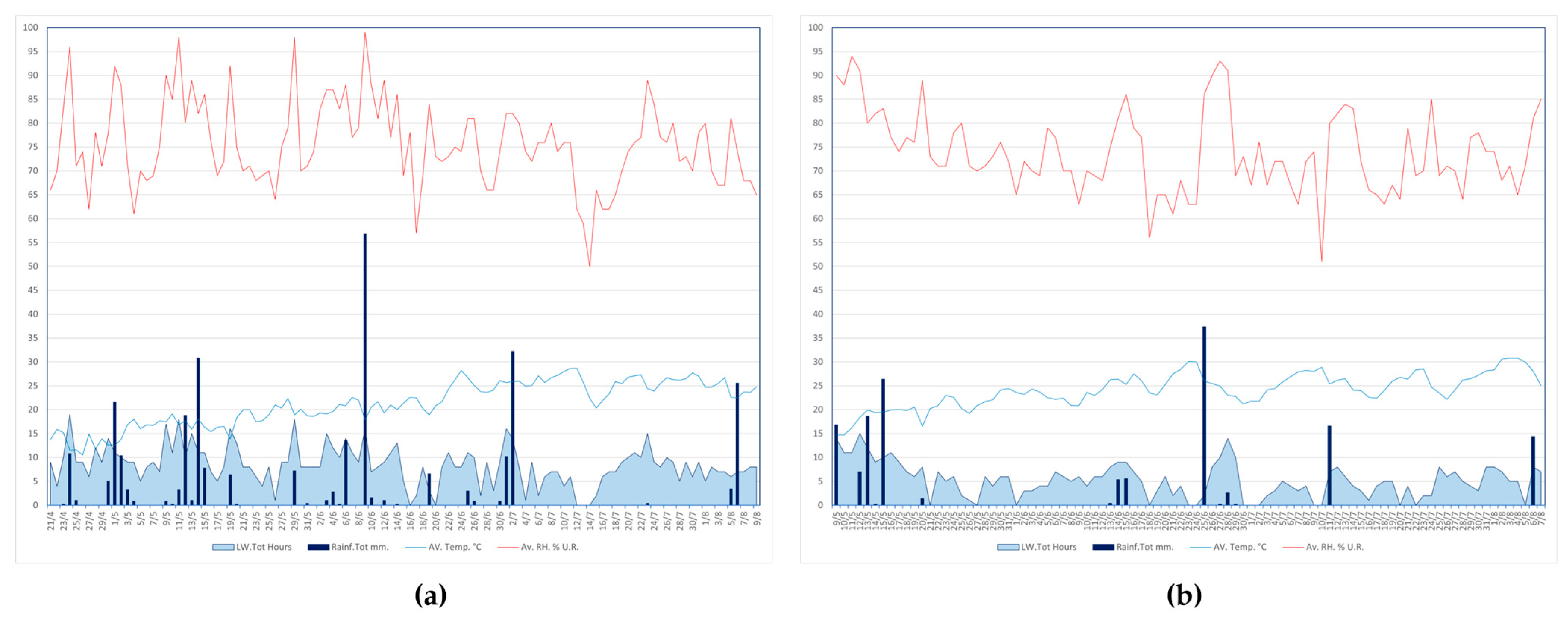
2.1. Field Experiments
2.2. Isolation of S. vesicarium
2.3. In Vitro Sensitivity Tests
2.4. DNA Extraction and Downstream Procedures for the Determination of Mutations
3. Results
3.1. Field Experiments
3.2. Sensitivity Assay
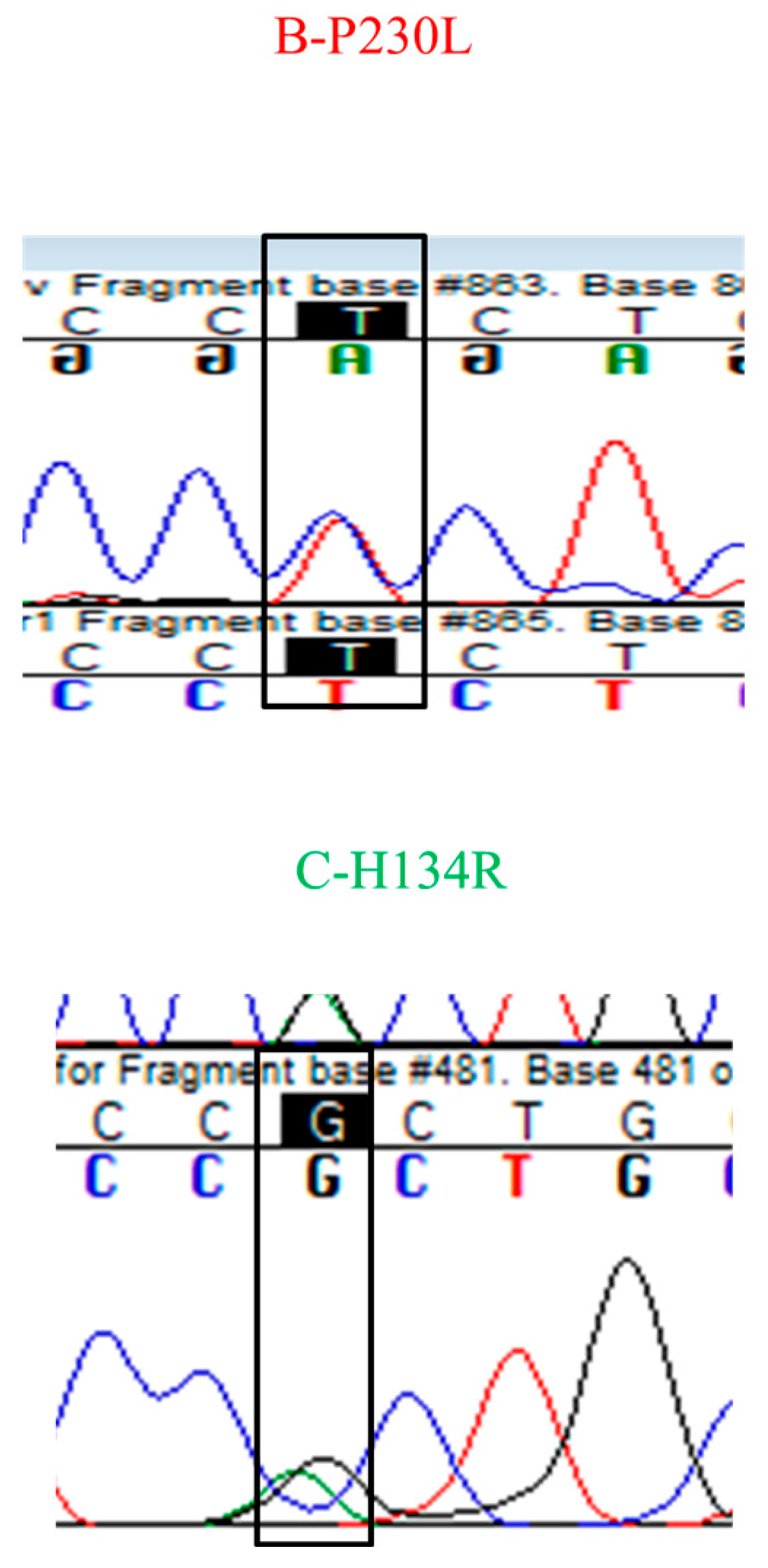
3.3. Identification of SDHB P230L and SDHC H134R Mutations
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| a.i. | Active ingredient |
| BSP | Brown spot of pear |
| FRAC | Fungicide Resistance Action Committee |
| h | Hour |
| MoAs | Modes of actions |
| MIs | Monoconidial isolates |
| SDHIs | Succinate dehydrogenase inhibitors |
References
- Woudenberg, J.H.C.; Hanse, B.; van Leeuwen, G.C.M.; Groenewald, J.Z.; Crous, P.W. Stemphylium revisited. Stud. Mycol. 2017, 87, 77–103. [Google Scholar] [CrossRef]
- Blancard, D.; Piquemal, J.P.; Gindrat, D. La stemphyliose de l’asperge. Rev. Hortic. 1984, 248, 27–30. [Google Scholar]
- Basallote Ureba, M.J.; Prados-Ligero, A.M.; Melero-Vara, J.M. Aetiology of leaf spot of garlic and onion caused by Stemphylium vesicarium in Spain. Plant Pathol. 1999, 48, 139–145. [Google Scholar] [CrossRef]
- Falloon, P.G.; Falloon, L.M.; Grogan, R.G. Etiology and epidemiology of Stemphylium leaf spot and purple spot of asparagus in California. Phytopathology 1987, 77, 407–413. [Google Scholar] [CrossRef]
- Lamprecht, S.C.; Baxter, A.; Thompson, A.H. Stemphylium vesicarium on Medicago spp. in South Africa. Phytophylactica 1984, 16, 73–75. [Google Scholar]
- Rossi, V.; Pattori, E.; Giosué, S.; Bugiani, R. Growth and sporulation of Stemphylium vesicarium, the causal agent of brown spot of pear, on herb plants of orchard lawns. Eur. J. Plant Pathol. 2005, 111, 361–370. [Google Scholar] [CrossRef]
- Suheri, H.; Price, T.V. Infection of onion leaves by Alternaria porri and Stemphylium vesicarium and disease development in controlled environments. Plant Pathol. 2000, 49, 375–382. [Google Scholar] [CrossRef]
- Ponti, I.; Cavanni, P.; Brunelli, A. Brown spot of pear: Aetiology and protection. Inf. Fitopatol. 1982, 32, 35–40. [Google Scholar]
- Blancard, D.; Allard, E.; Brest, P. La stemphyliose du poirier ou “macules brunes”. Phytoma 1989, 406, 35–37. [Google Scholar]
- Heijne, B.; Van Mourik, J. Zwartvruchtrot op peer neemt toe. Fruitt. Den Haag 2001, 91, 18–19. [Google Scholar]
- Llorente, I.; Moragrega, C.; Ruz, L.; Montesinos, E. An update on control of brown spot of pear. Trees 2012, 26, 239–245. [Google Scholar] [CrossRef] [PubMed]
- Vilardell, P. Stemphylium vesicarium en plantaciones de peral. Frutic. Prof. 1988, 18, 51–55. [Google Scholar]
- Llorente, I.; Montesinos, E. Brown spot of pear: An emerging disease of economic importance in Europe. Plant Dis. 2006, 92, 99–104. [Google Scholar] [CrossRef]
- Llorente, I.; Moragrega, C.; Ruz, L.; Santamaría, G.; Vilardell, A.; Vilardell, P.; Montesinos, E. Basis for new strategies in integrated control of brown spot of pear (Stemphylium vesicarium, telemorpoh Pleospora allii). IOBC/WPRS Bull. 2010, 54, 35–39. [Google Scholar]
- Rossi, V.; Pattori, E.; Bugiani, R. Sources and seasonal dynamics of inoculum for brown spot disease of pear. Eur. J. Plant Pathol. 2008, 121, 147–159. [Google Scholar] [CrossRef]
- Montesinos, E.; Moragrega, C.; Llorente, I.; Vilardell, P.; Bonaterra, A.; Ponti, I.; Bugiani, R.; Cavanni, P.; Brunelli, A. Development and evaluation of an infection model for Stemphylium vesicarium on pear based on temperature and wetness duration. Phytopathology 1995, 85, 586–592. [Google Scholar] [CrossRef]
- Cugier, J.P.; Humbert, W. Stemphyliose du poirier. Etude de la biologie du parasite et recherches des fongicides actifs. Phytoma Déf. Vég. 1991, 431, 47–50. [Google Scholar]
- Montesinos, E.; Vilardell, P. Evaluation of FAST as a forecasting system for scheduling fungicide sprays for control of Stemphylium vesicarium on pear. Plant Dis. 1992, 76, 1221–1226. [Google Scholar] [CrossRef]
- Singh, P.; Bugiani, R.; Cavanni, P.; Nakajima, H.; Kodama, M.; Otani, H.; Kohmoto, K. Purification and biological characterization of host-specific SV-toxins from Stemphylium vesicarium causing brown spot of European pear. Phytopathology 1999, 89, 947–953. [Google Scholar] [CrossRef]
- Singh, P.; Park, P.; Bugiani, R.; Cavanni, P.; Nakajiama, H.; Kodama, M.; Otani, H.; Kohmoto, K. Effect of host- selective SV-toxin from Stemphyilium vesicarium, the cause of brown spot of European pear plants, on ultrasctucture of leaf cells. J. Phytopathol. 2000, 148, 87–93. [Google Scholar] [CrossRef]
- Cavanni, P.; Ponti, I. Maculatura bruna del pero: Una micopatia sempre d’attualità. Riv. Fruttic. Ortofloric. 1994, 12, 37–42. [Google Scholar]
- Montesinos, E.; Moragrega, C.; Llorente, I.; Vilardell, P. Susceptibility of selected European pear cultivars to infection by Stemphylium vesicarium and influence of leaf and fruit age. Plant Dis. 1995, 79, 471–473. [Google Scholar] [CrossRef]
- Cappai, F.; De Franceschi, P.; Ciriani, A.; Collina, M.; Dondini, L. QTLs for susceptibility to Stemphylium vesicarium in pear. Mol. Breed. 2018, 38, 24. [Google Scholar] [CrossRef]
- Gazzetti, K.; Diaconu, E.L.; Nanni, I.M.; Ciriani, A.; Collina, M. Genome sequence resource for Stemphylium vesicarium, causing brown spot disease of pear. Mol. Plant-Microbe Interact. 2019, 32, 935–938. [Google Scholar] [CrossRef]
- Rossi, V.; Pattori, E. Inoculum reduction of Stemphylium vesicarium, the causal agent of brown spot of pear, through application of Trichoderma-based products. Biol. Control 2009, 49, 52–57. [Google Scholar] [CrossRef]
- Puig, M.; Moragrega, C.; Ruz, L.; Montesinos, E.; Llorente, I. Controlling brown spot of pear by a synthetic antimicrobial peptide under field conditions. Plant Dis. 2015, 99, 1816–1822. [Google Scholar] [CrossRef]
- Brunelli, A.; Di Marco, G.; Contarelli, G.; Ponti, I. Prove di lotta contro la maculatura bruna delle pere. Atti Giornate Fitopatol. 1984, 2, 203–212. [Google Scholar]
- Brunelli, A.; Rovesti, R.; Di Marco, S.; Ponti, I. Attività di diversi fungicidi contro la maculatura bruna del pero. Riv. Fruttic. Ortofloric. 1986, 1, 51–54. [Google Scholar]
- Ponti, I.; Brunelli, A.; Tosi, C.; Basaglia, M.; Bevilacqua, T.; Emiliani, G.; Cont, C.; Viccinelli, R. Verifica dell’attività di diversi preparati contro la maculatura bruna del pero. Inf. Fitopatol. 1993, 43, 45–52. [Google Scholar]
- Ponti, I.; Brunelli, A.; Tosi, C.; Cavallini, G.; Mazzini, F. Chemical control trials on pear brown spot (Stemphylium vesicarium). Atti Giornate Fitopatol. 1996, 2, 165–172. [Google Scholar]
- Llorente, I.; Montesinos, E. Development and field evaluation of a model to estimate the maturity of pseudothecia of Pleospora allii on pear. Plant Dis. 2004, 88, 215–219. [Google Scholar] [CrossRef]
- Llorente, I.; Vilardell, P.; Bugiani, R.; Gherardi, I.; Montesinos, E. Evaluation of BSPcast disease warning system in reduced fungicide use programs for management of brown spot of pear. Plant Dis. 2000, 84, 631–637. [Google Scholar] [CrossRef][Green Version]
- Alberoni, G.; Collina, M.; Pancaldi, D.; Brunelli, A. Resistance to dicarboximide fungicides in Stemphylium vesicarium of Italian pear orchards. Eur. J. Plant Pathol. 2005, 113, 211–219. [Google Scholar] [CrossRef]
- Alberoni, G.; Cavallini, D.; Collina, M.; Brunelli, A. Characterization of the first Stemphylium vesicarium isolates resistant to strobilurins in Italian pear orchards. Eur. J. Plant Pathol. 2010, 126, 453–457. [Google Scholar] [CrossRef]
- Alberoni, G.; Collina, M.; Cavallini, D.; Brunelli, A. Field strains of Stemphylium vesicarium with a resistance to dicarboximide fungicides correlated with changes in a two-component histidine kinase. Eur. J. Plant Pathol. 2010, 128, 171–184. [Google Scholar] [CrossRef]
- Sierotzki, H.; Scalliet, G. A review of current knowledge of resistance aspects for the next-generation succinate dehydrogenase inhibitor fungicides. Phytopathology 2013, 103, 880–887. [Google Scholar] [CrossRef]
- Carolina Duarte Hospital; Tete, A.; Debizet, K.; Imler, J.; Tomkiewicz-Raulet, C.; Blanc, E.B.; Barouki, R.; Coumoul, X.; Bortoli, S. SDHI fungicides: An example of mitotoxic pesticides targeting the succinate dehydrogenase complex. Environ. Int. 2023, 180, 108219. [Google Scholar] [CrossRef] [PubMed]
- Brunelli, A.; Fabbri, M.; Casagrandi, F.; Geminiani, E.; Troiano, P.P. Attività di recenti fungicidi inibitori della Succinato Deidrogenasi (SDHI) contro la maculatura bruna del pero. Atti Giornate Fitopatol. 2016, 2, 377–384. [Google Scholar]
- Fungicide Resistance Action Committee SDHI Fungicides Page. Available online: https://www.frac.info/frac-teams/working-groups/sdhi-fungicides/#open-tour (accessed on 15 April 2025).
- Fungicide Resistance Action Committee List of Species Resistant to SDHIs April 2015 Page. Available online: https://www.frac.info/media/k5yocwty/list-of-species-resistant-to-sdhis-april-2015.pdf (accessed on 15 April 2025).
- Alberoni, G.; Cavallini, D.; Collina, M.; Brunelli, A. Baseline sensitivity of Stemphylium vesicarium, the causal agent of pear brown spot, to boscalid. Commun. Agric. Appl. Biol. Sci. 2009, 74, 797–800. [Google Scholar] [PubMed]
- Ciriani, A.; Gazzetti, K.; Collina, M. Baseline sensitivity of Stempylium vesicarium of pear to SDHIs and fluazinam. In Book of Abstracts: XXII National Congress of the Italian Society of Plant Pathology; ETS: Pisa, ltaly, 2016; p. 85. [Google Scholar]
- Mair, W.; Lopez-Ruiz, F.; Stammler, G.; Clark, W.; Burnett, F.; Hollomon, D. Proposal for a unified nomenclature for target site mutations associated with resistance to fungicides. Pest Manag. Sci. 2016, 72, 1449–1459. [Google Scholar] [CrossRef]

| Block No. | Commercial Product | Active Ingredient (Concentration) | Rate (g or mL/100 L) |
|---|---|---|---|
| 1 | Cantus WG | boscalid (50%) | 27 g |
| 2 | Fontelis SC | penthiopyrad (200 g/L) | 75 mL |
| 3 | Xemium | fluxapyroxad (300 g/L) | 20 mL |
| 4 | Switch WG | cyprodinil + fludioxonil (37.5 + 25%) | 80 g |
| Block No. | Commercial Product | Active Ingredient (Concentration) | Rate (g or mL/100 L) |
|---|---|---|---|
| 1 | Cantus WG | boscalid (50%) | 27 g |
| 2 | Fontelis SC | penthiopyrad (200 g/L) | 75 mL |
| 3 | Xemium | fluxapyroxad (300 g/L) | 20 mL |
| 4 | Luna Privilege | fluopyram (500 g/L) | 28 mL |
| 5 | Switch WG | cyprodinil + fludioxonil (37.5 + 25%) | 80 g |
| EC50 Value (mg/L) | Strain | Notation |
|---|---|---|
| <2.5 | Sensitive | value (mg/L) |
| <5 | Resistant | value (mg/L) |
| <20 | Resistant | >2.5 |
| >20 | Resistant | >>2.5 |
| Name | Target | Sequence (5′-3′) | Amplicon Size (bp) |
|---|---|---|---|
| SdhB_for | sdh B | CACTCTCCACTCCGCACTAC | 1192 |
| SdhB_rev | sdh B | TGTCTGGTGACACTCGCTG | 1192 |
| SdhC_for | sdh C | AGCAGAGTAATGCGAAGGCA | 863 |
| SdhC_rev | sdh C | GGCTTGACACATGGAGGATC | 863 |
| SdhD_for | sdh D | CGAGGCATTGTCGTCAAC | 788 |
| SdhD_rev | sdh D | CCAAATTACACAGGCTGTATGCT | 788 |
| Fer4_17_for | sdh B | GTGGCTGAAGTACTAACGACCG | 780 |
| Fer4_17_rev | sdh B | ACCCTCGAGAGTCCACTCAC | 780 |
| Treatment | Disease Incidence (%) 2016 | Efficacy (%) 2016 | Disease Incidence (%) 2017 | Efficacy (%) 2017 |
|---|---|---|---|---|
| Untreated | 25.8 a | - | 59.3 a | - |
| Boscalid | 6.5 b | 74.8 | 68.4 a | 0 |
| Penthiopyrad | 8.8 b | 65.9 | 45.1 a | 23.9 |
| Fluxapyroxad | 21.5 a | 16.7 | 54.2 a | 8.6 |
| Fluopyram | - | - | 66.6 a | 0 |
| Cyprodinil + fludioxonil | 4.6 b | 82.2 | 15.0 b | 74.7 |
| 2016 (EC50 mg/L) | 2017 (EC50 mL/L) | |||||||
|---|---|---|---|---|---|---|---|---|
| Boscalid | Fluxapyroxad | Penthiopyrad | Fluopyram | Boscalid | Fluxapyroxad | Penthiopyrad | Fluopyram | |
| Untreated July | 0.46 | 0.11 | 0.02 | 0.01 | - | - | - | - |
| Untreated August | 0.55 | 0.49 | 0.39 | 0.26 | >2.5 | >>2.5 | >>2.5 | >>2.5 |
| Boscalid | 0.43 | 0.43 | 0.3 | - | >2.5 | >>2.5 | >2.5 | >2.5 |
| Penthiopyrad | 0.77 | 0.5 | 0.49 | - | >>2.5 | >>2.5 | >>2.5 | >2.5 |
| Fluxapyroxad July | >>2.5 | >>2.5 | >>2.5 | >>2.5 | - | - | - | - |
| Fluxapyroxad August | >>2.5 | >>2.5 | >>2.5 | >>2.5 | >>2.5 | >>2.5 | >>2.5 | >>2.5 |
| Cyprodinil + Fludioxonil | 0.94 | 0.88 | 1.07 | - | - | - | - | - |
| Fluopyram | - | - | - | - | >>2.5 | >>2.5 | >>2.5 | >2.5 |
| Isolates | EC 50 (mg/L) | |||
|---|---|---|---|---|
| Boscalid | Fluxapyroxad | Penthiopyrad | Fluopyram | |
| 1 | >>2.5 | >>2.5 | 4.21 | >2.5 |
| 2 | >>2.5 | |||
| 3 | >>2.5 | |||
| 4 | >>2.5 | >2.5 | >>2.5 | >2.5 |
| 5 | >>2.5 | |||
| 6 | >>2.5 | |||
| 7 | >>2.5 | |||
| 8 | >>2.5 | |||
| 9 | >>2.5 | |||
| 10 | >>2.5 | |||
| 11 | >>2.5 | |||
| 12 | >>2.5 | |||
| 13 | >>2.5 | |||
| 14 | >>2.5 | >>2.5 | >>2.5 | >>2.5 |
| 15 | >>2.5 | |||
| Monoconidial Strains 2016 | EC 50 (mg/L) Fluxapyroxad |
|---|---|
| 1.1 | >2.5 |
| 1.2 | >>2.5 |
| 1.3 | >>2.5 |
| 4.1 | >>2.5 |
| 4.2 | >>2.5 |
| 4.3 | >2.5 |
| 14.1 | 3.47 |
| 14.2 | 2.36 |
| 14.3 | 1.34 |
| STRAIN | SNPs |
|---|---|
| Fluxapyroxad population August 2016 | B-P230L, C-H134R |
| Fluxapyroxad isolates August 2016 | |
| 1 | B-P230L |
| 2 | C-H134R |
| 3 | B-P230L |
| 4 | B-P230L |
| 5 | C-H134R |
| 6 | C-H134R |
| 7 | B-P230L |
| 8 | C-H134R |
| 9 | C-H134R |
| 10 | B-P230L |
| 11 | B-P230L |
| 12 | B-P230L |
| 13 | B-P230L |
| 14 | C-H134R |
| 15 | C-H134R |
| Fluxapyroxad monoconidial isolates 2016 | |
| 1.1 | B-P230L |
| 1.2 | B-P230L |
| 1.3 | B-P230L |
| 4.1 | B-P230L |
| 4.2 | B-P230L |
| 4.3 | B-P230L |
| 14.1 | C-H134R |
| 14.2 | C-H134R |
| 14.3 | C-H134R |
| Fluxapyroxad population 2017 | B-P230L, C-H134R |
| Fluopyram population 2017 | B-P230L, C-H134R |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gazzetti, K.; Menghini, M.; Nanni, I.M.; Ciriani, A.; Fabbri, M.; Venturi, P.; Collina, M. First Identification of P230L and H134R Mutations Conferring SDHIs Resistance in Stemphylium vesicarium Isolated from an Italian Experimental Pear Orchard. Agrochemicals 2025, 4, 15. https://doi.org/10.3390/agrochemicals4030015
Gazzetti K, Menghini M, Nanni IM, Ciriani A, Fabbri M, Venturi P, Collina M. First Identification of P230L and H134R Mutations Conferring SDHIs Resistance in Stemphylium vesicarium Isolated from an Italian Experimental Pear Orchard. Agrochemicals. 2025; 4(3):15. https://doi.org/10.3390/agrochemicals4030015
Chicago/Turabian StyleGazzetti, Katia, Massimiliano Menghini, Irene Maja Nanni, Alessandro Ciriani, Mirco Fabbri, Pietro Venturi, and Marina Collina. 2025. "First Identification of P230L and H134R Mutations Conferring SDHIs Resistance in Stemphylium vesicarium Isolated from an Italian Experimental Pear Orchard" Agrochemicals 4, no. 3: 15. https://doi.org/10.3390/agrochemicals4030015
APA StyleGazzetti, K., Menghini, M., Nanni, I. M., Ciriani, A., Fabbri, M., Venturi, P., & Collina, M. (2025). First Identification of P230L and H134R Mutations Conferring SDHIs Resistance in Stemphylium vesicarium Isolated from an Italian Experimental Pear Orchard. Agrochemicals, 4(3), 15. https://doi.org/10.3390/agrochemicals4030015







