Mechanistic Insights into the Metabolic Pathways Using High-Resolution Mass Spectrometry and Predictive Models in Pancreatic β-Cell Lines (β-TC-6) †
Abstract
:1. Background
2. Objectives
3. Methods
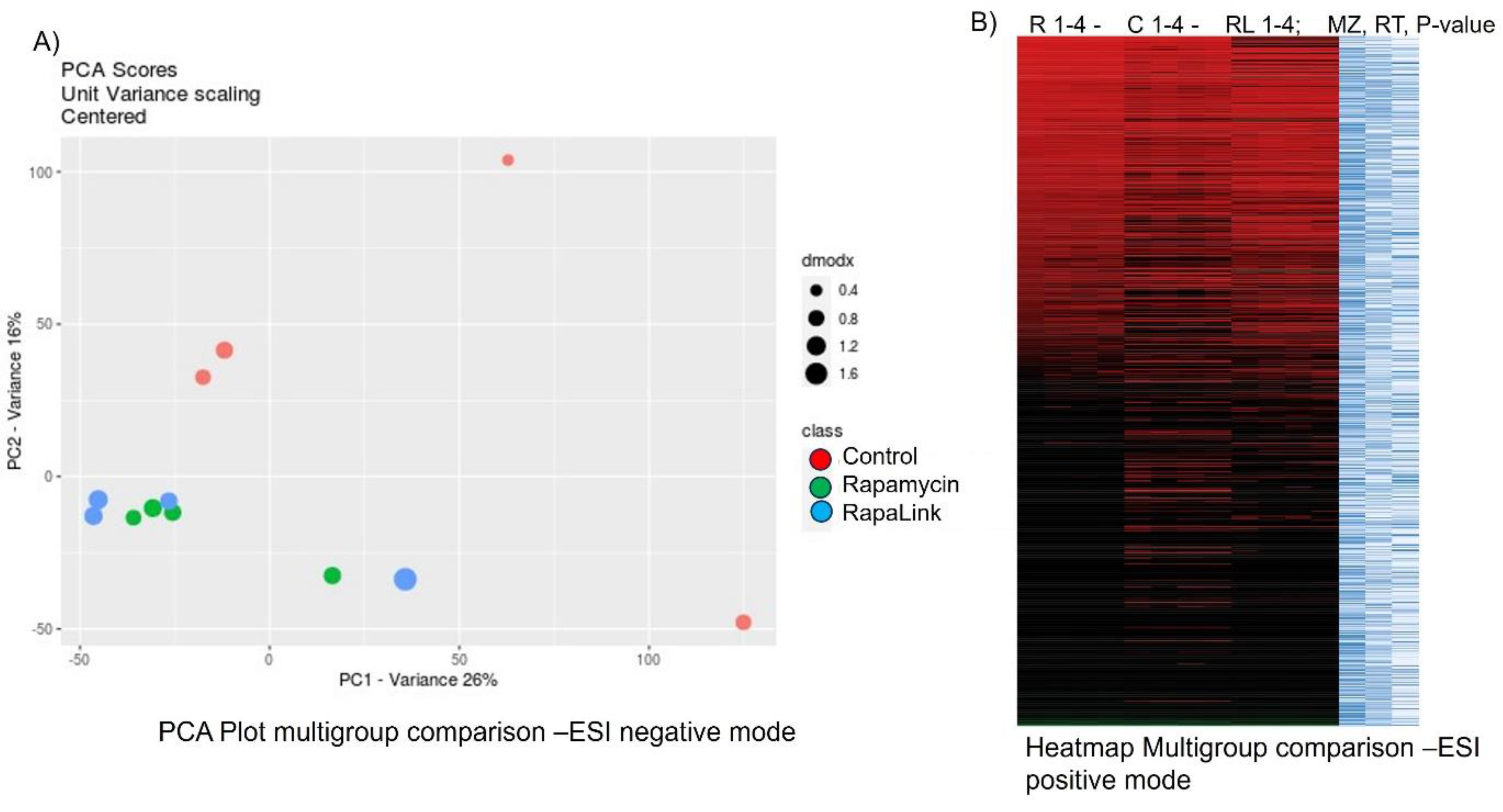
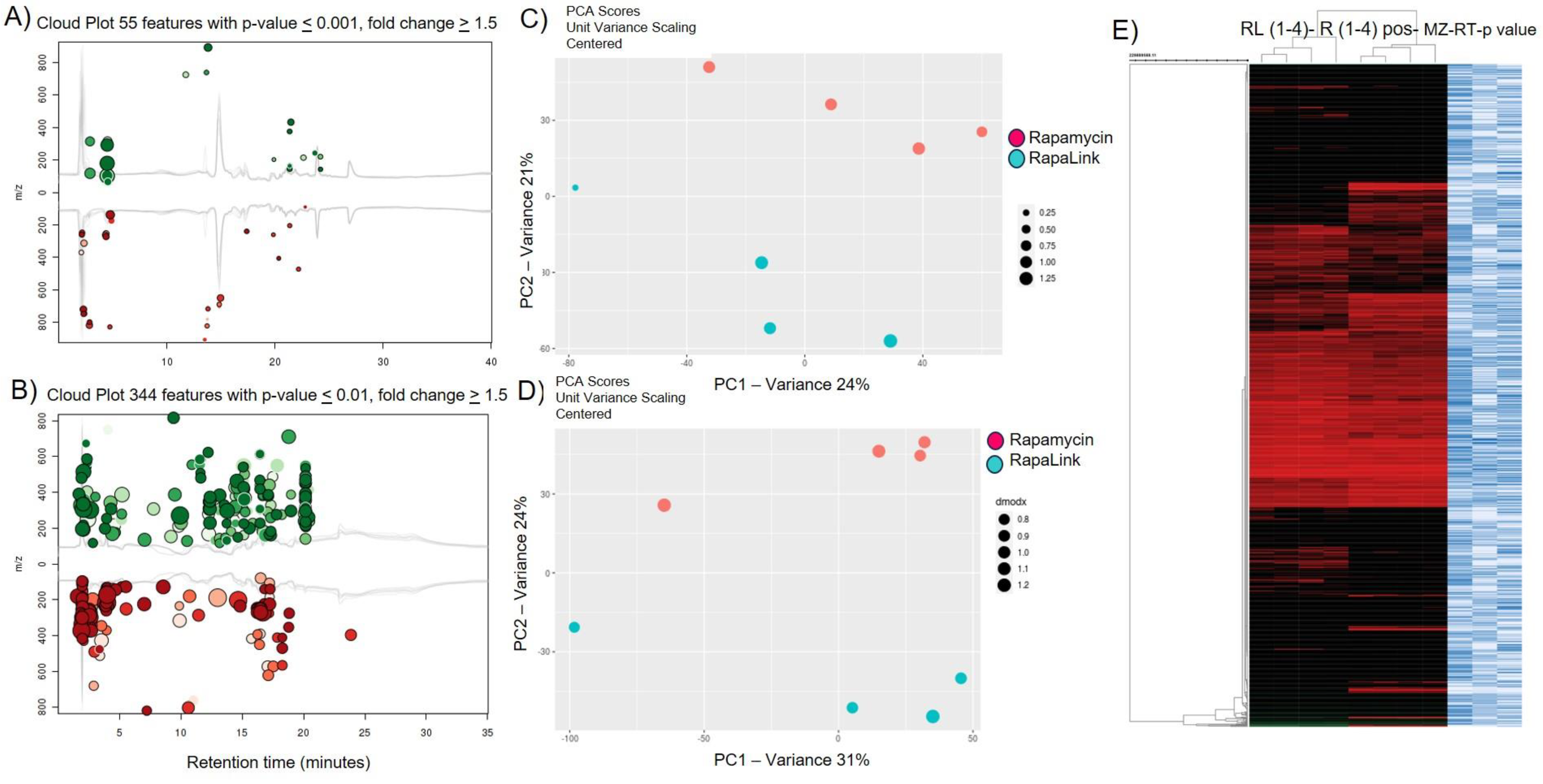
4. Results
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Battaglioni, S.; Benjamin, D.; Walchli, M.; Maier, T.; Hall, M.N. mTOR substrate phosphorylation in growth control. Cell 2022, 185, 1814–1836. [Google Scholar] [CrossRef]
- Soliman, G.A. The role of the mechanistic target of rapamycin (mTOR) complexes signaling in the immune responses. Nutrients 2013, 5, 2231–2257. [Google Scholar] [CrossRef] [PubMed]
- Soliman, G.A.; Abzalimov, R.R.; He, Y. mTORC1 and mTORC2 Complexes Regulate the Untargeted Metabolomics and Amino Acid Metabolites Profile through Mitochondrial Bioenergetic Functions in Pancreatic Beta Cells. Nutrients 2022, 14, 3022. [Google Scholar] [CrossRef]
- Domingo-Almenara, X.; Siuzdak, G. Metabolomics Data Processing Using XCMS. Methods Mol. Biol. 2020, 2104, 11–24. [Google Scholar] [PubMed]
- Domingo-Almenara, X.; Montenegro-Burke, J.R.; Ivanisevic, J.; Thomas, A.; Sidibe, J.; Teav, T.; Guijas, C.; Aisporna, A.E.; Rinehart, D.; Hoang, L.; et al. XCMS-MRM and METLIN-MRM: A cloud library and public resource for targeted analysis of small molecules. Nat. Methods 2018, 15, 681–684. [Google Scholar] [CrossRef] [PubMed]
- Gowda, H.; Ivanisevic, J.; Johnson, C.H.; Kurczy, M.E.; Benton, H.P.; Rinehart, D.; Nguyen, T.; Ray, J.; Kuehl, J.; Arevalo, B.; et al. Interactive XCMS Online: Simplifying advanced metabolomic data processing and subsequent statistical analyses. Anal. Chem. 2014, 86, 6931–6939. [Google Scholar] [CrossRef] [PubMed]
- Huan, T.; Forsberg, E.M.; Rinehart, D.; Johnson, C.H.; Ivanisevic, J.; Benton, H.P.; Fang, M.; Aisporna, A.; Hilmers, B.; Poole, F.L.; et al. Systems biology guided by XCMS Online metabolomics. Nat. Methods 2017, 14, 461–462. [Google Scholar] [CrossRef] [PubMed]
| Pathways | Overlap_Size | Pathway_Size | p-Value (raw) | p-Value |
|---|---|---|---|---|
| Pentose phosphate pathway (non-oxidative branch | 2 | 3 | 0.023334 | 0.0 |
| Dopamine degradation | 2 | 7 | 0.12893 | 0.00004 |
| Ubiquinol-8-biosynthesis (eukaryotic) | 2 | 11 | 0.2682 | 0.00074 |
| Arsenate detoxification I (glutaredoxin) | 1 | 4 | 0.32129 | 1 |
| Pathways | Overlap_Size | Pathway_Size | p-Value (raw) | p-Value |
|---|---|---|---|---|
| Nicotine degradation II | 4 | 10 | 0.0587 | 0.00289 |
| Phospholipases | 2 | 2 | 0.02513 | 0.0053 |
| Glycine degradation (creatine biosynthesis) | 3 | 7 | 0.08438 | 0.0058 |
| Creatine biosynthesis | 3 | 8 | 0.11986 | 0.00875 |
| D-myo-inositol (3,4,5,6)-tetrakisphosphate biosynthesis | 2 | 3 | 0.06755 | 0.01111 |
| D-myo-inositol (1,3,4)-trisphosphate biosynthesis | 2 | 3 | 0.06755 | 0.0111 |
| Glutathione biosynthesis | 2 | 3 | 0.06755 | 0.0111 |
| 1D-myo-inositol hexakisphosphate biosynthesis II (mammalian) | 2 | 4 | 0.12123 | 0.02083 |
| L-dopachrome biosynthesis | 2 | 4 | 0.12123 | 0.02083 |
| tRNA charging pathway | 3 | 12 | 0.29586 | 0.03833 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soliman, G.A.; He, Y.; Abzalimov, R. Mechanistic Insights into the Metabolic Pathways Using High-Resolution Mass Spectrometry and Predictive Models in Pancreatic β-Cell Lines (β-TC-6). Biol. Life Sci. Forum 2023, 29, 16. https://doi.org/10.3390/IECN2023-15878
Soliman GA, He Y, Abzalimov R. Mechanistic Insights into the Metabolic Pathways Using High-Resolution Mass Spectrometry and Predictive Models in Pancreatic β-Cell Lines (β-TC-6). Biology and Life Sciences Forum. 2023; 29(1):16. https://doi.org/10.3390/IECN2023-15878
Chicago/Turabian StyleSoliman, Ghada A., Ye He, and Rinat Abzalimov. 2023. "Mechanistic Insights into the Metabolic Pathways Using High-Resolution Mass Spectrometry and Predictive Models in Pancreatic β-Cell Lines (β-TC-6)" Biology and Life Sciences Forum 29, no. 1: 16. https://doi.org/10.3390/IECN2023-15878
APA StyleSoliman, G. A., He, Y., & Abzalimov, R. (2023). Mechanistic Insights into the Metabolic Pathways Using High-Resolution Mass Spectrometry and Predictive Models in Pancreatic β-Cell Lines (β-TC-6). Biology and Life Sciences Forum, 29(1), 16. https://doi.org/10.3390/IECN2023-15878







