Abstract
Patulin (PAT) is one of the most common mycotoxins produced by Penicillium and Aspergillus species and is often associated with fruits and fruit by-products, mostly apple derivatives, although it has been detected in infant food and cereals. This toxin has shown a mutagenic and carcinogenic effect. Thus, the development of rapid and accurate methods for PAT detection is of utmost importance. Currently, the most widely used methods for the analysis and detection of mycotoxins are based on chromatography, including liquid chromatography (HPLC) and gas chromatography coupled to a mass detector (GC–MS), since these techniques provide high precision, selectivity, and sensitivity. In this work we show the preliminary results in the development of a GC–MS method for the detection of PAT without derivatization. Usually, the detection of mycotoxins by GC–MS needs a derivatization of all non-volatile and polar compounds. This is one drawback of gas chromatography compared to the liquid chromatographic technique for the determination of mycotoxins. In this sense, the PAT monitoring method by GC–MS proposed here is an alternative and useful technique to maintain high-quality foodstuffs and to ensure food safety.
1. Introduction
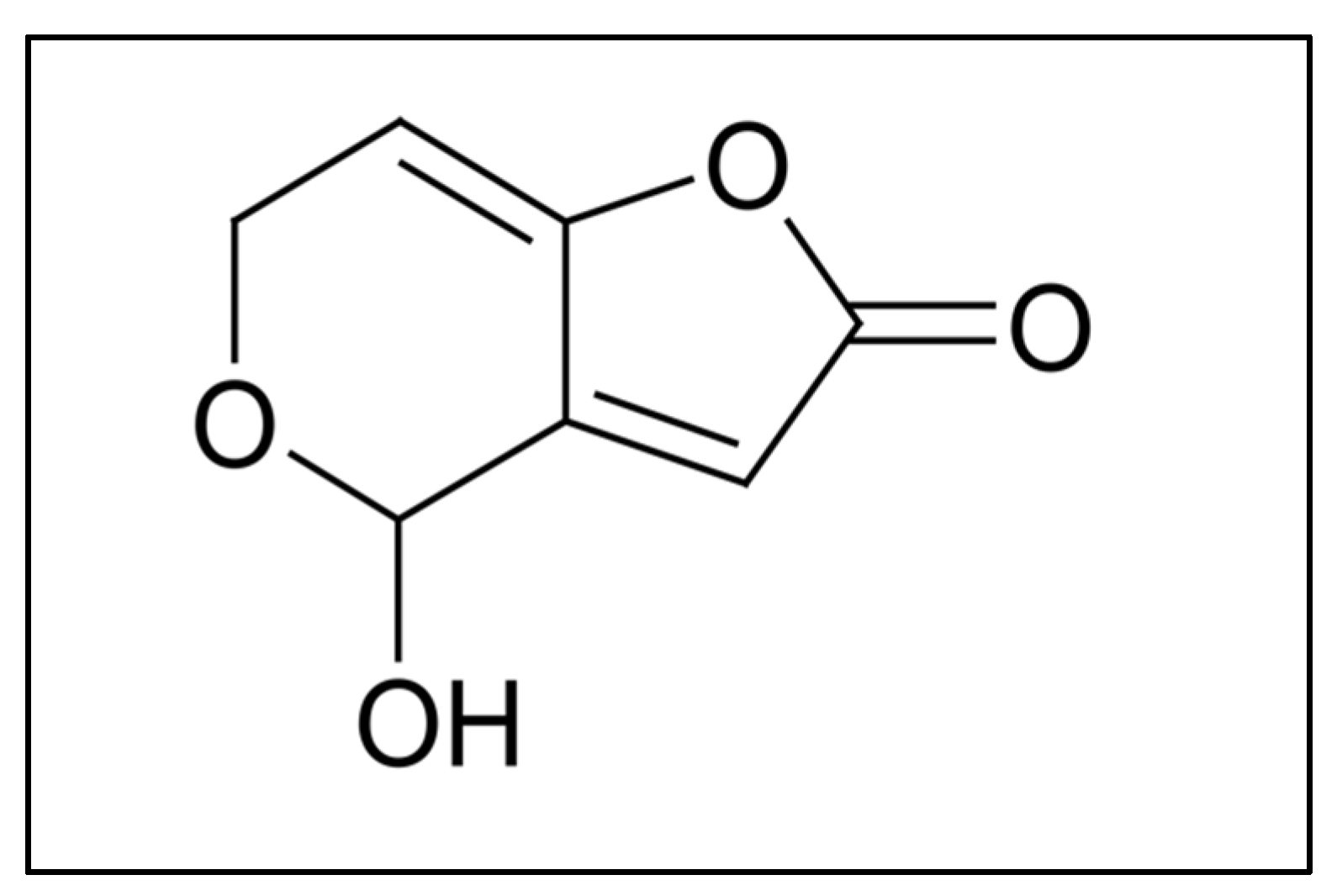
Food safety is a growing concern in our society due to the increase in chemical and biological contaminants in the environment. Among these contaminants, mycotoxins are highly toxic secondary metabolites of low molecular weight and volatility, synthesized by certain groups of fungi [1,2]. The contamination of agricultural and other food products with this type of toxins poses serious problems for both human and animal health [1]. Inadequate collection, drying, handling, packaging, storage, and distribution are some of the factors that contribute to mycotoxin contamination the most [1,3]. Among mycotoxins, patulin (PAT) is a highly polar lactone with low molecular weight (154.12 g/mol) (Figure 1). It is produced by many species of the genera Penicillium, Aspergillus and Byssochlamys, Penicillium expansum being one of the main producers and responsible for PAT contamination of apples and their derived products [2,4]. PAT is soluble in water and in various solvents such as ethanol, methanol, acetone, ethyl acetate, etc. It is slightly soluble in sulfuric acid and benzene, and stable under acidic conditions [1]. Compared to raw foods, the presence of PAT in processed foods is not frequent, since the clarification, filtration, and enzymatic treatment during juice processing and fermentation during winemaking significantly reduce its content [1,5]. Acute exposure to PAT can cause gastrointestinal symptoms including nausea, vomiting, ulcers, intestinal bleeding, and duodenal lesions, as well as impaired intestinal barrier function accompanied by kidney damage [2], while long-term (chronic) exposure includes neurotoxic, immunotoxic, immunosuppressive, genotoxic, teratogenic, and potentially carcinogenic effects [4]. In relation to the analytical determination, methods to confirm the presence of PAT usually include specific detection techniques, such as mass spectrometry (MS), previous separation by liquid chromatography (LC) or gas chromatography (GC). Today, with the majority of detection methods using the GC–MS methodology, PAT is detected as its trimethylsilyl derivative (TMS-patulin) [6,7,8]. In general, mycotoxins’ detection by GC implies their derivatization. However, the derivatization process takes time and leads to a delay in analysis, especially when a high number of food samples have to be monitored. In this context, the objective of this work is to collect information regarding the available chromatography methods for PAT detection using GC and go further in the use of this technique to detect PAT without derivatization, so that it will be the starting point for the development of a method which could be improved in speed.
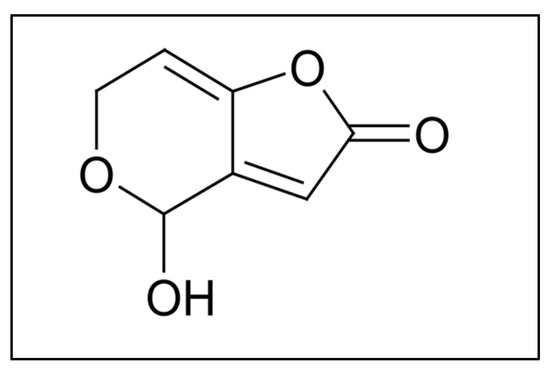
Figure 1.
Patulin structure.
2. Material and Methods
2.1. Chemicals and Materials
Dichloromethane (CH2Cl2), acetonitrile (MeCN), and methanol (MeOH) were purchased from Merck (Darmstadt, Germany). All chemical and reagents were of analytical grade (GC). The standard of PAT was obtained from Sigma-Aldrich (5 mg). A stock solution was prepared by dissolving 5 mg of PAT in 1 mL of pure methanol, obtaining a 5 mg/mL solution. Then, this stock solution was used in further dilutions in dichloromethane, acetonitrile, and methanol in order to obtain the appropriate working standard solutions (10 μg/mL and 100 μg/mL). All solutions were kept at −20 °C before analysis.
2.2. GC–MS Equipment and Methodology
The analysis of patulin was performed on the Thermo Scientific Trace 1300 GC system coupled with a Thermo Scientific ISQ 7000 single quadrupole mass spectrometer. The Trace 1300 GC module is integrated with the Chromeleon chromatography data system software. The separation was achieved on a HP-88 30 m × 0.25 mm × 0.2 μm capillary column from—Agilent. Helium was used as the carrier gas with a constant flow of 1 mL/min. The oven temperature program was initially set at 80 °C for 1 min, and increased to 245 °C at 60 °C/min. After 3 min of hold time at 245 °C, the temperature was increased to 260 °C at 3 °C/min and then increased to 270 °C at 10 °C/min. Finally, 270 °C was held for 10 min. Patulin was dissolved in dichloromethane, acetonitrile, and methanol; the injection volume was 1 μL and the solvent delay was 2 min. The method operated in split injection mode (1:50) with pulse injection at 35 psi for 0.5 min. The ion source temperature was 250 °C and the transfer line temperature was 250 °C.
3. Results and Discussion
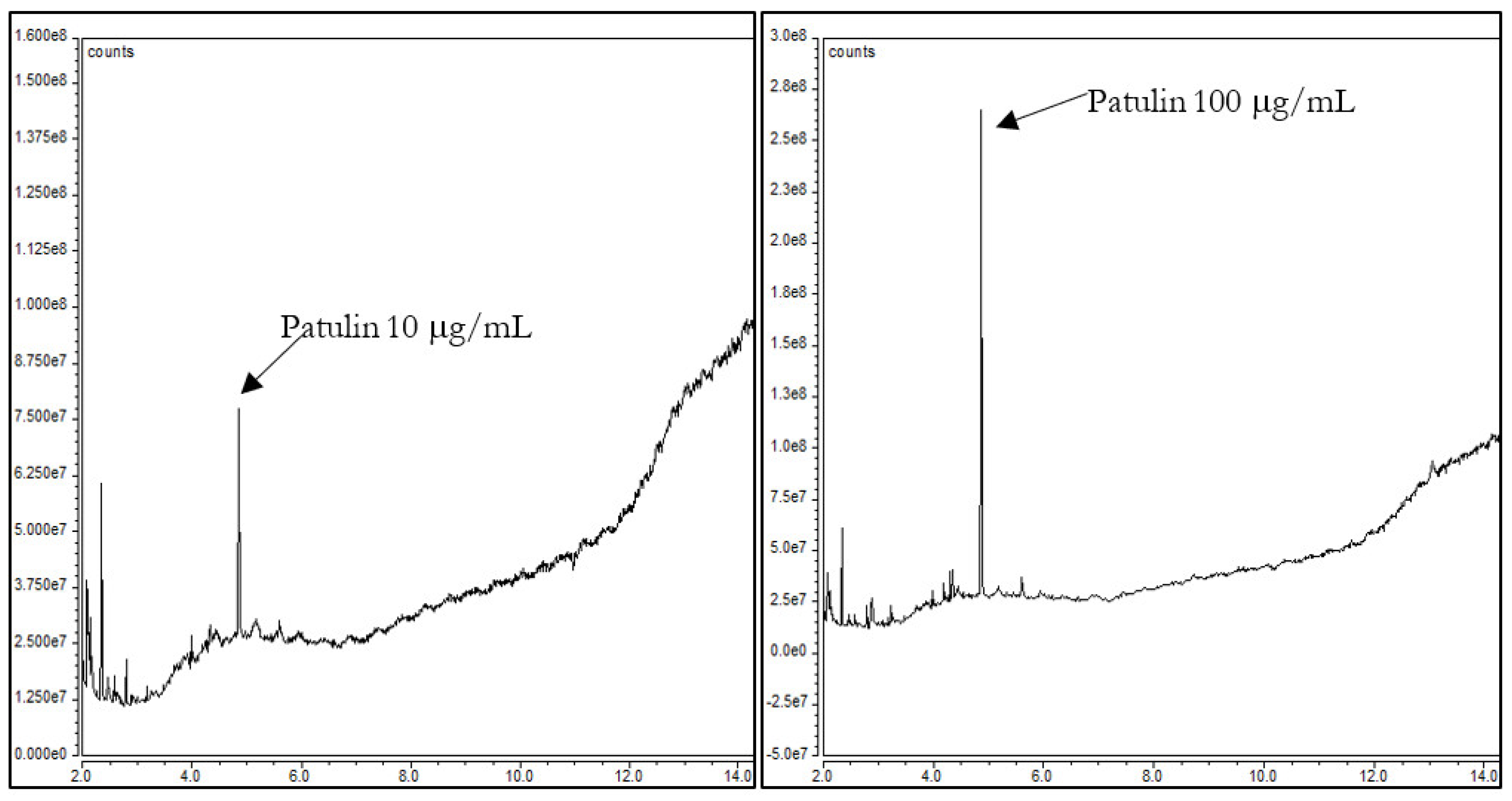
Although PAT detection by GC is usually performed by derivatization, the bibliography includes some works in which mycotoxins are not derivatized. To our knowledge, there are just two studies in which GC–MS with negative ion chemical ionization allowed the detection of underivatized PAT [9,10]. PAT without derivatization was successfully analyzed and this fact encouraged us to work in this line and develop a method for rapid detection of PAT without derivatization. For the method development, we first evaluated the influence of the sample solvent on the chromatogram signal. For this, three commonly used solvents were selected and compared; they were dichloromethane, acetonitrile, and methanol [5,6,11,12]. For each solvent, two working standard solutions of 10 μg/mL and 100 μg/mL were prepared and injected. Out of the three solvents used, dichloromethane produced the higher signal of PAT (Figure 2).
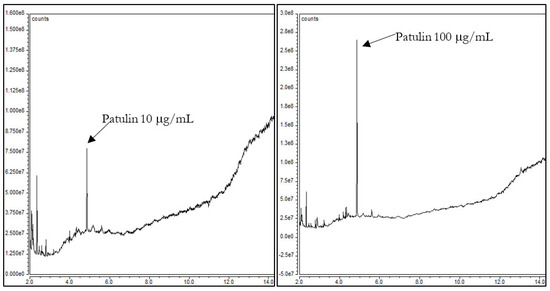
Figure 2.
Standard of patulin at 10 μg/mL and 100 μg/mL in dichloromethane without derivatization.
In this work, we showed that PAT can be analyzed without derivatization using a HP-88 30 m × 0.25 mm × 0.2 μm capillary column from Agilent. Different columns have been used to detect PAT. Table 1 shows the available GC–MS methodology for PAT. The most used column for identifying PAT in apple juice is the capillary column HP-5ms [8,9,11,13] with limits of detection (LOD) between 0.4 and 3 µg/L. Another study employs three capillary columns of moderate polarity, DB-17 (0.25 μm i.d. × 30 m, 0.25 μm J&W), DB-1701 (0.25 μm i.d. × 30 m, 0.25 μm, J&W), and BPX-35 (0.22 μm i.d. × 25 m, 0.25 μm, BSE), obtaining better LODs of 0.1 µg/L [7]. Future research will test the method with different food samples which will determine the sensitivity of our methodology. Most available methods for PAT are focused on apple and apple derivatives, although in the last decade, the GC method has been developed for the detection of PAT and other mycotoxins in cereals, wheat, rice, maize, spelt, oat, soy, and tapioca [14,15].

Table 1.
Available methodology for patulin detection by GC–MS.
4. Conclusions
PAT is one of the most toxic secondary metabolites produced by fungi. Their occurrence is growing significantly on vegetables and fruits like apples, pears, and grapes [1]. Toxicology data revealed that acute PAT intoxication results in ulceration, agitation, convulsions, oedema, vomiting, and DNA damage in the brain, kidney and liver. Thus, the EU Commission has established maximum levels for PAT in some foods including fruit juices, spirit drinks and cider (50 µg/kg), solid apple products like compote and puree (25 µg/kg), and products for infants and young children like apple juice and solid apple (10 µg/kg) [2], and FAO/WHO have suggested a provisional maximum tolerable daily intake (PMTDI) of 0.4 mg/kg body weight/day. Available data show that at least 30% of apple juices may be contaminated with PAT at levels higher than 50 mg/L and that consumers ingest as much as 250 mL of apple juice per day [1]. PAT has also been found in seafood such as shellfish and cereals, which is a concern for several sectors of the food industry. Thus, the monitoring of the contamination of PAT in several kinds of foods is of the utmost importance to guarantee the products’ safety and quality. In this context, we have found that the detection of PAT by GC–MS without derivatization can be a solid analytical procedure for their qualitative detection.
Author Contributions
Conceptualization, P.O.; methodology, A.A.M.; formal analysis, P.O.; investigation, A.T., F.C., M.A.P. and R.P.-G.; resources, J.S.-G.; data curation, P.O.; writing—original draft preparation, A.A.M.; writing—review and editing, P.O.; visualization, P.O.; supervision, P.O.; project admin-istration, J.S.-G. and P.O.; funding acquisition, J.S.-G. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by HORIZON-CL6-2022-FARM2FORK-01-09 grant number 101084344 (WHEATBIOME). Funder under Food, Bioeconomy Natural Resources, Agriculture and Environment.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
The authors are grateful to WHEATBIOME under grant agreement ID: 101084344 (HORIZON-CL6-2022-FARM2FORK-01) and to Bio Based Industries Joint Undertaking (JU) under grant agreement No 888003 UP4HEALTH Project (H2020-BBI-JTI-2019). The JU receives support from the European Union’s Horizon 2020 research and innovation program and the Bio Based Industries Consortium. The project SYSTEMIC Knowledge hub on Nutrition and Food Security has received funding from national research funding parties in Belgium (FWO), France (INRA), Germany (BLE), Italy (MIPAAF), Latvia (IZM), Norway (RCN), Portugal (FCT), and Spain (AEI), in a joint action of JPI HDHL, JPI-OCEANS, and FACCE-JPI launched in 2019 under the ERA-NET ERA-HDHL (n° 696295).
Conflicts of Interest
The authors declare no conflict of interest.
References
- Mahato, D.K.; Kamle, M.; Sharma, B.; Pandhi, S.; Devi, S.; Dhawan, K.; Selvakumar, R.; Mishra, D.; Kumar, A.; Arora, S.; et al. Patulin in food: A mycotoxin concern for human health and its management strategies. Toxicon 2021, 198, 12–23. [Google Scholar] [CrossRef] [PubMed]
- Vidal, A.; Ouhibi, S.; Ghali, R.; Hedhili, A.; De Saeger, S.; Boevre, M. De The mycotoxin patulin: An updated short review on occurrence, toxicity and analytical challenges. Food Chem. Toxicol. 2019, 129, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Bhat, R.; Rai, R.V.; Karim, A.A. Mycotoxins in Food and Feed: Present Status and Future Concerns. Compr. Rev. Food Sci. Food Saf. 2010, 9, 57–81. [Google Scholar] [CrossRef] [PubMed]
- Moake, M.M.; Padilla-Zakour, O.I.; Worobo, R.W. Comprehensive review of patulin control methods in foods. Compr. Rev. Food Sci. Food Saf. 2005, 4, 8–21. [Google Scholar] [CrossRef] [PubMed]
- Cunha, S.C.; Faria, M.A.; Fernandes, J.O. Determination of patulin in apple and quince products by GC-MS using 13C5-7 patulin as internal standard. Food Chem. 2009, 115, 352–359. [Google Scholar] [CrossRef]
- Rupp, H.S.; Turnipseed, S.B. Confirmation of patulin and 5-hydroxymethylfurfural in apple juice by gas chromatography/mass spectrometry. J. AOAC Int. 2000, 83, 612–620. [Google Scholar] [CrossRef] [PubMed]
- Tabata, S.; Iida, K.; Suzuki, J.; Kimura, K.; Ibe, A.; Saito, K. A quantification and confirmation method of patulin in apple juice by GC/MS. J. Food Hyg. Soc. Japan 2004, 45, 245–249. [Google Scholar] [CrossRef] [PubMed]
- Xiao, H.; Fu, S. A sensitive gas chromatography-mass spectrometry method for the determination of patulin in apple juice. J. AOAC Int. 2012, 95, 1709–1712. [Google Scholar] [CrossRef] [PubMed]
- Llovera, M.; Viladrich, R.; Torres, M.; Canela, R. Analysis of underivatizated patulin by a GC-MS technique. J. Food Prot. 1999, 62, 202–205. [Google Scholar] [CrossRef] [PubMed]
- Roach, J.A.; White, K.D.; Trucksess, M.W.; Thomas, F.S. Capillary Gas Chromatography/Mass Spectrometry with Chemical Ionization and Negative Ion Detection for Confirmation of Identity of Patulin in Apple Juice. J. AOAC Int. 2000, 83, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Marks, H.S. Rapid gas chromatography/mass spectrometry determination and confirmation of patulin in apple juice. J. AOAC Int. 2007, 90, 879–883. [Google Scholar] [PubMed]
- Moukas, A.; Panagiotopoulou, V.; Markaki, P. Determination of patulin in fruit juices using HPLC-DAD and GC-MSD techniques. Food Chem. 2008, 109, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Kharandi, N.; Babri, M.; Azad, J. A novel method for determination of patulin in apple juices by GC–MS. Food Chem. 2013, 141, 1619–1623. [Google Scholar] [CrossRef]
- Rodríguez-Carrasco, Y.; Berrada, H.; Font, G.; Mañes, J. Multi-mycotoxin analysis in wheat semolina using an acetonitrile-based extraction procedure and gas chromatography-tandem mass spectrometry. J. Chromatogr. A 2012, 1270, 28–40. [Google Scholar] [CrossRef]
- Rodríguez-Carrasco, Y.; Moltó, J.C.; Berrada, H.; Mañes, J. A survey of trichothecenes, zearalenone and patulin in milled grain-based products using GC-MS/MS. Food Chem. 2014, 146, 212–219. [Google Scholar] [CrossRef] [PubMed]
- Marsol-Vall, A.; Balcells, M.; Eras, J.; Canela-Garayoa, R. A rapid gas chromatographic injection-port derivatization method for the tandem mass spectrometric determination of patulin and 5-hydroxymethylfurfural in fruit juices. J. Chromatogr. A 2016, 1453, 99–104. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).