1. Introduction
Cutaneous melanoma is a highly aggressive malignancy, causing the vast majority of skin-cancer-related deaths and displaying rising incidence globally [
1]. Nevertheless, the prognosis has improved significantly due to advances in treatment options [
2]. In this context, early diagnosis and prompt identification of adverse prognostic factors are of the utmost importance for choosing the best therapeutic approach.
Cutaneous melanoma progression can be unpredictable and is influenced by several clinical and histopathological factors. The most frequently discussed clinical prognostic factors for cutaneous melanoma are age, gender, and the location of the primary tumor. However, their prognostic value is still a matter of debate. Some studies correlated older age and male gender with decreased survival [
3,
4,
5], while others found no association [
6,
7]. The primary location of the melanoma also has debatable prognostic value, but it appears location on the lower limbs is associated with a more favorable outcome [
8] in comparison to an acral location [
9] or head and neck location [
10].
Among the histopathological prognostic factors, the Breslow depth of invasion remains the most significant and is used to stage cutaneous melanomas [
11]. Patients with thin cutaneous melanomas (pT1, with Breslow depth less than 1 mm, and pT2, with Breslow depth between 1 and 2 mm) usually have excellent prognosis and patients with thick melanomas (pT4) still present high mortality rates. In contrast, patients with intermediate thickness (pT3) display highly variable outcomes [
12]. In addition to the Breslow depth of invasion, other factors influence the prognosis of primary cutaneous melanomas, including ulceration, mitotic count, regression, microsatellites, lymphovascular invasion, and perineural invasion [
12,
13,
14,
15,
16]. Recent research has shown that a new parameter, the width of invasion, could also be an independent prognostic factor [
17,
18], but the evidence is still limited.
In this context, we conducted a study on the aforementioned clinical and histopathological prognostic factors, including the width of invasion, for evaluating the progression-free survival (PFS) and overall survival (OS) in primary cutaneous melanomas with depth of invasion between 2 and 4 mm (pT3).
4. Discussion
During the follow-up time, 22.45% of the patients died, which is in concordance with data reported in the literature for pT3 cutaneous melanomas [
12]. Among the clinical characteristics, age was the only factor correlated with both PFS and OS on univariate analysis, but not on multivariate analysis. Age is considered an important prognostic factor in melanomas. Still, the poor outcomes of elderly patients may be explained by factors unrelated to the tumor itself, such as age-related comorbidities [
5]. Female gender has been associated with increased survival by some authors [
19], while, as in our study, others found no correlation between gender and prognosis [
20,
21]. We found no correlation between the primary tumor location and PFS or OS, and a clear connection is still debatable. A poorer prognosis has been reported in acral locations [
20] or the head and neck [
22] in comparison to location on the extremities [
23]. On the contrary, some studies demonstrated that head and neck location is an adverse prognostic factor only in elderly patients. In contrast, tumor location does not seem to influence disease evolution in younger individuals [
24]. Furthermore, other authors reported no association at all between tumor location and prognosis [
25].
The presence of ulceration is used to stage melanomas according to the AJCC Cancer Staging Manual [
11]. Interestingly, ulceration was more frequently noted in patients with progressive disease and in patients who died during follow-up, but these associations were not statistically significant. Similar results concerning the lack of association between ulceration and prognosis have been reported for thick cutaneous melanomas (Breslow > 4 mm) [
26], and more studies would be beneficial for evaluating this parameter in intermediate-thickness melanomas.
Regression is a controversial parameter associated with both decreased and increased survival [
16,
27,
28]. In the present study, the presence of regression was more often noted in patients with progressive disease and deceased patients than in patients without progressive disease, but the difference was not significant.
The presence of microsatellites was correlated with decreased PFS and OS on univariate analysis, and similar findings are reported in various other studies [
15,
25,
29]. Similarly, perineural invasion was also associated with PFS and OS on univariate analysis. Nevertheless, the impact of perineural invasion on cutaneous melanoma prognosis has not been extensively studied, but recent evidence has shown this parameter to be a significant predictor for poor prognosis [
26,
29,
30,
31]. Therefore, the presence of perineural invasion should be reported as it is also a risk factor for local recurrence, and in some instances, treatment strategies may include wider excision margins and/or radiotherapy [
11,
32].
Interestingly, lymphovascular invasion was only correlated with PFS on univariate analysis. These results may seem surprising as lymphovascular invasion is widely regarded as an important prognostic factor for cutaneous melanomas [
33]. Nevertheless, its predictive impact on pT4 cutaneous melanomas is questionable [
34,
35,
36], and further studies are needed to better assess the relationship between lymphovascular invasion and pT3 cutaneous melanomas.
Mitotic counts are an important prognostic factor that should be reported according to the AJCC Staging Manual. Nonetheless, the impact of this parameter may vary based on tumor stage and it is no longer used for sub-classifying pT1 tumors [
11]. Unexpectedly, in our study, there was no correlation between mitotic index and PFS or OS, even though mitotic figures tended to be slightly more numerous in patients with metastatic disease than in patients with PFS. Furthermore, the highest values were noted in patients who died during follow-up. Even though the lack of correlation between mitotic counts and prognosis might seem surprising, this might be explained by the fact that mitotic counts are correlated with Breslow depth [
37]. Since the cases included in our study had similar Breslow depths, there was no significant variation in mitotic counts among the three groups, possibly explaining the unexpectedly insignificant differences. Additionally, mitotic counts have been shown to display significant interobserver variation, which further challenges the value of this parameter [
38].
The Breslow depth of invasion is the most important prognostic factor in cutaneous melanomas as it is used for disease staging [
39]. Nevertheless, within each particular stage based on Breslow depth, patient outcomes can vary significantly. For instance, in pT4 cutaneous melanomas, increasing Breslow depth continues to be correlated with PFS and OS [
26,
27,
34,
40]. For pT3 cutaneous melanomas (Breslow between 2 and 4 mm), we have demonstrated that increasing depth is correlated with PFS and OS on univariate analysis. This is an interesting finding as it could help improve our understanding of disease progression and subsequent staging of cutaneous melanomas.
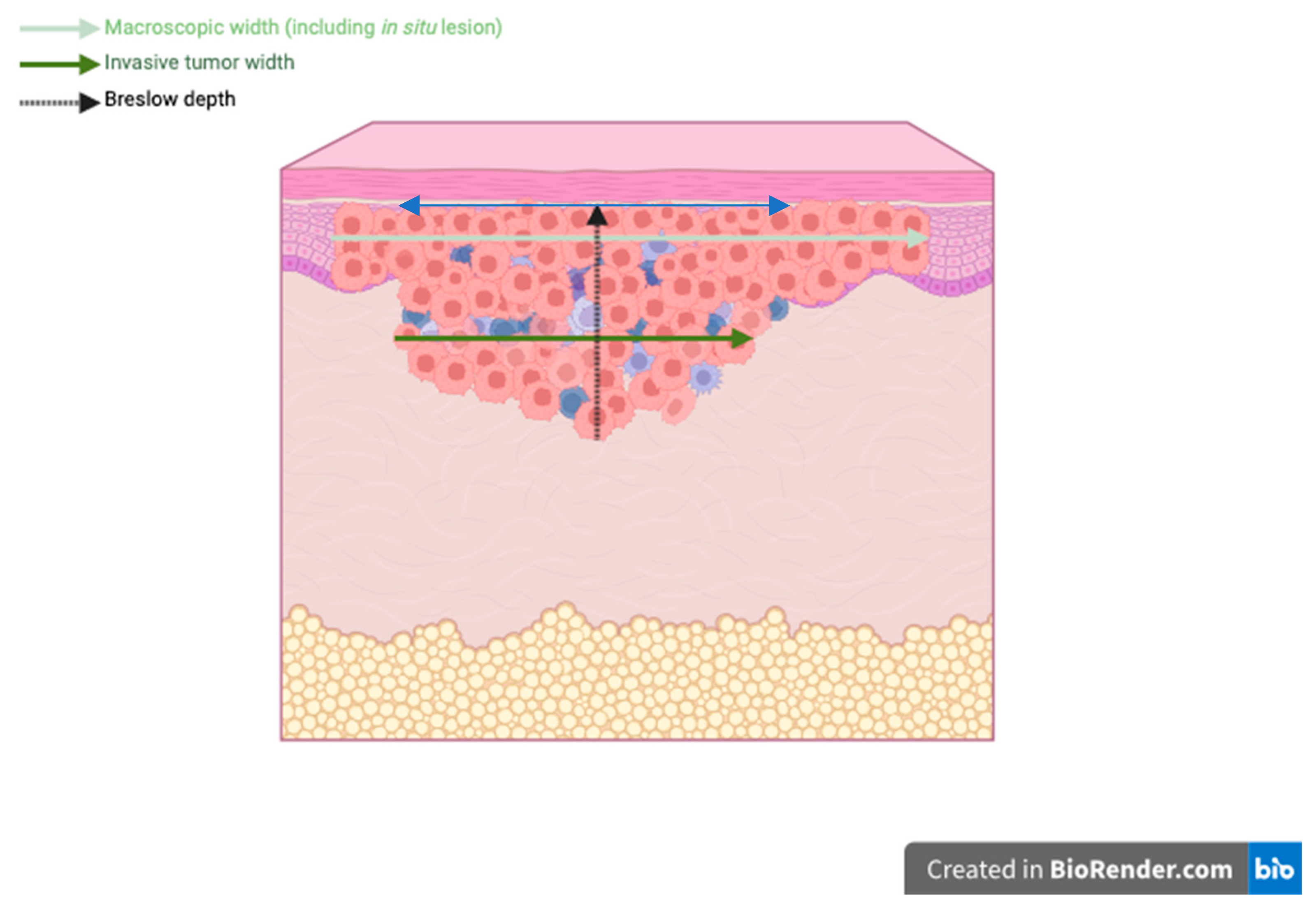
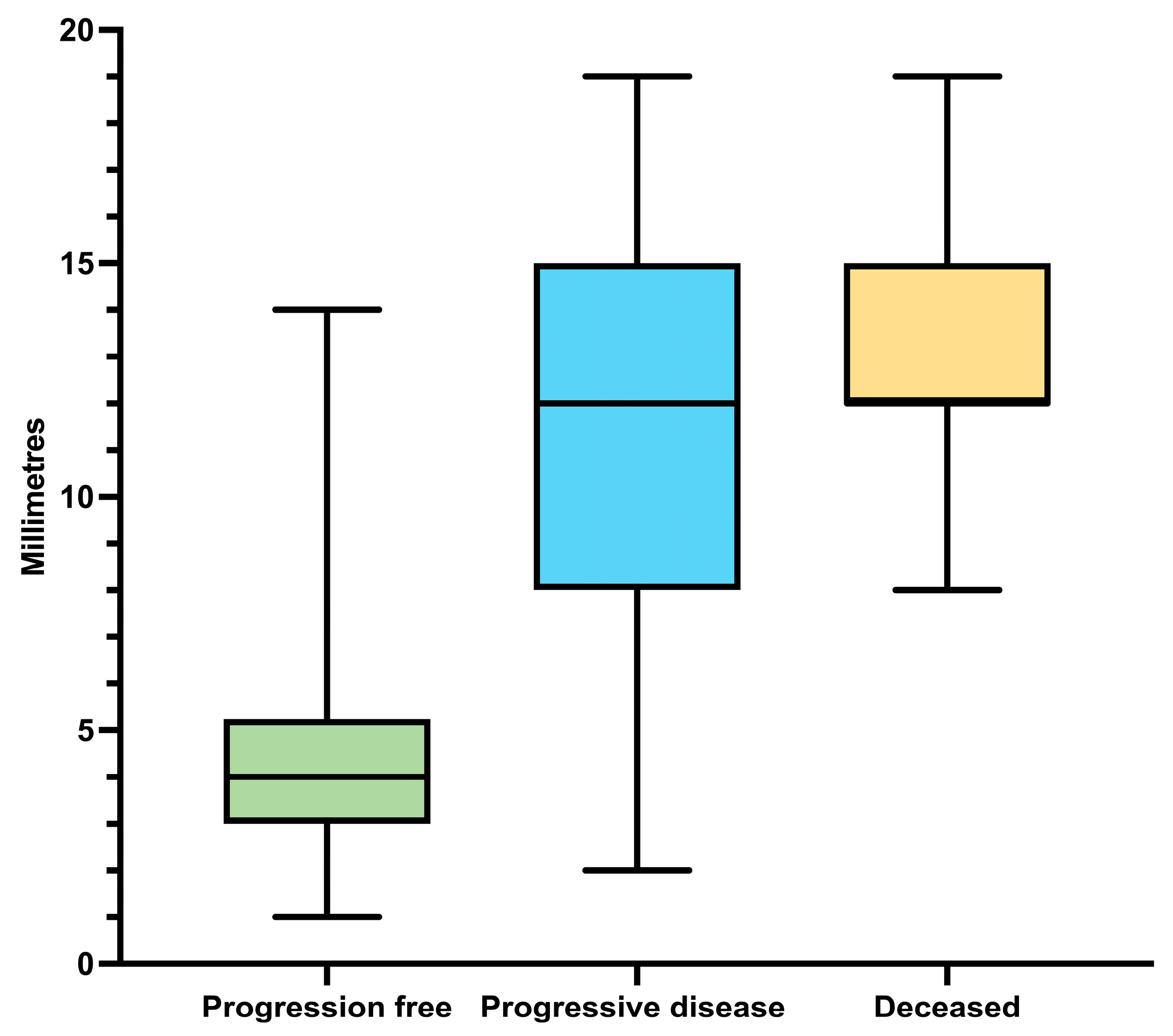
Finally, the strongest and only independent factor correlated with PFS and OS in multivariate analysis was the width of invasion. Until now, there has been insufficient data regarding the prognostic role of microscopic tumor width in cutaneous melanomas. Various studies have tried to evaluate the macroscopic tumor width and correlated it with Breslow depth [
41,
42]. A few authors have reported that tumor size and tumor volume can be regarded as independent prognostic factors and may even be superior to Breslow depth [
43,
44,
45]. However, calculating tumor volume may be time-consuming and prone to interobserver variability as there is no standard for evaluating this parameter, and different authors have reported slightly different methods, also including macroscopic measurements [
44,
45]. Macroscopic and microscopic tumor dimensions may vary significantly, as shown by Bamford M. et al. in a study on 718 cutaneous melanomas. Both macroscopic and microscopic width of invasion are correlated with survival, but the microscopic analysis is superior in this instance. The median microscopic width was 3.7 mm smaller than the macroscopic width as the former only accounts for invasive width, without in situ lesions [
18]. This measurement method was first reported in 2020 by Saldanha G. et al., who demonstrated that the microscopic measurement of tumor width is an independent prognostic factor for PFS and OS, in contrast to Breslow depth, which was not independently associated with any outcome [
17]. Our study used the same method for reporting tumor width, and the results confirmed the findings reported by Saldanha G. et al. Furthermore, these authors also addressed the prognostic value of calculated tumor area (CTA), a two-dimensional feature approximating the area of invasive melanoma cells measured on the same slide used for reporting Breslow depth [
46]. CTA is a strong independent prognostic factor, superior to Breslow depth [
17,
46]. Despite the promising results concerning tumor width and CTA, a 2023 study argues that these parameters are not superior to Breslow depth in predicting sentinel lymph node metastases and, hence, the overall outcome of the patients [
47]. Additionally, calculating the tumor area is also time-consuming and may be affected by observer estimations. These disadvantages could be solved in the future with the help of digital pathology, as computer-assisted measurement of tumor area has been correlated with recurrence-free survival [
48]. In the same context, another related novel parameter, Breslow density, has been shown to be superior to Breslow depth for estimating PFS and OS. Still, this method bears the same disadvantages as CTA: its calculation is tedious and dependent on the observer [
49]. Therefore, at present, we believe that tumor width is a more valuable parameter as it is easily determined. Having considered everything, invasive width could become a precious prognostic factor in cutaneous due to its estimated strong association with patient outcome and straightforward estimation. Nevertheless, more studies on larger populations, integrating all stages of cutaneous melanoma, are required to fully validate these findings and potentially integrate the width of invasion in future disease staging.
In spite of the promising results presented in this paper, the study also has some limitations. One such restraint is that the cases included were limited to a single center. A future multi-center study would be beneficial in order to confirm these findings on a significantly higher number of patients. Another limitation is that this study focuses solely on histopathological features of the primary tumor and it does not address the predictive value of lymph node invasion. The patients were included in this study regardless of lymph node invasion due to the fact that over half of them did not benefit from a sentinel lymph node biopsy immediately after their melanoma diagnoses. The future study also intends to address this parameter.
At present, patients with pT3 cutaneous melanomas and lymph node metastasis (stage IIIB/IIIC) are usually treated with neoadjuvant therapy, which may also include BRAF/MEK inhibitors and immune modulators [
50,
51,
52]. Nevertheless, not all pT3 cases undergo sentinel lymph node biopsy and even among those that do the positivity rate is around 11% [
53]. Therefore, a significant number of pT3 CM patients remain untreated after surgical intervention. Still, the mortality rates in pT3 cases are much higher, as proven both by this study and other authors [
12]. In this context, identifying high-risk patients regardless of lymph node status is of the utmost importance in order to choose the best therapeutic options. Assessing the width of the invasion may further increase the accuracy of identifying such patients.