Exercise-Induced Gastrointestinal Symptoms in Endurance Sports: A Review of Pathophysiology, Symptoms, and Nutritional Management
Abstract
:1. Introduction
2. Methods
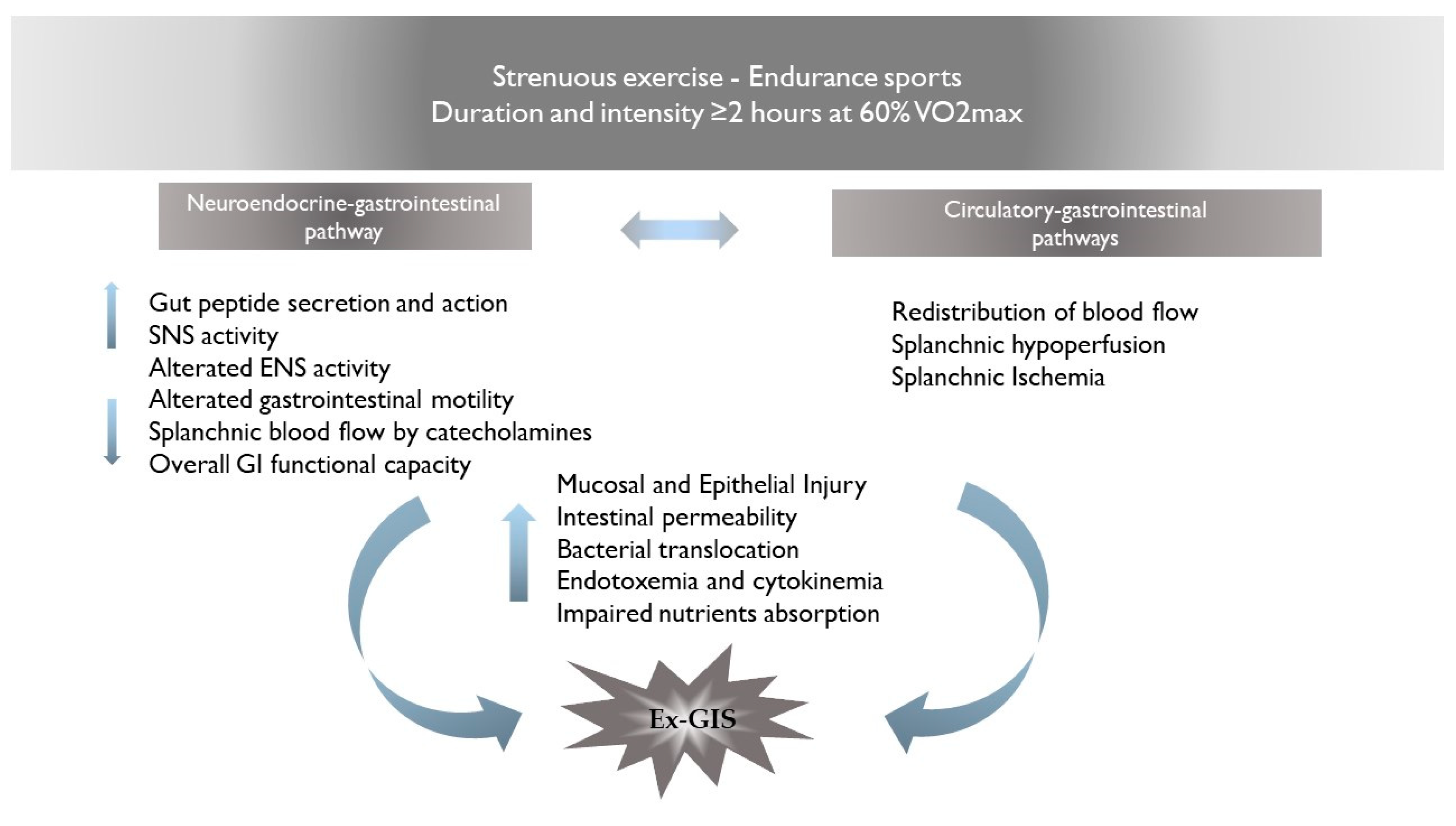
3. Pathophysiology of Ex-GIS: Proposed Mechanisms for GI Distress
3.1. Neuroendocrine–Gastrointestinal Pathway
3.2. Circulatory–Gastrointestinal Pathways
4. Gastrointestinal Symptoms during Exercise
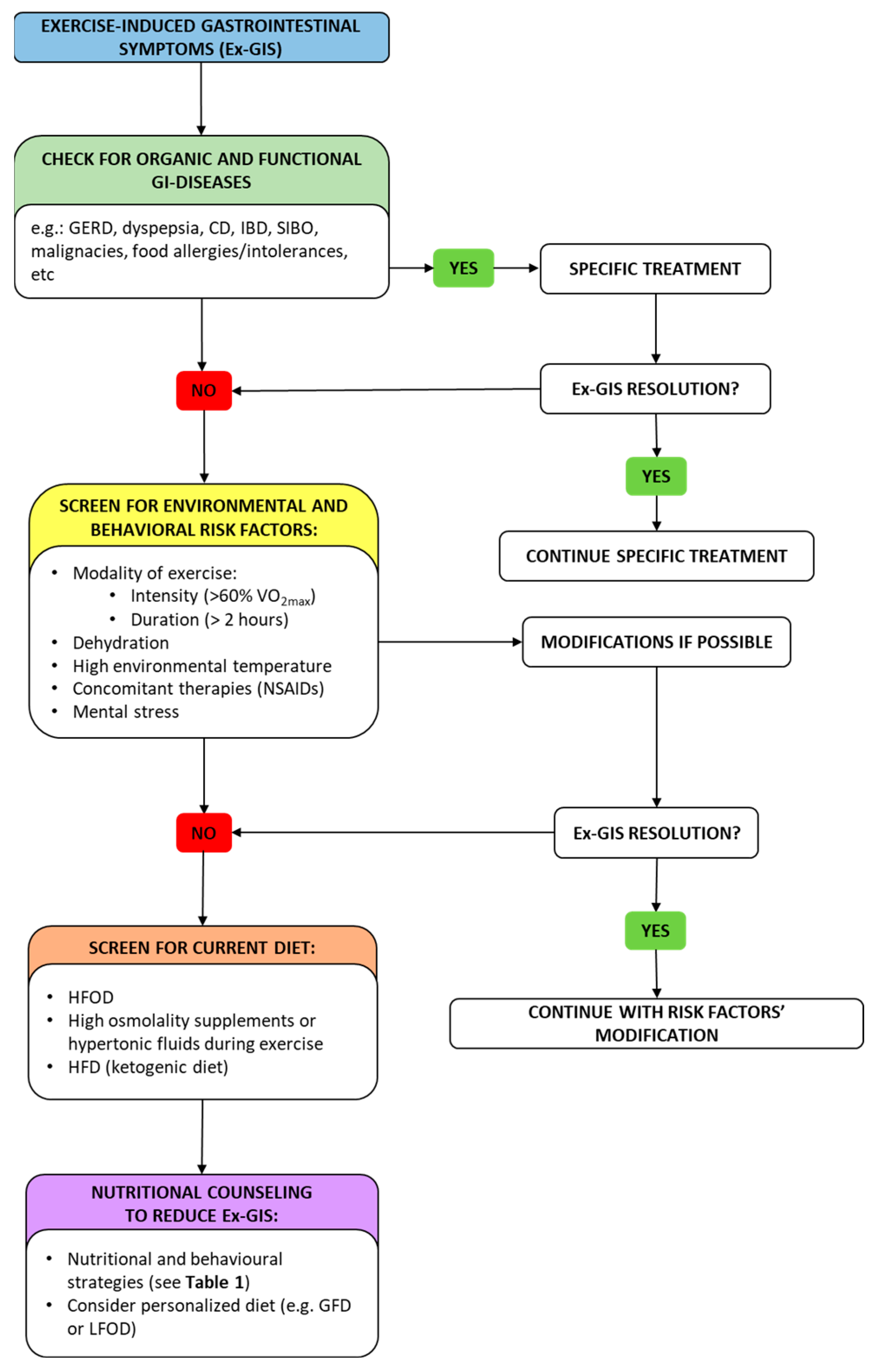
5. Nutritional and Behavior Strategies to Reduce Ex-GIS
6. Efficacy of Specific Diets Applied by Endurance Athletes to Avoid Ex-GIS
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- de Oliveira, E.P.; Burini, R.C. The impact of physical exercise on the gastrointestinal tract. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 533–538. [Google Scholar] [CrossRef]
- Slattery, M.L. Physical activity and colorectal cancer. Sports Med. 2004, 34, 239–252. [Google Scholar] [CrossRef]
- Lira, C.A.B.; Viana, R.B.; Mesquista, K.P.; Santos, D.A.T.; Campos, M.H.; Andrade, M.S.; Vancini, R.L. Frequency and intensity of gastrointestinal symptoms in exercisers individuals at rest and during physical exercise: An internet-based survey. Intest. Res. 2019, 17, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.J.S.; Mika, A.S.; McCubbin, A.J. The impact of exercise modality on exercise-induced gastrointestinal syndrome and associated gastrointestinal symptoms. J. Sci. Med. Sport 2022, 25, 788–793. [Google Scholar] [CrossRef]
- Costa, R.J.S.; Snipe, R.M.J.; Kitic, C.M.; Gibson, P.R. Systematic review: Exercise-induced gastrointestinal syndrome-implications for health and intestinal disease. Aliment. Pharmacol. Ther. 2017, 46, 246–265. [Google Scholar] [CrossRef]
- World Health Organisation. Global Recommendations on Physical Activity for Health; WHO Library Cataloguing-in-Publication Data Press, World Health Organization: Geneva, Switzerland, 2010. [Google Scholar]
- Bull, F.C. The Expert Working Groups. Physical Activity Guidelines in the U.K.: Review and Recommendations; School of Sport, Exercise and Health Sciences, Loughborough University: Loughborough, UK, 2010. [Google Scholar]
- Mach, N.; Fuster-Botella, D. Endurance exercise and, G.M. A review. J. Sport Health Sci. 2017, 6, 179–197. [Google Scholar] [CrossRef]
- Zhao, X.; Zhang, Z.; Hu, B.; Huang, W.; Yuan, C.; Zou, L. Response of GM to Metabolite Changes Induced by Endurance Exercise. Front. Microbiol. 2018, 9, 765. [Google Scholar] [CrossRef]
- van Wijck, K.; Lenaerts, K.; Grootjans, J.; Wijnands, K.A.P.; Poeze, M.; van Loon, L.J.C.; Dejong, C.H.C.; Buurman, W.A. Physiology and pathophysiology of splanchnic hypoperfusion and intestinal injury during exercise: Strategies for evaluation and prevention. Am. J. Physiol. 2012, 303, G155–G168. [Google Scholar] [CrossRef] [PubMed]
- van Wijck, K.; Lenaerts, K.; van Loon, L.J.; Peters, W.H.M.; Buurman, W.A.; Dejong, C.H.C. Exercise-induced splanchnic hypoperfusion results in gut dysfunction in healthy men. PLoS ONE 2011, 6, e22366. [Google Scholar] [CrossRef]
- Smith, K.A.; Pugh, J.N.; Duca, F.A.; Close, G.L.; Ormsbee, M.J. Gastrointestinal pathophysiology during endurance exercise: Endocrine, microbiome, and nutritional influences. Eur. J. Appl. Physiol. 2021, 121, 2657–2674. [Google Scholar] [CrossRef] [PubMed]
- Horner, K.M.; Schubert, M.M.; Desbrow, B.; Byrne, N.M.; King, N.A. Acute exercise and gastric emptying: A meta-analysis and implications for appetite control. Sports Med. 2015, 45, 659–678. [Google Scholar] [CrossRef]
- Costa, M.; Brookes, S.J.; Hennig, G.W. Anatomy and physiology of the enteric nervous system. Gut 2000, 47 (Suppl. S4), iv15–iv19; discussion iv26. [Google Scholar] [CrossRef] [PubMed]
- Moran, T. Gut peptides in the control of food intake. Int. J. Obes. 2009, 33 (Suppl. S1), S7–S10. [Google Scholar] [CrossRef] [PubMed]
- Côté, C.D.; Zadeh-Tahmasebi, M.; Rasmussen, B.A.; Duca, F.A.; Lam, T.K.T. Hormonal signaling in the gut. J. Biol. Chem. 2014, 289, 11642–11649. [Google Scholar] [CrossRef]
- Schwartz, G.J.; McHugh, P.R.; Moran, T.H. Gastric loads and cholecystokinin synergistically stimulate rat gastric vagal afferents. Am. J. Physiol. 1993, 265, R872–R876. [Google Scholar] [CrossRef]
- Schwartz, G.J.; Tougas, G.; Moran, T.H. Integration of vagal afferent responses to duodenal loads and exogenous CCK in rats. Peptides 1995, 16, 707–711. [Google Scholar] [CrossRef] [PubMed]
- Liddle, R.A.; Goldfine, I.D.; Rosen, M.S.; Taplitz, R.A.; Williams, J.A. Cholecystokinin bioactivity in human plasma. Molecular forms, responses to feeding, and relationship to gallbladder contraction. J. Clin. Investig. 1985, 75, 1144–1152. [Google Scholar] [CrossRef]
- Adrian, T.E.; Ferri, G.L.; Bacarese-Hamilton, A.J.; Fussel, H.S.; Polak, J.M.; Bloom, S.R. Human distribution and release of a putative new gut hormone, peptide, Y.Y. Gastroenterology 1985, 89, 1070–1077. [Google Scholar] [CrossRef]
- Holst, J.J. Glucagon like peptide 1: A newly discovered gastrointestinal hormone. Gastroenterology 1994, 107, 1848–1855. [Google Scholar] [CrossRef]
- Harada, K.; Kitaguchi, T.; Tsuboi, T. Integrative function of adrenaline receptors for glucagon-like peptide-1 exocytosis in enteroendocrine L cell line GLUTag. Biochem. Biophys. Res. Commun. 2015, 460, 1053–1058. [Google Scholar] [CrossRef]
- Brechet, S.; Plaisancié, P.; Dumoulin, V.; Chayvialle, J.A.; Cuber, J.C.; Claustre, J. Involvement of beta1- and beta2- but not beta3-adrenoceptor activation in adrenergic PYY secretion from the isolated colon. J. Endocrinol. 2001, 168, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Goyal, R.K.; Guo, Y.; Mashimo, H. Advances in the physiology of gastric emptying. Neurogastroenterol. Motil. 2019, 31, e13546. [Google Scholar] [CrossRef]
- Travagli, R.A.; Anselmi, L. Vagal neurocircuitry and its influence on gastric motility. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 389–401. [Google Scholar] [CrossRef]
- Halliday, T.M.; White, M.H.; Hild, A.K.; Conroy, M.B.; Melanson, E.L.; Cornier, M.A. Appetite and Energy Intake Regulation in Response to Acute Exercise. Med. Sci. Sports Exerc. 2021, 53, 2173–2181. [Google Scholar] [CrossRef]
- Broom, D.R.; Batterham, R.L.; King, J.A.; Stensel, D.J. Influence of resistance and aerobic exercise on hunger, circulating levels of acylated ghrelin, and peptide YY in healthy males. American journal of physiology. Regul. Integr. Comp. Physiol. 2009, 296, R29–R35. [Google Scholar] [CrossRef] [PubMed]
- Balaguera-Cortes, L.; Wallman, K.E.; Fairchild, T.J.; Guelfi, K.J. Energy intake and appetite-related hormones following acute aerobic and resistance exercise. Appl. Physiol. Nutr. Metab. = Physiol. Appl. Nutr. Metab. 2011, 36, 958–966. [Google Scholar] [CrossRef]
- Mittal, R.; Debs, L.H.; Patel, A.P.; Nguyen, D.; Patel, K.; O’Connor, G.; Grati, M.; Mittal, J.; Yan, D.; Eshraghi, A.A.; et al. Neurotransmitters: The Critical Modulators Regulating Gut-Brain Axis. J. Cell. Physiol. 2017, 232, 2359–2372. [Google Scholar] [CrossRef]
- Coxon, J.P.; Cash, R.F.H.; Hendrikse, J.J.; Rogasch, N.C.; Stavrinos, E.; Suo, C.; Yucel, M. GABA concentration in sensorimotor cortex following high-intensity exercise and relationship to lactate levels. J. Physiol. 2018, 596, 691–702. [Google Scholar] [CrossRef]
- Kjaer, M. Epinephrine and some other hormonal responses to exercise in man: With special reference to physical training. Int. J. Sports Med. 1989, 10, 2–15. [Google Scholar] [CrossRef] [PubMed]
- Binienda, A.; Storr, M.; Fichna, J.; Salaga, M. Efficacy and Safety of Serotonin Receptor Ligands in the Treatment of Irritable Bowel Syndrome: A Review. Curr. Drug Targets 2018, 19, 1774–1781. [Google Scholar] [CrossRef]
- Peters, H.P.; Wiersma, J.W.; Koerselman, J.; Akkermans, L.M.; Bol, E.; Mosterd, W.L.; de Vries, W.R. The effect of a sports drink on gastroesophageal reflux during a run-bike-run test. Int. J. Sports Med. 2000, 21, 65–70. [Google Scholar] [CrossRef] [PubMed]
- Fordtran, J.S.; Saltin, B. Gastric emptying and intestinal absorption during prolonged severe exercise. J. Appl. Physiol. 1967, 23, 331–335. [Google Scholar] [CrossRef] [PubMed]
- Leiper, J.B.; Prentice, A.S.; Wrightson, C.; Maughan, R.J. Gastric emptying of a carbohydrate-electrolyte drink during a soccer match. Med. Sci. Sports Exerc. 2001, 33, 1932–1938. [Google Scholar] [CrossRef]
- Camilleri, M.; Malagelada, J.R.; Kao, P.C.; Zinsmeister, A.R. Gastric and autonomic responses to stress in functional dyspepsia. Dig. Dis. Sci. 1986, 31, 1169–1177. [Google Scholar] [CrossRef] [PubMed]
- Song, G.Q.; Sun, Y.; Foreman, R.D.; Chen, J.D. Therapeutic potential of spinal cord stimulation for gastrointestinal motility disorders: A preliminary rodent study. Neurogastroenterol. Motil. 2014, 26, 377–384. [Google Scholar] [CrossRef]
- Rao, K.A.; Yazaki, E.; Evans, D.F.; Carbon, R. Objective evaluation of small bowel and colonic transit time using pH telemetry in athletes with gastrointestinal symptoms. Br. J. Sports Med. 2004, 38, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Clarke, S.F.; Murphy, E.F.; O’Sullivan, O.; Lucey, A.J.; Humphreys, M.; Hogan, A.; Hayes, P.; O’Reilly, M.; Jeffery, I.B.; Wood-Martin, R.; et al. Exercise and associated dietary extremes impact on gut microbial diversity. Gut 2014, 63, 1913–1920. [Google Scholar] [CrossRef] [PubMed]
- van Nieuwenhoven, M.A.; Brouns, F.; Brummer, R.J. The effect of physical exercise on parameters of gastrointestinal function. Neurogastroenterol. Motil. 1999, 11, 431–439. [Google Scholar] [CrossRef]
- van Nieuwenhoven, M.A.; Brouns, F.; Brummer, R.-J.M. Gastrointestinal profile of symptomatic athletes at rest and during physical exercise. Eur. J. Appl. Physiol. 2004, 91, 429–434. [Google Scholar] [CrossRef] [PubMed]
- Matheson, P.J.; Wilson, M.A.; Garrison, R.N. Regulation of intestinal blood flow. J. Surg. Res. 2000, 93, 182–196. [Google Scholar] [CrossRef]
- Wright, H.; Collins, M.; Villiers, R.D.; Schwellnus, M.P. Are splanchnic hemodynamics related to the development of gastrointestinal symptoms in ironman triathletes? A prospective cohort study. Clin. J. Sport Med. 2011, 21, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Qamar, M.I.; Read, A.E. Effects of exercise on mesenteric blood flow in man. Gut 1987, 28, 583–587. [Google Scholar] [CrossRef]
- Otte, J.A.; Oostveen, E.; Geelkerken, R.H.; Groeneveld, A.B.; Kolkman, J.J. Exercise induces gastric ischemia in healthy volunteers: A tonometry study. J. Appl. Physiol. 2001, 91, 866–871. [Google Scholar] [CrossRef]
- Kolkman, J.J.; Groeneveld, A.B.; van der Berg, F.G.; Rauwerda, J.A.; Meuwissen, S.G. Increased gastric PCO2 during exercise is indicative of gastric ischaemia: A tonometric study. Gut 1999, 44, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Kenney, W.L.; Ho, C.W. Age alters regional distribution of blood flow during moderate-intensity exercise. J. Appl. Physiol. 1995, 79, 1112–1119. [Google Scholar] [CrossRef] [PubMed]
- Derikx, J.P.; Matthijsen, R.A.; de Bruine, A.P.; van Bijnen, A.A.; Heineman, E.; van Dam, R.M.; Dejong, C.H.; Buurman, W.A. Rapid reversal of human intestinal ischemia-reperfusion induced damage by shedding of injured enterocytes and reepithelialisation. PLoS ONE 2008, 3, e3428. [Google Scholar] [CrossRef]
- Papantoniou, K.; Michailides, C.; Bali, M.; Papantoniou, P.; Thomopoulos, K. Gastrointestinal bleeding in athletes. Ann. Gastroenterol. 2023, 36, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Pelsers, M.M.; Hermens, W.T.; Glatz, J.F. Fatty acid-binding proteins as plasma markers of tissue injury. Clin. Chim. Acta 2005, 352, 15–35. [Google Scholar] [CrossRef]
- Berkes, J.; Viswanathan, V.K.; Savkovic, S.D.; Hecht, G. Intestinal epithelial responses to enteric pathogens: Effects on the tight junction barrier, ion transport, and inflammation. Gut 2003, 52, 439–451. [Google Scholar] [CrossRef]
- Baumgart, D.; Dignass, A. Intestinal barrier function. Curr. Opin. Clin. Nutr. Metab Care 2002, 5, 685–694. [Google Scholar] [CrossRef]
- Lambert, G.P. Intestinal barrier dysfunction, endotoxemia, and gastrointestinal symptoms: The ‘canary in the coal mine’ during exercise-heat stress? Med. Sport Sci. 2008, 53, 61–73. [Google Scholar] [CrossRef]
- Brock-Utne, J.; Gaffin, S.; Wells, M.; Gathiram, P.; Sohar, E.; James, M.; Morrell, D.; Norman, R. Endotoxemia in exhausted runners after a long-distance race. S. Afr. Med. J. 1988, 73, 533–536. [Google Scholar] [PubMed]
- Hart, T.L.; Townsend, J.R.; Grady, N.J.; Johnson, K.D.; Littlefield, L.A.; Vergne, M.J.; Fundaro, G. Resistance Exercise Increases Gastrointestinal Symptoms, Markers of Gut Permeability, and Damage in Resistance-Trained Adults. Med. Sci. Sports Exerc. 2022, 54, 1761–1770. [Google Scholar] [CrossRef] [PubMed]
- Lambert, G.P.; Murray, R.; Eddy, D.; Scott, W.; Laird, R.; Gisolfi, C.V. Intestinal permeability following the 1998 Ironman triathlon. Med. Sci. Sports Exerc. 1999, 31, S318. [Google Scholar] [CrossRef]
- Lambert, G.P.; Lang, J.A.; Bull, A.J.; Pfeifer, P.C.; Eckerson, J.M.; Moore, G.A.; Lanspa, S.J.; O’Brien, J.J. Fluid restriction increases GI permeability during running. Int. J. Sports Med. 2008, 29, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Jeukendrup, A.E.; Vet-Joop, K.; Sturk, A.; Stegen, J.H.; Senden, J.; Saris, W.H.; Wagenmakers, A.J. Relationship between gastro-intestinal complaints and endotoxemia, cytokine release and the acute-phase reaction during and after a long-distance triathlon in highly trained men. Clin. Sci. 2000, 98, 47–55. [Google Scholar] [CrossRef]
- Gill, S.K.; Hankey, J.; Wright, A.; Marczak, S.; Hemming, K.; Allerton, D.M.; Ansley-Robson, P.; Costa, R.J.S. The impact of a 24-hour ultramarathon on circulatory endotoxin and cytokine profile. Int. J. Sports Med. 2015, 36, 688–695. [Google Scholar]
- Motiani, K.K.; Collado, M.C.; Eskelinen, J.J.; Virtanen, K.A.; Löyttyniemi, E.; Salminen, S.; Nuutila, P.; Kalliokoski, K.K.; Hannukainen, J.C. Exercise Training Modulates GM Profile and Improves Endotoxemia. Med. Sci. Sports Exerc. 2020, 52, 94–104. [Google Scholar] [CrossRef]
- Silva, J.S.C.; Seguro, C.S.; Naves, M.M.V. GM and physical exercise in obesity and diabetes—A systematic review. Nutr. Metab Cardiovasc. Dis. 2022, 32, 863–877. [Google Scholar] [CrossRef]
- Bowyer, R.C.E. An association between chronic widespread pain and the gut microbiome. Rheumatology 2021, 60, 3727–3737. [Google Scholar] [CrossRef]
- Mariadason, J.M.; Catto-Smith, A.; Gibson, P.R. Modulation of distal colonic epithelial barrier function by dietary fibre in normal rats. Gut 1999, 44, 394–399. [Google Scholar] [CrossRef] [PubMed]
- Axelrod, C.L.; Brennan, C.J.; Cresci, G.; Paul, D.; Hull, M.; Fealy, C.E.; Kirwan, J.P. UCC118 supplementation reduces exercise-induced gastrointestinal permeability and remodels the gut microbiome in healthy humans. Physiol. Rep. 2019, 7, e14276. [Google Scholar] [CrossRef] [PubMed]
- Schreiber, C.; Tamir, S.; Golan, R.; Weinstein, A.; Weinstein, Y. The effect of probiotic supplementation on performance, inflammatory markers and gastro-intestinal symptoms in elite road cyclists. J. Int. Soc. Sports Nutr. 2021, 18, 36. [Google Scholar] [CrossRef] [PubMed]
- van Wijck, K.; Pennings, B.; van Bijnen, A.A.; Senden, J.M.G.; Buurman, W.A.; Dejong, C.H.C.; van Loon, L.J.C.; Lenaerts, K. Dietary protein digestion and absorption are impaired during acute postexercise recovery in young men. Am. J. Phyiol. Regul. Integr. Comp. Physiol. 2013, 304, R356–R361. [Google Scholar] [CrossRef] [PubMed]
- Costa, R.J.S.; Miall, A.; Khoo, A.; Rauch, C.; Snipe, R.; Camões-Costa, V.; Gibson, P. Gut-training: The impact of two weeks repetitive gut-challenge during exercise on gastrointestinal status, glucose availability, fuel kinetics, and running performance. Appl. Physiol. Nutri. Metab 2017, 42, 547–557. [Google Scholar] [CrossRef]
- Sperber, A.D. Review article: Epidemiology of IBS and other bowel disorders of gut-brain interaction (DGBI). Aliment. Pharmacol. Ther. 2021, 54 (Suppl. S1), S1–S11. [Google Scholar] [CrossRef]
- Chey, W.D.; Kurlander, J.; Eswaran, S. Irritable bowel syndrome: A clinical review. JAMA 2015, 313, 949–958. [Google Scholar] [CrossRef]
- Killian, L.A.; Lee, S.Y. Irritable bowel syndrome is underdiagnosed and ineffectively managed among endurance athletes. Appl. Physiol. Nutr. Metab 2019, 44, 1329–1338. [Google Scholar] [CrossRef]
- Hungin, A.P.S.; Chang, L.; Locke, G.R.; Dennis, E.H.; Barghout, V. Irritable bowel syndrome in the United States: Prevalence, symptom patterns and impact. Aliment. Pharmacol. Ther. 2005, 21, 1365–1375. [Google Scholar] [CrossRef]
- ter Steege, R.W.; Van der Palen, J.; Kolkman, J.J. Prevalence of gastrointestinal complaints in runners competing in a long-distance run: An internet-based observational study in 1281 subjects. Scand. J. Gastroenterol. 2008, 43, 1477–1482. [Google Scholar] [CrossRef]
- Pfeiffer, B.; Stellingwerff, T.; Hodgson, A.B.; Randell, R.; Pöttgen, K.; Res, P.; Jeukendrup, A.E. Nutritional intake and gastrointestinal problems during competitive endurance events. Med. Sci. Sports Exerc. 2012, 44, 344–351. [Google Scholar] [CrossRef] [PubMed]
- Peters, H.P.; Bos, M.; Seebregts, L.; Akkermans, L.M.; van Berge Henegouwen, G.P.; Bol, E.; Mosterd, W.L.; de Vries, W.R. Gastrointestinal symptoms in longdistance runners, cyclists, and triathletes: Prevalence, medication, and etiology. Am. J. Gastroenterol. 1999, 94, 1570–1581. [Google Scholar] [CrossRef] [PubMed]
- Zachwieja, J.J.; Costill, D.L.; Widrick, J.J.; Anderson, D.E.; McConell, G.K. Effects of drink carbonation in the gastric emptying characteristics of water and flavored water. Int. J. Sport Nutr. 1991, 1, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Horn, M.K.; Osterberg, K.L.; Stofan, J.R.; Zachwieja, J.J.; Horswill, C.A.; Passe, D.H.; Murray, R. Gastrointestinal discomfort during intermittent high-intensity exercise: Effect of carbohydrate-electrolyte beverage. Int. J. Sport Nutr. Exerc. Metab 2004, 14, 673–683. [Google Scholar] [CrossRef] [PubMed]
- Carrio, I.; Estorch, M.; Serra-Grima, R.; Ginjaume, M.; Notivol, R.; Calabuig, R.; Vilardell, F. Gastric emptying in marathon runners. Gut 1989, 30, 152–155. [Google Scholar] [CrossRef]
- Morton, D.P.; Callister, R. Factors influencing exercise-related transient abdominal pain. Med. Sci. Sports Exerc. 2002, 34, 745–749. [Google Scholar] [CrossRef]
- van Nieuwenhoven, M.A.; Brouns, F.; Kovacs, E.M. The effect of two sports drinks and water on GI complaints and performance during an 18-km run. Int. J. Sports Med. 2005, 26, 281–285. [Google Scholar] [CrossRef]
- de Oliveira, E.P.; Burini, R.C.; Jeukendrup, A. Gastrointestinal complaints during exercise: Prevalence, etiology, and nutritional recommendations. Sports Med. 2014, 44 (Suppl. S1), S79–S85. [Google Scholar] [CrossRef]
- ter Steege, R.W.; Kolkman, J.J.; Huisman, A.B.; Geelkerken, R.H. Gastrointestinal ischaemia during physical exertion as a cause of gastrointestinal symptoms. Ned. Tijdschr. Geneeskd. 2008, 152, 1805–1808. [Google Scholar]
- Gil, S.M.; Yazaki, E.; Evans, D.F. Aetiology of running-related gastrointestinal dysfunction: How far is the finishing line? Sports Med. 1998, 26, 365–378. [Google Scholar] [CrossRef]
- Parnell, J.A.; Wagner-Jones, K.; Madden, R.F.; Erdman, K.A. Dietary restrictions in endurance runners to mitigate exercise-induced gastrointestinal symptoms. J. Int. Soc. Sports Nutr. 2020, 17, 32. [Google Scholar] [CrossRef]
- Riddoch, C.; Trinick, T. Gastrointestinal disturbances in marathon runners. Br. J. Sports Med. 1988, 22, 71–74. [Google Scholar] [CrossRef]
- Miall, A.; Khoo, A.; Rauch, C.; Snipe, R.M.J.; Camões-Costa, V.L.; Gibson, P.R.; Costa, R.J.S. Two weeks of repetitive gut-challenge reduce exercise-associated gastrointestinal symptoms and malabsorption. Scand. J. Med. Sci. Sport 2018, 28, 630–640. [Google Scholar] [CrossRef]
- ten Haaf, D.S.M.; van der Worp, M.P.; Groenewoud, H.M.M.; Leij-Halfwerk, S.; der Sanden, M.W.G.N.-V.; Verbeek, A.L.M.; Staal, J.B. Nutritional indicators for gastrointestinal symptoms in female runners: The “Marikenloop study”. BMJ Open 2014, 4, e005780. [Google Scholar] [CrossRef]
- Fossati, C.; Torre, G.; Vasta, S.; Giombini, A.; Quaranta, F.; Papalia, R.; Pigozzi, F. Physical Exercise and Mental Health: The Routes of a Reciprocal Relation. Int. J. Environ. Res. Public Health 2021, 18, 12364. [Google Scholar] [CrossRef]
- Chang, L. The role of stress on physiological responses and clinical symptoms in irritable bowel syndrome. Gastroenterology 2011, 140, 761–765. [Google Scholar] [CrossRef]
- Haug, T.T.; Mykletun, A.; Dahl, A.A. Are anxiety and depression related to gastrointestinal symptoms in the general population? Scand. J. Gastroenterol. 2002, 37, 294–298. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.B. Perceived life stress and anxiety correlate with chronic gastrointestinal symptoms in runners. J. Sports Sci. 2018, 36, 1713–1719. [Google Scholar] [CrossRef] [PubMed]
- Kostic-Vucicevic, M.; Marinkovi, D.; Dikic, N.; Stojmenovic, T.; Andjelkovic, M.; Nikolic, I.; Vukasinovic-Vesic, M.; Malic, T. Is there connection between food intolerance and sports performance in elite athletes? Br. J. Sports Med. 2016, 50, A20. [Google Scholar] [CrossRef]
- Fedewa, A.; Rao, S.S. Dietary fructose intolerance, fructan intolerance and FODMAPs. Curr. Gastroenterol. Rep. 2014, 16, 370. [Google Scholar] [CrossRef]
- Tiller, N.B.; Roberts, J.D.; Beasley, L.; Chapman, S.; Pinto, J.M.; Smith, L.; Wiffin, M.; Russell, M.; Sparks, S.A.; Duckworth, L.; et al. International Society of Sports Nutrition Position Stand: Nutritional considerations for single-stage ultra-marathon training and racing. J. Int. Soc. Sports Nutr. 2019, 16, 50. [Google Scholar] [CrossRef]
- Patel, R.K.; Brouner, J.; Spendiff, O. Dark chocolate supplementation reduces the oxygen cost of moderate intensity cycling. J. Int. Soc. Sports Nutr. 2015, 12, 47. [Google Scholar] [CrossRef] [PubMed]
- Muller-Lissner, S.A.; Kaatz, V.; Brandt, W.; Keller, J.; Layer, P. The perceived effect of various foods and beverages on stool consistency. Eur. J. Gastroenterol. Hepatol. 2005, 17, 109–112. [Google Scholar] [CrossRef]
- Thomas, D.T.; Erdman, K.A.; Burke, L.M. Position of the Academy of Nutrition and Dietetics, Dietitians of Canada, and the American College of Sports Medicine: Nutrition and athletic performance. J. Acad. Nutr. Diet 2016, 116, 501–528. [Google Scholar] [CrossRef]
- Wilson, P.B. Dietary and non dietary correlates of gastrointestinal distress during the cycle and run of a triathlon. Eur. J. Sport Sci. 2016, 16, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Spriet, L.L. Exercise and sport performance with low doses of caffeine. Sports Med. 2014, 44 (Suppl. S2), S175–S184. [Google Scholar] [CrossRef]
- Gunja, N.; Brown, J.A. Energy drinks: Health risks and toxicity. Med. J. Aust. 2012, 196, 46–49. [Google Scholar] [CrossRef] [PubMed]
- Murray, R. Training the gut for competition. Curr. Sports Med. Rep. 2006, 5, 161–164. [Google Scholar] [CrossRef] [PubMed]
- Cox, G.R.; Clark, S.A.; Cox, A.J.; Halson, S.L.; Hargreaves, M.; Hawley, J.A.; Jeacocke, N.; Snow, R.J.; Yeo, W.K.; Burk, L.M. Daily training with high carbohydrate availability increases exogenous carbohydrate oxidation during endurance cycling. J. Appl. Physiol. 2010, 109, 126–134. [Google Scholar] [CrossRef]
- Arribalzaga, S.; Viribay, A.; Calleja-González, J.; Fernández-Lázaro, D.; Castañeda-Babarro, A.; Mielgo-Ayuso, J. Relationship of Carbohydrate Intake during a Single-Stage One-Day Ultra-Trail Race with Fatigue Outcomes and Gastrointestinal Problems: A Systematic Review. Int. J. Environ. Res. Public Health 2021, 18, 5737. [Google Scholar] [CrossRef]
- Rowe, J.T.; King, R.F.G.J.; King, A.J.; Morrison, D.J.; Preston, T.; Wilson, O.J.; O’Hara, J.P. Glucose and Fructose Hydrogel Enhances Running Performance, Exogenous Carbohydrate Oxidation, and Gastrointestinal Tolerance. Med. Sci. Sports Exerc. 2022, 54, 129–140. [Google Scholar] [CrossRef]
- Costa, R.J.S.; Hoffman, M.D.; Stellingwerff, T. Considerations for ultra-endurance activities: Part 1—nutrition. Res. Sports Med. 2019, 27, 166–181. [Google Scholar] [CrossRef] [PubMed]
- Scrivin, R.; Costa, R.J.S.; Pelly, F.; Lis, D.; Slater, G. An exploratory study of the management strategies reported by endurance athletes with exercise-associated gastrointestinal symptoms. Front. Nutr. 2022, 9, 1003445. [Google Scholar] [CrossRef] [PubMed]
- Trapp, D.; Knez, W.; Sinclair, W. Could a vegetarian diet reduce exercise-induced oxidative stress? A review of the literature. J. Sports Sci. 2010, 28, 1261–1268. [Google Scholar] [CrossRef]
- Burke, L.M. Ketogenic low-CHO, high-fat diet: The future of elite endurance sport? J. Physiol. 2021, 599, 819–843. [Google Scholar] [CrossRef]
- Somerville, V.S.; Braakhuis, A.J.; Hopkins, W.G. Effect of flavonoids on upper respiratory tract infections and immune function: A systematic review and meta-analysis. Adv. Nutr. 2016, 7, 488–497. [Google Scholar] [CrossRef]
- Devrim-Lanpir, A.; Hill, L.; Knechtle, B. Efficacy of Popular Diets Applied by Endurance Athletes on Sports Performance: Beneficial or Detrimental? A Narrative Review. Nutrients 2021, 13, 491. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Shan, W.; Zhu, F.; Wu, J.; Wang, Q. Ketone bodies in neurological diseases: Focus on neuroprotection and underlying mechanisms. Front. Neurol. 2019, 10, 585. [Google Scholar] [CrossRef] [PubMed]
- Dearlove, D.J.; Harrison, O.K.; Hodson, L.; Jefferson, A.; Clarke, K.; Cox, P.J. The Effect of Blood Ketone Concentration and Exercise Intensity on Exogenous Ketone Oxidation Rates in Athletes. Med. Sci. Sports Exerc. 2021, 53, 505–516. [Google Scholar] [CrossRef] [PubMed]
- Cox, P.J.; Kirk, T.; Ashmore, T.; Willerton, K.; Evans, R.; Smith, A.; Murray, A.J.; Stubbs, B.; West, J.; McLure, S.W.; et al. Nutritional Ketosis Alters Fuel Preference and Thereby Endurance Performance in Athletes. Cell. Metab 2016, 24, 256–268. [Google Scholar] [CrossRef]
- Lis, D.M.; Kings, D.; Larson-Meyer, D.E. Dietary practices adopted by track-andfield athletes: Gluten-free, low FODMAP, vegetarian, and fasting. Int. J. Sport Nutr. Exerc. Metab 2019, 29, 236–245. [Google Scholar] [CrossRef] [PubMed]
- Lis, D.M.; Stellingwerff, T.; Shing, C.M.; Ahuja, K.D.; Fell, J.W. Exploring the popularity, experiences, and beliefs surrounding gluten-free diets in nonceliac athletes. Int. J. Sport Nutr. Exerc. Metab 2015, 25, 37–45. [Google Scholar] [CrossRef] [PubMed]
- Erdman, K.A.; Jones, K.W.; Madden, R.F.; Gammack, N.; Parnell, J.A. Dietary Patterns in Runners with Gastrointestinal Disorders. Nutrients 2021, 13, 448. [Google Scholar] [CrossRef]
- Lis, D.M.; Stellingwerff, T.; Kitic, C.M.; Fell, J.W.; Ahuja, K.D.K. Low FODMAP: A preliminary strategy to reduce gastrointestinal distress in athletes. Med. Sci. Sports Exerc. 2018, 50, 116–123. [Google Scholar] [CrossRef] [PubMed]
- Fritscher, K.; Harris, M.; Meyer, N. Survey of Gluten-free diet and its effects on performance amongst cyclists. In Proceedings of the American College of Sports Medicine, Indianapolis, ID, USA, 28 May 2012–1 June 2013. [Google Scholar]
- Lis, D.; Stellingwerff, T.; Kitic, C.M.; Ahuja, K.D.; Fell, J. No Effects of a Short-Term Gluten-free Diet on Performance in Nonceliac Athletes. Med. Sci. Sports Exerc. 2015, 47, 2563–2570. [Google Scholar] [CrossRef] [PubMed]
- Halson, S.L.; Martin, D.T. Lying to win-placebos and sport science. Int. J. Sports Physiol. Perform. 2013, 8, 597–599. [Google Scholar] [CrossRef]
- Aziz, I.; Hadjivassiliou, M.; Sanders, D.S. The spectrum of noncoeliac gluten sensitivity. Nat. Rev. Gastroenterol. Hepatol. 2015, 12, 516–526. [Google Scholar] [CrossRef]
- Killian, L.A.; Muir, J.G.; Barrett, J.S.; Burd, N.A.; Lee, S.Y. High Fermentable Oligosaccharides, Disaccharides, Monosaccharides, and Polyols (FODMAP) Consumption Among Endurance Athletes and Relationship to Gastrointestinal Symptoms. Front. Nutr. 2021, 8, 637160. [Google Scholar] [CrossRef]
- Biesiekierski, J.L.; Rosella, O.; Rose, R.; Liels, K.; Barrett, J.S.; Shepherd, S.J.; Gibson, P.R.; Muir, J.G. Quantification of fructans, galacto-oligosacharides and other short-chain carbohydrates in processed grains and cereals. J. Hum. Nutr. Diet 2011, 24, 154–176. [Google Scholar] [CrossRef]
- Whelan, K.; Abrahmsohn, O.; David, G.J.; Staudacher, H.; Irving, P.; Lomer, M.C.; Ellis, P.R. Fructan content of commonly consumed wheat, rye and gluten-free breads. Int. J. Food Sci. Nutr. 2011, 62, 498–503. [Google Scholar] [CrossRef]
- Skodje, G.I.; Sarna, V.K.; Minelle, I.H.; Rolfsen, K.L.; Muir, J.G.; Gibson, P.R.; Veierød, M.B.; Henriksen, C.; Lundin, K.E.A. Fructan, rather than gluten, induces symptoms in patients with self-reported non-celiac gluten sensitivity. Gastroenterology 2018, 154, 529–539. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P.R.; Muir, J.G.; Newnham, E.D. Other dietary confounders: FODMAPS. Dig. Dis. 2015, 33, 269–276. [Google Scholar] [CrossRef] [PubMed]
- De Giorgio, R.; Volta, U.; Gibson, P.R. Sensitivity to wheat, gluten and FODMAPs in IBS: Facts or fiction? Gut 2015, 65, 169–178. [Google Scholar] [CrossRef]
- Lis, D.M. Exit Gluten-Free and Enter Low FODMAPs: A Novel Dietary Strategy to Reduce Gastrointestinal Symptoms in Athletes. Sports Med. 2019, 49 (Suppl. S1), 87–97. [Google Scholar] [CrossRef] [PubMed]
- Staudacher, H.M.; Lomer, M.C.E.; Farquharson, F.M.; Louis, P.; Fava, F.; Franciosi, E.; Whelan, K. A diet low in FODMAPs reduces symptoms in patients with irritable bowel syndrome and a probiotic restores bifidobacterium species: A randomized controlled trial. Gastroenterology 2017, 153, 936–947. [Google Scholar] [CrossRef]
- Black, C.J.; Staudacher, H.M.; Ford, A.C. Efficacy of a low FODMAP diet in irritable bowel syndrome: Systematic review and network meta-analysis. Gut 2022, 71, 1117–1126. [Google Scholar] [CrossRef]
- Killian, L.; Lee, S.-Y. Nutritional Habits and FODMAPs in Relation to Gastrointestinal Issues of Endurance Athletes. Gastroenterology 2017, 152, S751. [Google Scholar] [CrossRef]
- Staudacher, H.M.; Irving, P.M.; Lomer, M.C.; Whelan, K. Mechanisms and efficacy of dietary FODMAP restriction in IBS. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 256–266. [Google Scholar] [CrossRef]
- Böhn, L.; Störsrud, S.; Liljebo, T.; Collin, L.; Lindfors, P.; Törnblom, H.; Simrén, M. Diet Low in FODMAPs Reduces Symptoms of Irritable Bowel Syndrome asWell as Traditional Dietary Advice: A Randomized Controlled Trial. Gastroenterology 2015, 149, 1399–1407.e2. [Google Scholar] [CrossRef]
- Barrett, J.S. How to institute the low-FODMAP diet. J. Gastroenterol. Hepatol. 2017, 32, 8–10. [Google Scholar] [CrossRef]
- Gaskell, S.K.; Taylor, B.; Muir, J.; Costa, R.J.S. Impact of 24-h high and low fermentable oligo-, di-, monosaccharide, and polyol diets on markers of exercise-induced gastrointestinal syndrome in response to exertional heat stress. Appl. Physiol. Nutr. Metab. 2020, 45, 569–580. [Google Scholar] [CrossRef] [PubMed]
- Gaskell, S.K.; Costa, R.J.S. Applying a Low-FODMAP dietary intervention to a female ultraendurance runner with irritable bowel syndrome during a multistage ultramarathon. Int. J. Sport Nutr. Exerc. Metab. 2019, 29, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Wiffin, M.; Smith, L.; Antonio, J.; Johnstone, J.; Beasley, L.; Roberts, J. Effect of a short-term low fermentable oligiosaccharide, disaccharide, monosaccharide and polyol (FODMAP) diet on exercise-related gastrointestinal symptoms. J. Int. Soc. Sports Nutr. 2019, 16, 1. [Google Scholar] [CrossRef] [PubMed]
- Schumann, D.; Klose, P.; Lauche, R.; Dobos, G.; Langhorst, J.; Cramer, H. Low fermentable, oligo-, di-, mono-saccharides and polyol diet in the treatment of irritable bowel syndrome: A systematic review and meta-analysis. Nutrition 2018, 45, 24–31. [Google Scholar] [CrossRef]
- Craddock, J.C.; Neale, E.P.; Peoples, G.E.; Probst, Y.C. Plant-based eating patterns and endurance performance: A focus on inflammation, oxidative stress and immune responses. Nutr. Bull. 2020, 45, 123–132. [Google Scholar] [CrossRef]
- Saturni, L.; Ferretti, G.; Bacchetti, T. The gluten-free diet: Safety and nutritional quality. Nutrients 2010, 2, 16–34. [Google Scholar] [CrossRef]
- Stevens, L.; Rashid, M. Gluten-free and regular foods: A cost comparison. Can. J. Diet Pract. Res. 2008, 69, 147–150. [Google Scholar] [CrossRef]
| GI Symptoms | Nutritional and Behavior Strategies |
|---|---|
Upper-GI symptoms
| Avoid high-calorie and fatty meals three hours prior to exercise [50,53] Avoid hypertonic fluids during exercise [50,53] |
Lower-GI symptoms
| Defecate prior to exercise to prevent the urge to defecate during exercise [71] Drink small amounts of hypotonic carbohydrate fluids to prevent the risk of osmotic diarrhea [72] Avoid hypertonic solutions, including fatty, high-calorie meals, or high-glycemic-index foods three hours before exercising and during exercise [73] Train the gut to ingest a high amount of carbohydrates and fluids before competition [70] Avoidance of food and fluid intake at least two hours prior to exercise [3] |
| Side stitch or ETAP | Wait 2–3 h before exercising after a meal or drink [74] Take small amounts of drink during exercise and abstain from hypertonic fluids [3] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ribichini, E.; Scalese, G.; Cesarini, A.; Mocci, C.; Pallotta, N.; Severi, C.; Corazziari, E.S. Exercise-Induced Gastrointestinal Symptoms in Endurance Sports: A Review of Pathophysiology, Symptoms, and Nutritional Management. Dietetics 2023, 2, 289-307. https://doi.org/10.3390/dietetics2030021
Ribichini E, Scalese G, Cesarini A, Mocci C, Pallotta N, Severi C, Corazziari ES. Exercise-Induced Gastrointestinal Symptoms in Endurance Sports: A Review of Pathophysiology, Symptoms, and Nutritional Management. Dietetics. 2023; 2(3):289-307. https://doi.org/10.3390/dietetics2030021
Chicago/Turabian StyleRibichini, Emanuela, Giulia Scalese, Alessandra Cesarini, Chiara Mocci, Nadia Pallotta, Carola Severi, and Enrico Stefano Corazziari. 2023. "Exercise-Induced Gastrointestinal Symptoms in Endurance Sports: A Review of Pathophysiology, Symptoms, and Nutritional Management" Dietetics 2, no. 3: 289-307. https://doi.org/10.3390/dietetics2030021
APA StyleRibichini, E., Scalese, G., Cesarini, A., Mocci, C., Pallotta, N., Severi, C., & Corazziari, E. S. (2023). Exercise-Induced Gastrointestinal Symptoms in Endurance Sports: A Review of Pathophysiology, Symptoms, and Nutritional Management. Dietetics, 2(3), 289-307. https://doi.org/10.3390/dietetics2030021






