Effects of Salinity and Drought Stress on Seed Germination of Common Purslane (Portulaca oleracea) †
Abstract
:1. Introduction
2. Material and Methods
2.1. Seed Collection
2.2. Seed Germination and Treatments
2.3. Determination of Germination Parameters
2.4. Experimental Design and Data Analysis
3. Results and Discussions
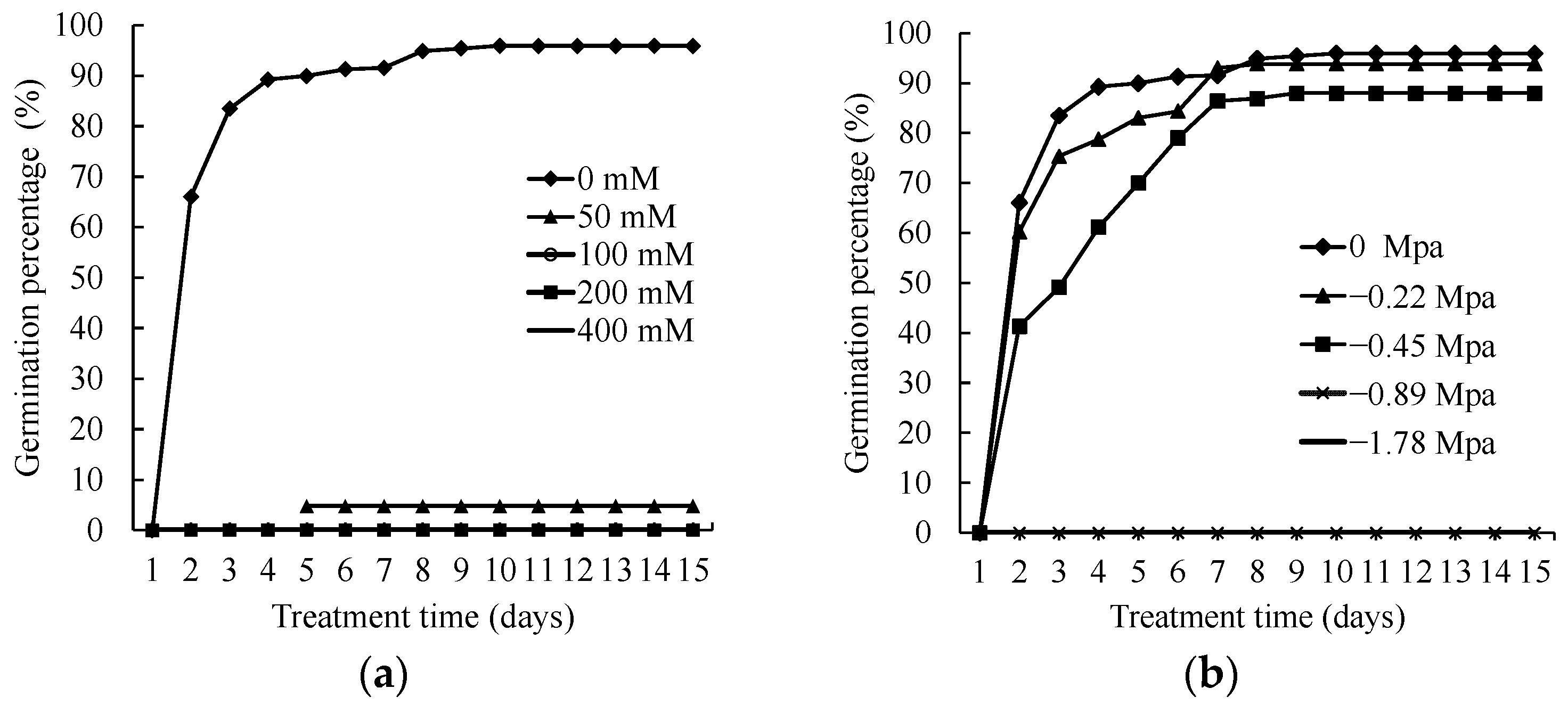
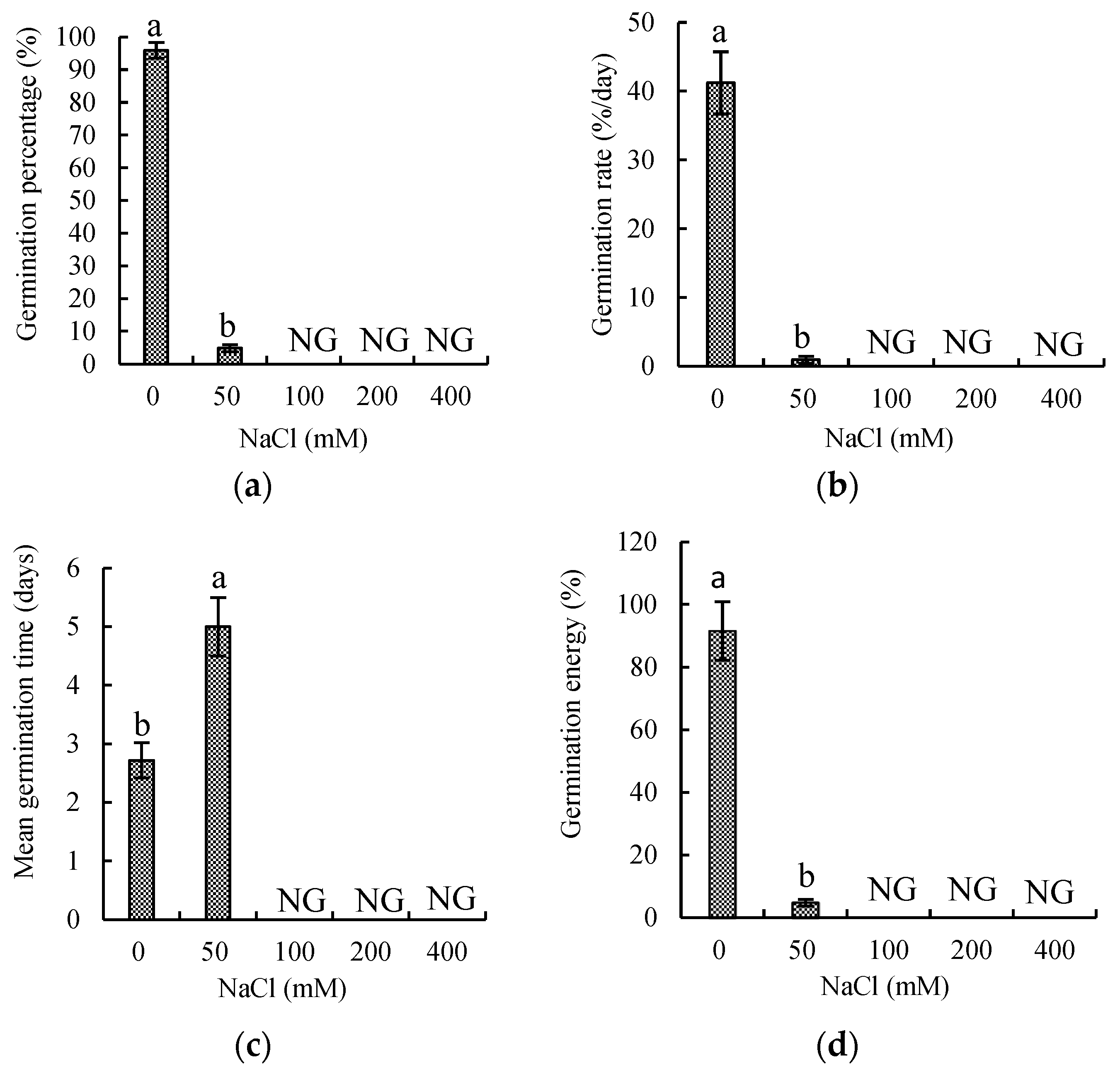
3.1. Effects of Salinity Stress on Germination of P. oleracea Seed
3.2. Effects of Drought Stress on Germination of P. oleracea Seed
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Athar, H.R.; Ashraf, M. Strategies for crop improvement against salinity and drought stress: An overview. In Salinity and Water Stress; Ashraf, M., Ozturk, M., Athar, H., Eds.; Springer: Dordrecht, The Netherlands, 2009; p. 44. [Google Scholar] [CrossRef]
- Shiade, S.R.G.; Boelt, B. Seed germination and seedling growth parameters in nine tall fescue varieties under salinity stress. Acta Agric. 2020, 70, 485–494. [Google Scholar] [CrossRef]
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef] [PubMed]
- Gheidary, S.; Akhzari, D.; Pessarakli, M. Effects of salinity, drought, and priming treatments on seed germination and growth parameters of Lathyrus sativus L. J. Plant Nutr. 2017, 40, 1507–1514. [Google Scholar] [CrossRef]
- Bewley, J.D.; Black, M. Seeds: Physiology of Development and Germination; Springer Science & Business Media: New York, NY, USA, 2015. [Google Scholar]
- Alam, M.A.; Juraimi, A.S.; Rafii, M.Y.; Hamid, A.A.; Aslani, F.; Alam, M.Z. Effects of salinity and salinity-induced augmented bioactive compounds in purslane (Portulaca oleracea L.) for possible economical use. Food Chem. 2015, 169, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Yazici, I.; Türkan, I.; Sekmen, A.H.; Demiral, T. Salinity tolerance of purslane (Portulaca oleracea L.) is achieved by enhanced antioxidative system, lower level of lipid peroxidation and proline accumulation. Environ. Exp. Bot. 2007, 61, 49–57. [Google Scholar] [CrossRef]
- Petrović, G.; Jovičić, D.; Nikolić, Z.; Tamindžić, G.; Ignjatov, M.; Milošević, D.; Milošević, B. Comparative study of drought and salt stress effects on germination and seedling growth of pea. Genet. Belgrade 2016, 48, 373–381. [Google Scholar] [CrossRef]
- Tran, Q.D.; Pham, C.A.; Nguyen, T.T.T.; Vo, C.T. Characteristics of seed morphology and germination of Launaea sarmentosa. J. Sci. Technol. Univ. Danang 2022, 21, 87–92. [Google Scholar]
- Kader, M.A. A comparison of seed germination calculation formulae and the associated interpretation of resulting data. J. Proceeding R. Soc. New South Wales 2005, 138, 65–75. [Google Scholar] [CrossRef]
- Bina, F.; Bostani, A. Effect of salinity (NaCl) stress on germination and early seedling growth of three medicinal plant species. Adv. Life Sci. 2017, 4, 77–83. [Google Scholar]
- Parvaneh, R.; Shahrokh, T.; Meysam, H.S. Studying of salinity stress effect on germination, proline, sugar, protein, lipid and chlorophyll content in purslane (Portulaca oleracea L.) leaves. J. Stress Physiol. Biochem. 2012, 8, 182–193. [Google Scholar]
- Zandi Esfahan, E.; Azarnivand, H. Effect of water stress on seed germination of Agropyron elongatum, Agropyron desertourm & Secale Montanum. Desert 2012, 17, 249–253. [Google Scholar]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pham, A.C.; Vo, T.C.; Vu, H.D.; Tran, D.Q. Effects of Salinity and Drought Stress on Seed Germination of Common Purslane (Portulaca oleracea). Biol. Life Sci. Forum 2023, 27, 31. https://doi.org/10.3390/IECAG2023-14974
Pham AC, Vo TC, Vu HD, Tran DQ. Effects of Salinity and Drought Stress on Seed Germination of Common Purslane (Portulaca oleracea). Biology and Life Sciences Forum. 2023; 27(1):31. https://doi.org/10.3390/IECAG2023-14974
Chicago/Turabian StylePham, Anh Cong, Tuan Chau Vo, Hoang Duc Vu, and Dan Quang Tran. 2023. "Effects of Salinity and Drought Stress on Seed Germination of Common Purslane (Portulaca oleracea)" Biology and Life Sciences Forum 27, no. 1: 31. https://doi.org/10.3390/IECAG2023-14974
APA StylePham, A. C., Vo, T. C., Vu, H. D., & Tran, D. Q. (2023). Effects of Salinity and Drought Stress on Seed Germination of Common Purslane (Portulaca oleracea). Biology and Life Sciences Forum, 27(1), 31. https://doi.org/10.3390/IECAG2023-14974





