Assessing Microplastic Prevalence and Dispersion from Saigon Urban Canals via Can Gio Mangrove Reserve to East Sea by Raman Scattering Microscopy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Equipment
2.2. Study Areas and Sampling Methods
2.3. The Workflow for Marine Microplastic Extraction and Analysis
2.4. Quality Control
2.5. Statistical Analysis
3. Results and Discussion
3.1. Combined Qualification and Quantification of Microplastics by Raman Microscopy
3.1.1. Visual Identification
3.1.2. Spectroscopic Identification
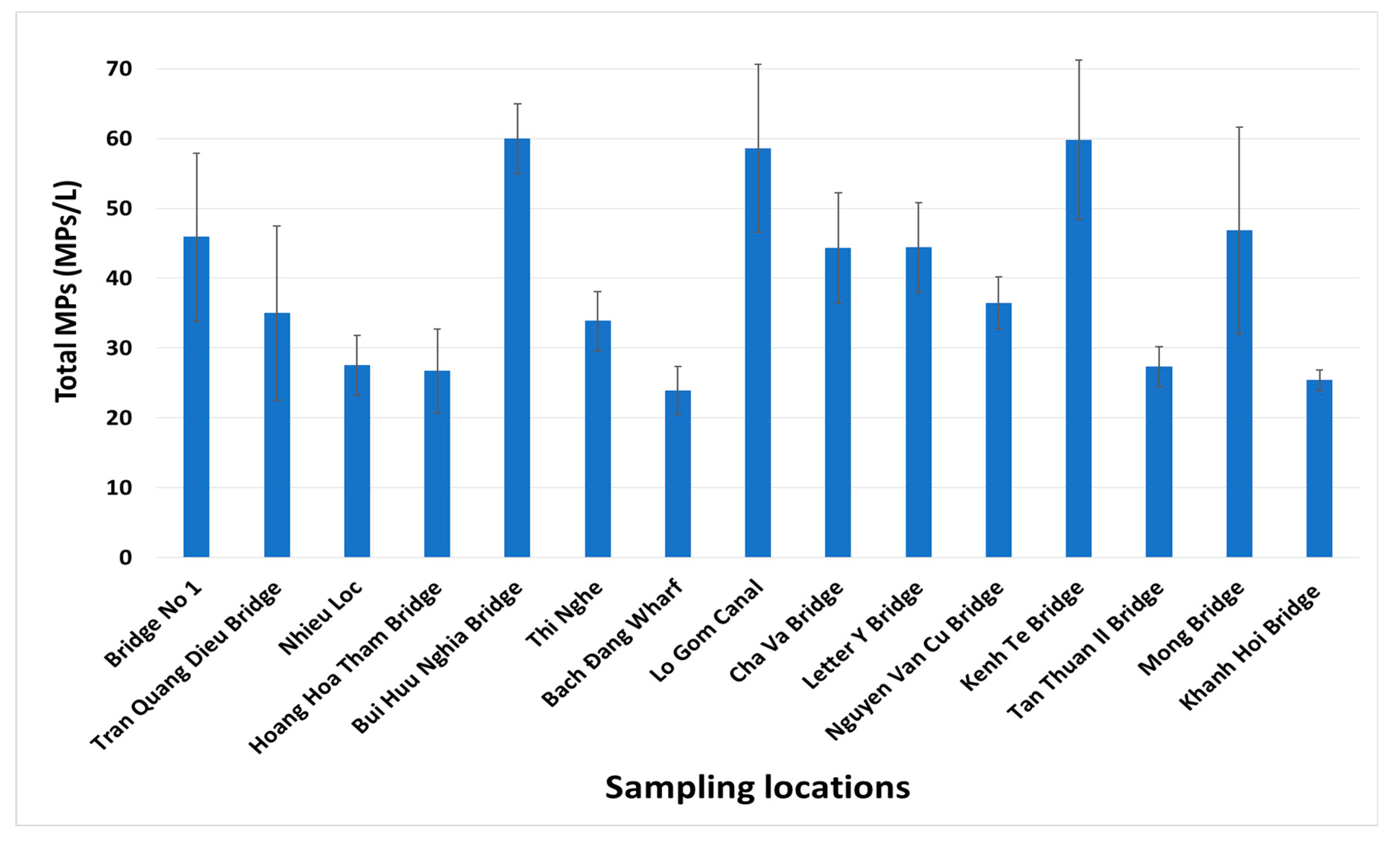
3.2. Microplastic Pollution in the Freshwater at Saigon Urban Canals
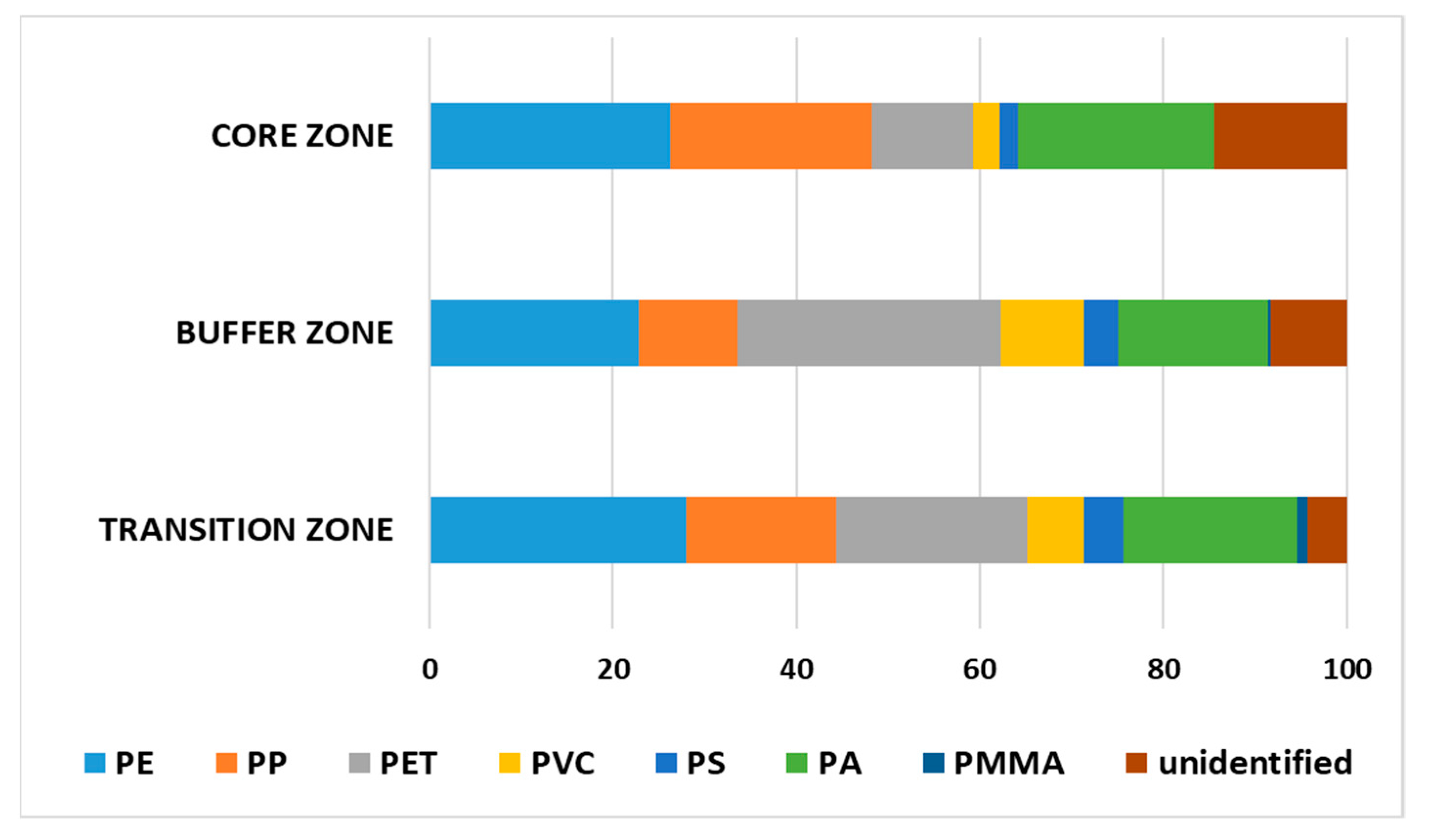
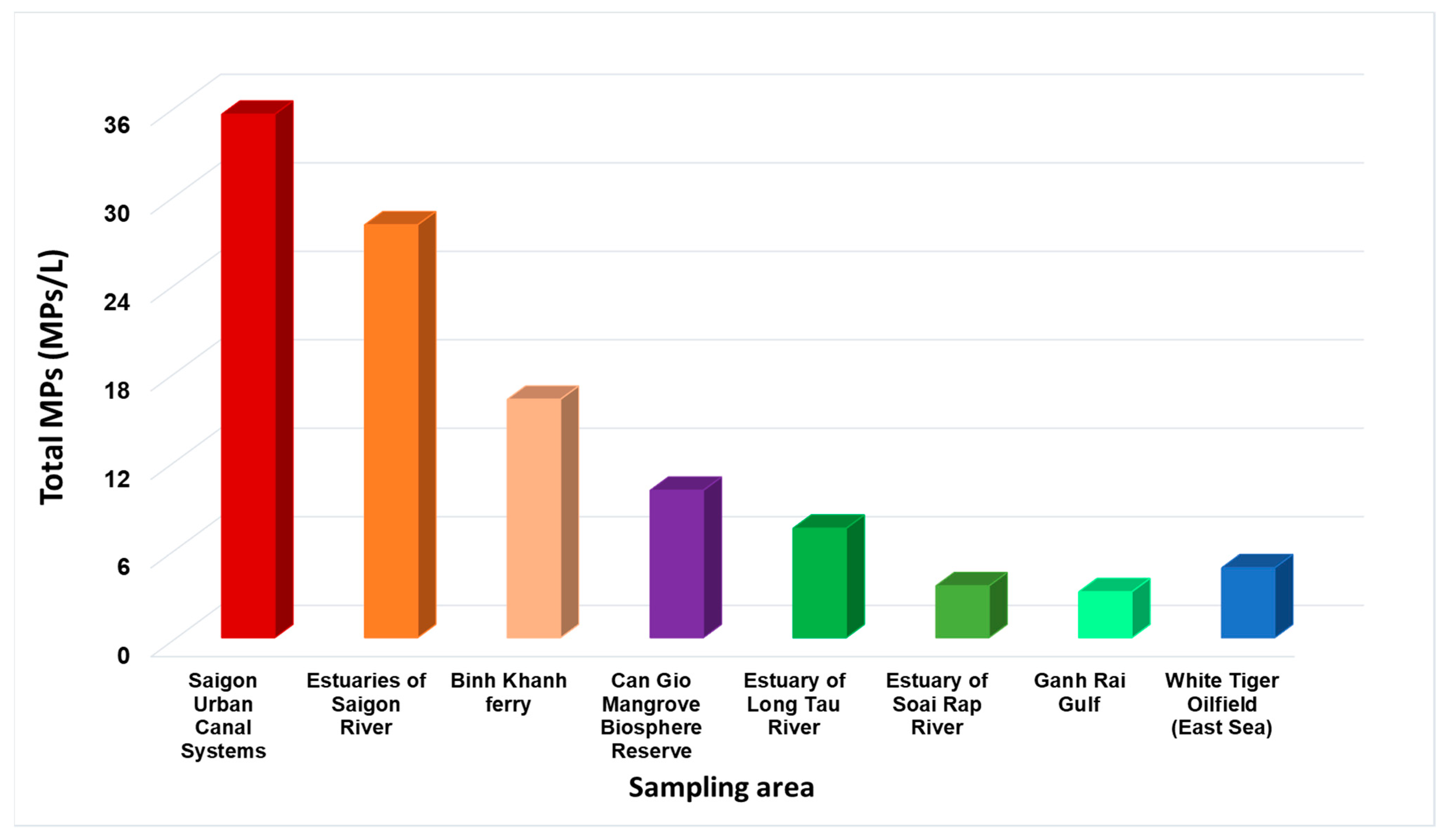
3.3. Spatial Distribution of Marine Microplastic from Can Gio Biosphere Reserve to the East Sea
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| The Abbreviations of Plastic Types | |
| MPs | Microplastics, total microplastics |
| PE | Poly-Ethylene |
| LDPE | Low-Density Polyethylene |
| MDPE | Medium-Density Polyethylene |
| HDPE | High-Density Polyethylene |
| PP | Poly-Propylene |
| PS | Poly-Styrene |
| PVC | Poly-Vinyl Chloride |
| PET (PETE) | Poly-Ethylene Terephthalate |
| PA | Poly-Amides, Nylon |
| PMMA | Poly Methyl Metacrylate |
| Geographic Terms and Abbreviations | |
| SG, HCMC | Hồ Chí Minh City, “Saigon” was also used alternatively |
| NL-TN | Nhiêu Lộc–Thị Nghè canal in Saigon city |
| BN | Bến Nghé canal in Saigon city |
| KT | Tẻ canal in Saigon city |
| LG | Lò Gốm canal in Saigon city |
| CG | Cần Giờ—a coastal suburban district of Ho Chi Minh City, UNESCO Can Gio Mangrove Biosphere Reserve |
| East Sea | East Sea, a name of Vietnamese people for South China Sea |
| CT | Can Gio Mangrove–Transition zone |
| CB | Can Gio Mangrove–Buffer zone |
| CC | Can Gio Mangrove–Core zone |
References
- Plastics Europe. Plastics—The Facts 2017: An Analysis of European plastics Production, Demand and Waste Data. 2020. Available online: https://www.plasticseurope.org/application/files/5716/0752/4286/AF_Plastics_the_facts-WEB-2020-ING_FINAL.pdf (accessed on 3 June 2022).
- Scalenghe, R. Resource or waste? A perspective of plastics degradation in soil with a focus on end-of-life options. Heliyon 2018, 4, e00941. [Google Scholar] [CrossRef] [PubMed]
- Le, D.T. Overview of Marine Plastic Debris in Vietnam in Relation to International Context FIG Working Week 2019. In Proceedings of the Geospatial Information for a Smarter Life and Environmental Resilience Hanoi, Hanoi, Vietnam, 22–26 April 2019. [Google Scholar]
- Jambeck, J.R.; Geyer, R.; Wilcox, C.; Siegler, T.R.; Perryman, M.; Andrady, A.; Narayan, R.; Law, K.L. Plastic waste inputs from land into the ocean. Science 2015, 347, 768–771. [Google Scholar] [CrossRef] [PubMed]
- Verma, R.L.; Borongan, G.; Memon, M. Municipal Solid Waste Management in Ho Chi Minh City, Viet Nam, Current Practices and Future Recommendation. Procedia. Environ. Sci. 2016, 35, 127–139. [Google Scholar] [CrossRef]
- Kieu-Le, T.C.; Strady, E.; Perset, M. Life Cycle of Floating Debris in the Canals of Ho Chi Minh City. PADDI. 2016. Available online: https://hal.archives-ouvertes.fr/hal-02357363/document (accessed on 3 June 2022).
- Cormier-Salem, M.C.; Nguyen, V.T.; Burgos, A.; Durand, J.D.; Bettarel, Y.; Klein, J.; Hoang, D.H.; Panfili, J. The mangrove’s contribution to people: Interdisciplinary pilot study of the Can Gio Mangrove Biosphere Reserve in Viet Nam. C. R. Geosci. 2017, 349, 341–350. [Google Scholar] [CrossRef]
- Ng, E.-L.; Lwanga, E.H.; Eldridge, S.M.; Johnston, P.; Hu, H.-W.; Geissen, V.; Chen, D. An overview of microplastic and nanoplastic pollution in agroecosystems. Sci. Total Environ. 2018, 627, 1377–1388. [Google Scholar] [CrossRef] [PubMed]
- Mai, L.; Bao, L.-J.; Shi, L.; Wong, C.S.; Zeng, E.Y. A review of methods for measuring microplastics in aquatic environments. Environ. Sci. Pollut. Res. Int. 2018, 25, 11319–11332. [Google Scholar] [CrossRef]
- Gregory, M.R. Environmental implications of plastic debris in marine settings—entanglement, ingestion, smothering, hangers-on, hitch-hiking and alien invasions. Phil. Trans. R. Soc. B. 2009, 364, 2013–2025. [Google Scholar] [CrossRef]
- Botterell, Z.L.R.; Beaumont, N.; Dorrington, T.; Steinke, M.; Thompson, R.C.; Lindeque, P.K. Bioavailability and effects of microplastics on marine zooplankton: A review. Environ. Pollut. 2019, 245, 98–110. [Google Scholar] [CrossRef]
- Udayakumar, K.V.; Gore, P.M.; Kandasubramanian, B. Foamed materials for oil-water separation. Chem. Eng. J. Adv. 2021, 5, 100076. [Google Scholar] [CrossRef]
- Auta, H.S.; Emenike, C.U.; Fauziah, S.H. Distribution and importance of microplastics in the marine environment: A review of the sources, fate, effects, and potential solutions. Environ. Int. 2017, 102, 165–176. [Google Scholar] [CrossRef] [PubMed]
- Braun, U.; Stein, U.; Schritt, H.; Altmann, K.; Bannick, C.; Becker, R.; Bitter, H.; Dierkes, G.; Enders, K.; Kyriakos, A.E. Analysis of Microplastics—Sampling, Preparation and Detection Methods. Status Report within the Framework Program Plastics in the Environment. 2021. Available online: https://bmbf-plastik.de/sites/default/files/2021-12/Status%20Report_Analysis%20of%20Microplastics_PidU_May_2021_0.pdf (accessed on 3 June 2022).
- Enders, K.; Lenz, R.; Stedmon, C.A.; Nielsen, T.G. Abundance, size and polymer composition of marine microplastics ≥ 10 μm in the Atlantic Ocean and their modelled vertical distribution. Mar. Pollut. Bull. 2015, 100, 70–81. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, T.M.; Kärrman, A.; Rotander, A.; Hassellöv, M. Comparison between manta trawl and in situ pump filtration methods, and guidance for visual identification of microplastics in surface waters. Environ. Sci. Pollut. Res. 2020, 27, 5559–5571. [Google Scholar] [CrossRef] [PubMed]
- De Witte, B.; Devriese, L.; Bekaert, K.; Hoffman, S.; Vandermeersch, G.; Cooreman, K.; Robbens, J. Quality assessment of the blue mussel (Mytilus edulis): Comparison between commercial and wild types. Mar. Pollut. Bull. 2014, 85, 146–155. [Google Scholar] [CrossRef]
- Kosuth, M.; Mason, S.A.; Wattenberg, E.V. Anthropogenic contamination of tap water, beer, and sea salt. PLoS ONE 2018, 13, e0194970. [Google Scholar] [CrossRef] [PubMed]
- Shim, W.J.; Song, Y.K.; Hong, S.H.; Jang, M. Identification and quantification of microplastics using Nile Red staining. Mar. Pollut. Bull. 2016, 113, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Fries, E.; Jens, H.D.; Willmeyer, J.; Nuelle, M.T.; Ebert, M.; Remy, D. Identification of polymer types and additives in marine microplastic particles using pyrolysis-GC/MS and scanning electron microscopy. Environ. Sci. Process Impacts 2013, 15, 1949–1956. [Google Scholar] [CrossRef]
- Erni-Cassola, G.; Gibson, M.I.; Thompson, R.C.; Christie-Oleza, J.A. Lost, but Found with Nile Red: A Novel Method for Detecting and Quantifying Small Microplastics (1 mm to 20 μm) in Environmental Samples. Environ. Sci. Technol. 2017, 51, 13641–13648. [Google Scholar] [CrossRef]
- Song, Y.K.; Hong, S.H.; Jang, M.; Han, G.M.; Shim, W.J. Occurrence and distribution of microplastics in the sea surface microlayer in Jinhae Bay, South Korea. Arch. Environ. Contam. Toxicol. 2015, 69, 279–287. [Google Scholar] [CrossRef]
- Cincinelli, A.; Scopetani, C.; Chelazzi, D.; Lombardini, E.; Martellini, T.; Katsoyiannis, A.; Fossi, M.C.; Corsolini, S. Microplastic in the surfac e waters of the Ross Sea (Antarctica): Occurrence, distribution and characterization by FTIR. Chemosphere 2017, 175, 391–400. [Google Scholar] [CrossRef]
- Schymanski, D.; Goldbeck, C.; Humpf, H.-C.; Fürst, P. Analysis of microplastics in water by micro-Raman spectroscopy: Rele ase of plastic particles from different packaging into mineral water. Water Res. 2018, 129, 154–162. [Google Scholar] [CrossRef]
- Gillibert, R.; Balakrishnan, G.; Deshoules, Q.; Tardivel, M.; Magazzù, A.; Donato, M.G.; Maragò, O.M.; de La Chapelle, M.L.; Colas, F.J.; Lagarde, F.; et al. Raman Tweezers for Small Microplastics and Nanoplastics Identification in Seawater. Environ. Sci. Technol. 2019, 53, 9003–9013. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro-Claro, P.; Nolasco, M.M.; Catarina, A. Chapter 5—Characterization of Microplastic by Raman Spectroscopy. Compr. Anal. Chem. 2017, 75, 119–151. [Google Scholar]
- Käppler, A.; Fischer, D.; Oberbeckmann, S.; Schernewski, G.; Labrenz, M.; Eichhorn, K.-J.; Voit, B. Analysis of environmental microplastics by vibrational microspectroscopy: FTIR, Raman or both? Anal. Bioanal. Chem. 2016, 408, 8377–8391. [Google Scholar] [CrossRef]
- Shim, W.J.; Hongab, S.H.; Eo, S.E. Identification methods in microplastic analysis: A review. Anal. Methods 2017, 9, 1384–1391. [Google Scholar] [CrossRef]
- Hermabessiere, L.; Himber, C.; Boricaud, B.; Kazour, M.; Amara, R.; Cassone, A.-L.; Laurentie, M.; Paul-Pont, I.; Soudant, P.; Dehaut, A.; et al. Optimization, performance, and application of a pyrolysis-GC/MS method for the identification of microplastics. Anal. Bioanal. Chem. 2018, 410, 6663–6676. [Google Scholar] [CrossRef] [PubMed]
- Lahens, L.; Strady, E.; Le, K.T.C.; Dris, R.; Boukerma, K.; Rinnert, E.; Gasperi, J.; Tassin, B. Macroplastic and microplastic contamination assessment of a tropical river (Saigon River, Vietnam) transversed by a developing megacity. Environ. Pollut. 2018, 236, 661–671. [Google Scholar] [CrossRef]
- Strady, E.; Kieu-Le, T.C.; Gasperi, J.; Tassin, B. Temporal dynamic of anthropogenic fibers in a tropical river-estuarine system. Environ. Pollut. 2020, 259, 113897. [Google Scholar] [CrossRef]
- Strady, E.; Dang, T.H.; Dao, T.D.; Dinh, H.N.; Do, T.T.D.; Duong, T.N.; Duong, T.T.; Hoang, D.A.; Kieu-Le, T.C.; Le, T.P.Q.; et al. Baseline assessment of microplastic concentrations in marine and freshwater environments of a developing Southeast Asian country, Viet Nam. Mar. Pollut. Bull. 2021, 162, 111870. [Google Scholar] [CrossRef]
- Su, L.; Xue, Y.; Li, L.; Yang, D.; Kolandhasamy, P.; Li, D.; Shi, H. Microplastics in Taihu Lake, China. Environ. Pollut. 2016, 216, 711–719. [Google Scholar] [CrossRef]
- Lusher, A.L.; Burke, A.; O ′Connor, I.; Officer, R. Microplastic pollution in the Northeast Atlantic Ocean: Validated and opportunistic sampling. Mar. Pollut. Bull. 2014, 88, 325–333. [Google Scholar] [CrossRef]
- Zhao, S.; Zhu, L.; Wang, T.; Li, D. Suspended microplastics in the surface water of the Yangtze Estuary System, China: First observations on occurrence, distribution. Mar. Pollut. Bull. 2014, 86, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Khuyen, V.T.K.; Le, D.V.; Anh, L.H.; Fischer, A.R.; Dornack, C. Investigating the Correlation of Microplastic Pollution Between Seawater and Marine Salt Using Micro-Raman Spectroscopy. Front. Mar. Sci. 2021, 8, 735975. [Google Scholar] [CrossRef]
- Zhao, S.; Zhu, L.; Li, D. Microscopic anthropogenic litter in terrestrial birds from Shanghai, China: Not only plastics but also natural fibers. Sci. Total Environ. 2016, 550, 1110–1115. [Google Scholar] [CrossRef] [PubMed]
- Chubarenko, I.; Bagaev, A.; Zobkov, M.; Esiukova, E. On some physical and dynamical properties of microplastic particles in marine environment. Mar. Pollut. Bull. 2016, 108, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Ismail, A.; Adilah, N.M.B.; Nurulhudha, M.J. Plastic pellets along Kuala Selangor-Sepang coastline. Malays. Appl. Biol. 2009, 38, 85–88. Available online: http://psasir.upm.edu.my/id/eprint/16233 (accessed on 3 June 2022).
- Frias, J.P.G.L.; Sobral, P.; Ferreira, A.M. Organic pollutants in microplastics from two beaches of the Portuguese coast. Mar. Pollut. Bull. 2010, 60, 1988–1992. [Google Scholar] [CrossRef]
- Zhu, L.; Bai, H.; Chen, B.; Sun XQu, K.; Xia, B. Microplastic pollution in North Yellow Sea, China: Observations on occurrence, distribution and identification. Sci. Total Environ. 2018, 636, 20–29. [Google Scholar] [CrossRef]
- Kunz, A.; Walther, B.A.; Löwemark, L.; Lee, Y.C. Distribution and quantity of microplastic on sandy beaches along the northern coast of Taiwan. Mar. Pollut. Bull. 2016, 111, 126–135. [Google Scholar] [CrossRef]
- Crawford, C.B.; Quinn, B. Microplastic Pollutants, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 101–130. [Google Scholar]
- Endo, S.; Takizawa, R.; Okuda, K.; Takada, H.; Chiba, K.; Kanehiro, H.; Ogi, H.; Yamashita, R.; Date, T. Concentration of polychlorinated biphenyls (PCBs) in beached resin pellets: Variability among individual particles and regional differences. Mar. Pollut. Bull. 2005, 50, 1103–1114. [Google Scholar] [CrossRef]
- Prata, J.C.; Costa, J.P.; Duarte, A.C.; Rocha-Santos, T. Methods for sampling and detection of microplastics in water and sediment: A critical review. TrAC Trends Anal. Chem. 2019, 110, 150–159. [Google Scholar] [CrossRef]
- Al-Azzawi, M.S.M.; Kefer, S.; Weißer, J.; Reichel, J.; Schwaller, C.; Glas, K.; Knoop, O.; Drewes, J.E. Validation of Sample Preparation Methods for Microplastic Analysis in Wastewater Matrices—Reproducibility and Standardization. Water 2020, 12, 2445. [Google Scholar] [CrossRef]
- Hu, C.; Chen, X.; Chen, J.; Zhang, W.; Zhang, M.Q. Observation of mutual diffusion of macromolecules in PS/PMMA binary films by confocal Raman microscopy. Soft Matter 2012, 8, 4780. [Google Scholar] [CrossRef]
- Bruckmoser, K.; Resch, K.; Kisslinger, T.; Lucyshyn, T. Measurement of interdiffusion in polymeric materials by applying Raman spectroscopy. Polym. Test. 2015, 46, 122–133. [Google Scholar] [CrossRef]
- Gopanna, A.; Mandapati, R.N.; Thomas, S.P.; Rajan, K.; Chavali, M. Fourier transform infrared spectroscopy (FTIR), Raman spectroscopy and wide angle X ray scattering (WAXS) of polypropylene (PP)/cyclic olefin copolymer (COC) blends for qualitative and quantitative analysis. Polym. Bull. 2019, 76, 4259–4274. [Google Scholar] [CrossRef]
- Daniel, J.S.; Wiebeck, H. Predicting LDPE/HDPE blend composition by CARS-PLS regression and confocal Raman spectroscopy. Polímeros 2019, 29, 1–11. [Google Scholar] [CrossRef]
- Gündoğdu, S. Contamination of table salts from Turkey with microplastics. Food Addit. Contam. Part A 2018, 35, 1006–1014. [Google Scholar] [CrossRef]
- Milani, A. Unpolarized and Polarized Raman Spectroscopy of Nylon-6 Polymorphs: A Quantum Chemical Approach. J. Phys. Chem. B. 2015, 119, 3868–3874. [Google Scholar] [CrossRef]
- Käppler, A.; Windrich, F.; Löder, M.G.J.; Malanin, M.; Fischer, D.; Labrenz, M.; Eichhorn, K.J.; Voit, B. Identifiation of microplastics by FTIR and Raman microscopy: A novel silicon fiter substrate opens the important spectral range below 1300 cm−1 for FTIR transmission measurements. Anal. Bioanal. Chem. 2015, 407, 6791–6801. [Google Scholar] [CrossRef]
- Alexiou, F.V.; Mathioudakis, G.N.; Andrikopoulos, K.S.; Beobide, A.S.; Voyiatzis, G.A. Poly(ethylene Terephthalate) Carbon-Based Nanocomposites: A Crystallization and Molecular Orientation Study. Polymers 2020, 12, 2626. [Google Scholar] [CrossRef]
- Solodovnichenko, V.S.; Polyboyarov, V.A.; Zhdanok, A.A.; Arbuzov, A.B.; Zapevalova, E.S.; Kryazhev Yu, G.; Likholobov, V.A. Synthesis of carbon materials by the short-term mechanochemical activation of polyvinyl chloride. Procedia Eng. 2016, 152, 747–752. [Google Scholar] [CrossRef]
- Emmerik, T.V.; Strady, E.; Kieu-Le, T.C.; Nguyen, L.; Gratiot, N. Seasonality of riverine macroplastic transport. Sci. Rep. 2019, 9, 13549. [Google Scholar] [CrossRef]
- Frère, L.; Paul-Pont, I.; Moreau, J.; Soudant, P.; Lambert, C.; Huvet, A.; Rinnert, E. A semi-automated Raman micro-spectroscopy method for morphological and chemical characterizations of microplastic litter. Mar. Pollut. Bull. 2016, 113, 461–468. [Google Scholar] [CrossRef]
- Barnes, K.A.D.; Galgani, F.; Thompson, R.C.; Barlaz, M. Accumulation and fragmentation of plastic debris in global environments. Phil. Trans. R. Soc. B. 2009, 364, 1985–1998. [Google Scholar] [CrossRef]
- Lenz, R.; Enders, K.; Stedmon, C.A.; Mackenzie, D.M.A.; Nielsen, T.G. A critical assessment of visual identification of marine microplastic using Raman spectroscopy for analysis improvement. Mar. Pollut. Bull. 2015, 100, 82–91. [Google Scholar] [CrossRef]
- Khuyen, V.T.K.; Le, D.V.; Fischer, A.R.; Dornack, C. Comparison of microplastic pollution in beach sediment and seawater at UNESCO Can Gio Mangrove Biosphere Reserve. Glob. Chall. 2021, 5, 2100044. [Google Scholar] [CrossRef]
- Lusher, A.; Tirelli, V.; O’Connor, I.; Officer, R. Microplastics in Arctic polar waters: The first reported values of particles in surface and sub-surface samples. Sci. Rep. 2015, 5, 14947. [Google Scholar] [CrossRef]
- Mazda, Y.; Wolanski, E. Chapter 8 Hydrodynamics and Modeling of Water Flow in Mangrove Areas. In Coastal Wetlands: An Integrated Ecosystem Approach, 1st ed.; Perillo, G., Wolanski, E., Cahoon, D., Brinson, M., Eds.; Elsevier: Amsterdam, The Netherlands, 2009; p. 244. ISBN 978-0-444-53103-2. [Google Scholar]
- Li, R.; Zhang, L.; Xue, B.; Wang, Y. Abundance and characteristics of microplastics in the mangrove sediment of the semi- enclosed Maowei Sea of the South China Sea: New implications for location, rhizosphere, and sediment compositions. Environ. Pollut. 2019, 244, 685–692. [Google Scholar] [CrossRef]
- Valiela, I.; Cole, M.L. Comparative evidence that saltmarshes and mangroves may protect sea grass meadow from land-derived nitrogen loads. Ecosystems 2002, 5, 92–102. [Google Scholar] [CrossRef]
- Adame, F.M.; Neil, D.; Wright, S.F.; Lovelock, C.E. Sedimentation within and among mangrove forests along a gradient of geomorphological settings. Estuar. Coast. Shelf Sci. 2010, 86, 21–30. [Google Scholar] [CrossRef]
- Di, M.; Wang, J. Microplastics in surface waters and sediments of the Three Gorges Reservoir, China. Sci. Total Environ. 2018, 616, 1620–1627. [Google Scholar] [CrossRef]
- Miller, R.Z.; Watts, A.J.; Winslow, B.O.; Galloway, T.S.; Barrows, A.P. Mountains to the sea: River study of plastic and non-plastic microfiber pollution in the northeast USA. Mar. Pollut. Bull. 2017, 124, 245–251. [Google Scholar] [CrossRef]
- Sheavly, S.B. Marine debris—An overview of a critical issue for our oceans. In Proceedings of the Sixth Meeting of the UN Open-ended Informal Consultative Processes on Oceans & the Law of the Sea, New York, NY, USA, 6–10 June 2005. [Google Scholar]
- Hasnat, M.A.; Rahman, M.A. A review paper on the hazardous effect of plastic debris on marine biodiversity with some possible remedies. Asian J. Med. Biol. Res. 2018, 4, 233–241. [Google Scholar] [CrossRef]
- Browne, M.A. Chapter 9 Sources and Pathways of Microplastics to Habitats. In Marine Anthropogenic Litter, 1st ed.; Melanie, B., Lars, G., Michael, K., Eds.; Springer Open: Gothenburg, Germany, 2015; pp. 229–244. [Google Scholar] [CrossRef]
- Weber, A.; Scherer, C.; Brennholt, N.; Reifferscheid, G.; Wagner, M. PET microplastics do not negatively affect the survival, development, metabolism and feeding activity of the freshwater invertebrate Gammarus pulex. Environ. Pollut. 2018, 234, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Issac, M.N.; Kandasubramanian, B. Effect of microplastics in water and aquatic systems. Environ. Sci. Pollut. Res. 2021, 28, 19544–19562. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Tan, Z.; Peng, J.; Qiu, Q.; Li, M. The behaviours of microplastics in the marine environment. Mar. Environ. Res. 2016, 113, 7–17. [Google Scholar] [CrossRef]
- Castillo, A.B.; Al-Maslamani, I.; Obbard, J.P. Prevalence of microplastics in the marine waters of Qatar. Mar. Pollut. Bull. 2016, 111, 260–267. [Google Scholar] [CrossRef]
- Nor, N.H.; Obbard, J.P. Microplastics in Singapore’s coastal mangrove ecosystems. Mar. Pollut. Bull. 2014, 79, 278–283. [Google Scholar] [CrossRef]

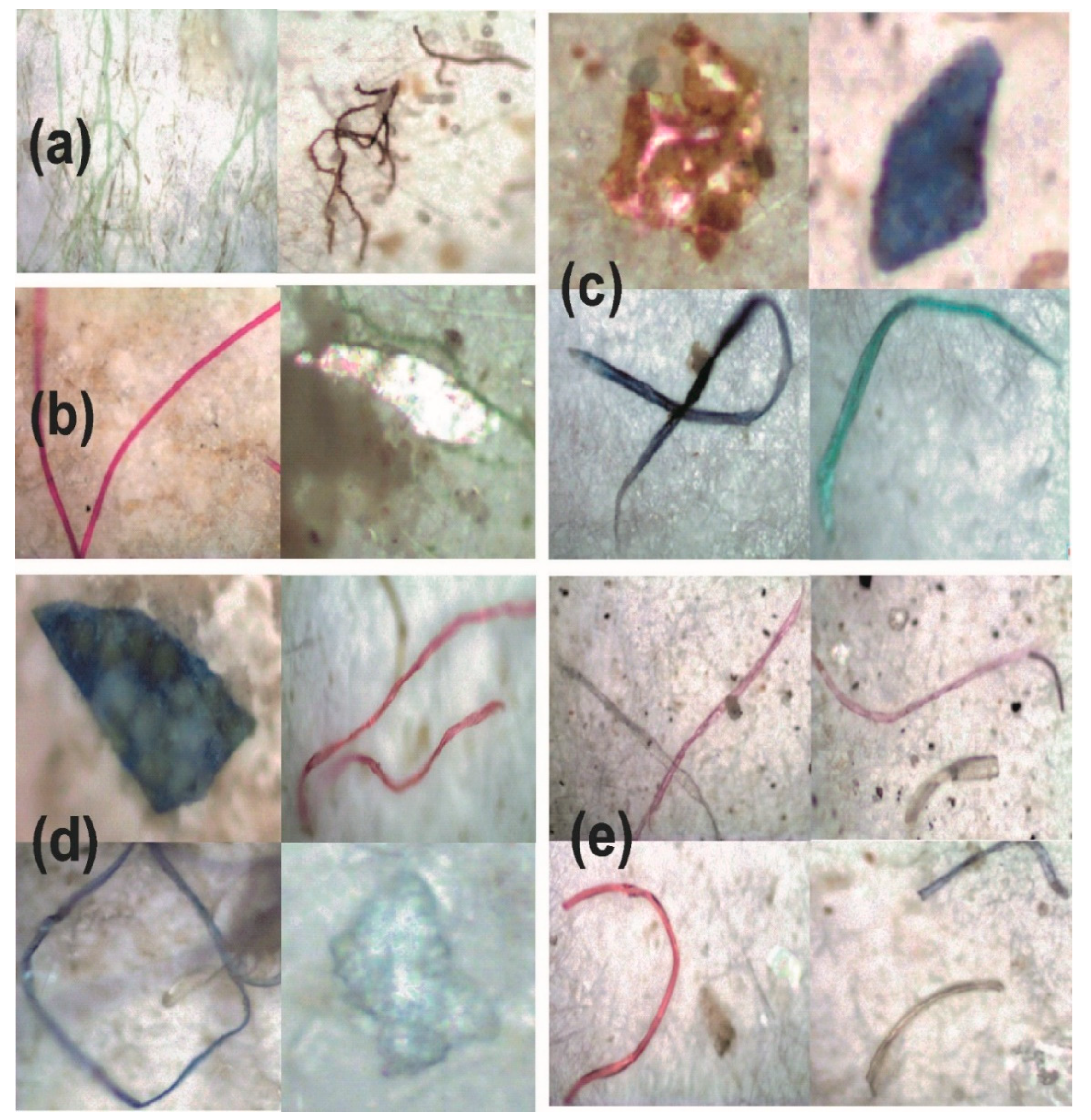
| Polymer | Raman Bands (cm−1) | Raman Bands (cm−1) | Bond Assignment |
|---|---|---|---|
| of Sampled MPs | of Plastic Standards | ||
| Polystyrene (PS) | 1612.87 | 1604.24 | phenyl ring stretch [47,48] |
| 1015.03 | 1001.75 | C–C in-plane ring deformation +, C–H out-of-plane deformation [47] | |
| 622.43 | 621.1 | in-plane ring deformation [47,48] | |
| Poly (methyl methacrylate) (PMMA) | 1679.85 | 1723.71 | C–O–C symmetric stretch [47] |
| 1512.1 | 1447.51 | –CH2– deformation [48] | |
| 1154.13 | 1181.31 | C–C–C–C stretch [48] | |
| 835.31 | 808.67 | C=O stretch [47] | |
| Polypropylene (PP) | 2886.5 | 2889.14 | –CH3 stretch [49] |
| 1451.79 | 1458.89 | –CH2– deformation [48] | |
| 1325.16 | 1328.97 | C–C stretch [49] | |
| 1150.51 | 1153.82 | –CH3 rock [49] | |
| 805.23 | 808.67 | –CH2– rock [48] | |
| Polyethylene (LDPE) | 2928.9 | 2884.5 | –CH2– stretch [50] |
| 1442.63 | 1441.9 | –CH2– wag [50] | |
| 1293.13 | 1296.25 | –CH2– twist [50] | |
| 1172.35 | 1124.36 | C–C stretch [50] | |
| 1069.69 | 1060.97 | C–C stretch [50] | |
| Polyamide-6, nylon-6 (PA-6) | 2895.67 | 2900.2 | –CH2– stretch [51] |
| 1680.05 | 1633.39 | unknown group [51] | |
| 1440.22 | 1441.9 | –CH2– [51,52] | |
| 1383.02 | 1430.49 | –CH2– bend [51,52] | |
| 1094.93 | 928.73 | –CH2– twist [52] | |
| Poly-ethylene terephthalate (PET) | 1753.59 | 1725.34 | C=O stretch [53,54] |
| 1602.3 | 1614.52 |  C=C [54] C=C [54] | |
| 1302.58 | 1288.21 | C–C | |
| 846.1 | 855.4 | C=C [53] | |
| 693.92 | 628.46 |  [53] [53] | |
| Polyvinyl chloride (PVC) | 2892.78 | 2852.09 | C–H stretch [55] |
| 1436.72 | 1430.19 | –CH2– bend [51,55] | |
| 1360.33 | 1319.11 | unknown group [55] | |
| 693.92 | 632.64 | C–Cl |
| Plastic Composition | Nhieu Loc– Thi Nghe (NL) | Tau Hu (TH) | Ben Nghe (BN) | Te Canal (KT) |
|---|---|---|---|---|
| Total microplastics (MPs/L) | 35.58 ± 4.75 | 45.19 ± 10.30 | 33.83 ± 10.54 | 43.41 ± 7.93 |
| PE (%) | 14.35 | 12.23 | 14.68 | 17.41 |
| PP (%) | 13.48 | 12.66 | 11.51 | 11.34 |
| PET (%) | 21.74 | 18.34 | 8.73 | 7.283 |
| PVC (%) | 5.65 | 6.11 | 11.51 | 4.86 |
| PS (%) | 3.04 | 2.18 | 4.76 | 1.21 |
| PA (%) | 22.17 | 23.58 | 14.68 | 17.41 |
| PMMA (%) | 1.31 | 0.87 | 0.79 | 1.21 |
| Unidentifed (%) | 18.26 | 24.02 | 33.33 | 39.27 |
| Parameters | Saigon Urban Canals (Saigon River) | Can Gio Reserve | Downstream of Soai Rap River | Ganh Rai Gulf (East Sea) | White Tiger Oilfield (East Sea) |
|---|---|---|---|---|---|
| Plastic compositions | PA > PET > PE > PP > PVC > PS > PMMA | PET > PE > PA > PP > PVC > PS > PMMA | PE > PP > PET > PA > PVC = PS > PMMA | PET > PE > PA > PP > PVC ≥ PS > PMMA | PET > PA > PP > PE > PVC > PS = PMMA |
| Common shapes (%) | Fiber: 35.4 Fragment: 20.6 | Fiber: 27.7 Fragment: 34.2 | Fiber: 31.3 Particle: 21.8 | Fiber: 42.4 Particle: 20.14 | Fiber: 44.1 Particle: 26.5 |
| Common colors | white > gray > brown > green > pink | white > gray > blue > pink > brown | white > pink > blue > purple > yellow | white > blue > pink > gray > green | white > gray > pink > blue > purple |
| Size range (μm) | Fiber: 14.1–561.2 Non-fibers: 15.7–188.4 | Fiber: 25–60 Non-fibers: 14–53.4 | Fiber: 19.7–58.3 Non-fibers: 21.9–94.5 | Fiber: 25–436.9 Non-fibers: 23.5–265 | Fiber: 36.1–184.7 Non-fibers: 22–92 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khuyen, V.T.K.; Le, D.V.; Le, H.A.; Fischer, A.R.; Dornack, C. Assessing Microplastic Prevalence and Dispersion from Saigon Urban Canals via Can Gio Mangrove Reserve to East Sea by Raman Scattering Microscopy. Microplastics 2022, 1, 536-553. https://doi.org/10.3390/microplastics1030038
Khuyen VTK, Le DV, Le HA, Fischer AR, Dornack C. Assessing Microplastic Prevalence and Dispersion from Saigon Urban Canals via Can Gio Mangrove Reserve to East Sea by Raman Scattering Microscopy. Microplastics. 2022; 1(3):536-553. https://doi.org/10.3390/microplastics1030038
Chicago/Turabian StyleKhuyen, Vo Thi Kim, Dinh Vu Le, Hung Anh Le, Axel René Fischer, and Christina Dornack. 2022. "Assessing Microplastic Prevalence and Dispersion from Saigon Urban Canals via Can Gio Mangrove Reserve to East Sea by Raman Scattering Microscopy" Microplastics 1, no. 3: 536-553. https://doi.org/10.3390/microplastics1030038
APA StyleKhuyen, V. T. K., Le, D. V., Le, H. A., Fischer, A. R., & Dornack, C. (2022). Assessing Microplastic Prevalence and Dispersion from Saigon Urban Canals via Can Gio Mangrove Reserve to East Sea by Raman Scattering Microscopy. Microplastics, 1(3), 536-553. https://doi.org/10.3390/microplastics1030038






