1. Introduction
For more than a century, empirical data in mammals has shown that blood pressure does not vary with the mass of the animal. In addition, for more than a century, empirical data has shown that blood mass (volume) is a somewhat fixed value, around 8% of body mass, irrespective of the size of the animal [
1,
2,
3,
4]. Despite the fact that these two variables, namely blood pressure and volume, are central to all other relationships of the cardiovascular system and other organic systems as well, no endeavor has been able to explain the reasons why blood volume scales linearly with body mass while blood pressure remains constant.
Relationships between sizes or between a size and a function are of the utmost importance in practically all scientific areas, particularly in Biology and Engineering [
5,
6,
7,
8,
9]. The relationships involving sizes are termed “scaling”, and a scaling rule is given as an equation of the following form [
6]:
where
L and
Y are the sizes and
α and
β are constants of the relationship. The scaling rule itself is related to the parameter
β, while
α is a reference value. Therefore, when only the scaling matters, the expression above is written as
where ∝ indicates “proportional to”.
Some scaling of the cardiovascular system has become widely known, for example, the decrease in heart rate with size [
10,
11] which results in a somewhat similar number of heart beats along the lifespans of both a 20 g mouse and of a 2 ton elephant. However, other cardiovascular scaling variables are barely mentioned beyond the realm of certain fields.
Considering body mass as a proxy of size, some organs scale linearly with the overall organism size, such as the bone mass [
12,
13,
14] of vertebrates, whereas many other organs do not scale linearly with the overall organism size [
15,
16]. The size-to-size linear relationship is termed isometry (
β = 1), and size-to-size non-linear scaling is termed allometry (
β ≠ 1; in particular, when
β = 0, the variable Y has a constant value irrespective of the value of
L) (e.g., [
17]). Therefore, blood volume scales isometrically with body mass, and, as we stated above, to date, there has been no explanation for such a relationship. Blood pressure, on the other hand, is non-linearly related to body mass (
β = 0), and we still lack an adequate explanation for why this is so.
Why are blood pressure and volume so important? In energetic terms, about 90% of the external cardiac work is to rise the enthalpy of the arterial system (i.e., its pressure). In volumetric terms, the entire branching of the system relies on the volume it can, or must, accommodate. In essence, the pressure dictates perfusion and, ultimately, rates, while the volume dictates lengths and branching.
There was a renewed interest in the scaling of the cardiovascular system at the end of the 20th century due to a tentative explanation of the scaling of basal metabolic rate as related to blood supply in organisms [
18]. That tentative explanation generated a prolific series of models, addressing in a general sense [
19,
20,
21,
22,
23,
24,
25,
26,
27,
28,
29] the topology of supply networks from leaf venation and blood microvasculature to human transportation systems. However, none of these studies addressed the isometry of blood volume or the constancy of blood pressure. This was primarily because these studies aimed to merely obtain the local architecture of a supply network; as a corollary, the driving force (e.g., pressure) is a fixed value from the beginning, or it is not important for solving the problem. The same applies to volume: it is either fixed from the start or unimportant in the solution. In a different line of thought, there are authors who seek more general scaling rules of the vasculature, without concerns regarding the topology of the network [
30,
31,
32]. In their studies, however, the isometry of blood volume and body mass is assumed directly from empirical data. In fact, the only attempt to obtain the linear scaling of blood volume with body mass comes from the study that sparked renewed interest in the topic two decades ago [
18]; unfortunately, the solution specified in this study was flawed [
33].
By the same token, the scaling of blood pressure with body mass also lacks formal proof. Dimensional analysis, with its non-dimensional invariant numbers (not to be confused with the invariance of blood pressure itself; see the Buckingham-Pi theorem [
8]) has often been put forward as a general approach [
34,
35]. An invariant number, such as the Reynolds number, is a criterion of similarity among systems, but it cannot furnish the scaling of the variables themselves (and those who applied dimensional analysis in approaching the cardiovascular system never claimed that). In this sense, dimensional analysis might suggest the constancy of blood pressure, though it cannot prove it.
Therefore, these two most relevant variables of a circulatory system, which are taken as roots for deriving the scaling rules of its other variables (see [
19,
20,
21,
22,
23,
24,
25,
26,
27,
28,
29,
30,
31,
32]), still remain without a theoretical basis to explain their own scaling.
An adequate method to address an optimization problem in biology would likely involve minimization of energy or a power function, both considered putative tenets of natural selection as it is currently understood [
36]. This was the method sought by most of the studies previously cited. Undeniable as a principle, power optimization is better placed within a more generic context, such as the entropy generation or exergy destruction approach [
37,
38,
39,
40]. This is because, embedded in such an approach is the loss of available work. That is, entropy generation (or exergy destruction) indicates the loss of an amount of power that would otherwise be useful. In a step beyond Carnot’s efficiency, this approach combines the first (conservation of energy) and the second (entropy changes) laws of thermodynamics into a single framework, summarized by the Gouy-Stodola theorem [
38,
41,
42]. From this comprehensive perspective, a system aims simultaneously to obtain a maximum of flow and to destroy less exergy (these ideas are well developed in Lotka’s principle, associated with near-stable systems, and in constructal theory [
9,
39,
43]).
In a very unspecific sense, flows in a system are dictated by a set of parameters belonging to the system. These parameters might or might not be under a control, whether central or local. Then, changing parameter values causes changes in flow, in a very broad sense. A flow is the result of a certain thermodynamic force times a certain conductance. For instance, electric current (the flow) is the result of the difference in electric potential (voltage, the force) times the electric conductance (inverse of resistance) of the wiring. In this very simple scenario, one could consider the resistance and the voltage as resulting from a set of parameters, and therefore changing these parameters would change the current through the system.
Entropy production (the time derivative of entropy generation [
41]) is given by the product of the flow times the force divided by the temperature (see
Section 2.1). Therefore, changing the conductance or the force changes both the flow and the entropy production. Thus, the problem that needs to be solved can be read in two stages:
- (1)
how to maximize the relationship between blood flow and entropy production (see Equation (10)); and
- (2)
under which scaling rules this optimization occurs when treating body mass as a parameter (see Equation (13)). by treating body mass as a parameter, what scaling rule is obtained (see equation 13).
2. Methods
In this section, the thermodynamic background for approaching the problem is presented in two subsections. In the first subsection, entropy production is presented, and its components are further parametrized. In the second subsection, the concept of elasticity is applied to the parametrized function of entropy production, obtaining the impacts of changes of the parameter on flow and on entropy production simultaneously. This serves as a guide to determine the optimized ways to increase flow in the system.
2.1. Entropy Production
An open system in a non-equilibrium but steady-state condition has flow(s) of energy and/or matter through its borders and internally. Entropy production is the result of this flow(s) and is computed as [
41,
42]
where
Xi is the general thermodynamic force for term
i (e.g., electric potential, concentration, etc.) and
Ji is the associated flow.
T is the temperature of the system (or at a given specific location at its border where flow
i takes place). Related to each flow i in the system, there is a certain entropy production. For the sake of notation, the index “
i” will be omitted in what follows, and it is tacitly assumed that blood flow is the subject under analysis. Without loss of generality, coupling between forces and flows does not take part in the analysis.
In turn, a flow is the result of
where
G is the conductance for that flow. Thus, Equation (2) reads as
Consider a given parameter
k, which, when changed, imparts changes in the flow through changes in
X and/or in
G. In addition, changes in k might cause changes in
T. In fact,
k can be seen as a vector composed of three orthogonal components: one related to
X, another related to
G and the third related to
T. In this sense, the changes in
k can be decomposed as
2.2. Elasticities
Elasticity
ε is the relative fractional change in a function. Borrowing the idea of “flux control coefficient” from metabolic control analysis [
43] and inserting Equation (3) for flow, the elasticity of the flow in relation to
k is
In a similar way, the elasticity of entropy production is
Dividing
εσ by
εJ, one obtains the relative change in entropy production per unit of change in flow in the face of a change in the parameter
k:
Equation (8) is separated into two pieces. The first right-hand-side term contains only changes in force and in conductance. The second term, within the square brackets, contains possible changes in
X,
G and temperature (see Equations (5a–c)). For ease of the initial approach, we consider that changes in
k do not lead to changes in
T (i.e.,
τ = 0). Therefore, Equation (8) now reads as
where ^ indicates the cases where
τ = 0.
In order understand the information that Equation (9) brings, consider γ = 0. That is, changes in the parameter k only cause changes in the force X. Then, . This means that each unitary increase in flow doubles the entropy production. On the other hand, consider χ = 0 (changes in k only lead to changes in the conductance G). Then, , and for each unitary increase in flow there is a unitary increase in entropy production.
Clearly, the latter seems a much more profitable scenario to address increases in flow. In fact, as will be shown, is the fairest condition once a system is optimized.
Bearing this in mind, we consider the possible outcomes for . A simple inspection of Equation (9) reveals two possibilities:
- (A).
χ < 0, γ > 0; with
- (B).
γ < 0, χ > 0; with
For both (A) and (B), ; however, flow increases in case (A), whereas it decreases in case (B). In other words, case (B) depicts a situation where the system is “paying more to obtain less”. Thus, we will focus on case (A).
Case (A) indicates that the best scenario to obtain increases in flow is to have the driving force decreasing while the conductance increases. A naïve approach would be to consider the optimal solution as X = 0 and G→∞, resulting in an unrealistic physical outcome. Nevertheless, case (A) shows what could be expected if an optimization of flow occurs.
3. Results and Pointwise Discussions
The results are separated in two subsections for clarity.
Section 3.1 presents results concerning the optimization referred to in the preceding section, i.e., how to obtain increases in flow and simultaneously minimize entropy production. From these results, the consequences in the scaling of the cardiovascular system of mammals is presented in
Section 3.2. In this section, the main goals of this study are reached, i.e., the scaling of pressure and of blood volume with body mass is derived. Then, in
Section 3.3, we further discuss the consequences for the entropy production in the cardiovascular system in the transition from an ectothermic-heterothermic condition to an endothermic-homeothermic one.
3.1. Simultaneous Optimization of Entropy Production and Blood Flow
Part of the total blood flow is directed to the maintenance of structures, which results in the force (the heart) and in the conductance (the vascular bed and blood). Therefore, blood flow cannot be unboundedly maximized because, as a consequence, more and more would be directed to the maintenance of those structures instead of supplying the rest of the system.
Consider a function
f, akin to
or to
, written as a ratio between flow and entropy production:
where
X is the force (pressure),
G is the conductance of the vascular system, and
CX and
CG are the unitary maintenance costs of the respective structures. The denominator of function
f is entropy production, where
T is the temperature.
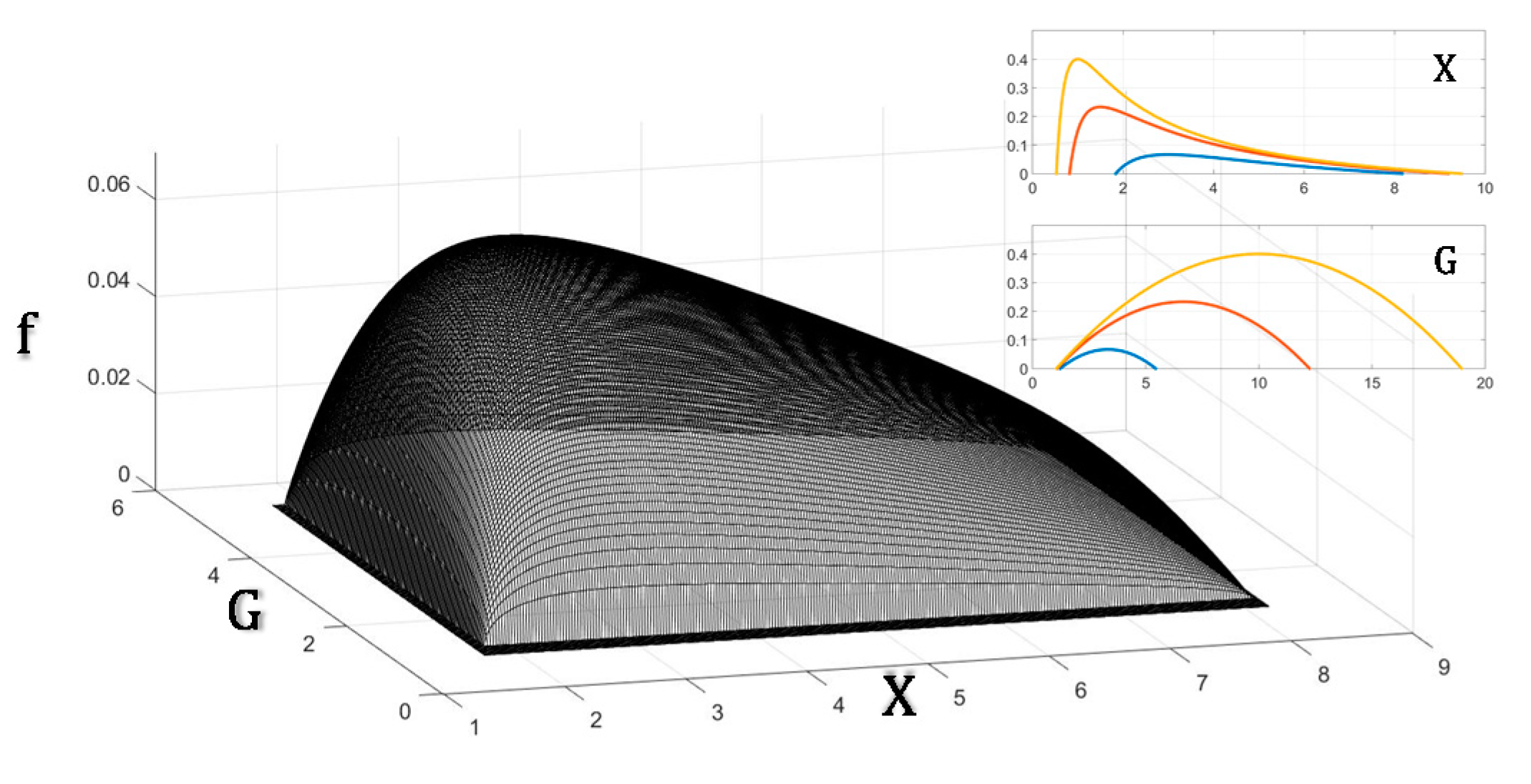
Figure 1 graphically summarizes the key problem solved in the evolution of the cardiovascular system: since blood flow is crucial to sustain the aerobic metabolic rate of organisms, how would pressure and conductance combine to maximize flow given that maintaining pressure and conductance also imposes a metabolic demand? The analytical solution is presented in the following text.
Then, an extremum of
f must be found, having
X ⋅ G = J as a restriction (i.e., to find an extremum given an arbitrary flow
J as the boundary condition). Temperature is not important at this point. Incorporating the restriction in the form of
, deriving in
X and equating to zero, results in
Next, incorporating the restriction in the form of
, deriving in
G and equating to zero, results in
Notice that the second derivative of f in relation to X and in relation to G at the extremum is negative, indicating that the point is a maximum.
The results in Equation (11a,b) are the solutions to where the conditions contained in case (A) depicted above lead the system: the force tends to be a fixed value, and the conductance tends to become linearly proportional to the flow.
Notice that by increasing the unitary cost of conductance, CG, the force is expected to increase while conductance is expected to decrease, to generate a given flow J. In addition, the unitary cost to generate force does not matter with respect to the values of X and G, which will be optimal. Because force is expected to be kept constant in case the system is optimized, this implies that χ = 0, and as stated earlier, is the fairest condition that can possibly be attained. In other words, once the relationship between flow J and entropy production σ has been maximized, changes in J and σ are of equal magnitude.
Therefore, given a certain unitary cost of the conductance structure, the optimal solution is a fixed value of force (pressure) and a conductance linearly related to the flow. Given that the properties of the blood and vessels are basically the same in a shrew and in a whale (i.e., CG does not change), these results prove that the best solution for optimizing flow and entropy production is to keep blood pressure constant irrespective of the size of the animal.
3.2. Scaling
The force term, blood pressure in the present case, is considered an intensive property in a system. Volume, on the other hand, is an extensive property. Thus, the next step is to move from the unitary frame pictured above to a size-varying one. In order to reach this goal, the size of the structures that generate force X and conductance G must be taken into account.
To maintain the size of a given structure, a given flow must be displaced to that structure. Generally speaking, there is a cost in maintaining an overall structure that allows for the flow to travel between points in the system (for instance, the electrical grid from the power plant to the city to the houses), and there is a cost in maintaining an overall structure that generates the force (for instance, the power plant). These costs can be related to the flows that go to the structures. In this sense, the function
f can be rewritten as
Notice that Equation (12) does not allow for the optimization procedure that was previously implemented. This means that it is already known that
X is a constant value (and
T does not matter for the moment). In Equation (12),
J is the overall flow in the system, and
JX and
JG are the flows needed to maintain the structures that generate pressure and conductance, respectively. Now, the flows must be expressed as scaling rules of the respective structure mass:
Inserting these scaling rules in Equation (12), we get
Notice that “1” is a constant, and therefore it is a fixed value irrespective of mass;
X and
T are also constants. Therefore, these variables are all expressed as ∝
M0. The function
f is at an extremum, and hence
f must be size-independent as well:
f ∝
M0. The mass of the conductance structure and the mass of the generating-force structure are independent of each other. Because of this, in order to have
f ∝
M0, each ratio in the numerator of Equation (13) must scale as ∝
M0. This implies that
That is, the mass MG of the structure that generates the conductance in the system scales linearly with the mass of the organism. Since the blood fills the conductance system (the vessels) and also takes part in the conductance, blood mass (or volume) must scale linearly with body mass, as was expected.
Another important scaling rule comes from this approach. The mass
MX of the structure that generates pressure must scale linearly with the size of the organism. That is, the mass of the heart scales isometrically with body mass, as empirical findings have also shown [
3,
15].
Summarizing the results obtained, Equations (11a) and (14c) show that blood pressure and that blood volume .
3.3. Temperature Changes
The final step is to understand the transition from an ecto-heterothermic (ectoHE) to an endo-homeothermic (endoHO) condition, for instance, from synapsid ancestors to primitive mammals [
44].
In a system in steady state, supply and demand must match. Therefore, the increase in metabolic rate from an ectoHE to an endoHO needs an increase in blood flow. However, the ancestor form, as any extant species, was expected to operate near its optimum, with the conductance system for its size corresponding to the value allowed by its unitary cost. Consequently, to increase flow, an increase in force must occur, i.e., an increase in blood pressure must take place as it is observed between extant endoHO and ectoHE [
45]. This increase in pressure implies a poor scenario in terms of entropy production and a very high price (non-efficient) would be paid (see
Section 2.2, also see discussion of energy waste in endotherms [
3]). However, two concomitant changes might partially mitigate this increase in exergy destruction.
Examining Equation (8), it can be seen that even slight changes (increases) in conductance (i.e.,
γ > 0) would turn the transition into a less demanding process. Given the near-optimum condition of the ancestor form, this must occur as a qualitative change in the conductance system as the qualitative change in erythrocytes (note: this has nothing to do with oxygen transport; the point is related to the blood flow conductance [
46]), and an increase in capillary density, as do occurs between extant ectoHE and endoHO [
47,
48]. These allow for an increase in flow to depend on a lower increase in pressure.
Equation (8) reveals another facet of the issue: the increase in body temperature (i.e., τ > 0) also brings the combined elasticity of entropy production and blood flow to a lesser value. This decrease in the fractional change of entropy production occurs because part of the power that would be dissipated is retained in the system as an increase in its internal energy. That is, speaking in thermodynamic terms, the system retains part of its capability to perform work. Therefore, the transition from an ectoHE to an endoHO condition is less inefficient than it would be if the increase in blood pressure were not accompanied by an increase in body temperature.
4. Discussion
The main goal of the present study was to prove, from a theoretical standpoint, why blood volume should scale linearly with body mass and blood pressure should be kept constant along mammalian phylogeny. These scaling rules are empirically known for more than a century and their theoretical explanations had constantly escaped of be derived.
The procedures described herein can be applied to any system. The primordial step is to recognize which are the relevant flow(s), force(s) and conductance(s) for the system under analysis. The cardiovascular system, for instance, comes with a bonus. Due to its intrinsically mechanical features, flow, conductance and force are inherent to the functioning of the system. Therefore, these variables come at ease in the analysis.
On the other hand, consider, for instance, the liver and its role in maintaining glucose levels. Blood flow is essential to the functioning/maintenance of the organ, but it is the interconversion of glucose-glycogen that is the flow of interest related to its function. If this distinction is not made clear, one could wrongly conclude that the mass of every organ should scale linearly with the mass of the organism. Or, in general terms, one could conclude equivocally that all structures that generate forces and that also generate conductance should scale linearly with the size of the system to which they belong.
Three other facets must also be kept clear. First, pressure, here, is so-called dynamic pressure. Thus, the hydrostatic component of pressure that is due to the increase in height of organisms is not relevant to the analysis. Second, conductance is related to the morphological size of the cardiovascular system and not the functional state of peripheral resistance in a given particular condition. Third, our analysis was aimed at the cardiovascular system of mammals, which forms a closed circuit, operates continually and has enough data to derive empirical scaling. The scaling of other circulatory systems can be approached using the procedures developed in this paper, but care must be taken as to what would be considered the conductance, where the system is open, and the pressure, where the system does not operate continuously.
In conclusion, the present study develops a new method to approach optimization in flow-generating systems, combining the maximization of flow and the minimization of entropy production. The analysis and results of the present study offer a suitable thermodynamic explanation of the scaling rules of blood pressure, blood volume and cardiac mass in mammals and also shows their plausibility from an evolutionary standpoint.