Abstract
Diabetes is a long-term (chronic), challenging lifestyle (metabolic) disorder in which the body cannot regulate the amount of sugar in the blood and majorly affecting endocrine system and metabolic functions. Its complications majorly affect the neurons (as developing neuropathy), kidney (as nephropathy), and eye (as retinopathy). Diabetes also results in other associated problems, such as diabetic foot ulcers, sexual dysfunction, heart diseases etc. In the traditional medicines, the search for effective hypoglycemic agents is a continuous and challenging approach. Plant-derived bioactives, including alkaloids, phenols, glycosides, anthocyanins, flavonoids, saponins, tannins, polysaccharides, and terpenes, have been established to target cellular and molecular mechanisms involved in carbohydrate metabolism. Numerous diabetic patients select the herbal or traditional medicine system as an alternative therapeutic approach along with the mainstream anti-diabetic drugs. However, due to restrictive hurdles related to solubility and bioavailability, the bioactive compound cannot deliver the requisite effect. In this review, information is presented concerning well researched phytoconstituents established as potential hypoglycemic agents for the prevention and treatment of diabetes and its associated disorders having restricted solubility and bioavailability related issues. This information can be further utilized in future to develop several value added formulation and nutraceutical products to achieve the desired safety and efficacy for the prevention and treatment of diabetes and its related diseases.
1. Introduction
The World Health Organization (WHO) declares Diabetes mellitus as one of the commonest types of metabolic disorders with multiple etiologies involving chronic hyperglycemia, abnormalities in the metabolic rate of carbohydrates, proteins and fats, as well as fluctuations in the secretion of insulin and insulin activity [1]. Diabetes is a chronic condition that contributes to abnormalities in blood sugar levels due to the dysregulation of insulin, leading to insufficient insulin production or insufficient use of insulin due to inefficient pancreatic function [2]. As per the available literature it is categorized as one of the largest epidemics disease/disorders worldwide. According to the International Diabetes Federation (IDF), Southeast Asia, approximately, 537 million people were affected with diabetes worldwide, 90 million people in Southeast Asia, and 774 million cases were reported in India in 2022, mostly among the adult age group (8.3%) [3].
According to the latest predictions, diabetes will threaten 9.3% (463 million populations under the ages of 20 to 79 years) by 2019 and increase to 10.2% (578 million) through 2030 and 700 million through 2045 [3,4]. Throughout the world, people living with diabetes experience a severe, long-term condition that has a significant impact on their lives, families, and societies. According to estimates, it caused four million deaths globally in 2017. Diabetes accounted for USD 727 billion loss in global health expenditures in 2017 [5]. Among diabetes, type-2 Diabetes mellitus (T2DM) accounts for 90% of the total number of cases, which is continues to rise daily due to overburden of associated factors such as aging, rapid urbanization and obesity [6]. In recent years, T2DM was reported to be growing in young adults and matter of concern. Currently, there is no clear explanation for the rising number of diabetic patients. The increased prevalence of diabetes is also due to improved survival rates (in some populations) due to early detection, better diabetes management, and a reduction in premature death rate. Diabetes reports numerous side effects due to the abnormal functioning of insulin or absence of insulin such as coronary artery disease, retinopathy, foot ulcers, nephropathy and neuropathy [7]. Interestingly, the utilization of ethanobotanicals and traditional remedies for separation of several phytoconstituents has been currently well established for the treatment and management of diabetes and its related complications. Therefore, several such phytoconstituents exhibiting potent anti-hyperglycemic action, reviewed in the present communication, might play a significant role in the prevention and treatment of diabetes and its associated complications along with their molecular mechanism.
2. Classification of Diabetes mellitus
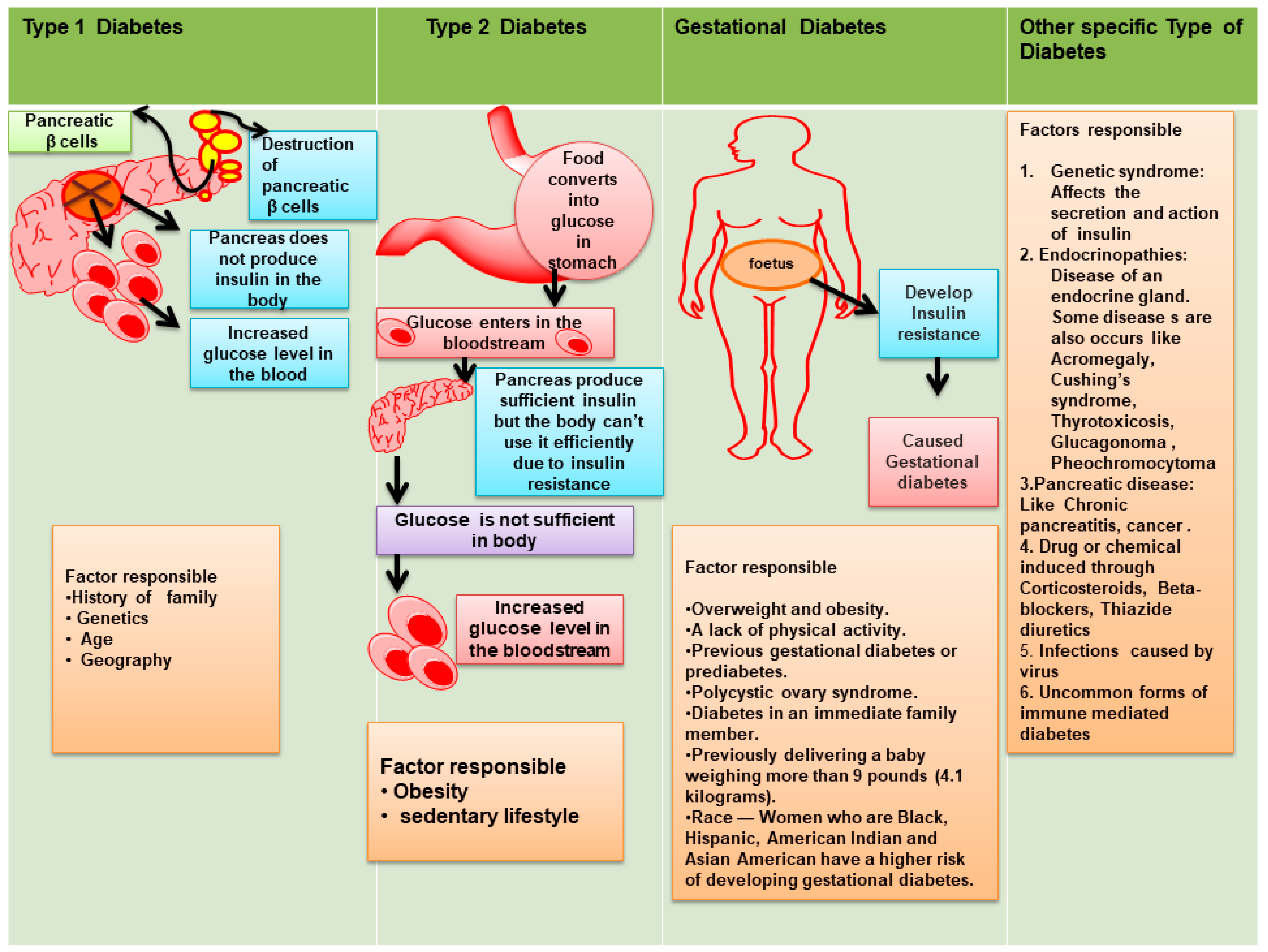
Earlier Diabetes were classified as: Insulin Dependent Diabetes mellitus (IDDM) and Non-Insulin Dependent Diabetes mellitus (NIDDM), respectively. Currently, diabetes has been replaced by a new classification system, shown in Figure 1.
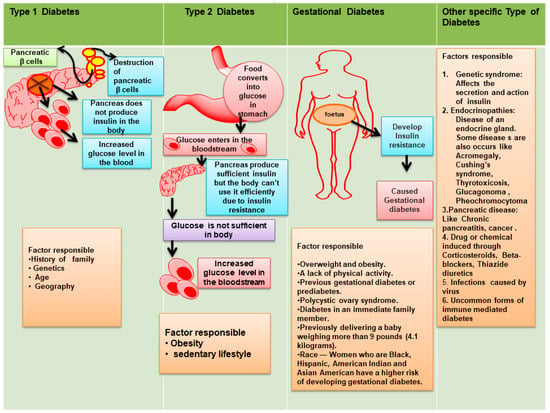
Figure 1.
Types of Diabetes mellitus (new classification system).
Type 1 Diabetes mellitus (T1DM), T2DM, others (as related with secondary conditions, genetic disorders of β-cell functions, pancreatic disease, endocrine disease), and gestational diabetes [8]. In brief:
- T1DM is recognized by autoimmune destruction of β-cell responsible for production of insulin. T2DM leads to the development of insulin resistance [9].
- Gestational diabetes mellitus (GDM) represents an increase in glucose level and is commonly seen during second or third trimesters of pregnancy.
- Monogenic diabetes caused through various genetic reasons and grouped together under the term “Other Specific Types” [10].
2.1. T1DM
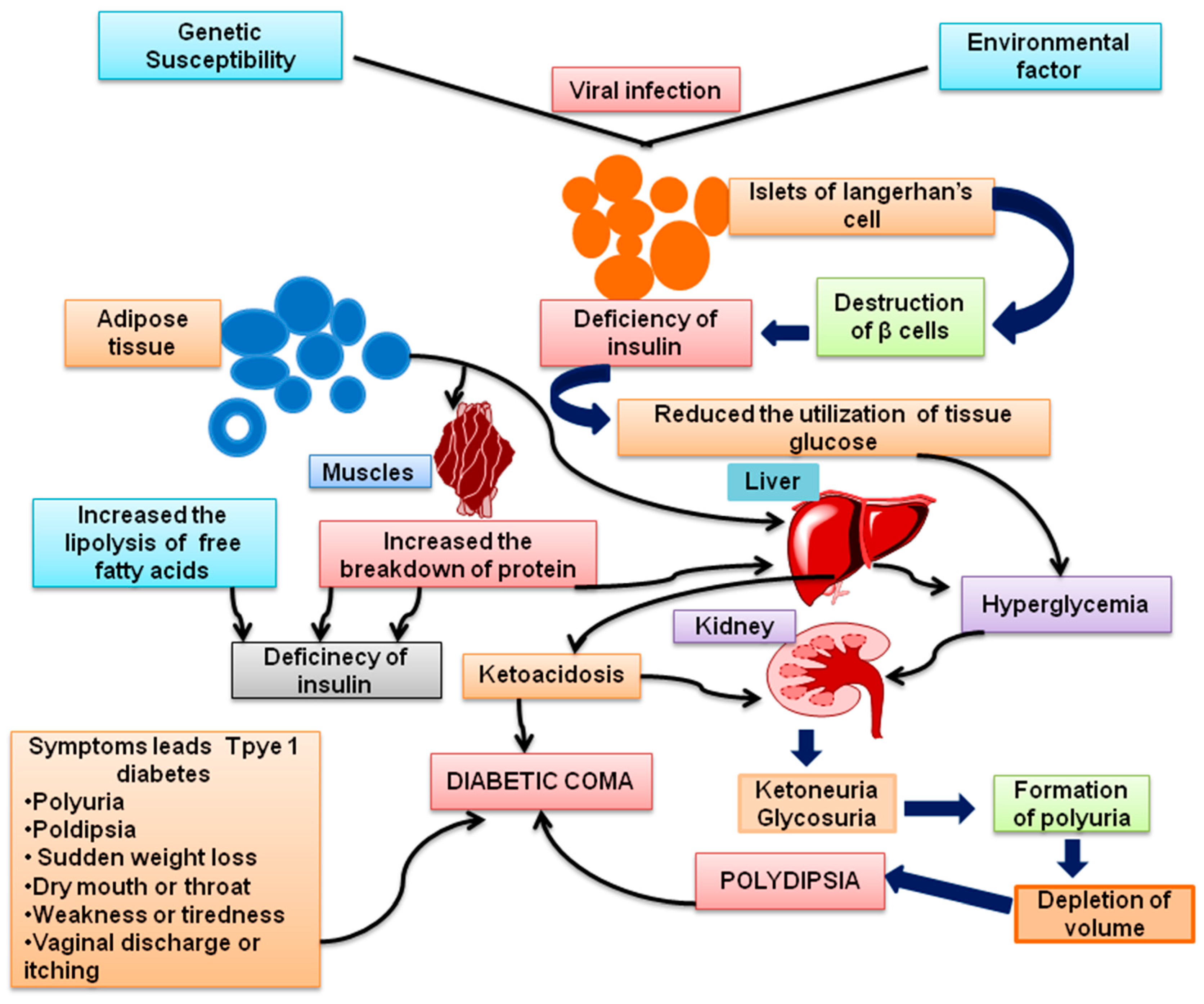
T1DM is characterized as an autoimmune disorder caused by destruction of pancreatic β-cells responsible for insulin-production. The detailed pathophysiology of T1DM was shown in Figure 2. A decrease in insulin increases the body’s production of ketone bodies in cases of high blood sugar levels, resulting in severe side effects, including liver damage, hypertension, renal failure, coma, and death in severe cases [2,11].
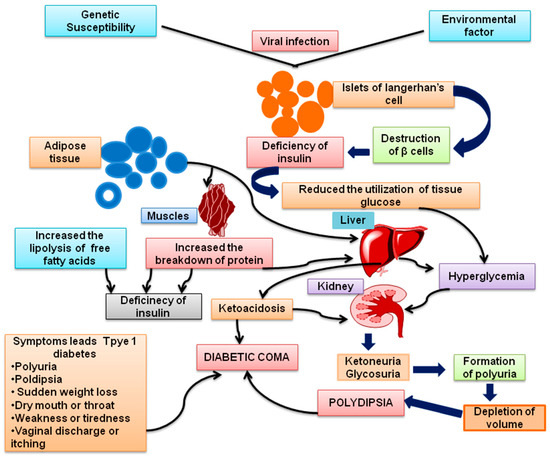
Figure 2.
Pathophysiology of Type 1 Diabetes mellitus (T1DM).
The progression of T1DM are as follows:
Phase 1 is asymptomatic, refers with normal glucose tolerance and fasting glucose conditions followed by the presence of 2 or >2 pancreatic auto antibodies.
Phase 2 signs and symptoms include the existence of 2 or >2 pancreatic auto antibodies and dysglycemia, which is condition of impairment of fasting glucose condition (glucose of 100–125 mg/dL) or impairment in glucose tolerance (140–199 mg/dL after 2-h) or a ranges of hemoglobin A1c approximately 5.7–6.4%. This is also the condition where individuals become asymptomatic.
Phase 3 is the phase of clinical signs and symptoms which seem to occur and presence of two or more than two pancreatic auto antibodies [12,13].
T1DM in pediatrics characteristically shows hyperglycemic symptoms. In youth the symptoms may occur if not assessed and treated immediately, this might result in a health emergency. Hyperglycemic patients severely affected with polydipsia, polyuria, and polyphagia. If these symptoms are ignored, the patient may develop DKA (Diabetic ketoacidosis), which necessitates hospitalizations and treatments with intravenous fluids, potassium, and insulin with attentive monitoring. The development of sign and symptoms in people with T1DM might be more varied. The diagnosis is confirmed either glucose level in plasma or hemoglobin A1c (HbA1c) monitoring. If individual has sudden onset of symptoms, the plasma glucose level should be used for diagnosis rather than HbA1c. T1DM can be diagnosed when a patient’s random plasma glucose level is more than or equal to 200 mg/dl followed with characteristic symptoms. A fasting glucose level in plasma more than or equivalent to 126 mg/dL can also be utilized for diagnosis. However, the oral glucose tolerance test (OGTT) is only used in exceptional cases [14].
2.2. T2DM
T2DM, commonly known as non-insulin dependent diabetes, or adult-onset diabetes, is majorly caused due to insulin resistance. However, in recent decades, a reduction in the functions of β-cell has been recognized as a key problem associated with T2DM. Moreover, T2DM progresses when β-cells no longer have the capability of producing sufficient insulin, leading to weight gain and obesity. T2DM is further associated with increase glucagon levels, decrease adiponectin levels, enhanced leptin levels, and decreased concentrations of interleukin-6, cytokine signal suppressors, and retinal binding proteins. There are several factors associated with T2DM, including a combination of life style choices and genetics. Some of the common factors responsible for causing diabetes include living a sedentary lifestyle, drinking alcohol, smoking, and inactivity, which may result in a reduction of insulin sensitivity and glucose tolerance, leading to obesity [8,15]. The pathophysiology of T2DM includes proper functioning of adipose tissues (endocrine organ), which may be responsible for the secretion of several types of hormones and cytokines (e.g., IL-6, TNF-α, and resistin) followed by chronic inflammation and insulin resistance. Additionally, over-weight patients associated with metabolic syndrome commonly present low levels of adiponectins and develop resistance to leptin. Leptin is a hormone responsible for orexigenic action, which regulates the energy balanced via suppression of starvation [10], whereas adiponectin (peptide) is synthesized by adipocytes having insulin sensitizing properties with anti-atherogenic, and anti-inflammatory effects [16]. This type of metabolic dysfunctions leads to insulin-resistance which predominantly affects adipose, muscles, and hepatic tissues [10]. During this period, insulin does not exhibit anti-lipolytic activity due to an increase in the production and secretion of free fatty acids in the blood [17]. Further, elevations in plasma free fatty acid (FFA) concentration are associated with increases in the synthesis and secretion of glucose in the liver [10]. Subsequently, the concentrations of cholesterol and triglycerides are also increased, particularly in LDL (low-density lipoprotein) [18], followed by obstructive impacts on the cardiovascular system [19]. Additionally, numerous genes that raise the risk of T2DM have been found as a result of GWAS (genome-wide association study), and only a few genes were found necessary for the function of β-cells and its development, as well as regulation of β-cell mass. These outcomes confirm the actual contribution of insulin resistance and also in dysfunctioning of β-cell that are involved in the pathophysiology of T2DM. Particularly, in the primary phase, T2DM pathologies might be asymptomatic with quite minor sign and symptoms, which might be neglected. The sign and symptoms, including extreme dehydration and starvation, frequent urination, blurred visualization, fatigue, slow restoration of injuries, stinging and discomfort or numbness in the hand/feet. Diagnosis is performed by determination of FPG (≥126 mg/dL), OGTT (≥200 mg/dL) or by HbA1 (≥48 mmol/mol or 6.5%). Subsequently, chronic hyperglycemic condition is exposed to tissue, i.e., mainly blood vessel, increases the risk of the developments of co-morbidities including micro-vascular and macro-vascular complications. All these types of complications are responsible for reduction in life expectancy by up to ten years with diabetic patients [10].
2.3. GDM
GDM is not considered as pathophysiological condition and develops mainly during pregnancy. T1DM develops during pregnancy, but not detected and mainly undiagnosed. However, T2DM, which was discovered during pregnancy and typically occurs in the third trimester of pregnancy, are classified as GDM [10].
2.4. Other Specific Type of Diabetes (Monogenic Diabetes)
This group includes deficiencies of β-cell (formerly recognized as MODY or maturity onset diabetes in youth) or other related defects in action of insulin. Additionally, diseases associated with exocrine pancreas, e.g., inflammation in pancreas (pancreatitis) and cystic fibrosis in pancreas are also included. People with pancreatic dysfunction caused by various types of medicines, chemicals, and infections constitute less than 10% of all Diabetes mellitus cases. However, people associated with dysfunction owing to endocrinopathies (e.g., acromegaly) other than diabetes are also included [10].
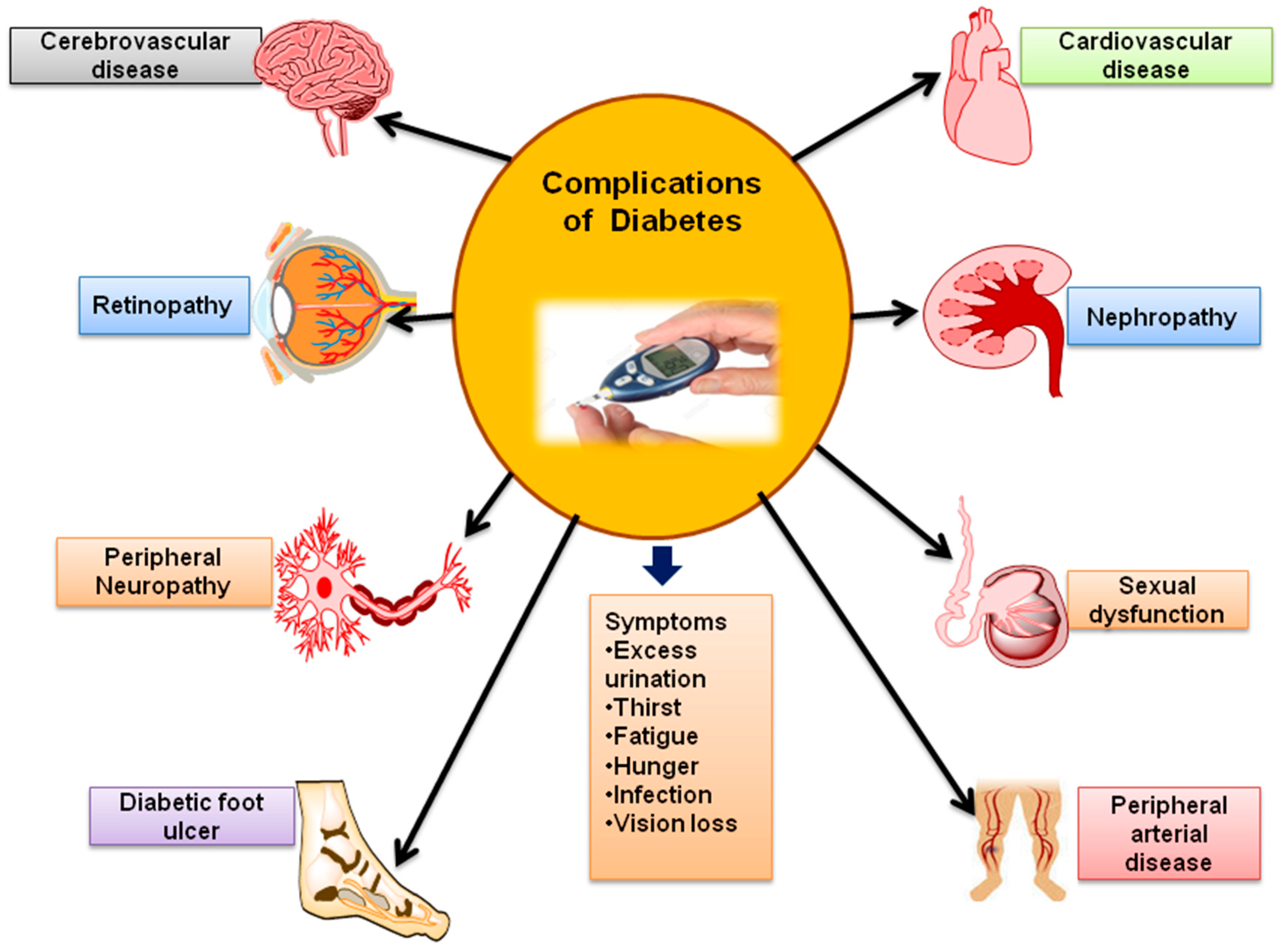
3. Complications of Diabetes
Various organs affected by diabetes, eventually lead to serious complications as shown and discussed in Figure 3 and Figure 4. Diabetes complications are mainly divided into two types i.e., microvascular and macrovascular. A microvascular complication includes damage to the nervous system (neuropathy), renal system (nephropathy) and eye (retinopathy). While macrovascular complications, including cardiac disease, stroke, and peripheral vascular disease. Particularly, peripheral vascular disease leads to non-healable bruising or trauma followed by gangrene and amputation. Diabetes complications during pregnancy includes tooth disorder, low level immunity against influenza, pneumonia and macrosomia infections followed by other birth related complications. T1DM and T2DM patients can experience same types of complications, but the timing and frequency of their occurrences can vary [20]. The details of these complications are as follows:
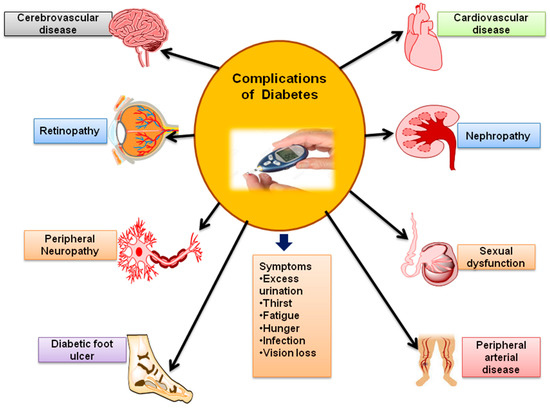
Figure 3.
Complications associated with Diabetes mellitus.
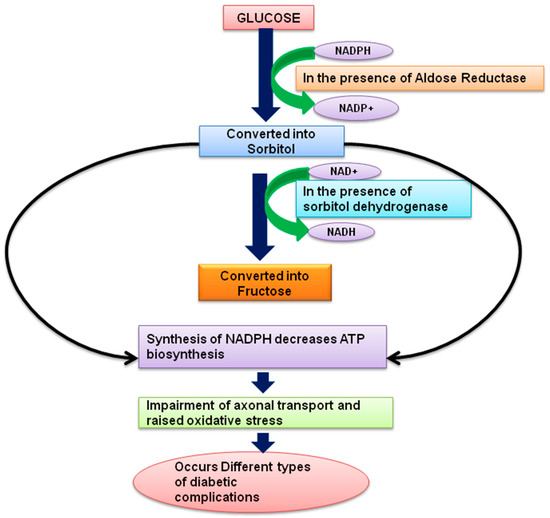
Figure 4.
Pathophysiology of Diabetes complications.
3.1. Diabetic Nephropathy (DN)
Clinically, DN is slow in progression (approximately 10–20 years) and majorly leads to the development of proteinuria followed by declination in glomerular filtration rate (GFR). If untreated, the resulting uremia can be fatal. The kidneys help regulate the levels of fluids and salts in the body, which is vital for controlling blood pressure and protecting cardiovascular health. Therefore, DN can also referred as chronic kidney disease (CKD) characterized by the presence of pathological quantities of urine albumin excretion, diabetic glomerular lesions, and GFR loss in diabetics [21]. These physiological changes majorly contribute further progression and development of nephropathy and its development. In diabetes, the alteration of toxin filtration into the blood is most commonly affected. Furthermore, kidney regulates fluid balance and blood pressure by releasing hormone such as erythropoietin. Additionally, vitamin D cell activation in the kidneys is directly affected by high glucose concentrations in several ways including smooth muscle cells, mesangial cells, endothelial cells, podocytes, muscle fibroblasts, cells of tubular and collecting duct systems [22].
Several studies reported that kidneys exhibit glomerular hyperfiltration as a result of changes in hemodynamic brought though changing blood pressure within and outside the kidney. Initial studies suggested that hyperfiltration of the glomerulus caused the most serious kidney damage including harms to glomerulus, main kidney’s filtration system and pre-glomerular vessels. Recent data suggest that diabetic individuals whose glomerular filtration rate is normal or even higher are effectively protected against end-stage kidney problems [23]. It has been further suggested that these hemodynamic changes result from changes in the metabolic conditions, signal transduction, release of vasoactive factors, and intrinsic abnormalities in glomerular arterioles. Proteuria, the accumulation of albumin in the urine, is sometimes linked to changes in renal hemodynamic and is associated with alteration within the glomerular filtration difficulties, specifically, podocytes which are glomerular epithelial cells. Hyperplasia and hypertrophy (during early diabetic conditions) of the kidney are typically seen along with enlargement of the kidney, when diabetes is diagnosed. As a result of glomerular hypertrophy, expanded mesangial cells and thickens the basement membrane of the glomerular are observed within the glomeruli. In diabetes, the proximal tubule is responsible for the greatest change in growth, constituting more than 90% of the cortical mass. Several studies have proposed that the development of extracellular matrix in the tubular component of the kidney (tubule interstitial fibrosis) is a significant contributor to the progression of renal disease in diabetic patients [24].
3.2. Diabetic Retinopathy (DR)
Diabetic retinopathy majorly leads to blindness or loss of vision and is generally attributed to diabetic macular edema (DME) due to impaired central vision and proliferative diabetic retinopathy (PDR), in turn due to the development of new blood vessels and fibrous tissues, resulting in the fractional detachment of retinal and preretinal or vitreous hemorrhage. The progression of DR involves numerous pathophysiological pathways, including oxidative stress, stimulation of the growth factors in the eye’s vasculature, as well as inflammation and activation of both protein kinase C and hexosamine pathway [25]. Further, DR is also considered the chief vascular disease and proposed to be caused by diabetic retinal neurodegeneration (DRN) as per recent study [26]. A meta-analysis included 35 studies have been conducted world-wide from 1980 to 2008, suggested the occurrence of DR and PDR among patients with diabetes to be 35.4 and 7.5% respectively [27]. However, according to recent systematic review, the annual prevalence of DR all over the world ranges from 2.2% to 12.7% and the annual prevalence of disease progression ranges from 3.4% to 12.3%, respectively [28]. Further, alteration of blood sugar level, blood pressure, and dyslipidemia are well known risk factors associated with development and progression of DR. Further, DR is classified as non-proliferative diabetic retinopathy (NPDR) and proliferative diabetic retinopathy (PDR). NPDR is additionally categorized as mild, moderate and severe non-proliferative diabetic retinopathy. Mild non-proliferative diabetic retinopathy is caused by the existence of microaneurysms (bulging in the walls of blood capillaries), and hemorrhages (dots and blots) occurs in the retina. Patients belonging to this category must be observed during every 9–12 months of period. However, severe non-proliferative diabetic retinopathy develops the retinal hypoxia, and subsequently, changes in circulation lead to the development of more severe lesions. Particularly, hemorrhages are widespread during later stages of non-proliferative diabetic retinopathy, as indicated by bleeding in venous, cotton-wool spots (localized infractions of the nerve fiber layer in the retina) and intra retinal microvascular abnormalities (IRMA). In another way, the progression of DR during proliferative stage forms new blood vessels on the disc or in another place on the retina. This could be ruptured suddenly or even after vigorous exercises resulted to preretinal or vitreous hemorrhage. Such a type of patient must be referred for immediate treatment because they have a higher risk of losing their vision. Additionally, remission only occurs after the activation of neovascularization for the development of fibrous tissues but sometime leads to fear of retinal detachment [24].
3.3. Diabetic Peripheral Neuropathy (DPN)
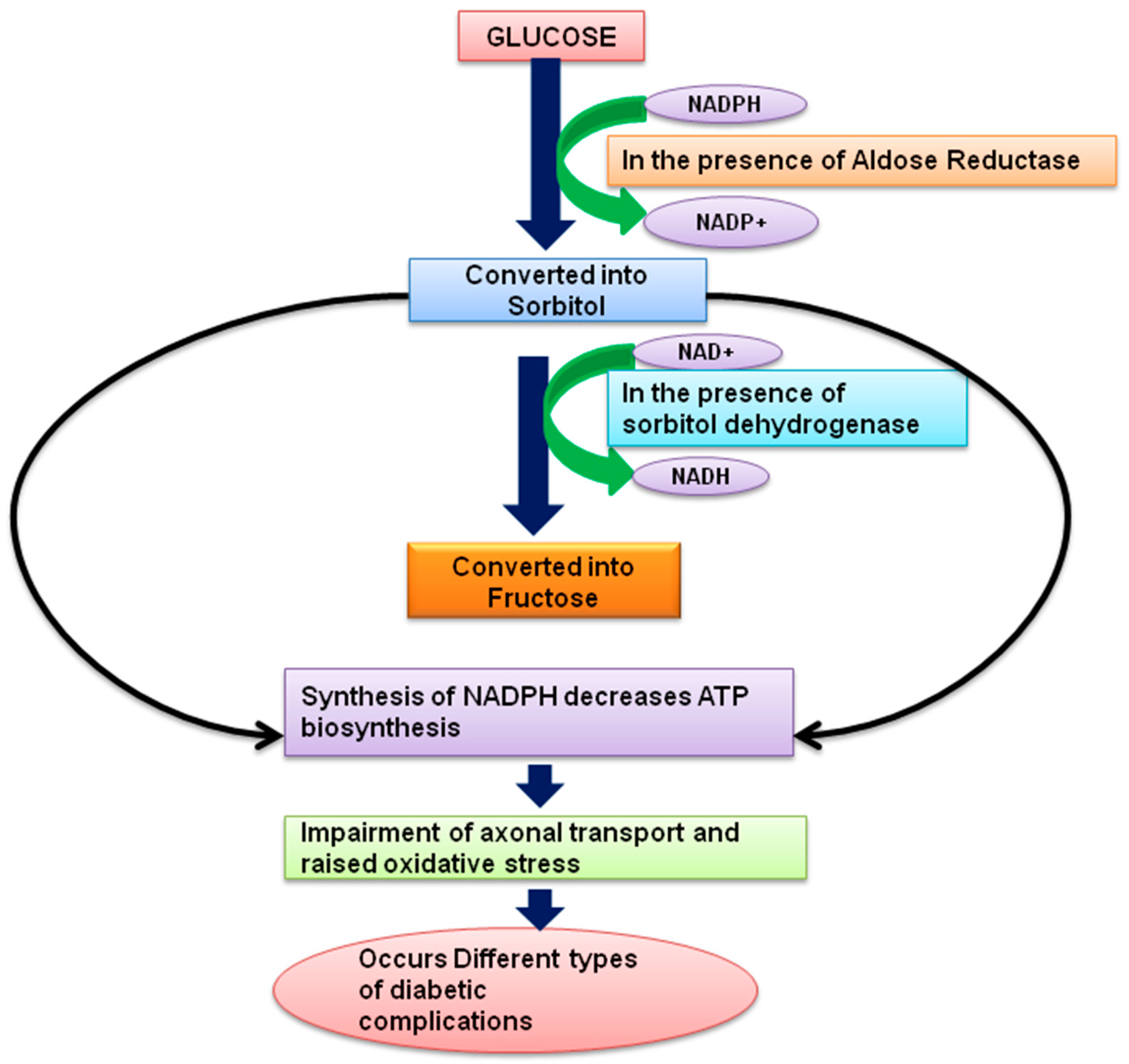
DPN encompasses a wide variety of medical pathology that can cause dysfunction of the peripheral nervous system [26]. Patients suffering from DPN present with symptoms, such as tingling, numbness, and burning sensations, respectively. DPN is caused by a variety of factors and is considered as one of the most important prevalence subtypes leading to severe complications. Consistently, a high level of glucose in blood serum seems to cause damage to minor blood vessels that provide oxygen and nutrients to the nerves. The distal sensory and autonomic nerve fibers are damaged first, resulting in the gradual loss of protective senses in both the skin and foot joints [27,28,29].
DPN is often associated with the impairment of motor, sensory, and autonomic nerve systems. An important cause of damage to peripheral nerves include oxidative stress, sorbitol accumulation, advanced glycosylation end products, disruption of hexosamine, PKC, and polymerase pathways. Neurovascular destruction with poor repair process and improper function of endothelial cells have also been implicated [30].
3.4. Sexual Dysfunction
Sexual dysfunction occurs frequently in T2DM patients, despite the fact that it has a highly significant impact on quality of life. The factors that contribute to erectile dysfunction (ED) in diabetic patients includes neuropathic, vasculopathic, and hormonal changes, respectively. Additionally, ED could be used as a diagnostic tools and can be utilized as an early biomarker of diabetic complications. In brief, ED (range 35–90%) is associated with inability or incapacity to obtain and maintain an erection during sexual activity observe for a minimum of three-month time frame [24,31]. Additionally, diabetic patients suffer from ED more frequently than non-diabetics. However, it is estimated that half of them will be affected within 10 years of diabetes diagnosis [24,32]. A study conducted to understand the prevalence of ED on recently diagnosed T2DM patients suggested 33.3% without ED, 19.4% had mild ED, 15.4% had mild-to-moderate ED, 10.4% had moderate, and 21.6% had severe ED, indicating that ED can appear early in T2DM [33].
Diabetic women are more likely to experience depression as compared to non-diabetics, and BMI was the only factor that correlated with female sexual dysfunction (FSD) [34]. Whereas, FSD is characterized by a persistent reduction of sexual desire, persistent decline in arousal, dyspareunia, and difficulty obtaining orgasmic effects [24]. Studies suggest women with T2DM are more likely to develop FSD (2.49 odds ratios) [34], while earlier studies indicated that women with T2DM are more likely to develop FSD between 12–88%. Neither the pathogenesis of FSD nor the risk factors for its development are fully understood in diabetic women. The presence of T2DM in women with FSD is higher with greater risk for blood sugar imbalances [35].
3.5. Heart Stroke and Heart Diseases
Approximately 65% of death in people with diabetes is due to cardiovascular disorder. The main causes of mortality associated with diabetes are ischemic heart disease and stroke. People with diabetes also have a 2–4-fold higher death rate due to heart disease and strokes, when compared to those without diabetes. A diabetic person often suffers from hypertension or hyperlipidemia, which are risk factors associated to cardiovascular diseases, similar to those for non-diabetics [36]. Diabetes increases the risk of coronary arterial diseases because diabetics are generally at greater risk of atherosclerosis, myocardial infarctions, strokes, and weakening of cardiac functions. Therefore, diabetes patients are generally prescribed for both management and treatment strategies, including glycemic control, renin-angiotensin-receptor enforcement (using statins or fibrates), and antiplatelet therapy such as aspirin [37]. Diabetes can also damage the myocardium without hypertension or coronary artery disease, and this condition has been termed as diabetic cardiomyopathy, which was further characterized as diastolic dysfunction [38]. Diastolic dysfunction refers to the inability of the heart to get relax properly and fill properly throughout the diastolic phase of the cardiac cycle. Despite this, long-term blood glucose control, as determined by biochemical HbA1C measures, is still the best predictor of cardiovascular diseases risks in both T1DM and T2DM conditions [14,16,39].
3.6. Diabetic Foot Ulcer
A diabetic foot ulcer (DFU) is a major health concern among people with diabetes due to its negative effects on physical and psychological well-being. Further, DFU involves increased risks of recurrence, amputation, and mortality [40]. In brief, DFU, is defined as a wound that penetrates the dermis (the deep vascular and collagenous inner layer of the skin) and spreads downward below the ankle in a diabetic patient. According to the WHO, limb amputation has a relative 68% mortality rate during five years and second in place after lung cancer. In diabetic patients with foot ulcers, impaired arterial blood flow accounts for about 80%, neuropathy for 50%, while approximately 20% have both the conditions. DFU is primarily associated with vascular insufficiency, peripheral neuropathies and reduces the ability to resist infections. Certainly, the problem gets more complicated with the induction of oxidative stress. Diabetic foot problems tend to occur due to physical or mechanical trauma resulting in cutaneous ulcers [41]. In patients with diabetes, neutrophils, fibroblasts, and leukocytes exhibit impaired activity, while inflammatory cells migrate and act less effectively at the ulcer site [42,43].
Additionally, other risk factors that contribute to macrovascular diseases, including hyperlipidemia, smoking, and high blood pressure, increase the risk of foot problems by disrupting the foot physiology, blood supply, and immune response to traumas and infection. The predisposing factors for DFU in diabetic’s shows characteristic features in male rather than females, who have more than 10-year history of previous ulcers or diabetes. Additionally, vascular disorders, neural disorders, and other related factors, including a sedentary lifestyle, smoking, poor glycemic control, etc., are other associated factors for the progression of DFU [43].
4. Molecular Mechanism of Phytoconstituents Exhibiting Antihyperglycemic Action Might Be Useful for Prevention and Treatment of Diabetes and Its Associated Complications
Diabetes and its complications have been treated with different categories of drugs, such as biguanides, sulfonylureas, α-glucosidase inhibitors and glinides either individually or in combination as evidence from past literature. The major challenges include severe side effects associated with the use of these drugs [44]. There are number of medications that prevents absorption of glucose from gut, such as chlorpropamide, tolazamide, and glibenclamide etc. [45]. From both past and current research, several ethnobotanicals and traditional remedies have been thoroughly investigated for their separation of several bioactive phytoconstituents for the treatment and management of diabetes and its related complications. Therefore, several such phytoconstituents are reviewed in the present communication exhibiting a significant role in the treatment and prevention of diabetes and its associated complications. In Table 1, phytoconstituents effective for treatment of diabetes and related complications are discussed along with their molecular mechanism as per past literature.

Table 1.
Phytoconstituents exhibiting antihyperglycemic action.
These phytoconstituents (some time non-nutritive and anti-pathogenic) are produced by plants in order to protect themselves, but they may also protect humans against several diseases [203,204]. Interestingly, phytoconstituents, based on their biological activity, are one of the most effective sources for prevention and treatment of Diabetes mellitus. Currently, several herbal products are well established in the market, e.g., pancreatic tonic, diabecon, diasulin, bitter gourd powder, chakrapani, dia-care, epinsulin, and syndrex etc. [205].
According to the WHO, approximately 65–80% of the total population in developing countries rely on medicinal herbs. The developments of new products from natural sources are also currently encouraged as only 15% out of 300,000 plant species have been evaluated scientifically for the establishment of any pharmacological potential.
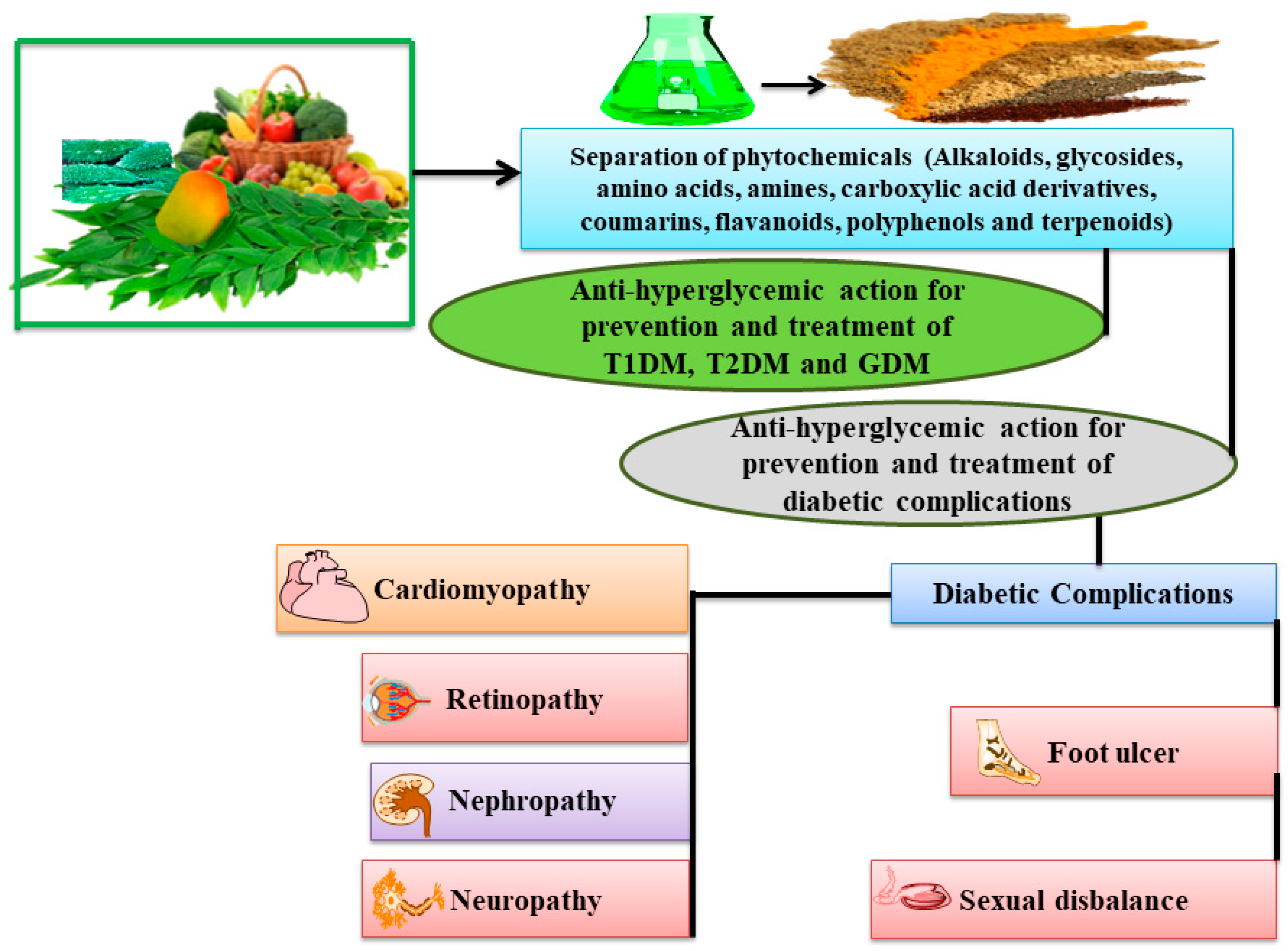
Since ancient times, various extraction procedures have been used to produce extracts from medicinal plants including maceration, infusion, percolation and decoction [206]. Researchers have been found about 800 plants having potential to prevent and inhibit diabetes progression [44]. WHO data regarding prevention and treatment of diabetes with traditional medicinal plants are very clear and majorly based on safety and efficacy parameters [205]. There are large number of phytoconstituents which have been reported and discussed as potential agents to inhibit diabetes progression, such as polyphenols, alkaloids, glycosides, amino acids, terpenoids, peptidoglycans, glycolipids, saponins, and other compounds (Figure 5). These phytoconstituents can relieve metabolic dysfunctioning by reversing them as well as delaying the progression of diabetes [207]. Some of the selective phytoconstituents that have been already established for the treatment and prevention of diabetes and its complication are discussed herewith:
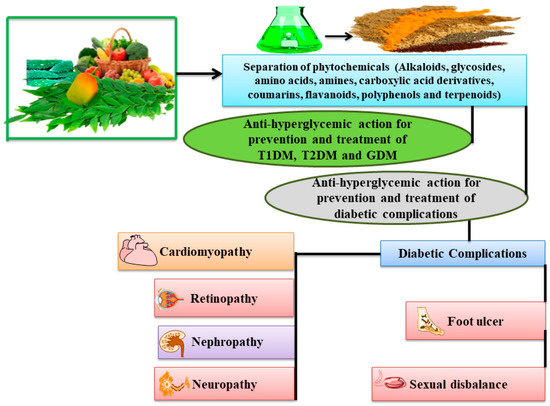
Figure 5.
An overview for prevention and treatment of diabetes and its associated complications by phytoconstituents.
4.1. Flavonoids
Flavonoids are plant metabolites that belongs to the family of water-soluble polyphenolic compounds and is composed of two benzene rings connected by a short three carbon chain [208,209]. There are several phenolic compounds classified by their chemical structure, including: flavones (kaempferol, quercetin, isorhamnetin and myricetin etc.), flavan-3-ols (e.g., epicatechin, catechin and gallocatechin etc.), anthocyanidins (e.g., malvidin, peonidin, cyanidin, delphinidin, and petunidin etc.), flavones (e.g., apigenin, luteolin, baicalein, chrysin), isoflavones (daidzein, genistein, glycitein and biochanin A) and flavanones (eriodictyol, hesperedin and naringenin) [208,210,211]. A number of flavonoids possess anti-hyperglycemic properties via antioxidant capacity and ability to modulate certain cell signaling pathways. A flavonoid can be found in vegetables, herbs, fruits, beverages, chocolates, and plants [143,212]. Researchers have isolated the flavonoids from plants described below that have anti-hyperglycemic properties.
4.1.1. Diosmin
Diosmin is naturally occurring flavonoid found mainly in citrus plants. Diosmin is an anti-hyperglycemic principle isolated from several plants including Scrophularia nodosa. Researchers suggested that it has an inhibitory effect on diabetes, which are mediated by decreases the levels of glycosylated hemoglobin and an increased in glutathione peroxidase activity. A reduction in the concentrations of plasma glucose, an increase in liver glycogenolysis, and an improvement in insulin levels were observed in patients getting treatment with diosmin. Aside from that, it enhanced the levels of both vitamins C and E [213]. In another study, a potential mechanism of action that is responsible in the improvement of CVD after treatment with diosmin was suggested due to a considerable reduction in pro-inflammatory mediators, such as IL-6 and TNF-α, as well as pro-angiogenetic factors [214].
4.1.2. Morin
Morin is a flavonol isolated from several plants, including Prunus dulcis, Chlorophora tinctoria, and Psidium guajava (Guava leaves) [213]. This flavonol showed increased antioxidant effects for improving the anti-hyperglycemic properties and reducing the insulin resistance via improvement in oxidative stress; normalizing the lipid and lipoprotein levels, and decreased TNF-α [215]. Studies on morin have shown it to reduce the inflammatory action of cytokines IL-1 and IL-6 in diabetic rats [216]. The mechanisms by which morin showed its anti-hyperglycemic effects have been attributed due to the restoration of both insulin and leptin sensitivity followed by reduction in hyperlipidemia and accumulation of liver lipids [217]. In another study, morin was found to reduced glucose-6-phosphatase activities, increased hexokinase activity, and decreased insulin levels [167]. In a very recent study, the potential significance of morin supplements in protecting against hyperglycemia–induced skeletal impairments due to the activation of insulin/IGF-1 signaling was suggested [171].
4.1.3. Fisetin
Fisetin is a flavonoid found in apples, grapes, strawberries, onions, cucumbers, and grape sap. Fisetin has been found to have a potential activity in prevention of diabetes by inhibiting mitochondrial pyruvate transport and decreasing the redox mechanism of cytosolic NADH/NAD. It works by decreasing glycogen breakdown, lowering blood glucose levels, increasing insulin levels, and securing the expression of gluconeogenic genes, like phosphoenol pyruvate carboxykinase. Further, Fisetin was found to inhibit hyperglycemia via lowering nuclear factors like IL-β, NF-kB p65 unit levels, and serum nitric oxide during study on rat model [213,218]. A recent study reported that fisetin alleviates high blood glucose levels on podocyte damage and with streptozotocin-induced diabetic nephropathy mice. The effect was due to restoring cyclin-dependent kinase inhibitor 1B (CDKN1B)/ribosomal protein S6 kinase β-1 (P70S6K)-mediated autophagy and inhibiting NLRP3 inflammasome. The study further provided a new vision for the renoprotective properties of fisetin in diabetic persons [219].
4.1.4. Hesperidin
Hesperidin is a naturally occurring flavonoid found in citrus plants, particularly in Citrus aurantium [213]. Hesperidin decreases the risk of diabetes by inhibiting the synthesis of free radicals and pro-inflammatory cytokines [220]. Hesperidin treatment has been found to normalize lipid levels, increase adiponectin, reduces the level of thio-barbituric acid reactive substances (TBARS), and also decreases lactate dehydrogenase (LDH) activities [221]. Recently, it was reported that hesperidin treatment reduced oxidative stress and mitochondrial dysfunction in HG-persuaded insulin resistance, partially by suppressing miR-149 silencing mediated by DNA (cytosine-5)-methyl transferase 1 (DNMT1) [222].
4.1.5. Eriodictyol
Eriodictyol, is a natural flavonoid compound mostly found in lemon with profound use in the prevention and treatment of diabetes and obesity [223]. Furthermore, it has been reported that eriodictyol have shown potent anti-hyperglycemic action via inhibition of oxidative stress, promotes mRNA expression of peroxisome proliferation-activated receptor gamma (PPARγ) and adipocyte-specific fatty acid-binding protein (AFABP) along with the protein expression of PPARγ in differentiated 3T3-L1 adipocytes. It also reduces endothelial nitric oxide synthase (eNOS) and vascular endothelial growth factors (VEGF), intercellular adhesion molecules (ICAM-1), and lipid peroxides [159,224,225,226]. It was further demonstrated that eriodictyol acts as an innovative insulin secretagogue with a glucose-dependent insulintropic activity via a cAMP/PKA pathway as an evidence from in vitro and in vivo model [227]. It was further reported that eriodictyol inhibits the invasion, proliferation, migration, and stimulation of apoptosis in retinoblastoma by regulating the PI3K/Akt pathway [228].
4.1.6. Naringenin
Most of the citrus fruits and tomatoes contain abundant amounts of naringenin, known for significant antioxidant action [213]. Naringenin inhibits the activity of α-glucosidase and interferes with genes convoluted in the metabolism of lipids and inhibits the uptake of glucose. Further, naringenin was found to act through the activating AMP-activated protein kinase (AMPK), the improvement of antioxidant properties, decreasing insulin resistance, and improving hepatic function markers [229]. In a very recent study, treatment with naringenin suppressed the nuclear translocation of activating transcription factor-4 (ATF-4) and cyclophosphamide, hydroxyl daunorubicin, oncovin and prednisone (CHOP) in both hyperglycemic renal cells and diabetic kidneys. Additionally, apoptosis was inhibited by naringenin in hyperglycemic renal cells and diabetic kidney tissues by downregulating expression of apoptotic marker proteins. It was further concluded that naringin protected from hyperglycemic renal damage via oxidative stress reduction and improvement in antioxidant defense response [163].
4.1.7. Apigenin
Apigenin is a flavonoid which is extensively found in citrus, vegetables, tea, and nuts. It acts by improving antioxidant parameters, improving glucose transporter 4 (GLUT4) translocation, and preserving beta cells [143]. A study explored effects of Apigenin (20 mg/kg) in diabetic rats, finding that it improved the renal impairment, reduced the oxidative stress and fibrosis (reduction in type IV collagen, fibronectin, and transforming growth factor-1) [230]. It also controlled inflammation (reduced production of IL-6, NF-κB and TNF-α) and apoptosis by dramatically preventing MAPK activation (by increasing Bcl-2 expression and decreasing Bax and caspase-3 production). Apigenin alleviated kidney damage caused by diabetic nephropathy through reduce oxidative stress and fibrosis and blocked the mitogen- activated protein kinase (MAPK) pathway.
4.1.8. Kaempferol
A flavonol which is found mainly in Ginkgo biloba, grapefruits, tea, edible berries, and vegetables. Kaempferol was reported to have apoptosis inhibition, reducing the activity of caspase-3 in β-cells, improving cAMP signaling and increasing the production and secretion of insulin. Additionally, it results in reducing IL-1β, TNF-α, lipid peroxidation, nitrite and glycosylated hemoglobin levels [143,231]. Another study investigated the activity of kaempferol on rat renal proximal tubular epithelial cell (NRK-52E) and primary human renal proximal tubule epithelial cells (RPTEC). Finding of study suggested that kaempferol was able to inhibit hyperglycemia-induced RhoA (Ras Homolog Family Member A) activation, reduce oxidative stress, pro-inflammatory cytokines (TNF-α and IL-1β), and fibrosis (expression of TGF-β1, and extracellular matrix protein). As a result, kaempferol was concluded to possess a therapeutic effect for the treatment of diabetic nephropathy [232]. In another study, kaempferol present in the unripe soybean leaf have been demonstrated the ability to reduce glycosylated hemoglobin levels via the downregulation of liver fatty acid synthase activity [143].
4.1.9. Chrysin
Chrysin is the flavonoid and an important phytoconstituents of Oroxylum indicum. Studies reveals number of biological sources of chrysin, which includes Pelargonium peltatum, Passiflora caerulea and Tilia tomentosa. Chrysin, when administered orally resulted to prevents nephropathy via decreased the levels of TGF-β, collagen-IV protein expression and fibronectin in the kidneys followed by significant reduction of IL-1β and IL-6 levels [233]. Chrysin therapy was further established to improve both lipid peroxidation and insulin levels [234]. In another study, chrysin was discovered to have similar effects as metformin, both of them able to decrease the levels of glucose and triglycerides, inhibit the production of pro-inflammatory cytokines (involves in the progression of diabetes), and thus improve atherosclerosis and other related cardiovascular conditions [235].
4.1.10. Baicalein
The fruit of Oroxylum indicum and the roots of Scutellaria baicalensis are one the rich sources of baicalein content. Baicalein was found to lower food intake significantly, improve body weight and levels of fasting blood glucose, followed by normalization of renal function and renal oxidative stress. Molecularly, these events are co-related with inhibition of NF-κB activation, reduction of expression of inducible nitric oxide synthase (iNOS) and transforming growth factor beta (TGF-β), followed by up-regulation of AMPK [236]. Recently, baicalein in the treatment of diabetic nephropathy was studied by reducing inflammation and oxidative stress via activation of the Nrf2-mediated antioxidant signaling pathway and suppression of the MAPK-mediated inflammatory signaling pathway [237].
4.1.11. Luteolin
Luteolin is a rich source of flavonoid in vegetables and fruits, including carrots, peppers, cabbage, and apples. The phytoconstituents luteolin promotes insulin action and PPARγ transcriptional activation. Luteolin was found to reduces inflammatory cytokines including monocyte chemotactic protein-1 (MCP-1), increased the levels of adiponectin and improving the secretion of insulin [234,238]. A study demonstrated the protective effect of luteolin on cardiac tissues of STZ-induced diabetic mice via altering Nrf2-mediated oxidative stress and NF-κB mediated inflammatory responses, suggesting the usefulness of luteolin as a potent therapeutic agent for the treatment of diabetic cardiomyopathy [239].
4.1.12. Tangeretin
Targeretin is a flavonoid most abundantly found in peels of citrus fruits. An oral administration of tangeretin (200 mg/kg bw) reduced total cholesterol, blood glucose, resistin, leptin, IL-6, and MCP-1 [143,240]. Subsequently, tangerine (100 mg/kg bw) when administered orally to diabetic rats for 30 days, the levels of HbA1c and plasma glucose got decreased while the levels of insulin and hemoglobin got increased, respectively. Additionally, it was shown that tangeretin increases the glycolytic enzymes and regulates glucose metabolism in the hepatic tissues of diabetic rats by promoting insulin production from the β-cells of the pancreas by antioxidant mechanism [143,175]. In 3T3-L1 adipocytes, tangeretin boosted the production of the insulin-sensitizing protein adiponectin while concurrently reducing the secretion of the insulin-resistance protein monocyte chemotactic protein-1 (MCP-1), as evidenced by another study [234]. Recently, tangeretin was demonstrated to significantly reduce high glucose levels, inducing the expression of VEGF, IL-1β, interleukin 6 (IL-6), and transforming growth factor beta 1 (TGF-β1) to improve diabetes [241].
4.1.13. Isorhamnetin
The isorhamnetin compound has been identified and isolated from Hippophae rhamnoides, Oenanthe javanicus, and Ginkgo biloba, and will be used to ameliorate diabetes. Mechanistically, isorhamnetin was able to reduce the oxidative stress, production of sorbitol, and interfere the metabolism of lipids in a diet induced mouse model [242]. Recently, isorhamnetin was demonstrated to improve the condition of insulin resistance in a T2DM induced rat model [243].
4.1.14. Wogonin
Scutellaria baicalensis is a rich source of wogonin, which has the potential to improve the lipid metabolism and insulin sensitivity by inhibiting AMPK and PPARα [234,244]. In a very recent study, wogonin was found to prevent glomerulopathy and podocytes injury by modulating the Bcl-2-mediated interaction between autophagy and apoptosis. Additionally, wogonin demonstrated as a potential therapeutic agent against diabetic kidney disease [245].
4.1.15. Rutin
Rutin is commonly present in various edible fruits such as grapes, lemons, berries, oranges, buckwheat, limes and peaches. Rutin was found to enhance insulin secretion, restore glycogen content, decreased oxidative stress, inhibit the productivity of AGEs, downregulate IL-6 production, reduce TNF-α levels, and improve liver antioxidant activity [234,246]. In another study, rutin was able to reduce the oxidative stress, apoptosis, and inflammation and exhibit cardiac and myocardial functions in streptozotocin induced diabetic rats [247].
4.1.16. Quercetin
Various fruits and vegetables, including onions, berries, apples, pepper, and coriander, are a rich source of quercetin. A study on quercetin found that it inhibits GLUT-2, increases the levels of antioxidant enzymes, and reduces the absorption of intestinal glucose, suggesting its anti-diabetic effects. Further, quercetin was found to induce cell proliferation by blocking tyrosine kinase enzyme [143,234]. The effectiveness of quercetin on 50 male SD rats including five groups viz. normal control, 50 mg/kg STZ-induced diabetes, and three (30 mg/kg for Quercetin (QE), 10 mg/kg for Resveratrol (RS), and combination) was assessed. The results showed that QE, RS, and combined treatments effectively decreased high serum blood glucose levels, insulin levels, and dyslipidemia in diabetic rats. Further, QE significantly reduced oxidative stress, tissue injuries biomarkers and improve hematological parameters, associated with ketoacidosis and hyperlactatemia majorly responsible for diabetes related complications [248]. Quercetin 3-o-glycoside and quercetin, a component of Vaccinium vitis, shows hypoglycemic effects by stimulating AMPK [233].
4.1.17. Genistein
This flavonoid is derived from a soya-isoflavones that occurs naturally in legumes and abundantly found in Genista tinctoria and Sophora subprostrala. Genistein significantly reduces renal thiobarbituric acid reactive substances (TBARS), increases blood insulin levels, and improves glucose tolerance without adversely affecting body weight [249]. Another study shown the beneficial effects of genistein by improving brain insulin signaling, increases neurotrophic support, and alleviates AD-related pathology in the obese diabetic mouse brain [149].
4.1.18. Daidzein
Daidzein is a Soy isoflavones flavonoid that naturally occurs in soybeans, fruits, and nuts. A study found that daidzein improves the metabolism of lipids and glucose level, insulin sensitivity, and phosphorylated AMPK in muscles [250]. Daidzein was found to act on VEGFA, INS, INSR, IL-6, TNF, and AKT1. In brief, Daidzein participates in the AGE-RAGE signaling pathway, insulin resistance, HIF-1 signaling pathway, FoxO signaling pathway, and MAPK signaling pathway. It may also control type 2 diabetes, inflammation, atherosclerosis, and diabetes complications, such as diabetic retinopathy, diabetic nephropathy, and diabetic cardiomyopathy [251].
4.1.19. Delphinidin
Delphinidin showed anti-diabetic properties mediated by antioxidant activity and decreases in albumin and hemoglobin glycation. It is most commonly available in eggplants, carrots, berries, grapes tomatoes, sweet potatoes, red cabbages, and red onions [252]. It exhibits, antioxidant, anti-mutagenic, anti-angiogenic, and anti-inflammatory effects [253]. Another study showed that the administration of 100 mg/kg of delphinidin to diabetic mice for eight weeks was able to decrease the albumin rate and HbA1c glycation [191]. The anti-diabetic effect of delphinidin due to the inhibition of the uptake of glucose in mice jejunal tissue and human intestinal cell lines via free fatty acid receptor 1 (also known as GPR40) was further established [254].
4.1.20. Cyanidin
Cyanidin is an anthocyanin flavonoid used in treating diabetes through regulating the enzymes, alpha-glucosidase, pancreatic alpha-amylase, inhibiting apoptosis and improvement in antioxidant status. Furthermore, it also inhibits apoptosis while simultaneously enhancing antioxidant activity [252]. In another study, cyanidin chloride liposomal formulations were formulated and found to be effective against diabetes mellitus complications [191]. Similarly, a study reported the potential effect of cyanidin on STZ-induced diabetic rats via pancreatic apoptosis inhibition, insulin receptor phosphorylation activation, and improvement in healthier β-cells [255].
4.1.21. Pelargonidin
Pelargonidin is an antioxidant molecule and was found to be present in blackberries, cranberries, raspberries, and blueberries [252]. Subsequently, pelargonidin was found to improve hyperglycemia, oxidative stress level, glucose in-tolerance, and insulin resistance as evidenced in a study conducted using db/db mice. Furthermore, pelargonidin enhanced serum lipid levels, impaired hepatic dysfunction, and mitigated type-2-diabetes via modulating glucose and lipid metabolism [256].
4.2. Saponins
Saponins are glycosides mostly found in plants as surface-active compounds consisting of sugars connected to a hydrophobic aglycone moiety called sapogenins also known by triterpenoid or steroid. Saponins are the active chemical moiety present in several herbs exhibiting anti-hyperglycemic action. In a study saponins was found to be responsible for the hypo-glycemic abilities of Anabasis articulata via improvement in insulin production. Additionally, Astragalus membranaceus, saponins was found to greatly ameliorates oxidative stress and AGES formation [233]. The saponins obtained from Entada phaseoloides elevate serum insulin level, alleviate hyperglycemia, and decrease lipids [257]. In another study, saponins (from Garcinia kola) were able to significantly reduce the levels of glucose in plasma and insulin in blood using alloxan induced diabetes rats [258]. Similarly, saponins obtained from Helicteres isora reduce serum lipid and plasma glucose levels with increased GLUT-4 at the same time. Momordica charantia has natural saponins and effective in improving diabetes [259]. Comparatively, saponins significantly induce AMP-activated protein kinase (AMPK) activation as similar as troglitazone [260]. Moreover, Solanum anguivi Lam. fruits were found to exhibit potent anti-hyperglycemic, hypolipidemic, and antioxidant effects due to the existence of saponins in an alloxan-induced diabetic rats model [261]. A study further reported potent anti-hyperglycemic action of saponin extract from Solanum anguivi [262]. A study demonstrated that Platycodi radix extracts containing saponin shows recovery of AMPK/ACC phosphorylation in the muscle of obese mice to improve obesity and insulin resistance by activating AMPK/ACC pathways and decreasing adipocyte differentiation [263]. Subsequently, saponins rich fractions of Polygonatum odoratum were found to improve glucose uptake and relieve diabetes symptoms via the upregulation of superoxide dismutase activity and downregulation of melondialdehyde levels in another study [264]. Several saponins isolated and found to be effective as anti-hyperglycemic agents are discussed below:
4.2.1. Diosgenin
Diosgenin, a saponin isolated from Dioscorea rotundata, was found to improve glucose 6-phosphatase enzyme function [233]. Both diosgenin and fenofibrate were found to reduced lipid accumulation in the liver and decrease hepatic triglyceride (TG) content. Contrarily, while diosgenin considerably reduced the level of ALT, fenofibrate significantly increased the levels of ALT and aspartate aminotransferase. Additionally, diosgenin also improved AMPK and ACC phosphorylation followed by suppressing the liver X receptor (LXR) α in the liver [265].
4.2.2. Arjunolic Acid
Arjunolic acid is a triterpene saponin found in Terminalia arjuna that protects against from several metabolic diseases, including diabetes and its complications. A recent study shown that arjunolic acid could be a promising candidate for the protection of retinal cells in STZ-induced inflammation and oxidative stress via AMPK/mTOR/HO-1 mediated autophagy pathways [266]. Further, the effect of orally administered arjunolic acid in preclinical model of Type 2 diabetes mellitus was found to shown significant reduction towards normalization of both fasted and random blood glucose samples, restore body weight and improved the conditions of polyphagia, polydipsia and glucose tolerance, respectively [267]. Additionally, arjunolic acid was found to reduce serum HbA1c, triglyceride and low-density lipoproteins without altering high density lipoproteins.
4.2.3. Platyconic Acid
Platyconic acid was isolated from Platycodi radix have found to regulates insulin-induced uptake of glucose in 3T3-L1 adipocytes. This substance is well known for increasing glycogen storage and decreasing disaccharide storage in the liver. It also improved insulin signaling, increased GLUT4 and adiponectin expression in adipose tissues [268].
4.3. Alkaloids
Alkaloids are secondary plant metabolites isolated from bacteria, fungi and animals. An alkaloid is a natural compound containing basic nitrogen atoms. Few researchers have worked with alkaloids having specific types of amines. The categories of alkaloids are mentioned below [269]:
- True alkaloids (Atropine, morphine and, nicotine etc.)
- Protoalkaloids (Adrenaline, ephedrine and, mescaline etc.)
- Polyamine alkaloids: Example; Spermidine, epinephrine, putrescine and spermine.
- Pseudoalkaloids: Example; Caffeine, theophylline and, theobromine, etc.
Alkaloids have potent hyper-glycemic effects that can be demonstrated by their significant impact on various experimental models as evidenced in several studies conducted in the past. A study demonstrated that an alkaloid containing extract obtained from Fenugreek was found to have anti-hyperglycemic properties via elevated serum insulin levels, decreased serum lipids and lipid peroxidation, using streptozotocin-induced diabetic rats [233,270]. Some of the potent alkaloids exhibiting antihyperglycemic effects are discussed here:
4.3.1. Koenidine
A study demonstrated uptake of glucose and translocation of GLUT-4 are enhanced in L6-GLUT-4 myotubules after treatment with koenidine. Additionally, koenidine could be beneficial to diabetes and insulin resistance conditions as evidenced by an experiment performed on streptozotocin-induced diabetic rats [271].
4.3.2. Canthin-6-One Derivatives
A study demonstrated that the administration of a partially purified alkaloids fraction (Canthin-6-one derivatives) from roots of Aerva lanata was found to significantly reduce the levels of serum glucose in blood and thus prevent diabetes on streptozotocin-nicotinamide induced rats model [272].
4.3.3. Vindoline I, Vindolidine II, Vindocine III and Vindocine IV
In another study, alkaloids isolated from Catharanthus roseus were found to have a potent anti-hyperglycemic effect. The four alkaloids obtained from C. roseus, i.e., Vindoline I, Vindolidine II, Vindocine III and Vindocine IV, showed increased glucose transport to pancreatic βTC6 or myoblast C2C12 cells and hence exhibited the strongest activity. Further, vindocine III and IV inhibited the protein tyrosinase phosphatase-1B (PTP-1B), as well as treated oxidative damage caused by hydrogen peroxide [273].
4.4. Tannins
Tannins are the polyphenolic biomolecules which are naturally occurring in tea, coffee, grapes, wine, dry fruits, apricot, mint, barley, dry fruits, peaches, rosemary, basil, pomegranates, amla, cloves, rye, rice, oat and strawberries etc. [274].
Tannins are mainly classified as:
- Hydrolysable tannins (gallic acid),
- Non-hydrolysable or Condensed tannins (flavones), and
- Phlorotannins (phloroglucinol).
Some studies have reported that medicinal plants containing tannins exhibit anti-hyperglycemic effects [275]. Tannins play a significant role in the treatment of diabetic neuropathic pain (DNP) due to their higher antioxidant activity. Further, neuroprotective beneficial effects of tannins in diabetic problems due to its potential antioxidant and anti-inflammatory effects were also demonstrated [276]. Some of the potent studies on tannins are discussed herewith:
4.4.1. Catechin
A study showed that (25 mg/kg and 50 mg/kg of catechin administered orally for 28 days) reduced the levels of melondialdehyde (MDA) and improved GSH, SOD, and catalase levels in diabetic rats induced by STZ [277].
4.4.2. Epigallocatechin
In STZ-induced diabetic rats, treatments with epigallocatechin (EGCG) reduced hyperalgesia responses (as measured by hot plate, tail immersion, formalin, and carrageenan-induced paw edema tests). Further, treatment with epigallocatechin for 10 weeks caused STZ-induced diabetic rats reflected paw withdrawal threshold (PWT), suggesting an improvement of mechanical hyperalgesia and tactile allodynia [278,279].
4.5. Terpenes
Terpenoids are secondary metabolites composed of repeating isoprene units with a number of structural variants [280]. They are classified into monoterpenes, diterpene, triterpenes, tetraterpenes, hemiterpenes, sesquiterpenes, sesterpenes, and polyisoprenes. Terpenes and their derivatives protect against several diseases and have a number of beneficial characteristics [281]. Generally, terpenes were found to have an antihyperglycemic effects, as evidence by both in vitro and in vivo studies. They have an excellent antioxidant property and a potential inhibitory effect against α-glucosidase enzyme. Moreover, terpenes were also found to improve the levels of insulin, glucose uptake in tissues and inhibited several carbohydrate metabolism signaling pathways [280,281]. A number of monoterpenes have been shown to improve glucose uptake by up regulating the translocation of glucose transporter (GLUT4), improves the insulin signaling pathway, improved insulin secretion, ameliorating cytokines proinflammatory effects and, protecting pancreatic cells [280]. Additionally, terpenes are responsible for maintaining sugar level via increase in the levels of glycogen, fructose-1,6-bisphosphatase and glucose-6-phosphatase. Further, activities of glucokinase and glucose-6-phosphate dehydrogenase were reduced [280]. Sesquiterpenes are more efficient as compared to other terpenes due its tightly arrangements of the structure. On the other hand, triterpenes with more hydrophilic groups were preferred for multi-targeting effects as anti-hyperglycemic [281,282]. Chi et al. (2013) worked on limonene protection, which might be mediated by inhibiting nuclear factor-κB (NF-κB) [283]. Terpenes, e.g., limonene, 3-carene, β-caryophyllene, and α-pinene, have been reported to bind directly with gamma-aminobutyric acid receptors, resulting in decreased activity of acetylcholinesterase and lipoxygenase activities. Molecularly, terpenes reduce lipid peroxidation induced by ROS formation, NO release, transforming growth factor (TFG)-1, and type I procollagen secretions, phosphorylation of various MAPK-related signaling molecules, production of O2, and H2O2-inducedastrocytic cell death. Additionally, terpenes also increased superoxide dismutase, catalase and peroxidase activities, reduced glutathione content, and restored the mitochondrial membrane [284]. Several studies quoted earlier that both terpenes and terpenoids are involved in the in the suppression of microglia mediated inflammations in acute and chronic neurological diseases [285,286].
4.5.1. Linalool
Researchers have reported the antioxidant and anti-inflammatory activity of the monoterpene linalool by in vitro and in vivo studies showing that the molecules have a strong neuroprotective characteristic property, acting in NF-κB activation and preventing its nuclear translocation [287].
4.5.2. α-Pinene
Studies show the photoprotective effects of α-pinene (present in essential oils of coniferous trees) on human skin epidermal keratinocytes from inflammatory signaling [288]. The researchers determine that α-pinene inhibits the UVA-induced expression of inflammatory proteins, such as IL-6 and TNF-α. Furthermore, it has been recommended that the anti-inflammatory property of alpha-pinene is due to its inhibitory effects of MAPK pathways via the suppression of JNK and ERK phosphorylation [289].
4.6. Glycosides
The presence of glycosides in Gymnema sylvestre was found to exhibit anti-hyperglycemic effects on experimental model [233]. Similarly, residues isolated from Jatropha curcus, were found to have significant anti-hyperglycemic effects on streptozotocin induced animal model [290]. Activity was further correlated with events, such as lowering of serum α-amylase, lactate dehydrogenase activity, reduction of oxidative stress, and modulation of melondialdehyde levels. The cardiac glycosides extracted from the seeds of Securigera securidaca (Fabaceae) lower the blood glucose levels via the stimulation of insulin secretion [291].
5. Nutrivigilance (Compounds’ Adverse Effects)—As a Possible Limitation of Their Use
Among phytochemicals, several nutrivigilance studies have been conducted and some noticeable adverse effects have been observed [292]. In this context aloin has been reported to cause bloody diarrhea, cardiac dysrhythmias, peripheral edema, red coloration of the urine, weakness, weight loss, stomach cramps, itching, redness, rash, and pruritus [293]. Alanin was associated with paraesthesia [294]. An overdose of diadzein was stomach upset, constipation, flatulence and diarrhea [295]. Similarly, hesperidin is associated with stomach pain, diarrhea, and headache [296]. Naringenin is associated with cough, dizziness, headaches, flushing sensation, palpitation, angioedema, liver dysfunction, and myositis [297]. Apigenin is associated with upset stomach, muscle relaxation and sedation [298]. Baicalein was involved for some time in fatigue and shortness of breath [299]. Silybin was found to be associated with gastrointestinal problems, headache, dizziness and pruritus [300].
6. Conclusions
Diabetes and its related complications represent one of the challenging situations in today’s world, affecting many people of all age groups. Many studies are currently taking place to provide sustainable, safer, and effective solutions, including antidiabetic drugs and insulin therapy. Despite many years of research into the use of bioactive compounds as hypoglycemic agents, none of them have been used alone in the treatment of DM due to restricted solubility and bioavailability related hurdles. Therefore, it is necessary to say that there is a potential source of antidiabetic drugs, even as an idea can be converted through application of advanced drug delivery system. This source can be further utilized to understand the safety and efficacy in details via clinical research. This will support the development of several value-added nutraceutical and pharmaceutical products with potent antihyperglycemic action in the near future.
Author Contributions
Conceptualization, S.S.R. and B.K.; methodology, N.K.S.; software, A.K.; validation, N.K.S., B.K. and A.K.; data curation, S.S.R.; writing—original draft preparation, S.S.R.; writing—review and editing, B.K.; visualization, N.K.S.; supervision, B.K.; project administration, N.K.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Acknowledgments
All authors acknowledge the administrative and technical support provided by DIT University, Dehradun, India for writing the current article.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
| ACC | Acetyl-CoA Carboxylase |
| AFABP | Adipocyte-specific Fatty Acid Binding Protein |
| AGEs | Advanced Glycation Endproducts |
| ALT | Alanine Aminotransferase |
| AMPK | AMP-activated Protein Kinase |
| ATF-4 | Activating Transcription Factor-4 |
| BDNF | Brain-Derived Neurotrophic Factor |
| BMI | Body Mass Index |
| Bw | Body weight |
| cAMP | Cyclic Adenosine Monophosphate |
| CDKN1B | Cyclin-Dependent Kinase Inhibitor 1B |
| CHOP | Cyclophosphamide, Hydroxyl daunorubicin, Oncovin and Prednisone |
| CKD | Chronic Kidney Disease |
| CVD | Cardio Vascular Diseases |
| DFU | Diabetic Foot Ulcer |
| DKA | Diabetic Keto Acidosis |
| DME | Diabetic Macular Edema |
| DN | Diabetic Nephropathy |
| DNMT1 | DNA (cytosine-5)-methyl transferase 1 |
| DPN | Diabetic Peripheral Neuropathy |
| DR | Diabetic Retinopathy |
| DRN | Diabetic Retinal Neurodegeneration |
| ED | Erectile Dysfunction |
| EGCG | Epigallocatechin |
| eNOS | Endothelial Nitric Oxide Synthase |
| ERK | Extracellular signal-regulated kinase |
| FFA | Free Fatty Acids |
| Fox | Forkhead Box |
| FPG | Fasting Plasma Glucose |
| FSD | Female Sexual Dysfunction |
| GDM | Gestational Diabetes Mellitus |
| GFR | Glomerular Filtration Rate |
| GLUT4 | Glucose Transporter 4 IL-1beta |
| GSH | Glutathione |
| GWAS | Genome-Wide Association Studies |
| HbA1 | Hemoglobin A1 |
| HbA1c | Hemoglobin A1c |
| HDL | High Density Lipoprotein |
| HG | High Glucose |
| HIF-1 | Hypoxia-Inducible Factors-1 |
| HO-1 | Heme Oxygenase |
| HUVECs | Human Umbilical Vein Endothelial Cells |
| ICAM-1 | Intercellular Adhesion Molecules |
| IDDM | Insulin Dependent Diabetes Mellitus |
| IDF | International Diabetes Federation |
| IL-1beta | Interleukin 1-beta |
| IL-6 | Interleukin-6 |
| iNOS | Inducible Nitric Oxide Synthase |
| INS | Insulin |
| INSR | Insulin Receptor |
| IRMA | Intra Retinal Microvascular Abnormalities. |
| IRSs | Insulin Receptor Substrate |
| JNK | c-Jun N-terminal kinase |
| LDH | Lactate Dehydrogenase |
| LDL | Low-Density Lipoprotein |
| LPS | Lipopolysaccharides |
| LXR | Liver X Receptor |
| MAPK | Mitogen Activated Protein Kinase |
| MCP-1 | Monocyte Chemotactic Protein-1 |
| MDA | Melondialdehyde |
| MG | Methyl Glyoxal |
| MODY | Maturity Onset Diabetes of the Young |
| NAD | Nicotinamide Adenine Dinucleotide |
| NADH | Nicotinamide Adenine Dinucleotide Hydrogen |
| NF-kB | Nuclear Factor Kappa B |
| NGF | Nerve Growth Factor |
| NIDDM | Non-Insulin Dependent Diabetes Mellitus |
| NLRP3 | NLR family pyrin domain containing 3 |
| NPDR | Nonproliferative Diabetic Retinopathy |
| Nrf2 | Nuclear factor erythroid 2 related factor |
| OGTT | Oral Glucose Tolerance Test |
| PAI-1 | Plasminogen activator inhibitor-1 |
| PDR | Proliferative Diabetic Retinopathy |
| PI3K-PKB/Akt | Phosphoinositide-3-kinase–protein kinase B/Akt |
| PKA | Protein kinase |
| PKC | Protein kinase C |
| PKR | Protein kinase R |
| PPARγ | Peroxisome proliferation-activated receptor gamma |
| PTP-1B | Protein tyrosinase phosphatase-1B |
| PWT | Paw withdrawal threshold |
| QE | Quercetin |
| RAGE | Receptor for advanced glycation endproducts |
| RGC-5 | Retinal ganglion cell-5 |
| RhoA | Ras Homolog Family Member A |
| ROS | Reactive Oxygen Species |
| RPTEC | Renal proximal tubule epithelial cells |
| SOD | Superoxide dismutase |
| SREBP-1c | Sterol Regulatory Element Binding Protein 1c |
| STZ | Streptozotocin |
| T1DM | Type 1 diabetes mellitus |
| T2DM | Type 2 diabetes mellitus |
| TBARS | Thio-barbituric acid reactive substances |
| TFG-1 | Transforming growth factor |
| TGF-β | Transforming growth factor beta |
| TNF-α | Tumor Necrosis Factor alpha |
| mTOR | Mammalian target of rapamycin |
| VEGF | Vascular endothelial growth factor |
| WHO | World Health Organization |
References
- WHO Diabetes Programme. WHO. 2019. Available online: https://www.who.int/diabetes/en/ (accessed on 18 May 2022).
- Canivell, S.; Gomis, R. Diagnosis and classification of autoimmune diabetes mellitus. Autoimmun. Rev. 2014, 13, 403–407. [Google Scholar] [CrossRef] [PubMed]
- IDF Website. International Diabetes Federation. 2022. Available online: https://idf.org/ (accessed on 18 May 2022).
- Saeedi, P.; Petersohn, I.; Salpea, P.; Malanda, B.; Karuranga, S.; Unwin, N.; Colagiuri, S.; Guariguata, L.; Motala, A.A.; Ogurtsova, K.; et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9th edition. Diabetes Res. Clin. Pract. 2019, 157, 107843. [Google Scholar] [CrossRef]
- International Diabetes Federation. IDF Diabetes Atlas, 8th ed.; International Diabetes Federation: Brussels, Belgium, 2017; Available online: https://diabetesatlas.org/upload/resources/previous/files/8/IDF_DA_8e-EN-final.pdf (accessed on 18 May 2022).
- Khan, M.A.B.; Hashim, M.J.; King, J.K.; Govender, R.D.; Mustafa, H.; Kaabi, J.A. Epidemiology of Type 2 Diabetes—Global Burden of Disease and Forecasted Trends. J. Epidemiol. Glob. Health 2020, 10, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Khunti, K.; Davies, M.J. Type 2 diabetes. Lancet Lond Engl. 2017, 389, 2239–2251. [Google Scholar] [CrossRef]
- Baynest, H.W. Classification, Pathophysiology, Diagnosis and Management of Diabetes Mellitus. J. Diabetes Metab. 2015, 6, 541. [Google Scholar] [CrossRef]
- Saisho, Y. Importance of Beta Cell Function for the Treatment of Type 2 Diabetes. J. Clin. Med. 2014, 3, 923–943. [Google Scholar] [CrossRef]
- Artasensi, A.; Pedretti, A.; Vistoli, G.; Fumagalli, L. Type 2 Diabetes Mellitus: A Review of Multi-Target Drugs. Molecules 2020, 25, 1987. [Google Scholar] [CrossRef]
- DiMeglio, L.A.; Molina, C.E.; Oram, R.A. Type 1 diabetes. Lancet 2018, 391, 2449–2462. [Google Scholar] [CrossRef]
- Roberts, M.S.; Burbelo, P.D.; Spichtig, D.E.; Perwad, F.; Romero, C.J.; Ichikawa, S.; Farrow, E.; Econs, M.J.; Guthrie, L.C.; Collins, M.T.; et al. Autoimmune hyperphosphatemic tumoral calcinosis in a patient with FGF23 autoantibodies. J. Clin. Investig. 2018, 128, 5368–5373. [Google Scholar] [CrossRef]
- Pozzilli, P.; Pieralice, S. Latent Autoimmune Diabetes in Adults: Current Status and New Horizons. Endocrinol. Metab. 2018, 33, 147–159. [Google Scholar] [CrossRef]
- Lucier, J.; Weinstock, R.S. Diabetes Mellitus Type 1. In StatPearls; StatPearls: Treasure Island, FL, USA, 2021. [Google Scholar]
- Kolb, H.; Martin, S. Environmental/lifestyle factors in the pathogenesis and prevention of type 2 diabetes. BMC Med. 2017, 15, 131. [Google Scholar] [CrossRef]
- Esmaili, S.; Hemmati, M.; Karamian, M. Physiological role of adiponectin in different tissues: A review. Arch. Physiol. Biochem. 2020, 126, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Sears, B.; Perry, M. The role of fatty acids in insulin resistance. Lipids Health Dis. 2015, 14, 121. [Google Scholar] [CrossRef] [PubMed]
- Verges, B. Pathophysiology of diabetic dyslipidaemia: Where are we? Diabetologia 2015, 58, 886–899. [Google Scholar] [CrossRef] [PubMed]
- Ference, B.A.; Ginsberg, H.N.; Graham, I.; Ray, K.K.; Packard, C.J.; Bruckert, E.; Hegele, R.A.; Krauss, R.M.; Raal, F.J.; Schunkert, H.; et al. Low-density lipoproteins cause atherosclerotic cardiovascular disease. Evidence from genetic, epidemiologic, and clinical studies. A consensus statement from the European Atherosclerosis Society Consensus Panel. Eur. Heart J. 2017, 38, 2459–2472. [Google Scholar] [CrossRef]
- Zheng, Y.; Ley, S.H.; Hu, F.B. Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 2017, 14, 88–98. [Google Scholar] [CrossRef]
- Lim, A.K.H. Diabetic nephropathy—Complications and treatment. Int. J. Nephrol. Renov. Dis. 2014, 7, 361–381. [Google Scholar] [CrossRef]
- Vallon, V.; Komers, R. Pathophysiology of the Diabetic Kidney. Compr. Physiol. 2011, 1, 1175–1232. [Google Scholar] [CrossRef]
- Anders, H.J.; Huber, T.B.; Isermann, B.; Schiffer, M. CKD in diabetes: Diabetic kidney disease versus nondiabetic kidney disease. Nat. Rev. Nephrol. 2018, 14, 361–377. [Google Scholar] [CrossRef]
- Faselis, C.; Katsimardou, A.; Imprialos, K.; Deligkaris, P.; Kallistratos, M.; Dimitriadis, K. Microvascular Complications of Type 2 Diabetes Mellitus. Curr. Vasc. Pharmacol. 2020, 18, 117–124. [Google Scholar] [CrossRef]
- Himasa, F.I.; Singhal, M.; Ojha, A.; Kumar, B. Prospective for Diagnosis and Treatment of Diabetic Retinopathy. Curr. Pharm. Des. 2022, 28, 560–569. [Google Scholar] [CrossRef]
- Wang, Y.; Li, W.; Peng, W.; Zhou, J.; Liu, Z. Acupuncture for postherpetic neuralgia: Systematic review and meta-analysis. Medicine 2018, 97, e11986. [Google Scholar] [CrossRef] [PubMed]
- Yau, J.W.; Rogers, S.L.; Kawasaki, R.; Lamoureux, E.L.; Kowalski, J.W.; Bek, T. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care 2012, 35, 556–564. [Google Scholar] [CrossRef] [PubMed]
- Sabanayagam, C.; Banu, R.; Chee, M.L. Incidence and progression of diabetic retinopathy: A systematic review. Lancet Diabetes Endocrinol. 2019, 7, 140–149. [Google Scholar] [CrossRef]
- Sloan, G.; Shillo, P.; Selvarajah, D.; Wu, J.; Wilkinson, L.D.; Tracey, I.; Anand, P.; Tesfaye, S. A new look at painful diabetic neuropathy. Diabetes Res. Clin. Pract. 2018, 144, 177–191. [Google Scholar] [CrossRef]
- Edwards, J.L.; Vincent, A.; Cheng, T.; Feldman, E.L. Diabetic Neuropathy: Mechanisms to Management. Pharmacol Ther. 2008, 120, 1–34. [Google Scholar] [CrossRef]
- NIH Consensus Development Panel on Impotence, Impotence—NIH Consensus Conference. JAMA 1993, 270, 83–90. Available online: https://pubmed.ncbi.nlm.nih.gov/8510302/ (accessed on 18 May 2022). [CrossRef]
- Imprialos, K.P.; Stavropoulos, K.; Doumas, M.; Tziomalos, K.; Karagiannis, A.; Athyros, V.G. Sexual dysfunction, cardiovascular risk and effects of pharmacotherapy. Curr. Vasc. Pharmacol. 2018, 16, 130–142. [Google Scholar] [CrossRef]
- Corona, G.C.; Giorda, B.; Cucinotta, D.; Guida, P.; Nada, E. Sexual dysfunction at the onset of type 2 diabetes: The interplay of depression, hormonal and cardiovascular factors. J. Sex. Med. 2014, 11, 2065–2073. [Google Scholar] [CrossRef]
- Pontiroli, A.E.; Cortelazzi, D.; Morabito, A. Female sexual dysfunction and diabetes: A systematic review and meta-analysis. J. Sex. Med. 2013, 10, 1044–1045. [Google Scholar] [CrossRef]
- Maseroli, E.; Scavello, I.; Vignozzi, L. Cardiometabolic risk and female sexuality-part I. risk factors and potential pathophysiological underpinnings for female vasculogenic sexual dysfunction syndromes. Sex. Med. Rev. 2018, 6, 508–524. [Google Scholar] [CrossRef] [PubMed]
- Einarson, T.R.; Acs, A.; Ludwig, C.; Panton, U.H. Prevalence of cardiovascular disease in type 2 diabetes: A systematic literature review of scientific evidence from across the world in 2007–2017. Cardiovasc. Diabetol. 2018, 17, 83. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Mittal, S.; Aggarwal, R.; Chauhan, M.K. Diabetes and cardiovascular disease: Inter-relation of risk factors and treatment. Future J. Pharm. Sci. 2020, 6, 130. [Google Scholar] [CrossRef]
- Forbes, J.M.; Cooper, M.E. Mechanisms of Diabetic Complications. Physiol. Rev. 2013, 93, 137–188. [Google Scholar] [CrossRef]
- Segers, V.F.M.; Keulenaer, G.W.D. Pathophysiology of diastolic dysfunction in chronic heart failure. Future Cardiol. 2013, 9, 711–720. [Google Scholar] [CrossRef]
- Coffey, L.; Mahon, C.; Gallagher, P. Perceptions and experiences of diabetic foot ulceration and foot care in people with diabetes: A qualitative meta-synthesis. Int. Wound J. 2019, 16, 183–210. [Google Scholar] [CrossRef]
- Acosta, J.B.; Montequin, J.F.; Perez, C.V.; Gutierrez, W.S.; Marí, Y.M.; Ojalvo, A.G.; Cama, V.F.; Herrera, D.G.D.B.; Mayola, M.F.; Vázquez, H.P.S.E.P.; et al. Diabetic Foot Ulcers and Epidermal Growth Factor: Revisiting the Local Delivery Route for a Successful Outcome. BioMed Res. Int. 2017, 2017, 2923759. [Google Scholar] [CrossRef]
- Amin, N.; Doupis, J. Diabetic foot disease: From the evaluation of the “foot at risk” to the novel diabetic ulcer treatment modalities. World J. Diabetes 2016, 7, 153–164. [Google Scholar] [CrossRef]
- Ahmad, J. The diabetic foot. Diabetes Metab. Syndr. 2016, 10, 48–60. [Google Scholar] [CrossRef]
- Prabhakar, P.; Banerjee, M. Antidiabetic Phytochemicals: A comprehensive Review on Opportunities and Challenges in Targeted Therapy for Herbal Drug Development. Int. J. Pharm. Res. 2020, 14, 1673–1696. [Google Scholar] [CrossRef]
- Chaudhury, A.; Duvoor, C.; Dendi, V.S.R.; Kraleti, S.; Chada, A.; Ravilla, R.; Marco, A.; Shekhawat, N.S.; Montales, M.T.; Kuriakose, K.; et al. Clinical review of antidiabetic drugs: Implications for type 2 diabetes mellitus management. Front. Endocrinol. 2017, 8, 6. [Google Scholar] [CrossRef] [PubMed]
- Baska, A.; Leis, K.; Gałązka, P. Berberine in the Treatment of Diabetes Mellitus: A Review. Endocr. Metab. Immune Disord. Drug Targets 2021, 21, 1379–1386. [Google Scholar] [CrossRef] [PubMed]
- Sahibzada, M.U.K.; Sadiq, A.; Faidah, H.S.; Khurram, M.; Amin, M.U.; Haseeb, A.; Kakar, M. Berberine nanoparticles with enhanced in vitro bioavailability: Characterization and antimicrobial activity. Drug Des. Dev. Ther. 2018, 12, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Miao, Y.Q.; Fan, D.J.; Yang, S.S.; Lin, X.; Meng, L.K.; Tang, X. Bioavailability study of berberine and the enhancing effects of TPGS on intestinal absorption in rats. AAPS PharmSciTech 2011, 12, 705–711. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Gao, Z.; Liu, D.; Liu, Z.; Ye, J. Berberine improves glucose metabolism through induction of glycolysis. Am. J. Physiol. Endocrinol. Metab. 2008, 294, E148–E156. [Google Scholar] [CrossRef] [PubMed]
- Cicero, A.F.G.; Tartagni, E. Antidiabetic properties of berberine: From cellular pharmacology to clinical effects. Hosp. Pract. 2012, 40, 56–63. [Google Scholar] [CrossRef]
- Dong, H.; Wang, N.; Zhao, L.; Lu, F. Berberine in the treatment of type 2 diabetes mellitus: A systemic review and meta-analysis. Evid. Based Complement. Altern. Med. 2012, 2012, 591654. [Google Scholar] [CrossRef]
- Zhang, M.; Chen, L. Berberine in type 2 diabetes therapy: A new perspective for an old antidiarrheal drug? Acta Pharm. Sin. B 2012, 2, 379–386. [Google Scholar] [CrossRef]
- Chang, W.; Chen, L.; Hatch, G.M. Berberine as a therapy for type 2 diabetes and its complications: From mechanism of action to clinical studies. Biochem. Cell Biol. 2015, 93, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Khan, H. Berberine: As a therapeutic target for treating obese diabetes. SciForschen J. Diabetes Res. Ther. 2016, 2, 1–2. [Google Scholar] [CrossRef]
- Mahmoud, A.M.; Abdel-Rahman, M.M.; Bastawy, N.A.; Eissa, H.M. Modulatory effect of berberine on adipose tissue PPARγ, adipocytokines and oxidative stress in high fat diet/streptozotocin-induced diabetic rats. J. Appl. Pharm. Sci. 2017, 7, 1–10. [Google Scholar] [CrossRef][Green Version]
- Zhao, L.; Cang, Z.; Sun, H.; Nie, X.; Wang, N.; Lu, Y. Berberine improves glucogenesis and lipid metabolism in nonalcoholic fatty liver disease. BMC Endocr. Disord. 2017, 17, 13. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.; Chen, H.; Zhang, X.; Lou, W.; Zhang, P.; Qiu, Y.; Zhang, C.; Wang, Y.; Jing, W. The Effect of Berberine on Metabolic Profiles in Type 2 Diabetic Patients: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Oxidative Med. Cell. Longev. 2021, 2021, 2074610. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Liu, D.; Wei, G.; Ge, H. Berberine and Metformin in the Treatment of Type 2 Diabetes Mellitus: A Systemic Review and Meta-Analysis of Randomized Clinical Trials. Health 2021, 13, 1314–1329. [Google Scholar] [CrossRef]
- Yoshinari, O.; Igarashi, K. Chapter 85—Antidiabetic Effects of Trigonelline: Comparison with Nicotinic Acid. In Coffee in Health and Disease Prevention; Academic Press: Cambridge, MA, USA, 2015; pp. 765–775. [Google Scholar] [CrossRef]
- Mohammed, A.; Islam, M.S. Spice-Derived Bioactive Ingredients: Potential Agents or Food Adjuvant in the Management of Diabetes Mellitus. Front. Pharm. 2018, 9, 893. [Google Scholar] [CrossRef]
- Cheng, Z.X.; Wu, J.J.; Liu, Z.Q.; Lin, N. Development of a hydrophilic interaction chromatography-UPLC assay to determine trigonelline in rat plasma and its application in a pharmacokinetic study. Chin. J. Nat. Med. 2013, 11, 164–170. [Google Scholar] [CrossRef]
- Ghule, A.E.; Jadhav, S.S.; Bodhankar, S.L. Trigonelline ameliorates diabetic hypertensive nephropathy by suppression of oxidative stress in kidney and reduction in renal cell apoptosis and fibrosis in streptozotocin induced neonatal diabetic (nSTZ) rats. Int. Immunopharmacol. 2012, 14, 740–748. [Google Scholar] [CrossRef]
- Zhou, J.; Zhou, S. Protection of Trigonelline on Experimental Diabetic Peripheral Neuropathy. Evid. Based Complement. Altern. Med. 2012, 2012, 164219. [Google Scholar] [CrossRef]
- Hamden, K.; Bengara, A.; Amri, Z.; Elfeki, A. Experimental diabetes treated with trigonelline: Effect on key enzymes related to diabetes and hypertension, β-cell and liver function. Mol. Cell. Biochem. 2013, 381, 85–94. [Google Scholar] [CrossRef]
- Subramanian, S.P.; Prasath, G.S. Antidiabetic and antidyslipidemic nature of trigonelline, a major alkaloid of fenugreek seeds studied in high-fat-fed and low-dose streptozotocin-induced experimental diabetic rats. Biomed. Prev. Nutr. 2014, 4, 475–480. [Google Scholar] [CrossRef]
- Zhou, J.; Du, X.; Zhang, Z.; Qian, G. Trigonelline Inhibits Inflammation and Protects β Cells to Prevent Fetal Growth Restriction during Pregnancy in a Mouse Model of Diabetes. Pharmacology 2017, 100, 209–217. [Google Scholar] [CrossRef]
- Li, Y.; Li, Q.; Wang, C.; Lou, Z.; Li, Q. Trigonelline reduced diabetic nephropathy and insulin resistance in type 2 diabetic rats through peroxisome proliferator-activated receptor-γ. Exp. Ther. Med. 2019, 18, 1331–1337. [Google Scholar] [CrossRef]
- Singh, S.; Mazumder, A.; Chakaraborthy, G.S. Potential herbs against diabetes mellitus—An update. Int. J. Pharm. Sci. Res. 2019, 10, 3619–3626. [Google Scholar] [CrossRef]
- Yang, S.; Mi, J.; Liu, Z.; Wang, B.; Xia, X.; Wang, R.; Liu, Y.; Li, Y. Pharmacokinetics, Tissue Distribution, and Elimination of Three Active Alkaloids in Rats after Oral Administration of the Effective Fraction of Alkaloids from Ramulus Mori, an Innovative Hypoglycemic Agent. Molecules 2017, 22, 1616. [Google Scholar] [CrossRef]
- Gómez, L.; Molinar-Toribio, E.; Calvo-Torras, M.A.; Adelantado, C.; Juan, M.E.; Planas, J.; Cañas, X.; Lozano, C.; Pumarola, S.; Clapés, P.; et al. D-Fagomine lowers postprandial blood glucose and modulates bacterial adhesion. Br. J. Nutr. 2012, 107, 1739–1746. [Google Scholar] [CrossRef]
- Fang, C.; Zhang, B.; Han, L.; Gao, C.; Wang, M. d-Fagomine Attenuates High Glucose-Induced Endothelial Cell Oxidative Damage by Upregulating the Expression of PGC-1α. J. Agric. Food Chem. 2018, 66, 2758–2764. [Google Scholar] [CrossRef]
- Molinar-Toribio, E.; Pérez-Jiménez, J.; Ramos-Romero, S.; Gómez, L.; Taltavull, N.; Nogués, M.R.; Adeva, A.; Jáuregui, O.; Joglar, J.; Clapés, P.; et al. D-Fagomine attenuates metabolic alterations induced by a high-energy-dense diet in rats. Food Funct. 2015, 6, 2614–2619. [Google Scholar] [CrossRef]
- Parida, I.S.; Takasu, S.; Nakagawa, K.A. Comprehensive review on the production, pharmacokinetics and health benefits of mulberry leaf iminosugars: Main focus on 1-deoxynojirimycin, d-fagomine, and 2-O-ɑ-d-galactopyranosyl-DNJ. Crit. Rev. Food Sci. Nutr. 2021. Online ahead of print. [Google Scholar] [CrossRef]
- Chen, F.; Nakashima, N.; Kimura, I.; Kimura, M.; Asano, N.; Koya, S. Potentiating effects on pilocarpine-induced saliva secretion, by extracts and N-containing sugars derived from mulberry leaves, in streptozocin-diabetic mice. Biol. Pharm. Bull. 1995, 18, 1676–1680. [Google Scholar] [CrossRef]
- Tambe, V.S.; Waichal, D.D.; Chanshetty, R.R. Bioactivity enhanced isolated carpaine from Carica papaya leaves for platelet stimulating activity. Indian J. Pharm. Sci. 2021, 83, 723–731. [Google Scholar] [CrossRef]
- Hameda, A.N.E.; Abouelela, M.E.; Zowalaty, A.E.E.; Badrf, M.M.; Abdelkader, M.S.A. Chemical constituents from Carica papaya Linn. leaves as potential cytotoxic, EGFRwt and aromatase (CYP19A) inhibitors; a study supported by molecular docking. RSC Adv. 2022, 12, 9154–9162. [Google Scholar] [CrossRef]
- Bharti, S.K.; Krishnan, S.; Kumar, A. Antidiabetic phytoconstituents and their mode of action on metabolic pathways. Ther. Adv. Endocrinol. Metab. 2018, 9, 81–100. [Google Scholar] [CrossRef]
- Fazal, J.; Naz, L.; Sohail, S.; Yasmeen, G.; Khan, N.I.; Zehra, N. Anti-diabetic activity of Carica Papaya Linn in Alloxan-Induced diabetic rats. Int. J. Endorsing Health Sci. Res. 2022, 1, 42–48. [Google Scholar] [CrossRef]
- Dey, P.; Singh, J.; Suluvoy, J.K.; Dilip, K.J.; Nayak, J. Utilization of Swertia chirayita Plant Extracts for Management of Diabetes and Associated Disorders: Present Status, Future Prospects and Limitations. Nat. Prod. Bioprospect. 2020, 10, 431–443. [Google Scholar] [CrossRef]
- Vaidya, H.; Goyal, R.K.; Cheema, S.K. Anti-diabetic activity of swertiamarin is due to an active metabolite, gentianine, that upregulates PPAR-γ gene expression in 3T3-L1 cells. Phytother. Res. 2013, 27, 624–627. [Google Scholar] [CrossRef]
- Tiong, S.H.; Looi, C.Y.; Arya, A.; Wong, W.F.; Hazni, H.; Mustafa, M.R.; Awang, K. Vindogentianine, a hypoglycemic alkaloid from Catharanthus roseus (L.) G. Don (Apocynaceae). Fitoterapia 2015, 102, 182–188. [Google Scholar] [CrossRef]
- Yan, X.; Huang, N. Effects of Three Different Types of Aloin on Optical, Mechanical, and Antibacterial Properties of Waterborne Coating on Tilia europaea Surface. Coating 2021, 11, 1537. [Google Scholar] [CrossRef]
- Park, M.; Kwon, H.; Sung, M. Intestinal absorption of aloin, aloe-emodin, and aloesin; A comparative study using two in vitro absorption models. Nutr Res Pr. 2009, 3, 9–14. [Google Scholar] [CrossRef]
- Zhong, R.; Chen., L.; Liu, Y.; Xie, S.; Li, S.; Liu, B.; Zhao, C. Anti-diabetic effect of aloin via JNK-IRS1/PI3K pathways and regulation of gut microbiota. Food Sci. Hum. Wellness 2022, 11, 189–198. [Google Scholar] [CrossRef]
- Araya, T.Y.; Karim, A.; Hailu, G.S.; Periasamy, G.; Kahsay, G. Antihyperglycemic Activity of TLC Isolates from the Leaves of Aloe megalacantha Baker in Streptozotocin-Induced Diabetic Mice. Diabetes Metab. Syndr. Obes. 2021, 14, 1153–1166. [Google Scholar] [CrossRef]
- Froldi, G.; Baronchelli, F.; Marin, E.; Grison, M. Antiglycation Activity and HT-29 Cellular Uptake of Aloe-Emodin, Aloin, and Aloe arborescens Leaf Extracts. Molecules 2019, 24, 2128. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, S.; Narasimhaiah, A.L.; Kundu, S.; Anand, S. Anti-hyperglycemic study of natural inhibitors for Insulin receptor. Bioinformation 2012, 8, 1195–1201. [Google Scholar] [CrossRef] [PubMed]
- Beppu, H.; Shimpo, K.; Chihara, T.; Tamai, I.; Nomoto-Yamaji, S.; Ozaki, S.; Ito, S.; Kuzuya, H. Inhibitory effects of aloe carboxypeptidase fraction on streptozotocin-induced enhancement of vascular permeability in the pancreatic islets. Phytomedicine 2006, 13, 49–60. [Google Scholar] [CrossRef] [PubMed]
- Pérez, Y.Y.; Jiménez-Ferrer, E.; Zamilpa, A.; Hernández-Valencia, M.; Alarcón-Aguilar, F.J.; Tortoriello, J.; Román-Ramos, R. Effect of a polyphenol-rich extract from Aloe vera gel on experimentally induced insulin resistance in mice. Am. J. Chin. Med. 2007, 35, 1037–1046. [Google Scholar] [CrossRef]
- Eid, S.A.; Adams, M.; Scherer, T.; Torres-Gómez, H.; Hackl, M.T.; Kaplanian, M.; Riedl, R.; Luger, A.; Fürnsinn, C. Emodin, a compound with putative antidiabetic potential, deteriorates glucose tolerance in rodents. Eur. J. Pharmacol. 2017, 798, 77–84. [Google Scholar] [CrossRef]
- Wang, S.; Chen, T.; Chen, R.; Hu, Y.; Chen, M.; Wang, Y. Emodin loaded solid lipid nanoparticles: Preparation, characterization and antitumor activity studies. Int. J. Pharm. 2012, 430, 238–246. [Google Scholar] [CrossRef]
- Cao, X. A systematic evaluation of biopharmaceutics classification system of main efficacy and toxic ingredients in Polygoni Multiflori Radix Praeparata. Chin. Tradit. Herb. Drugs 2020, 24, 3451–3456. [Google Scholar]
- Duan, H.G.; Wei, Y.H.; Li, B.X.; Qin, H.; Wu, X. Improving the dissolution and oral bioavailability of the poorly water-soluble drug aloe-emodin by solid dispersion with polyethylene glycol 6000. Drug Dev. Res. 2009, 70, 363–369. [Google Scholar] [CrossRef]
- Jangra, S.; Sharma, B.; Singh, S. Aloe-emodin-loaded SBA-15 and its in vitro release properties and cytotoxicity to cervical cancer cells. Mater. Res. Innov. 2021, 25, 264–275. [Google Scholar] [CrossRef]
- Dong, X.; Fu, J.; Yin, X.; Cao, S.; Li, X.; Lin, L.; Ni, H.J. Emodin: A Review of its Pharmacology, Toxicity and Pharmacokinetics. Phytother. Res. 2016, 30, 1207–1218. [Google Scholar] [CrossRef]
- Dong, X.; Zeng, Y.; Liu, Y.; You, L.; Yin, X.; Fu, J.; Ni, J. Aloe-emodin: A review of its pharmacology, toxicity, and pharmacokinetics. Phytother. Res. 2020, 34, 270–281. [Google Scholar] [CrossRef] [PubMed]
- Martorell, M.; Castro, N.; Victoriano, M.; Capó, X.; Tejada, S.; Vitalini, S.; Pezzani, R.; Sureda, A. An Update of Anthraquinone Derivatives Emodin, Diacerein, and Catenarin in Diabetes. Evid. Based Complement. Altern. Med. 2021, 2021, 3313419. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Hu, Y.; Fu, Y.; Wang, R.; Zhang, H.; Li, M.; Li, Z.; Zhang, Y.; Xuan, L.; Li, X.; et al. Emodin improves glucose metabolism by targeting microRNA-20b in insulin-resistant skeletal muscle. Phytomedicine 2019, 59, 152758. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.; Li, S.; Li, X.; Liu, R. Advances in the study of emodin: An update on pharmacological properties and mechanistic basis. Chin. Med. 2021, 16, 102. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Zhang, H.; Li, X.; Yang, G.; Ru, J.; Liu, T. Emodin protects hyperglycemia-induced injury in PC-12 cells by up-regulation of miR-9. Mol. Cell. Endocrinol. 2018, 474, 194–200. [Google Scholar] [CrossRef]
- Chang, K.; Li, L.; Sanborn, T.M.; Shieh, B.; Lenhart, P.; Ammar, D.; LaBarbera, D.V.; Petrash, J.M. Characterization of Emodin as a Therapeutic Agent for Diabetic Cataract. J. Nat. Prod. 2016, 79, 1439–1444. [Google Scholar] [CrossRef]
- Liu, H.; Wang, Q.; Shi, G.; Yang, W.; Zhang, Y.; Chen, W.; Wan, S.; Xiong, F.; Wang, Z. Emodin Ameliorates Renal Damage and Podocyte Injury in a Rat Model of Diabetic Nephropathy via Regulating AMPK/mTOR-Mediated Autophagy Signaling Pathway. Diabetes Metab Syndr Obes. 2021, 14, 1253–1266. [Google Scholar] [CrossRef]
- Lee, W.; Ku, S.; Lee, D.; Lee, T.; Bae, J. Emodin-6-O-β-D--glucoside inhibits high-glucose-induced vascular inflammation. Inflammation 2014, 37, 306–313. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, R.; Lv., P.; Yang, J.; Deng, Y.; Xu, J.; Zhu, R.; Zhang, D.; Yang, Y. Emodin up-regulates glucose metabolism, decreases lipolysis, and attenuates inflammation in vitro. J. Diabetes 2015, 7, 360–368. [Google Scholar] [CrossRef]
- Mohammed, A.; Ibrahim, M.A.; Tajuddeen, N.; Aliyu, A.B.; Isah, M.B. Antidiabetic potential of anthraquinones: A review. Phytother. Res. 2020, 34, 486–504. [Google Scholar] [CrossRef]
- Gao, J.; Wang, F.; Wang, W.; Su, Z.; Guo, C.; Cao, S. Emodin suppresses hyperglycemia-induced proliferation and fibronectin expression in mesangial cells via inhibiting cFLIP. PLoS ONE 2014, 9, e93588. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Sheng, X.; Zhao, S.; Zou, L.; Han, X.; Gong, Y.; Yuan, H.; Shi, L.; Guo, L.; Jia, T.; et al. Nanoparticle-encapsulated emodin decreases diabetic neuropathic pain probably via a mechanism involving P2X3 receptor in the dorsal root ganglia. Purinergic Signal. 2017, 13, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Zhang, J.; Li, G.; Xu, H.; Yi, Y.; Wu, Q.; Song, M.; Bee, Y.M.; Huang, L.; Tan, M.; et al. Protection of vascular endothelial cells from high glucose-induced cytotoxicity by emodin. Biochem. Pharmacol. 2015, 94, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Feng, Y.; Huang, S.; Dou, W.; Zhang, S.; Chen, J.; Shen, Y.; Shen, J.; Leng, Y. Emodin, a natural product, selectively inhibits 11beta-hydroxysteroid dehydrogenase type 1 and ameliorates metabolic disorder in diet-induced obese mice. Br. J. Pharmacol. 2010, 161, 113–126. [Google Scholar] [CrossRef]
- Cao, Y.; Chang, S.; Dong, J.; Zhu, S.; Zheng, X.; Li, J.; Long, R.; Zhou, Y.; Cui, J.; Zhang, Y. Emodin ameliorates high-fat-diet induced insulin resistance in rats by reducing lipid accumulation in skeletal muscle. Eur. J. Pharmacol. 2016, 780, 194–201. [Google Scholar] [CrossRef]
- Lu, L.; Li, Y. Aloe-Emodin Ameliorates Diabetic Nephropathy by Targeting Interferon Regulatory Factor 4. Evid. Based Complement. Altern. Med. 2022, 2022, 2421624. [Google Scholar] [CrossRef]
- Chen, T.; Zheng, L.Y.; Xiao, W.; Gui, D.; Wang, X.; Wang, N. Emodin ameliorates high glucose induced-podocyte epithelial-mesenchymal transition in-vitro and in-vivo. Cell. Physiol. Biochem. 2015, 35, 1425–1436. [Google Scholar] [CrossRef]
- Wu, Z.; Chen, Q.; Ke, D.; Li, G.; Deng, W. Emodin protects against diabetic cardiomyopathy by regulating the AKT/GSK-3β signaling pathway in the rat model. Molecules 2014, 19, 14782–14793. [Google Scholar] [CrossRef]
- Xue, J.; Ding, W.; Liu, Y. Anti-diabetic effects of emodin involved in the activation of PPAR gamma on high-fat diet-fed and low dose of streptozotocin-induced diabetic mice. Fitoterapia 2010, 81, 173–177. [Google Scholar] [CrossRef]
- Jing, D.; Bai, H.; Yin, S. Renoprotective effects of emodin against diabetic nephropathy in rat models are mediated via PI3K/Akt/GSK-3β and Bax/caspase-3 signaling pathways. Exp. Ther. Med. 2017, 14, 5163–5169. [Google Scholar] [CrossRef]
- Li, X.; Liu, W.; Wang, Q.; Liu, P.; Deng, Y.; Lan, T.; Zhang, X.; Qiu, B.; Ning, H.; Huang, H. Emodin suppresses cell proliferation and fibronectin expression via p38MAPK pathway in rat mesangial cells cultured under high glucose. Mol. Cell. Endocrinol. 2009, 307, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Huang, H.; Liu, P.; Tang, F.; Qin, J.; Huang, W.; Chen, F.; Guo, F.; Liu, W.; Yang, B. Inhibition of phosphorylation of p38 MAPK involved in the protection of nephropathy by emodin in diabetic rats. Eur. J. Pharmacol. 2006, 553, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Qiao, G.; Li, B.; Chi, L.; Li, Z.; Lu, Y.; Yang, B. Hypoglycaemic and hypolipidemic effects of emodin and its effect on L-type calcium channels in dyslipidaemia-diabetic rats. Clin. Exp. Pharmacol. Physiol. 2009, 36, 29–34. [Google Scholar] [CrossRef]
- Arvindekar, A.; More, T.; Payghan, P.V.; Laddha, K.; Ghoshal, N. Evaluation of anti-diabetic and alpha glucosidase inhibitory action of anthraquinones from Rheum emodi. Food Funct. 2015, 6, 2693–2700. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Lu, Q.; Jiang, W.; Pei, X.; Sun, Y.; Hao, H.; Hao, K. Pharmacokinetics and pharmacodynamics of rhubarb anthraquinones extract in normal and disease rats. Biomed. Pharmacother. 2017, 91, 425–435. [Google Scholar] [CrossRef]
- Sohn, E.; Kim, J.; Kim, C.S.; Jo, K.; Kim, J.S. Extract of Rhizoma Polygonum cuspidatum reduces early renal podocyte injury in streptozotocin-induced diabetic rats and its active compound emodin inhibits methylglyoxal-mediated glycation of proteins. Mol. Med. Rep. 2015, 12, 5837–5845. [Google Scholar] [CrossRef]
- Hu, H.C.; Zheng, L.T.; Yin, H.Y.; Tao, Y.; Luo, X.Q.; Wei, K.S.; Yin, L.P. A Significant Association between Rhein and Diabetic Nephropathy in Animals: A Systematic Review and Meta-Analysis. Front. Pharmacol. 2019, 10, 1473. [Google Scholar] [CrossRef]
- Wang, J.; Qian, Y.; Qian, C.; Shi, F.; Yao, J.; Bi, X.; Chen, Z. A novel β-cyclodextrin-rhein conjugate for improving the water solubility and bioavailability of rhein. Carbohydr Res 2020, 490, 107958. [Google Scholar] [CrossRef]
- Li, G.; Chen, J.; Zhang, H.; Cao, X.; Sun, C.; Peng, F.; Yin, Y.; Lin, Z.; Yu, L.; Chen, Y.; et al. Update on Pharmacological Activities, Security, and Pharmacokinetics of Rhein. Evid. Based Complement. Alternat. Med. 2021, 2021, 4582412. [Google Scholar] [CrossRef]
- Pei, R.; Jiang, Y.; Lei, G.; Chen, J.; Liu, M.; Liu, S. Rhein Derivatives, A Promising Pivot? Mini Rev. Med. Chem. 2021, 21, 554–575. [Google Scholar] [CrossRef]
- Cheng, L.; Chen, Q.; Pi, R.; Chen, J. A research update on the therapeutic potential of rhein and its derivatives. Eur. J. Pharmacol. 2021, 899, 173908. [Google Scholar] [CrossRef]
- Waldmann, S.; Almukainzi, M.; Chacra, N.A.B.; Amidon, G.L.; Lee, B.J.; Feng, J.; Kanfer, I.; Zuo, J.Z.; Wei, H.; Bolger, M.B.; et al. Provisional Biopharmaceutical Classification of Some Common Herbs Used in Western Medicine. Mol. Pharm. 2012, 2, 815–822. [Google Scholar] [CrossRef] [PubMed]
- Cavallito, C.J.; Bailey, J.H. Allicin, the Antibacterial Principle of Allium sativum. I. Isolation, Physical Properties and Antibacterial Action. J. Am. Chem. Soc. 1944, 66, 1950–1951. [Google Scholar] [CrossRef]
- Subramanian, M.S.; Ms, G.N.; Nordin, S.A.; Thilakavathy, K.; Joseph, N. Prevailing Knowledge on the Bioavailability and Biological Activities of Sulphur Compounds from Alliums: A Potential Drug Candidate. Molecules 2020, 25, 4111. [Google Scholar] [CrossRef]
- Lawson, L.D.; Hunsaker, S.M. Allicin Bioavailability and Bioequivalence from Garlic Supplements and Garlic Foods. Nutrients 2018, 10, 812. [Google Scholar] [CrossRef]
- Arellano-Buendía, A.S.; Castañeda-Lara, L.G.; Loredo-Mendoza, M.L.; Fernando, E.; García-Arroyo, F.E.; Rojas-Morales, P.; Raúl Argüello-García, R.; Juárez-Rojas, J.G.; Tapia, E.; José Pedraza-Chaverri, J.; et al. Effects of Allicin on Pathophysiological Mechanisms during the Progression of Nephropathy Associated to Diabetes. Antioxidants 2020, 9, 1134. [Google Scholar] [CrossRef]
- Zhai, B.; Zhang, C.; Sheng, Y.; Zhao, C.; He, X.; Xu, W.; Huang, K.; Luo, Y. Hypoglycemic and hypolipidemic effect of S-allyl-cysteine sulfoxide (alliin) in DIO mice. Sci. Rep. 2018, 8, 3527. [Google Scholar] [CrossRef]
- Nasim, S.A.; Dhir, B.; Kapoor, R.; Fatima, S.; Mahmooduzzafar; Mujib, A. Alliin obtained from leaf extract of garlic grown under in situ conditions possess higher therapeutic potency as analyzed in alloxan-induced diabetic rats. Pharm. Biol. 2011, 49, 416–421. [Google Scholar] [CrossRef] [PubMed]
- EFSA FEEDAP Panel (EFSA Panel on Additives and Products or Substances used in Animal Feed). Scientific Opinion on the safety and efficacy of betaine (betaine anhydrous and betaine hydrochloride) as a feed additive for all animal species based on a dossier submitted by VITAC EEIG. EFSA J. 2013, 11, 3210. [Google Scholar] [CrossRef]
- Schwahn, B.C.; Hafner, D.; Hohlfeld, T.; Balkenhol, N.; Laryea, M.D.; Wendel, U. Pharmacokinetics of oral betaine in healthy subjects and patients with homocystinuria. Br. J. Clin. Pharmacol. 2003, 55, 6–13. [Google Scholar] [CrossRef]
- Szkudelska, K.; Szkudelski, T. The anti-diabetic potential of betaine. Mechanisms of action in rodent models of type 2 diabetes. Biomed. Pharmacother. 2022, 150, 112946. [Google Scholar] [CrossRef] [PubMed]
- Zhao, G.; He, F.; Wu, C.; Li, P.; Li, N.; Deng, J.; Zhu, G.; Ren, W.; Peng, Y. Betaine in inflammation: Mechanistic Aspects and Applications. Front. Immunol. 2018, 9, 1070. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, C.; Wehsling, M.; Mignon, M.L.; Wille, G.; Rey, Y.; Schnellbaecher, A.; Zabezhinsky, D.; Fischer, M.; Zimmer, A. Lactoyl leucine and isoleucine are bioavailable alternatives for canonical amino acids in cell culture media. Biotechnol. Bioeng. 2021, 118, 3395–3408. [Google Scholar] [CrossRef]
- Elovaris, R.A.; Bitarafan, V.; Agah, S.; Ullrich, S.S.; Lange, K.; Horowitz, M.; Feinle-Bisset, C.F. Comparative Effects of the Branched-Chain Amino Acids, Leucine, Isoleucine and Valine, on Gastric Emptying, Plasma Glucose, C-Peptide and Glucagon in Healthy Men. Nutrients 2021, 13, 1613. [Google Scholar] [CrossRef] [PubMed]
- Okun, J.G.; Rusu, P.M.; Chan, A.Y.; Wu, Y.; Yap, Y.W.; Sharkie, T.; Schumacher, J.; Schmidt, K.V.; Thomson, K.M.R.; Russell, R.D.; et al. Liver alanine catabolism promotes skeletal muscle atrophy and hyperglycaemia in type 2 diabetes. Nat. Metab. 2021, 3, 394–409. [Google Scholar] [CrossRef]
- Li, H.; Zhang, D.; Qu, W.; Wang, H.; Liu, Y.; Borjigdai, A.; Cui, J.; Dong, Z. Biopharmaceutics classification and absorption mechanisms primary study on four kinds of flavonoids. Zhongguo Zhong Yao Za Zhi 2016, 41, 1198–1203. [Google Scholar] [CrossRef]
- Barve, A.; Chen, C.; Hebbar, V.; Desiderio, J.; Saw, C.L.; Kong, A. Metabolism, oral bioavailability and pharmacokinetics of chemopreventive kaempferol in rats. Biopharm. Drug Dispos. 2009, 30, 356–365. [Google Scholar] [CrossRef]
- Vinayagam, R.; Xu, B. Antidiabetic properties of dietary flavonoids: A cellular mechanism review. Nutr. Metab. 2015, 12, 60. [Google Scholar] [CrossRef]
- Truzzi, F.; Tibaldi, C.; Zhang, Y.; Dinelli, G.; D’Amen, E. An Overview on Dietary Polyphenols and Their Biopharmaceutical Classification System (BCS). Int. J. Mol. Sci. 2021, 22, 5514. [Google Scholar] [CrossRef]
- Kaur, H.; Kaur, G. A Critical Appraisal of Solubility Enhancement Techniques of Polyphenols. J. Pharm. 2014, 2014, 180845. [Google Scholar] [CrossRef]
- Kaşıkcı, M.B.; Bağdatlıoğlu, N. Bioavailability of Quercetin. Curr. Res. Nutr. Food Sci. 2016, 4, 146–151. [Google Scholar] [CrossRef]
- ALTamimi, J.Z.; BinMowyna, M.N.; AlFaris, N.A.; Alagal, R.I.; El-Kott, A.F.; Al-Farga, A.M. Fisetin protects against streptozotocin-induced diabetic cardiomyopathy in rats by suppressing fatty acid oxidation and inhibiting protein kinase R. Saudi Pharm. J. 2021, 29, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Skiba, M.; Gasmi, H.; Milon, N.; Bounoure, F.; Malika, L.S. Water Solubility and Dissolution Enhancement of Fisetin by Spherical Amorphous Solid Dispersion in Polymer of Cyclodextrin. J. Adv. Biotechnol. Bioeng. 2021, 8, 1106. [Google Scholar] [CrossRef]
- Li, R.Z.; Ding, X.W.; Geetha, T.; Al-Nakkash, L.; Broderick, T.L.; Babu, J.R. Beneficial Effect of Genistein on Diabetes-Induced Brain Damage in the ob/ob Mouse Model. Drug Des. Dev. Ther. 2020, 14, 3325–3336. [Google Scholar] [CrossRef]
- Chen, F.; Peng, J.; Lei, D.; Liu, J.; Zhao, G. Optimization of genistein solubilization by κ-carrageenan hydrogel using response surface methodology. Food Sci. Hum. Wellness 2013, 2, 124–131. [Google Scholar] [CrossRef]
- Laddha, A.; Kulkarni, Y.A. Daidzein ameliorates diabetic retinopathy in experimental animals. Life Sci. 2020, 265, 118779. [Google Scholar] [CrossRef] [PubMed]
- Panizzon, G.P.; Bueno, F.G.; Nakamura, T.U.; Nakamura, C.V.; Filho, B.P.D. Manufacturing Different Types of Solid Dispersions of BCS Class IV Polyphenol (Daidzein) by Spray Drying: Formulation and Bioavailability. Pharmaceutics 2019, 11, 492. [Google Scholar] [CrossRef]
- Miura, A.; Sugiyama, C.; Sakakibara, H.; Simoi, K.; Goda, T. Bioavailability of isoflavones from soy products in equol producers and non-producers in Japanese women. J. Nutr. Intermed. Metab. 2016, 6, 41–47. [Google Scholar] [CrossRef]
- Khalid, A.; Naseem, I. 2022. Antidiabetic and antiglycating potential of chrysin is enhanced after nano formulation: An in vitro approach. J. Mol. Struct. 2022, 1261, 13906. [Google Scholar] [CrossRef]
- Baidya, D.; Kushwaha, J.; Mahadik, K.; Patil, S. Chrysin-loaded folate conjugated PF127-F68 mixed micelles with enhanced oral bioavailability and anticancer activity against human breast cancer cells. Drug Dev. Ind. Pharm. 2018, 45, 852–860. [Google Scholar] [CrossRef]
- Mohammadian, F.; Abhari, A.; Dariushnejad, H.; Nikanfar, A.; Soltanahmadi, Y.P.; Zarghami, N. Effects of Chrysin-PLGA-PEG Nanoparticles on Proliferation and Gene Expression of miRNAs in Gastric Cancer Cell Line. Iran. J. Cancer Prev. 2016, 9, e4190. [Google Scholar] [CrossRef] [PubMed]
- Crescenti, A.; Caimari, A.; Alcaide-Hidalgo, J.M.; Mariné-Casadó, R.; Valls, R.M.; Companys, J.; Salamanca, P.; Calderón-Pérez, L.; Pla-Pagà, L.; Pedret, A.; et al. Hesperidin Bioavailability Is Increased by the Presence of 2S-Diastereoisomer and Micronization-A Randomized, Crossover and Double-Blind Clinical Trial. Nutrients 2022, 14, 2481. [Google Scholar] [CrossRef]
- Rekha, S.S.; Pradeepkiran, J.A.; Bhaskar, M. Bioflavonoid hesperidin possesses the anti-hyperglycemic and hypolipidemic property in STZ induced diabetic myocardial infarction (DMI) in male Wister rats. J. Nutr. Intermed. Metab. 2019, 15, 58–64. [Google Scholar] [CrossRef]
- Lv, P.; Yu, J.; Xu, X.; Lu, T.; Xu, F. Eriodictyol inhibits high glucose-induced oxidative stress and inflammation in retinal ganglial cells. J. Cell. Biochem. 2019, 120, 5644–5651. [Google Scholar] [CrossRef]
- Rajan, V.K.; Muraleedharan, K.; Hussan, K.P.S. Structural Evaluation and Toxicological Study of a Bitter Masking Bioactive Flavanone, ‘Eriodictyol’. In Polyphenols: Prevention and Treatment of Human Disease; Elsevier: Amsterdam, The Netherlands, 2018. [Google Scholar] [CrossRef]
- Sharma, A.; Bhardwaj, P.; Arya, S.K. Naringin: A potential natural product in the field of biomedical applications. Carbohydr. Polym. Technol. Appl. 2021, 2, 100068. [Google Scholar] [CrossRef]
- Ji, P.; Yu, T.; Liu, Y.; Jiang, J.; Xu, J.; Zhao, Y.; Hao, Y.; Qiu, Y.; Zhao, W.; Wu, C. Naringenin-loaded solid lipid nanoparticles: Preparation, controlled delivery, cellular uptake, and pulmonary pharmacokinetics. Drug Des. Devel. Ther. 2016, 10, 911–925. [Google Scholar] [CrossRef]
- Khan, M.F.; Mathur, A.; Pandey, V.K.; Kakkar, P. Naringenin alleviates hyperglycemia-induced renal toxicity by regulating activating transcription factor 4-C/EBP homologous protein mediated apoptosis. J. Cell Commun. Signal. 2022, 16, 271–291. [Google Scholar] [CrossRef] [PubMed]
- Jain, D.; Bansal, M.K.; Dalvi, R.; Upganlawar, A.; Somani, R. Protective effect of diosmin against diabetic neuropathy in experimental rats. J. Integr. Med. 2014, 12, 35–41. [Google Scholar] [CrossRef]
- Maggioli, A. Chronic venous disorders: Pharmacological and clinical aspects of micronized purified flavonoid fraction. Phlebolymphology 2016, 23, 57–120. [Google Scholar]
- Russo, R.; Chandradhara, D.; Tommasi, N.D. Comparative Bioavailability of Two Diosmin Formulations after Oral Administration to Healthy Volunteers. Molecules 2018, 23, 2174. [Google Scholar] [CrossRef]
- Vanitha, P.; Uma, C.; Suganya, N.; Bhakkiyalakshmi, E.; Suriyanarayanan, S.; Gunasekaran, P.; Sivasubramanian, S.; Ramkumar, K.M. Modulatory effects of morin on hyperglycemia by attenuating the hepatic key enzymes of carbohydrate metabolism and β-cell function in streptozotocin-induced diabetic rats. Environ. Toxicol. Pharmacol. 2014, 37, 326–335. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Li, J.; Ju, Y.; Fu, Y.; Gong, T.; Zhang, Z. Mechanism of enhanced oral absorption of morin by phospholipid complex based self-nanoemulsifying drug delivery system. Mol. Pharm. 2015, 12, 504–513. [Google Scholar] [CrossRef] [PubMed]
- Rajput, S.A.; Wang, X.; Yan, H. Morin hydrate: A comprehensive review on novel natural dietary bioactive compound with versatile biological and pharmacological potential. Biomed. Pharmacother. 2021, 138, 111511. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Yang, Y.; Ning, E.; Peng, Y.; Zhang, J. Mechanisms of poor oral bioavailability of flavonoid Morin in rats: From physicochemical to biopharmaceutical evaluations. Eur. J. Pharm. Sci. 2019, 128, 290–298. [Google Scholar] [CrossRef]
- Abuohashish, H.M.; AlAsmari, A.F.; Mohany, M.; Ahmed, M.M.; Al-Rejaie, S.S. Supplementation of Morin Restores the Altered Bone Histomorphometry in Hyperglycemic Rodents via Regulation of Insulin/IGF-1 Signaling. Nutrients 2021, 13, 2365. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, D.; Huang, Y.; Gao, Y.; Qian, S. Biopharmaceutics classification and intestinal absorption study of apigenin. Int. J. Pharm. 2012, 436, 311–317. [Google Scholar] [CrossRef]
- Zhang, B.; Sun, W.; Yu, N.; Sun, J.; Yu, X.; Li, X.; Xing, Y.; Yan, D.; Ding, Q.; Xiu, Z.; et al. Anti-diabetic effect of baicalein is associated with the modulation of gut microbiota in streptozotocin and high-fat-diet induced diabetic rats. J. Funct. Foods 2018, 46, 256–267. [Google Scholar] [CrossRef]
- Jakab, G.; Bogdan, D.; Mazak, K.; Deme, R.; Mucsi, Z.; Mandity, I.M.; Noszal, B.; Szabo, N.K.; Antal, I. Physicochemical Profiling of Baicalin Along with the Development and Characterization of Cyclodextrin Inclusion Complexes. AAPS PharmSciTech 2019, 20, 314. [Google Scholar] [CrossRef]
- Sundaram, R.; Shanthi, P.; Sachdanandam, P. Tangeretin, a polymethoxylated flavone, modulates lipid homeostasis and decreases oxidative stress by inhibiting NF-κB activation and proinflammatory cytokines in cardiac tissue of streptozotocin-induced diabetic rats. J. Funct. Foods 2015, 16, 315–333. [Google Scholar] [CrossRef]
- Elhennawy, M.G.; Lin, H. Determination of Tangeretin in Rat Plasma: Assessment of Its Clearance and Absolute Oral Bioavailability. Pharmaceutics 2018, 10, 3. [Google Scholar] [CrossRef]
- Hung, W.L.; Chang, W.S.; Lu, W.C.; Wei, G.J.; Wang, Y.; Ho, C.T.; Hwang, L.S. Pharmacokinetics, bioavailability, tissue distribution and excretion of tangeretin in rat. J. Food Drug Anal. 2018, 26, 2849–2857. [Google Scholar] [CrossRef] [PubMed]
- Kalai, F.Z.; Boulaaba, M.; Ferdousi, F.; Isoda, H. Effects of Isorhamnetin on Diabetes and Its Associated Complications: A Review of in vitro and in vivo Studies and a Post Hoc Transcriptome Analysis of Involved Molecular Pathways. Int. J. Mol. Sci. 2022, 23, 704. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; Luo, H.; Duan, J.; Hong, C.; Ma, P.; Li, G.; Zhang, T.; Wu, T.; Ji, G. Phytic acid enhances the oral absorption of isorhamnetin, quercetin, and kaempferol in total flavones of Hippophae rhamnoides L. Fitoterapia 2014, 93, 216–225. [Google Scholar] [CrossRef]
- Zhao, G.; Duan, J.; Xie, Y.; Lin, G.; Luo, H.; Li, G.; Yuan, X. Effects of solid dispersion and self-emulsifying formulations on the solubility, dissolution, permeability and pharmacokinetics of isorhamnetin, quercetin and kaempferol in total flavones of Hippophae rhamnoides L. Drug Dev. Ind. Pharm. 2013, 39, 1037–1045. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Zhang, D.; Zhang, Y.; Li, M.; Wang, C. Wogonin attenuates diabetic cardiomyopathy through its anti-inflammatory and anti-oxidative properties. Mol. Cell. Endocrinol. 2016, 428, 101–108. [Google Scholar] [CrossRef]
- Li, J.; Zhang, H.; Yan, Y.; Sun, S. Study of the inclusion complex and antioxidating activity of Wogonin with b-cyclodextrin and hydroxypropyl-cyclodextrin. J. Incl. Phenom. Macrocycl. Chem. 2016, 84, 115–120. [Google Scholar] [CrossRef]
- Zhu, N.; Li, J.C.; Zhu, J.X.; Wang, X.; Zhang, J. Characterization and Bioavailability of Wogonin by Different Administration Routes in Beagles. Med. Sci. Monit. 2016, 22, 3737–3745. [Google Scholar] [CrossRef]
- Hasanein, P.; Emamjomeh, A.; Chenarani, N.; Bohlooli, M. Beneficial effects of rutin in diabetes-induced deficits in acquisition learning, retention memory and pain perception in rats. Nutr. Neurosci. 2018, 23, 563–574. [Google Scholar] [CrossRef]
- Mahajan, H.S.; Bhalkar, K.G. Development and Evaluation of Rutin- HPβCD Inclusion Complex Based Mouth Dissolving Tablets. Int. J. Pharm. Sci. Dev. Res. 2017, 3, 1–6. [Google Scholar] [CrossRef]
- Das, S.; Roy, P.; Pal, R.; Auddy, R.G.; Chakraborti, A.S.; Mukherjee, A. Engineered Silybin Nanoparticles Educe Efficient Control in Experimental Diabetes. PLoS ONE 2014, 9, e101818. [Google Scholar] [CrossRef]
- Sahibzada, M.U.K.; Sadiq, A.; Khan, H.S.; Naseemullah, F.; Khurram, M.; Amin, M.U.; Haseeb, A. Fabrication, characterization and in vitro evaluation of silibinin nanoparticles: An attempt to enhance its oral bioavailability. Drug Des. Dev. Ther. 2017, 15, 1453–1464. [Google Scholar] [CrossRef] [PubMed]
- Voroneanu, L.; Nistor, I.; Dumea, R.; Apetrii, M.; Covic, A. Silymarin in Type 2 Diabetes Mellitus: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Diabetes Res. 2019, 2019, 5147468. [Google Scholar] [CrossRef] [PubMed]
- Yousaf, A.M.; Malik, U.R.; Shahzad, Y.; Mahmood, T.; Hussain, T. Silymarin-laden PVP-PEG polymeric composite for enhanced aqueous solubility and dissolution rate: Preparation and in vitro characterization. J. Pharm. Anal. 2019, 9, 34–39. [Google Scholar] [CrossRef]
- Jose, M.A.; Abraham, A.; Narmadha, M.P. Effect of silymarin in diabetes mellitus patients with liver diseases. J. Pharmacol. Pharmacother. 2011, 2, 287–289. [Google Scholar] [CrossRef]
- Gharib, A.; Faezizadeh, Z.; Godarzee, M. Treatment of diabetes in the mouse model by delphinidin and cyanidin hydrochloride in free and liposomal forms. Planta Med. 2013, 79, 1599–1604. [Google Scholar] [CrossRef]
- Matsumoto, H.; Chiyanagi, T.I.; Iida, H.; Ito, K.; Tsuda, T.; Hirayama, M.; Konishi, T. Ingested delphinidin-3-rutinoside is primarily excreted to urine as the intact form and to bile as the methylated form in rats. J. Agric. Food Chem. 2000, 54, 578–582. [Google Scholar] [CrossRef]
- Pivari, F.; Mingione, A.; Brasacchio, C.; Soldati, L. Curcumin and Type 2 Diabetes Mellitus: Prevention and Treatment. Nutrients 2019, 11, 1837. [Google Scholar] [CrossRef]
- Hu, L.; Shi, Y.; Li, J.H.; Gao, N.; Ji, J.; Niu, F.; Chen, Q.; Yang, X.; Wang, S. Enhancement of Oral Bioavailability of Curcumin by a Novel Solid Dispersion System. AAPS PharmSciTech 2015, 16, 6. [Google Scholar] [CrossRef]
- Zhang, D.W.; Fu, M.; Gao, S.H.; Liu, J.L. Curcumin and Diabetes: A Systematic Review. Evid. Based Complement. Altern. Med. 2013, 2013, 636053. [Google Scholar] [CrossRef]
- Bacanli, M. Limonene and ursolic acid in the treatment of diabetes: Citrus phenolic limonene, triterpenoid ursolic acid, antioxidants and diabetes. In Diabetes (Second Edition) Oxidative Stress and Dietary Antioxidants; Elsevier: Amsterdam, The Netherlands, 2020. [Google Scholar] [CrossRef]
- Eloy, J.O.; Saraiva, J.; Albuquerque, S.D.; Marchetti, J.M. Preparation, characterization and evaluation of the in vivo trypanocidal activity of ursolic acid-loaded solid dispersion with poloxamer 407 and sodium caprate. Braz. J. Pharm. Sci. 2015, 51, 101–109. [Google Scholar] [CrossRef]
- Yu, D.; Kan, Z.; Shan, F.; Zang, J.; Zhou, J. Triple Strategies to Improve Oral Bioavailability by Fabricating Co-amorphous Forms of Ursolic Acid with Piperine: Enhancing Water-Solubility, Permeability and Inhibiting Cytochrome P450 Isozymes. Mol. Pharm. 2020, 17, 12. [Google Scholar] [CrossRef]
- Nugroho, A.E.; Andrie, M.; Warditiani, N.K.; Siswanto, E.; Pramono, S.; Lukitaningsih, E. Antidiabetic and antihyperlipidemic effect of Andrographis paniculata (Burm. f.) Nees and Andrographolide in high-fructose-fat-fed rats. Indian J. Pharmacol. 2012, 44, 377–381. [Google Scholar] [CrossRef]
- Agrawal, A.; Goldfarb, A.; Teodoridis, F. Solubility enhancement of Andrographolide and formulation development of Hollow microspheres. World J. Pharm. Res. 2016, 5, 628–638. [Google Scholar] [CrossRef]
- Yan, Y.; Fang, L.H.; Du, G.H. Andrographolide. In Natural Small Molecule Drugs from Plants; Springer: Berlin/Heidelberg, Germany, 2018; pp. 357–362. [Google Scholar] [CrossRef]
- Subramanian, R.; Asmawi, M.Z.; Sadikun, A. In vitro alpha-glucosidase and alpha-amylase enzyme inhibitory effects of Andrographis paniculata extract and andrographolide. Acta Biochim. Pol. 2008, 55, 391–398. [Google Scholar] [CrossRef]
- Altemimi, A.; Lakhssassi, N.; Baharlouei, A.; Watson, D.G.; Lightfoot, D.A. Phytochemicals: Extraction, Isolation, and Identification of Bioactive Compounds from Plant Extracts. Plants 2017, 6, 42. [Google Scholar] [CrossRef]
- Kennedy, D.O.; Wightman, E.L. Herbal Extracts and Phytochemicals: Plant Secondary Metabolites and the Enhancement of Human Brain Function. Adv. Nutr. 2011, 2, 32–50. [Google Scholar] [CrossRef]
- Kumar, S.; Shachi, K.; Prasad, N.K. Diabetes Mellitus and Allopathic Medication Increase the Risk of Cancer Malignancy, but no Side Effect Associated with the Use of Antidiabetic Herbal Medicine. Curr. Res. Diabetes Obes. J. 2020, 13, 555868. [Google Scholar] [CrossRef]
- Palhares, R.M.; Drummond, M.G.; Brasil, B.D.S.A.F.; Cosenza, G.P.; Brandao, M.D.G.L.; Oliveira, G. Medicinal Plants Recommended by the World Health Organization: DNA Barcode Identification Associated with Chemical Analyses Guarantees Their Quality. Safety for the Consumers of Medicinal. Plants 2015, 10, e0127866. [Google Scholar] [CrossRef]
- Choudhury, H.; Pandey, M.; Hua, C.K.; Mun, C.S.; Jing, J.K.; Kong, L.; Ern, L.Y.; Ashraf, N.A.; Kit, S.W.; Yee, T.S.; et al. An update on natural compounds in the remedy of diabetes mellitus: A systematic review. J. Tradit. Complement. Med. 2018, 8, 361–376. [Google Scholar] [CrossRef]
- Arfaoui, L. Dietary Plant Polyphenols: Effects of Food Processing on Their Content and Bioavailability. Molecules 2021, 26, 2959. [Google Scholar] [CrossRef]
- Varshney, M.; Kumar, B.; Rana, V.S.; Sethiya, N.K. An overview on therapeutic and medicinal potential of poly-hydroxy flavone viz. Heptamethoxyflavone, Kaempferitrin, Vitexin and Amentoflavone for management of Alzheimer’s and Parkinson’s diseases: A critical analysis on mechanistic insight. Crit. Rev. Food Sci. Nutr. 2021. Online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Kai, G.; Yamamoto, K.; Chen, X. Advance in dietary polyphenols as alpha glucosidases inhibitors: A review on structure-activity relationship aspect. Crit. Rev. Food Sci. Nutr. 2013, 53, 818–836. [Google Scholar] [CrossRef] [PubMed]
- Sarian, M.N.; Ahmed, Q.U.; So’ad, S.Z.M.; Alhassan, A.M.; Murugesu, S.; Perumal, V.; Mohamad, S.N.A.S.; Khatib, A.; Latip, J. Antioxidant and antidiabetic effects of flavonoids: A structure-activity relationship based study. BioMed Res. Int. 2017, 2017, 8386065. [Google Scholar] [CrossRef] [PubMed]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. Flavonoids: An overview. J. Nutr. Sci. 2016, 5, e47. [Google Scholar] [CrossRef]
- Jain, N.; Mohan, S.C.; Sumathi, S. Mechanisms of Action of Flavonoids in the Management of Diabetes mellitus. J. Drug Deliv. Ther. 2021, 11, 194–202. [Google Scholar] [CrossRef]
- Serra, R.; Ielapi, N.; Bitonti, A.; Candido, S.; Fregola, S.; Gallo, A.; Loria, A.; Muraca, L.; Raimondo, L.; Velcean, L.; et al. Efficacy of a Low-Dose Diosmin Therapy on Improving Symptoms and Quality of Life in Patients with Chronic Venous Disease: Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients 2021, 13, 999. [Google Scholar] [CrossRef]
- Sendrayaperumal, V.; Pillai, S.I.; Subramanian, S. Design, synthesis and characterization of zinc-morin, a metal flavonol complex and evaluation of its anti-diabetic potential in HFD-STZ induced type 2 diabetes in rats. Chem.-Biol. Interact. 2014, 219, 9–17. [Google Scholar] [CrossRef]
- Abuohashish, H.M.; Al-Rejaie1, S.S.; Al-Hosaini, K.A.; Parmar, M.Y.; Ahmed, M.M. Alleviating effects of morin against experimentally-induced diabetic osteopenia. Diabetol. Metab. Syndr. 2013, 5, 5. [Google Scholar] [CrossRef]
- Wang, X.; Zhang, D.M.; Gu, T.T.; Ding, X.Q.; Fan, C.Y.; Zhu, Q.; Shi, Y.W.; Hong, Y.; Kong, L.D. Morin reduces hepatic inflammation-associated lipid accumulation in high fructose-fed rats via inhibiting sphingosine kinase 1/sphingosine 1-phosphate signaling pathway. Biochem. Pharmacol. 2013, 86, 791–1804. [Google Scholar] [CrossRef]
- Prasath, G.S.; Pillai, S.I.; Subramanian, S.P. Fisetin improves glucose homeostasis through the inhibition of gluconeogenic enzymes in hepatic tissues of streptozotocin induced diabetic rats. Eur. J. Pharmacol. 2014, 740, 248–254. [Google Scholar] [CrossRef]
- Dong, W.; Jia, C.; Li, J.; Zhou, Y.; Luo, Y.; Liu, J.; Zhao, Z.; Zhang, J.; Lin, S.; Chen, Y. Fisetin Attenuates Diabetic Nephropathy-Induced Podocyte Injury by Inhibiting NLRP3 Inflammasome. Front. Pharmacol. 2022, 13, 783706. [Google Scholar] [CrossRef] [PubMed]
- Visnagri, A.; Kandhare, A.D.; Chakravarty, S.; Ghosh, P.; Bodhankar, S.L. Hesperidin, a flavanoglycone attenuates experimental diabetic neuropathy via modulation of cellular and biochemical marker to improve nerve functions. Pharm. Biol. 2014, 52, 814–828. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, Y.O.; Sharma, P.K.; Shrivastava, B.; Ojha, S.; Upadhya, H.M.; Arya, D.S.; Goyal, S.N. Hesperidin produces cardioprotective activity via PPARγ pathway in ischemic heart disease model in diabetic rats. PLoS ONE 2014, 9, e111212. [Google Scholar] [CrossRef]
- Tian, M.; Han, Y.B.; Zhao, C.C.; Liu, L.; Zhang, F.L. Hesperidin alleviates insulin resistance by improving HG-induced oxidative stress and mitochondrial dysfunction by restoring miR-149. Diabetol. Metab. Syndr. 2021, 13, 50. [Google Scholar] [CrossRef]
- Zeng, B.; Chen, K.; Du, P.; Wang, S.S.; Ren, B.; Ren, Y.L.; Yan, H.S.; Liang, Y.; Wu, F.H. Phenolic compounds from Clinopodium chinense (Benth.) O. Kuntze and their inhibitory effects on alpha-Glucosidase and vascular Endothelial cells injury. Chem. Biodivers. 2016, 13, 596–601. [Google Scholar] [CrossRef] [PubMed]
- He, P.; Yan, S.; Zheng, J.; Gao, Y.; Zhang, S.; Liu, Z.; Liu, X.; Xiao, C. Eriodictyol Attenuates LPS-Induced Neuroinflammation, Amyloidogenesis, and Cognitive Impairments via the Inhibition of NF-kappaB in Male C57BL/6J Mice and BV2 Microglial Cells. J. Agric. Food Chem. 2018, 66, 10205–10214. [Google Scholar] [CrossRef]
- Kwon, E.Y.; Choi, M.S. Dietary eriodictyol alleviates adiposity, hepatic steatosis, insulin resistance, and inflammation in diet-induced obese mice. Int. J. Mol. Sci. 2019, 20, 1227. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Du, Q.; Li, X.; Zheng, X.; Lv, F.; Xi, X.; Huang, G.; Yang, J.; Liu, S. Eriodictyol Inhibits Proliferation, Metastasis and Induces Apoptosis of Glioma Cells via PI3K/Akt/NF-κB Signaling Pathway. Front. Pharmacol. 2020, 11, 114. [Google Scholar] [CrossRef] [PubMed]
- Hameed, A.; Hafizur, R.M.; Hussain, N.; Raza, S.A.; Rehman, M.; Ashraf, S.; Ul-Haq, Z.; Khan, F.; Abbas, G.; Choudhary, M.I. Eriodictyol stimulates insulin secretion through cAMP/PKA signaling pathway in mice islets. Eur. J. Pharmacol. 2018, 820, 245–255. [Google Scholar] [CrossRef]
- Wen, S.; Hu, M.; Xiong, Y. Effect of Eriodictyol on Retinoblastoma via the PI3K/Akt Pathway. J. Healthc. Eng. 2021, 2021, 6091585. [Google Scholar] [CrossRef]
- Priscilla, D.H.; Jayakumar, M.; Thirumurugan, K. Flavanone naringenin: An effective antihyperglycemic and antihyperlipidemic nutraceutical agent on high fat diet fed streptozotocin induced type 2 diabetic rats. J. Funct. Foods 2015, 14, 363–373. [Google Scholar] [CrossRef]
- Malik, S.; Suchal, K.; Khan, S.I.; Bhatia, J.; Kishore, K.; Dinda, A.K.; Arya, D.S. Apigenin ameliorates streptozotocin-induced diabetic nephropathy in rats via MAPK-NF-κB-TNF-α and TGF- β1-MAPK-fibronectin pathways. Am. J. Physiol. Renal Physiol. 2017, 313, 414–422. [Google Scholar] [CrossRef] [PubMed]
- Al-Numair, K.S.; Chandramohan, G.; Veeramani, C.; Alsaif, M.A. Ameliorative effect of kaempferol, a flavonoid, on oxidative stress in streptozotocin-induced diabetic rats. Redox Rep. 2015, 20, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Sharma, D.; Gondaliya, P.; Tiwari, V.; Kalia, K. Kaempferol attenuates diabetic nephropathy by inhibiting RhoA/Rho-kinase mediated inflammatory signaling. Biomed. Pharmacother. 2019, 109, 1610–1619. [Google Scholar] [CrossRef] [PubMed]
- Aba, P.E.; Asuzu, I.U. Mechanisms of actions of some bioactive anti-diabetic principles from phytochemicals of medicinal plants: A review. Indian J. Nat. Prod. Resour. 2018, 9, 85–96. [Google Scholar]
- Sood, A.; Kumar, B.; Singh, S.K.; Parashar, P.; Gautam, A.; Gulati, M.; Pandey, N.K.; Melkani, I.; Awasthi, A.; Saraf, S.A.; et al. Flavonoids as Potential Therapeutic Agents for the Management of Diabetic Neuropathy. Curr. Pharm. Des. 2020, 26, 5468–5487. [Google Scholar] [CrossRef]
- Espinosa, J.J.R.; Rios, J.S.; Jimenez, S.G.; Molina, R.V.; Villarreal, G.A.; Ocampo, A.N.R.; Fernández, G.B.; Soto, S.E. Chrysin induces antidiabetic, Antidyslipidemic and anti-inflammatory effects in athymic nude diabetic mice. Molecules 2018, 23, 67. [Google Scholar] [CrossRef]
- Ahad, A.; Mujeeb, M.; Ahsan, H.; Siddiqui, W.A. Prophylactic effect of baicalein against renal dysfunction in type 2 diabetic rats. Biochimie. 2014, 106, 101–110. [Google Scholar] [CrossRef]
- Ma, L.; Wu, F.; Shao, Q.; Chen, G.; Xu, L.; Lu, F. Baicalin Alleviates Oxidative Stress and Inflammation in Diabetic Nephropathy via Nrf2 and MAPK Signaling Pathway. Drug Des. Dev. Ther. 2021, 15, 3207–3221. [Google Scholar] [CrossRef]
- Ding, Y.; Shi, X.; Shuai, X.; Xu, Y.; Liu, Y.; Liang, X.; Wei, D.; Su, D. Luteolin prevents uric acid-induced pancreatic β-cell dysfunction. J. Biomed. Res. 2014, 28, 292–298. [Google Scholar] [CrossRef]
- Li, L.; Luo, W.; Qian, Y.; Zhu, W.; Qian, J.; Li, J.; Jin, Y.; Xu, X.; Liang, G. Luteolin protects against diabetic cardiomyopathy by inhibiting NF-κB-mediated inflammation and activating the Nrf2-mediated antioxidant responses. Phytomedicine 2019, 59, 152774. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Hur, H.J.; Kwon, D.Y.; Hwang, J. Tangeretin stimulates glucose uptake via regulation of AMPK signaling pathways in C2C12 myotubes and improves glucose tolerance in high-fat diet-induced obese mice. Mol. Cell Endocrinol. 2012, 358, 127–134. [Google Scholar] [CrossRef]
- Qin, D.; Jiang, Y.R. Tangeretin Inhibition of High-Glucose-Induced IL-1β, IL-6, TGF-β1, and VEGF Expression in Human RPE Cells. J. Diabetes Res. 2020, 2020, 9490642. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, C.R.; Torres, N.; Uribe, J.A.G.; Noriega, L.G.; Villalvazo, I.T.; Diaz, A.M.L.; Ricardo, M.A.; Mota, C.M.; Ordaz, G.; Santoscoy, R.A.C.; et al. The effect of isorhamnetin glycosides extracted from Opuntia ficus indica in a mouse model of diet induced obesity. Food Funct. 2015, 6, 805–815. [Google Scholar] [CrossRef]
- Matboli, M.; Saad, M.; Hasanin, A.H.; Saleh, L.A.; Baher, W.; Bekhet, M.M.; Eissa, S. New insight into the role of isorhamnetin as a regulator of insulin signaling pathway in type 2 diabetes mellitus rat model: Molecular and computational approach. Biomed. Pharmacother. 2021, 135, 111176. [Google Scholar] [CrossRef] [PubMed]
- Eun-Jung Bak, E.J.; Kim, J.; Choi, Y.H.; Kim, J.H.; Lee, D.E.; Woo, G.H.; Cha, J.H.; Yoo, Y.J. Wogonin ameliorates hyperglycemia and dyslipidemia via PPARα activation in db/db mice. Clin. Nutr. 2014, 33, 156–163. [Google Scholar] [CrossRef]
- Liu, X.Q.; Jiang, L.; Li, Y.Y.; Huang, Y.B.; Hu, X.R.; Zhu, W.; Wang, X.; Wu, Y.G.; Meng, X.M.; Qi, X.M. Wogonin protects glomerular podocytes by targeting Bcl-2- mediated autophagy and apoptosis in diabetic kidney disease. Acta Pharmacol. Sin. 2022, 43, 96–110. [Google Scholar] [CrossRef]
- Ghorbani, A. Mechanisms of antidiabetic effects of flavonoid rutin Ahmad Ghorbani. Biomed. Pharmacother. 2017, 96, 305–312. [Google Scholar] [CrossRef]
- Guimaraes, J.F.C.; Muzio, B.P.; Rosa, C.M.; Nascimento, A.F.; Sugizaki, M.M.; Fernandes, A.A.H.; Cicogna, A.C.; Padovani, C.R.; Okoshi, M.P.; Okoshi, K. Rutin administration attenuates myocardial dysfunction in diabetic rats. Cardiovasc. Diabetol. 2015, 14, 90. [Google Scholar] [CrossRef]
- Yang, D.K.; Kang, H.S. Anti-Diabetic Effect of Cotreatment with Quercetin and Resveratrol in Streptozotocin-Induced Diabetic Rats. Biomol. Ther. 2018, 26, 130–138. [Google Scholar] [CrossRef]
- Weng, L.; Zhang, F.; Wang, R.; Ma, W.; Song, Y. A review on protective role of genistein against oxidative stress in diabetes and related complications. Chem. Biol. Interact. 2019, 1, 108665. [Google Scholar] [CrossRef] [PubMed]
- Cheong, S.H.; Furuhashi, K.; Ito, K.; Nagaoka, M.; Yonezawa, T.; Miura, Y.; Yagasaki, K. Daidzein promotes glucose uptake through glucose transporter 4 translocation to plasma membrane in L6 myocytes and improves glucose homeostasis in type II diabetic model mice. J. Nutr. Biochem. 2014, 25, 136–143. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Ge, Q.; Chen, L.; Chen, K. Studies of the Anti-Diabetic Mechanism of Pueraria lobata Based on Metabolomics and Network Pharmacology. Processes 2021, 9, 1245. [Google Scholar] [CrossRef]
- AL-Ishaq, R.K.; Abotaleb, M.; Kubatka, P.; Kajo, K.; Busselberg, D. Flavonoids and Their Anti-Diabetic Effects: Cellular Mechanisms and Effects to Improve Blood Sugar Levels. Biomolecules 2019, 9, 430. [Google Scholar] [CrossRef] [PubMed]
- Lim, W.; Song, G. Inhibitory effects of delphinidin on the proliferation of ovarian cancer cells via PI3K/AKT and ERK 1/2 MAPK signal transduction. Oncol. Lett. 2017, 14, 810–818. [Google Scholar] [CrossRef]
- Hidalgo, J.; Teuber, S.; Morera, F.J.; Ojeda, C.; Flores, C.A.; Hidalgo, M.A.; Nunez, L.; Villalobos, C.; Burgos, R.A. Delphinidin Reduces Glucose Uptake in Mice Jejunal Tissue and Human Intestinal Cells Lines through FFA1/GPR40. Int. J. Mol. Sci. 2017, 18, 750. [Google Scholar] [CrossRef]
- Nasri, S.; Roghani, M.; Baluchnejadmojarad, T.; Rabani, T.; Balvardi, M.M. Vascular mechanisms of cyanidin-3-glucoside response in streptozotocin-diabetic rats. Pathophysiology 2011, 18, 273–278. [Google Scholar] [CrossRef]
- Su, H.; Bao, T.; Xie, L.; Xu, Y.; Chen, W. Transcriptome profiling reveals the antihyperglycemic mechanism of pelargonidin-3-O-glucoside extracted from wild raspberry. J. Funct. Foods 2020, 64, 103657. [Google Scholar] [CrossRef]
- Zheng, T.; Shu, G.; Yang, Z.; Mo, S.; Zhao, Y.; Mei, Z. Antidiabetic effect of total saponins from Entada phaseoloides (L.) Merr.in type 2 diabetic rats. J. Ethnopharmacol. 2012, 139, 814–821. [Google Scholar] [CrossRef]
- Smith, Y.R.A.; Adanlawo, I.G.; Oni, O.S. Hypoglycaemic effect of saponin from the root of Garcinia kola on alloxan- induced diabetic rats. J. Drug Deliv. Ther. 2012, 2, 9–12. [Google Scholar] [CrossRef]
- Jia, S.; Shen, M.; Zhang, F.; Xie, J. Recent Advances in Momordica charantia: Functional Components and Biological Activities. Int. J. Mol. Sci. 2017, 18, 2555. [Google Scholar] [CrossRef] [PubMed]
- Chang, C.I.; Hsin, I.T.; Wen, L.Y.; Hung, Y.C.; Min, C.T.; Chen, L.C.; Ling, C.H. In vivo and in vitro studies to identify the hypoglycemic constituents of Momordica charantia wild variant WB24. Food Chem. 2011, 125, 521–528. [Google Scholar] [CrossRef]
- Elekofehinti, O.O.; Kamdem, J.P.; Kade, I.J.; Rocha, J.B.T.; Adanlawo, I.G. Hypoglycemic anti-per oxidative and antihyperlipidemic effects of saponins from Solanum anguivi Lam. fruits in alloxan-induced diabetic rats. South Afr. J. Bot. 2013, 88, 56–61. [Google Scholar] [CrossRef]
- Nakitto, A.M.S.; Muyonga, J.H.; Byaruhanga, Y.B.; Wagner, A.E. Solanum anguivi Lam. Fruits: Their Potential Effects on Type 2 Diabetes Mellitus. Molecules 2021, 26, 2044. [Google Scholar] [CrossRef]
- Lee, C.E.; Hur, H.J.; Hwang, J.T.; Sung, M.J.; Yang, H.J.; Kim, H.J.; Park, J.H.; Kwon, D.Y.; Kim, M.S. Long-Term Consumption of Platycodi Radix Ameliorates Obesity and Insulin Resistance via the Activation of AMPK Pathways. Evid. Based Complement. Altern. Med. 2012, 2012, 759143. [Google Scholar] [CrossRef]
- Deng, Y.; He, K.; Ye, X.; Chen, X.; Huang, J.; Li, X.; Yuan, L.; Jin, Y.; Jin, Q.; Li, P. Saponin rich fractions from Polygonatu modoratum (Mill.) druce with more potential hypoglycemic effects. J. Ethnopharmacol. 2012, 141, 228–233. [Google Scholar] [CrossRef]
- Cheng, S.; Liang, S.; Liu, Q.; Deng, Z.; Zhang, Y.; Du, J.; Zhang, Y.N.; Li, S.; Cheng, B.; Ling, C. Diosgenin prevents high-fat diet-induced rat non-alcoholic fatty liver disease through the AMPK and LXR signaling pathways. Int. J. Mol. Med. 2018, 41, 1089–1095. [Google Scholar] [CrossRef]
- Zhang, X.X.; Ji, Y.L.; Zhu, L.P.; Wang, Z.H.; Fang, C.Q.; Jiang, C.H.; Pan, K.; Zhang, J.; Yin, Z.Q. Arjunolic acid from Cyclocarya paliurus ameliorates diabetic retinopathy through AMPK/mTOR/HO-1 regulated autophagy pathway. J. Ethnopharmacol. 2022, 284, 114772. [Google Scholar] [CrossRef]
- Aamir, K.; Khan, H.U.; Hossain, C.F.; Afrin, M.R.; Jusuf, P.R.; Waheed, I.; Sethi, G.; Arya, A. Arjunolic acid downregulates elevated blood sugar and pro-inflammatory cytokines in streptozotocin (STZ)-nicotinamide induced type 2 diabetic rats. Life Sci. 2022, 289, 120232. [Google Scholar] [CrossRef]
- Kwon, D.Y.; Kim, S.; Ryu, S.K.; Choi, Y.H.; Cha, M.R.; Yang, H.J.; Park, S. Platyconic acid, a saponin from Platycodi radix, improves glucose homeostasis by enhancing insulin sensitivity in vitro and in vivo. Eur. J. Nutr. 2012, 51, 529–540. [Google Scholar] [CrossRef]
- Roy, A. A Review on the Alkaloids an Important Therapeutic Compound from Plants. Int. J. Plant Biotechnol. 2017, 3, 1–9. [Google Scholar] [CrossRef]
- Abou El-Soud Khalil, M.Y.; Hussein, F.S.H.; Hussein Farray, A.R. Antidiabetic effects of Fenugreek alkaloid extract in streptozotocin induced hyperglycemic rats. J. App. Sci. Res. 2007, 3, 1073–1083. [Google Scholar]
- Patel, O.P.S.; Mishra, A.; Maurya, R.; Saini, D.; Pandey, J.; Taneja, I.; Raju, K.S.R.; Kanojiya, S.; Shukla, S.K.; Srivastava, M.N.; et al. Naturally occurring Carbazole alkaloids from Murraya koenigii as potential anti-diabetic agents. J. Nat. Prod. 2015, 79, 1276–1284. [Google Scholar] [CrossRef]
- Agrawal, R.; Sethiya, N.K.; Mishra, S.H. Antidiabetic activity of alkaloids of Aerva lanata roots on streptozotocin-nicotinamide induced type II diabetes in rats. Pharm. Biol. 2013, 51, 635–642. [Google Scholar] [CrossRef]
- Tiong, S.H.; Looi, C.Y.; Hazni, H.; Arya, A.; Paydar, M.; Wong, W.F.; Cheah, S.C.; Mustafa, M.R.; Awang, K. Antidiabetic and antioxidant properties of alkaloids from Catharanthus roseus (L.) G. Don. Molecules 2013, 18, 9770–9784. [Google Scholar] [CrossRef]
- Ajebli, M.; Eddouks, M. The Promising Role of Plant Tannins as Bioactive Antidiabetic Agents. Curr. Med. Chem. 2018, 26, 4852–4884. [Google Scholar] [CrossRef]
- Omar, N.; Ismail, C.A.N.; Long, I. Tannins in the Treatment of Diabetic Neuropathic Pain: Research Progress and Future Challenges. Front. Pharmacol. 2022, 12, 805854. [Google Scholar] [CrossRef]
- Meng, J.M.; Cao, S.Y.; Wei, X.L.; Gan, R.Y.; Wang, Y.F.; Cai, S.X.; Xu, X.Y.; Zhang, P.Z.; Li, H.B. Effects and Mechanisms of tea for the Prevention and Management of Diabetes Mellitus and Diabetic Complications: An Updated Review. Antioxidants 2019, 8, 170. [Google Scholar] [CrossRef]
- Addepalli, V.; Suryavanshi, S.V. Catechin Attenuates Diabetic Autonomic Neuropathy in Streptozotocin Induced Diabetic Rats. Biomed. Pharmacother. 2018, 108, 1517–1523. [Google Scholar] [CrossRef]
- Abo-Salem, O.M.; Ali, T.M.; Harisa, G.I.; Mehanna, O.M.; Younos, I.H.; Almalki, W.H. Beneficial Effects of (-)-Epigallocatechin-3-O-Gallate on Diabetic Peripheral Neuropathy in the Rat Model. J. Biochem. Mol. Toxicol. 2020, 34, e22508. [Google Scholar] [CrossRef]
- Raposo, D.; Morgado, C.; Terra, P.P.; Tavares, I. Nociceptive Spinal Cord Neurons of Laminae I-III Exhibit Oxidative Stress Damage during Diabetic Neuropathy Which Is Prevented by Early Antioxidant Treatment with Epigallocatechin-Gallate (EGCG). Brain Res. Bull. 2015, 110, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Shehadeh, M.B.; Suaifan, G.A.R.Y.; Abu-Odeh, A.M. Plants Secondary Metabolites as Blood Glucose Lowering Molecules. Molecules 2021, 26, 4333. [Google Scholar] [CrossRef] [PubMed]
- Suchitra, K.; Panigrahy, S.K.; Bhatt, R.; Kumar, A. Targeting type II diabetes with plant terpenes: The new and promising antidiabetic therapeutics. Biologia 2021, 76, 241–254. [Google Scholar] [CrossRef]
- Xu, L.; Li, Y.; Dai, Y.; Peng, J. Natural products for the treatment of type 2 diabetes mellitus: Pharmacology and mechanisms. Pharmacol. Res. 2018, 130, 451–465. [Google Scholar] [CrossRef]
- Chi, G.; Wei, M.; Xie, X.; Soromou, L.W.; Liu, F.; Zhao, S. Suppression of MAPK and NF-Κb Pathways by Limonene Contributes to Attenuation of Lipopolysaccharide-Induced Inflammatory Responses in Acute Lung Injury. Inflammation 2013, 36, 501–511. [Google Scholar] [CrossRef]
- Kim, T.; Song, B.; Cho, K.S.; Lee, I.S. Therapeutic Potential of Volatile Terpenes and Terpenoids from Forests for Inflammatory Diseases. Int. J. Mol. Sci. 2020, 21, 2187. [Google Scholar] [CrossRef]
- Ong, W.Y.; Farooqui, T.; Koh, H.L.; Farooqui, A.A.; Ling, E.A. Protective Effects of Ginseng on Neurological Disorders. Front. Aging Neurosci. 2015, 7, 129. [Google Scholar] [CrossRef]
- Quintans, J.S.S.; Sanmugam, S.; Heimfarth, L.; Araújo, A.A.S.; Almeida, J.R.G.D.S.; Picot, L.; Júnior, L.Q. Monoterpenes Modulating Cytokines-A Review. Food Chem. Toxicol. 2019, 123, 233–257. [Google Scholar] [CrossRef]
- Audelo, M.L.D.P.; Cortes, H.; Floran, I.H.C.; Torres, M.G.; Guadarrama, L.E.; Chavez, S.A.B.; Gomez, D.M.G.; Magana, J.J.; Gomez, G.L. Therapeutic Applications of Terpenes on Inflammatory Diseases. Front. Pharmacol. 2021, 12, 704197. [Google Scholar] [CrossRef]
- Karthikeyan, R.; Kanimozhi, G.; Prasad, N.R.; Agilan, B.; Ganesan, M.; Srithar, G. Alpha Pinene Modulates UVA-Induced Oxidative Stress, DNA Damage and Apoptosis in Human Skin Epidermal Keratinocytes. Life Sci. 2018, 212, 150–158. [Google Scholar] [CrossRef]
- Fernandes, J. Antitumor Monoterpenes in Bioactive Essential Oils and Cancer; de Sousa, D.P., Ed.; Springer: Cham, Switzerland, 2015; pp. 175–200. [Google Scholar] [CrossRef]
- Baz, F.E.; Aly, H.; Abd-Alla, H.I.; Saad, S.A. Bioactive flavonoid glycosides and anti-diabetic activity of Jatropha curcas on streptozotocin-induced diabetic rats. Int. J. Pharm. Rev. Res. 2014, 29, 143–156. [Google Scholar]
- Tofighi, Z.; Afrapoli, F.M.; Ebrahimi, S.N.; Goodarzi, S.; Hadjiakhoondi, A.; Neuburger, M.; Hamburger, M.; Abdollahi, M.; Yassa, N. Securigenin glycosides as hypoglycemic principles of Securigera securidaca seeds. J. Nat. Med. 2016, 71, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Admassu, S. Potential Health Benefits and Problems Associated with Phytochemicals in Food Legumes. East Afr. J. Sci. 2009, 3, 116–133. [Google Scholar] [CrossRef]
- Anonymous. Aloeaceae. Meyler’s Side Effects of Drugs, The International Encyclopedia of Adverse Drug Reactions and Interactions, 16th ed.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 160–161. [Google Scholar] [CrossRef]
- Trexler, E.T.; Smith-Ryan, A.E.; Stout, J.R.; Hoffman, J.R.; Wilborn, C.D.; Sale, C.; Kreider, R.B.; Jäger, R.; Earnest, C.P.; Bannock, L.; et al. International society of sports nutrition position stand: Beta-Alanine. J. Int. Soc. Sports Nutr. 2015, 12, 30. [Google Scholar] [CrossRef] [PubMed]
- Khaodhiar, L.; Ricciotti, H.A.; Li, L.; Pan, W.; Schickel, M.; Zhou, J.; Blackburn, G.L. Daidzein-rich isoflavone aglycones are potentially effective in reducing hot flashes in menopausal women. Menopause 2008, 15, 125–132. [Google Scholar] [CrossRef]
- Galati, E.M.; Monforte, M.T.; Kirjavainen, S.; Forestieri, A.M.; Trovato, A.; Tripodo, M.M. Biological effects of hesperidin, a citrus flavonoid. (Note I): Antiinflammatory and analgesic activity. Farmaco 1994, 40, 709–712. [Google Scholar]
- Alam. M.A.; Subhan, N.; Rahman, M.M.; Uddin, S.J.; Reza, H.M.; Sarker, S.D. Effect of citrus flavonoids, naringin and naringenin, on metabolic syndrome and their mechanisms of action. Adv. Nutr. 2014, 5, 404–417. [Google Scholar] [CrossRef]
- Salehi, B.; Venditti, A.; Sharifi-Rad, M.; Kręgiel, D.; Sharifi-Rad, J.; Durazzo, A.; Lucarini, M.; Santini, A.; Souto, E.B.; Novellino, E.; et al. The Therapeutic Potential of Apigenin. Int. J. Mol. Sci. 2019, 20, 1305. [Google Scholar] [CrossRef]
- Varsha, K.; Sharma, A.; Kaur, A.; Madan, J.; Pandey, R.S.; Jain, U.K.; Chandra, R. Chapter 28—Natural plant-derived anticancer drugs nanotherapeutics: A review on preclinical to clinical success. In Micro and Nano Technologies, Nanostructures for Cancer Therapy; Elsevier: Amsterdam, The Netherlands, 2017; pp. 775–809. [Google Scholar] [CrossRef]
- Loguercio, C.; Festi, D. Silybin and the liver: From basic research to clinical practice. World J. Gastroenterol. 2011, 17, 2288–2301. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).