Effect of Salt Stress on Pollen Tube Growth in Two Medicago truncatula Ecotypes †
Abstract
:1. Introduction
2. Methods
2.1. Pollen Condition of Germination
2.2. Data Analysis
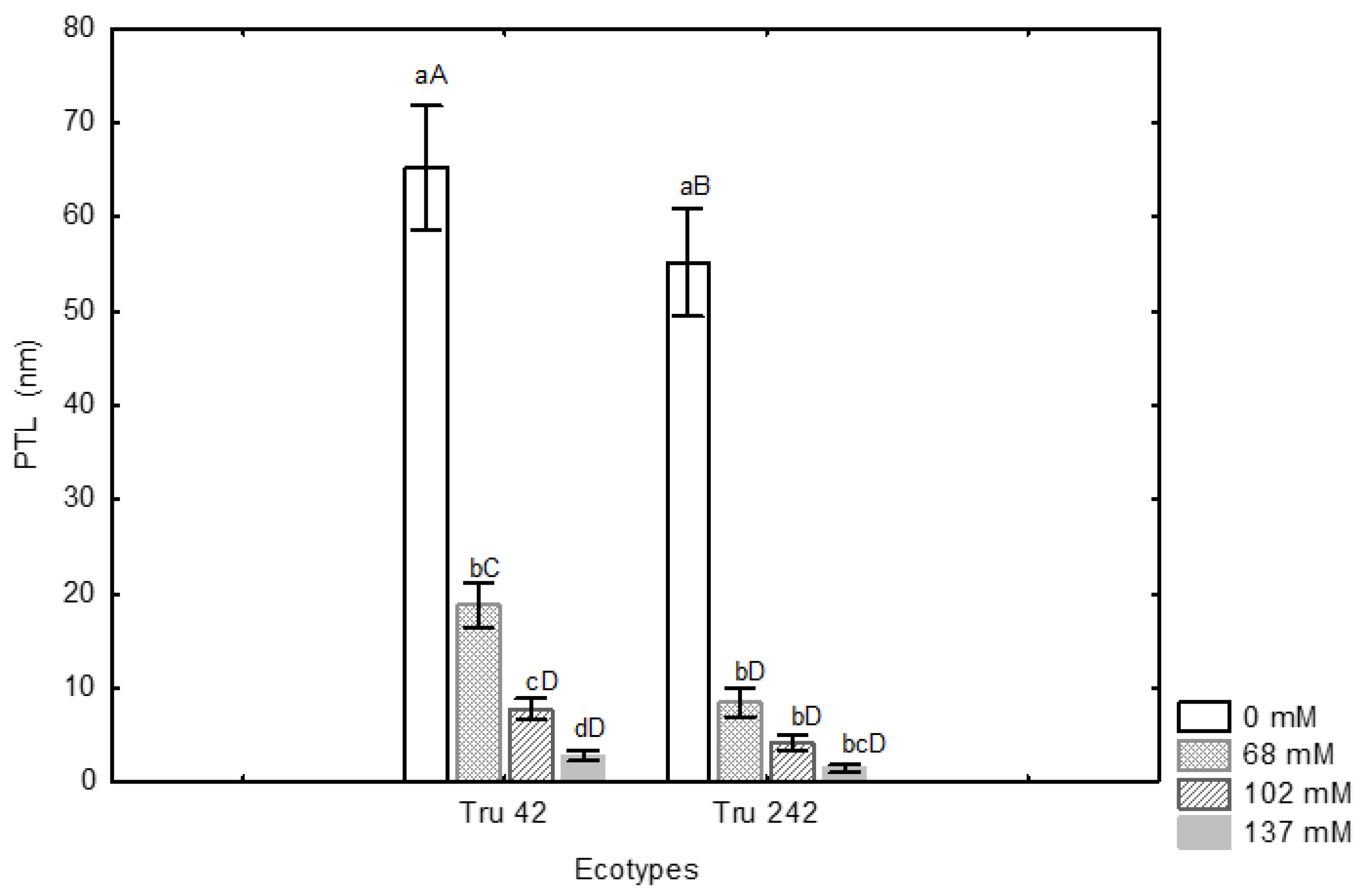
3. Results and Discussion
4. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hasanuzzaman, M.; Fujita, M. Plant Responses and Tolerance to Salt Stress: Physiological and Molecular Interventions. Int. J. Mol. Sci. 2022, 23, 4810. [Google Scholar] [CrossRef] [PubMed]
- Corwin, D.L.; Yemoto, K. Salinity: Electrical Conductivity and Total Dissolved Solids. Soil Sci. Soc. Am. J. 2020, 84, 1442–1461. [Google Scholar] [CrossRef]
- Laval-Martin, D.; Mazliak, P. Physiologie Végétale, Volume 1; Front Cover., Danielle; Plant Physiology; Hermann: Paris, France, 1995; 539p. [Google Scholar]
- Johnson, M.A.; Preuss, D. Plotting a course: Multiple signals guide pollen tubes to their targets. Dev. Cell 2002, 2, 273–281. [Google Scholar] [CrossRef] [PubMed]
- Fyad-Lameche, F.Z.; Nourredine, Y.; Fairouz, L.; Larbi, A. Tolérance au froid chez la tomate (Lycopersicum esculentum Mill.). Etude comparée du développement du gamétophyte mâle et du sporophyte. Acta Bot. Gallica 2007, 154, 251–263. [Google Scholar] [CrossRef]
- Vienne, D.E.D. Variabilité chez une espèce tétraploïde: Analyse iso-enzymatique et biométrique du pollen de quelques familles apparentées de luzerne. Ann. Amélior. Plantes 1979, 28, 289–307. [Google Scholar]
- Bassetti, P.; Westgate, M.E. Senescence and receptivity of maize silks. Crop Sci. 1993, 33, 275–278. [Google Scholar] [CrossRef]
- Khatun, S.; Flowers, T.J. Effects of salinity on seed set in rice. Plant Cell Environ. 1995, 18, 61–67. [Google Scholar] [CrossRef]
- Ghanem, M.E.; van Elteren, J.; Albacete, A.; Quinet, M.; Martínez-Andújar, C.; Kinet, J.M.; Pérez-Alfocea, F.; Lutts, S. Impact of salinity on early reproductive physiology of tomato (Solanum lycopersicum) in relation to a heterogeneous distribution of toxic ions in flower organs. Funct. Plant Biol. 2009, 36, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Aref, F.; Ebrahimi-Rad, H. Physiological characterization of rice under salinity stress during vegetative and reproductive stages. Indian J. Sci. Technol. 2012, 5, 2578–2586. [Google Scholar] [CrossRef]
- Amouri, A.A.; Fyad-Lamèche, F.Z. Comparative analysis of salinity tolerance of the male gametophyte and the sporophyte in Medicago at the germination stage. Acta Bot. Malacit. 2012, 37, 93–102. [Google Scholar] [CrossRef]
- Kiełkowska, A.; Grzebelus, E.; Lis-Krzyścin, A.; Maćkowska, K. Application of the salt stress to the protoplast cultures of the carrot (Daucus carota L.) and evaluation of the response of regenerants to soil salinity. Plant Cell Tissue Organ Cult. 2019, 137, 379–395. [Google Scholar] [CrossRef]
- Irakoze, W.; Prodjinoto, H.; Nijimbere, S.; Rufyikiri, G.; Lutts, S. NaCl and Na2SO4 Salinities Have Different Impact on Photosynthesis and Yield-Related Parameters in Rice (Oryza sativa L.). Agronomy 2020, 10, 864. [Google Scholar] [CrossRef]
- Ravikumar, R.L.; Patil, B.S.; Salimath, P.M. Drought tolerance in sorghum by pollen selection using osmotic stress. Euphytica 2003, 133, 371–376. [Google Scholar] [CrossRef]
- Li, Y.Q.; Zhang, H.Q.; Pierson, E.S.; Huang, F.Y.; Linskens, H.F.; Hepler, P.K.; Cresti, M. Enforced growth-rate fluctuation causes pectin ring formation in the cell wall of Lilium longiflorum pollen tubes. Planta 1996, 200, 41–49. [Google Scholar] [CrossRef]
- Biagini, G.; Faleri, C.; Cresti, M.; Cai, G. Sucrose concentration in the growth medium affects the cell wall composition of tobacco pollen tubes. Plant Reprod. 2014, 27, 129–144. [Google Scholar] [CrossRef] [PubMed]
- Habibi, N.; Fakoor, M.Y.; Faqiri, S.M.; Sharaf, Z.; Hotak, M.S.; Danishyar, N.; Haris, M.M.; Osmani, K.S.; Shinohara, T.; Terada, N.; et al. Enhancing salinity tolerance in tomatoes at the reproductive stage by increasing pollen viability. Revis Bionatura 2023, 8, 25. [Google Scholar] [CrossRef]
- Attioli, R.; Palombi, N.; Funck, D.; Trovato, M. Proline Accumulation in Pollen Grains as Potential Target for Improved Yield Stability Under Salt Stress. Front. Plant Sci. 2020, 11, 582877. [Google Scholar] [CrossRef] [PubMed]

| Species | Ecotypes | Origin | Altitude (m) | Pluviometry (mm) | Degree of Tolerance |
|---|---|---|---|---|---|
| Medicago truncatula Gaertn. | Tru 42 | Fetzara (Annaba) 36°47′50.2′′ N 7°30′44.5′′ E | 100 | 660 | Tolerant |
| Tru 242 | Kaïs (Khenchela) 35°29′30.1′′ N 6°55′42.2′′ E | 980 | 450 | Sensitive |
| Source of Variation | ddl | F | P |
|---|---|---|---|
| Ecotypes (Ec) | 1 | 28.6 | 0.000 ** |
| Treatment (T) | 3 | 515.6 | 0.000 ** |
| Interaction (Ec xT) | 3 | 3.7 | 0.011 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amouri, A.A. Effect of Salt Stress on Pollen Tube Growth in Two Medicago truncatula Ecotypes. Biol. Life Sci. Forum 2023, 27, 40. https://doi.org/10.3390/IECAG2023-16381
Amouri AA. Effect of Salt Stress on Pollen Tube Growth in Two Medicago truncatula Ecotypes. Biology and Life Sciences Forum. 2023; 27(1):40. https://doi.org/10.3390/IECAG2023-16381
Chicago/Turabian StyleAmouri, Adel Amar. 2023. "Effect of Salt Stress on Pollen Tube Growth in Two Medicago truncatula Ecotypes" Biology and Life Sciences Forum 27, no. 1: 40. https://doi.org/10.3390/IECAG2023-16381
APA StyleAmouri, A. A. (2023). Effect of Salt Stress on Pollen Tube Growth in Two Medicago truncatula Ecotypes. Biology and Life Sciences Forum, 27(1), 40. https://doi.org/10.3390/IECAG2023-16381





