Targeted Substituted-Phenol Production by Strategic Hydrogenolysis of Sugar-Cane Lignin
Abstract
1. Introduction
2. Experimental
2.1. Catalyst Preparation
2.2. Catalyst Reduction
2.3. Sugar-Cane Lignin (SCL) Extraction Process
2.4. SCL Depolymerization Process
2.5. Gas Chromatography Mass Spectroscopy Analysis
2.6. Product Identification and Yield Calculation
2.7. Gel Permeation Chromatography (GPC) and Elemental Analysis (CHN)
2.8. Thermogravimetric Analysis (TGA)
2.9. Surface Area and Pore Volume Determination (BET)
2.10. Raman and Atomic Absorption Spectroscopy Analysis
3. Results and Discussion
3.1. Catalyst Characterization
3.2. Sugar-Cane Lignin (SCL) Characterization
3.2.1. Elemental Analysis
3.2.2. Molecular Weight Distribution of SCL
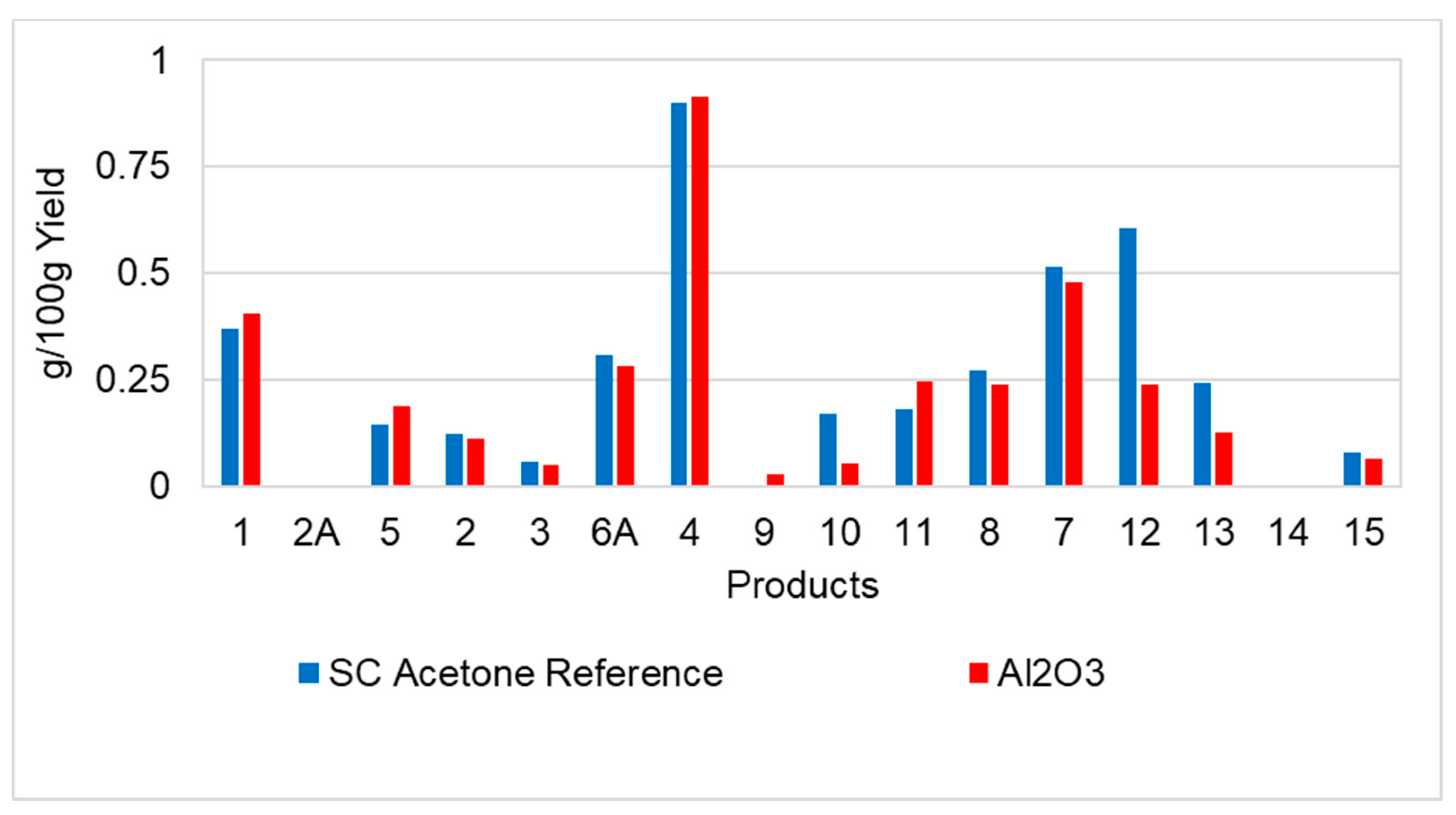
3.2.3. Product Distribution and Quantification by GC/MS
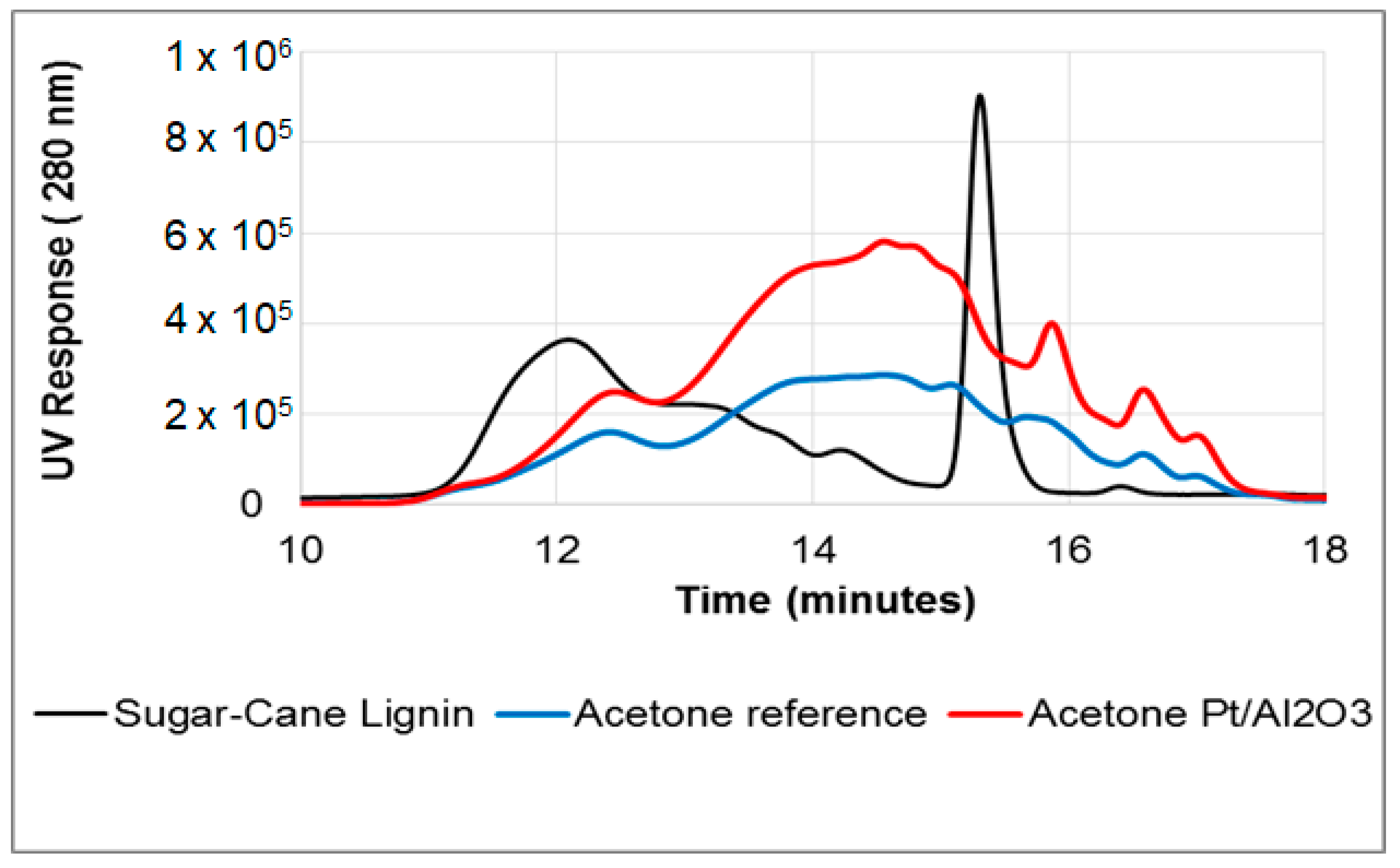
3.2.4. Depolymerisation of Sugar-Cane Lignin (SCL)
3.2.5. Catalyst Free System
3.2.6. Effect of Alumina Support
3.2.7. Catalysts Screening
3.2.8. Effect of Pt and Rh Noble Metal Catalysts
3.2.9. Effect of Ni and Fe Catalysts
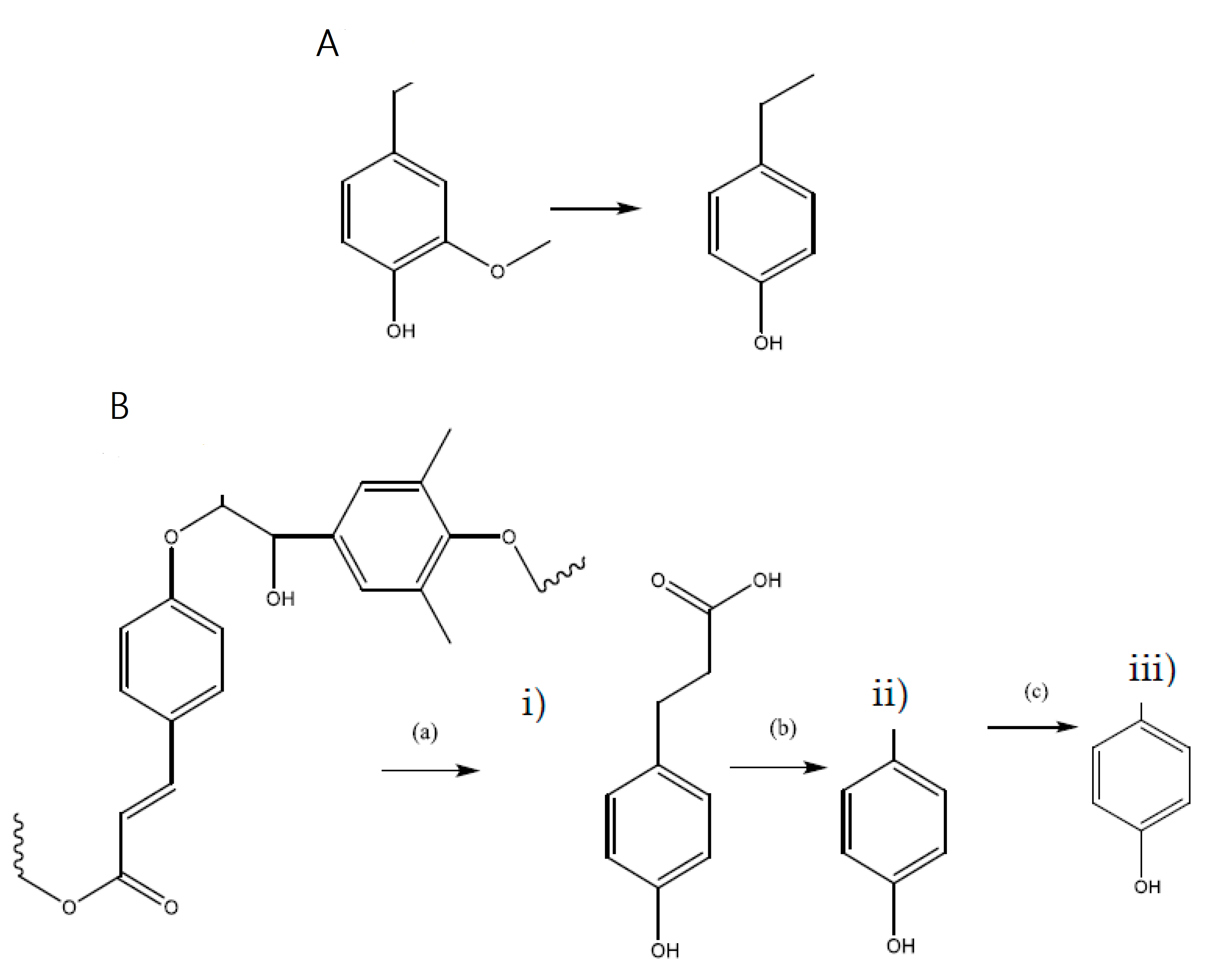
3.2.10. Catalytic Products
4. Post-Reaction Catalyst Evaluation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ventura, M.; Marinas, A.; Domine, M.E. Catalytic Processes for Biomass-Derived Platform Molecules Valorisation. Top. Catal. 2020, 63, 846–865. [Google Scholar] [CrossRef]
- Liu, X.; Bouxin, F.P.; Fan, J.; Budarin, V.L.; Hu, C.; Clark, J.H. Recent Advances in the Catalytic Depolymerization of Lignin towards Phenolic Chemicals: A Review. ChemSusChem 2020, 13, 4296–4317. [Google Scholar] [CrossRef]
- Crestini, C.; Lange, H.; Sette, M.; Argyropoulos, D.S. On the Structure of Softwood Kraft Lignin. Green Chem. 2017, 19, 4104–4121. [Google Scholar] [CrossRef]
- Sjostrom, E. Wood Chemistry: Fundamentals and Application, 2nd ed.; Press, A., Ed.; Elsevier: Orlando, FL, USA, 1993. [Google Scholar] [CrossRef]
- Abu-Omar, M.M.; Barta, K.; Beckham, G.T.; Luterbacher, J.S.; Ralph, J.; Rinaldi, R.; Román-Leshkov, Y.; Samec, J.S.M.; Sels, B.F.; Wang, F. Guidelines for Performing Lignin-First Biorefining. Energy Environ. Sci. 2021, 14, 262–292. [Google Scholar] [CrossRef]
- Xu, C.; Arancon, R.A.D.; Labidi, J.; Luque, R. Lignin Depolymerisation Strategies: Towards Valuable Chemicals and Fuels. Chem. Soc. Rev. 2014, 43, 7485–7500. [Google Scholar] [CrossRef] [PubMed]
- Pasquini, D.; Pimenta, M.T.B.; Ferreira, L.H.; Curvelo, A.A.D.S. Extraction of Lignin from Sugar Cane Bagasse and Pinus Taeda Wood Chips Using Ethanol-Water Mixtures and Carbon Dioxide at High Pressures. J. Supercrit. Fluids 2005, 36, 31–39. [Google Scholar] [CrossRef]
- Melati, R.B.; Schmatz, A.A.; Pagnocca, F.; Contiero, J.; Brienzo, M. Sugarcane Bagasse: Production, Composition, Properties, and Feedstock Potential. In Sugarcane: Production Systems, Uses and Economic Importance; Nova Science Publishers: Hauppauge, NY, USA, 2017; pp. 1–38. [Google Scholar]
- Goldemberg, J.; Coelho, S.T.; Guardabassi, P. The Sustainability of Ethanol Production from Sugarcane. Energy Policy 2008, 36, 2086–2097. [Google Scholar] [CrossRef]
- Pin, T.C.; Nascimento, V.M.; Costa, A.C.; Pu, Y.; Ragauskas, A.J.; Rabelo, S.C. Structural Characterization of Sugarcane Lignins Extracted from Different Protic Ionic Liquid Pretreatments. Renew. Energy 2020, 161, 579–592. [Google Scholar] [CrossRef]
- Huijgen, W.J.J.; Reith, J.H.; Den Uil, H. Pretreatment and Fractionation of Wheat Straw by an Acetone-Based Organosolv Process. Ind. Eng. Chem. Res. 2010, 49, 10132–10140. [Google Scholar] [CrossRef]
- Erdocia, X.; Prado, R.; Fernández-Rodríguez, J.; Labidi, J. Depolymerization of Different Organosolv Lignins in Supercritical Methanol, Ethanol, and Acetone to Produce Phenolic Monomers. ACS Sustain. Chem. Eng. 2016, 4, 1373–1380. [Google Scholar] [CrossRef]
- Wu, Q.Y.; Ma, L.L.; Long, J.X.; Shu, R.Y.; Zhang, Q.; Wang, T.J.; Xu, Y. Depolymerization of Organosolv Lignin over Silica-Alumina Catalysts. Chin. J. Chem. Phys. 2016, 29, 474–480. [Google Scholar] [CrossRef][Green Version]
- Bauer, S.; Sorek, H.; Mitchell, V.D.; Ibáñez, A.B.; Wemmer, D.E. Characterization of Miscanthus Giganteus Lignin Isolated by Ethanol Organosolv Process under Reflux Condition. J. Agric. Food Chem. 2012, 60, 8203–8212. [Google Scholar] [CrossRef]
- Lancefield, C.S.; Panovic, I.; Deuss, P.J.; Barta, K.; Westwood, N.J. Pre-Treatment of Lignocellulosic Feedstocks Using Biorenewable Alcohols: Towards Complete Biomass Valorisation. Green Chem. 2017, 19, 202–214. [Google Scholar] [CrossRef]
- Bouxin, F.P.; McVeigh, A.; Tran, F.; Westwood, N.J.; Jarvis, M.C.; Jackson, S.D. Catalytic Depolymerisation of Isolated Lignins to Fine Chemicals Using a Pt/Alumina Catalyst: Part 1—Impact of the Lignin Structure. Green Chem. 2015, 17, 1235–1242. [Google Scholar] [CrossRef]
- Lancefield, C.S.; Rashid, G.M.M.; Bouxin, F.; Wasak, A.; Tu, W.C.; Hallett, J.; Zein, S.; Rodríguez, J.; Jackson, S.D.; Westwood, N.J.; et al. Investigation of the Chemocatalytic and Biocatalytic Valorization of a Range of Different Lignin Preparations: The Importance of β-O-4 Content. ACS Sustain. Chem. Eng. 2016, 4, 6921–6930. [Google Scholar] [CrossRef]
- McVeigh, A.; Bouxin, F.P.; Jarvis, M.C.; Jackson, S.D. Catalytic Depolymerisation of Isolated Lignin to Fine Chemicals: Part 2—Process Optimisation. Catal. Sci. Technol. 2016, 6, 4142–4150. [Google Scholar] [CrossRef]
- Zhang, C.; Lu, J.; Zhang, X.; MacArthur, K.; Heggen, M.; Li, H.; Wang, F. Cleavage of the Lignin β-O-4 Ether Bond via a Dehydroxylation–Hydrogenation Strategy over a NiMo Sulfide Catalyst. Green Chem. 2016, 18, 6545–6555. [Google Scholar] [CrossRef]
- Yamaguchi, A.; Mimura, N.; Shirai, M.; Sato, O. Bond Cleavage of Lignin Model Compounds into Aromatic Monomers Using Supported Metal Catalysts in Supercritical Water. Sci. Rep. 2017, 7, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Rinaldi, R. Solvent Effects on the Hydrogenolysis of Diphenyl Ether with Raney Nickel and Their Implications for the Conversion of Lignin. ChemSusChem 2012, 5, 1455–1466. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Sun, H.; Liu, J.-X.; Zhang, J.-j.; Li, Z.-X.; Fu, Y. Selective Reductive Cleavage of C–O Bond in Lignin Model Compounds over Nitrogen-Doped Carbon-Supported Iron Catalysts. Mol. Catal. 2018, 452, 36–45. [Google Scholar] [CrossRef]
- Song, Q.; Wang, F.; Cai, J.; Wang, Y.; Zhang, J.; Yu, W.; Xu, J. Lignin Depolymerization (LDP) in Alcohol over Nickel-Based Catalysts via a Fragmentation-Hydrogenolysis Process. Energy Environ. Sci. 2013, 6, 994–1007. [Google Scholar] [CrossRef]
- Baral, N.R.; Slutzky, L.; Shah, A.; Ezeji, T.C.; Cornish, K.; Christy, A. Acetone-Butanol-Ethanol Fermentation of Corn Stover: Current Production Methods, Economic Viability and Commercial Use. FEMS Microbiol. Lett. 2016, 363, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Gelder, E.A. The Hydrogenation of Nitrobenzene of Metal Catalysts. Ph.D. Thesis, The University of Glasgow, Glasgow, UK, 2005. [Google Scholar]
- Munick de Albuquerque Fragoso, D.; Bouxin, F.P.; Montgomery, J.R.D.; Westwood, N.J.; Jackson, S.D. Catalytic Depolymerisation of Isolated Lignin to Fine Chemicals: Depolymerisation of Kraft Lignin. Bioresour. Technol. Rep. 2020, 9, 100400. [Google Scholar] [CrossRef]
- McVeigh, A. The Conversion of Lignin to Alkylphenolic Monomers Using Heterogeneous Catalysis. Ph.D. Thesis, The University of Glasgow, Glasgow, UK, 2016. [Google Scholar]
- Sarkanen, K.; Schuerch, C. Lignin Structure. XI: A Quantitative Study of the Alcoholysis of Lignin. J. Am. Chem. Soc. 1957, 79, 4203–4209. [Google Scholar] [CrossRef]
- Danielle Munick de Albuquerque Fragoso. Lignin Conversion to Fine Chemicals. Ph.D. Thesis, University of Glasgow, Glasgow, UK, 2018. [Google Scholar]
- Ekielski, A.; Mishra, P.K. Lignin for Bioeconomy: The Present and Future Role of Technical Lignin. Int. J. Mol. Sci. 2020, 22, 63. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.; Korányi, T.I.; Boot, M.D.; Hensen, E.J.M. Catalytic Depolymerization of Lignin in Supercritical Ethanol. ChemSusChem 2014, 7, 2276–2288. [Google Scholar] [CrossRef]
- Nguyen, T.D.H.; Maschietti, M.; Belkheiri, T.; Amand, L.E.; Theliander, H.; Vamling, L.; Olausson, L.; Andersson, S.-I. Catalytic Depolymerisation and Conversion of Kraft Lignin into Liquid Products Using Near-Critical Water. J. Supercrit. Fluids 2014, 86, 67–75. [Google Scholar] [CrossRef]
- Kawamata, Y.; Ishimaru, H.; Yamaguchi, K.; Yoshikawa, T.; Koyama, Y.; Nakasaka, Y.; Sato, S.; Masuda, T. Catalytic Cracking of Lignin Model Compounds and Degraded Lignin Dissolved in Inert Solvent over Mixed Catalyst of Iron Oxide and MFI Zeolite for Phenol Recovery. Fuel Process. Technol. 2020, 197, 106190. [Google Scholar] [CrossRef]
- Mostafa, M.R.; Youssef, A.M.; Hassan, S.M. Conversion of Ethanol and Isopropanol on Alumina, Titania and Alumina-Titania Catalysts. Mater. Lett. 1991, 12, 207–213. [Google Scholar] [CrossRef]
- Pillait, E.F.N.; Jain, R.A.I.; June, R.; Process, H. Catalytic Dehydration of Alcohols over Alumina: Mechanism of Ether Formation. J. Catal. 1967, 9, 322–330. [Google Scholar] [CrossRef]
- Knözinger, H.; Ratnasamy, P. Catalytic Aluminas: Surface Models and Characterization of Surface Sites. Catal. Rev. 1978, 17, 31–70. [Google Scholar] [CrossRef]
- Panov, A.G.; Fripiat, J.J. Acetone Condensation Reaction on Acid Catalysts. J. Catal. 1998, 178, 188–197. [Google Scholar] [CrossRef]
- Varma, R.S.; Kabalka, G.W.; Evans, L.T.; Pagni, R.M. Aldol Condensations on Basic Alumina: The Facile Syntheses of Chalcones and Enones in a Solvent-Free Medium. Synth. Commun. 1985, 15, 279–284. [Google Scholar] [CrossRef]
- NIST Chemistry WebBook. Acetone. Available online: https://webbook.nist.gov/cgi/cbook.cgi?ID=C67641&Mask=4 (accessed on 14 July 2018).
- Joffres, B.; Laurenti, D.; Charon, N.; Daudin, A.; Quignard, A.; Geantet, C. Thermochemical Conversion of Lignin for Fuels and Chemicals: A Review. Oil Gas Sci. Technol. Rev. IFP Energ. Nouv. 2013, 68, 753–763. [Google Scholar] [CrossRef]
- Bouxin, F.P.; Zhang, X.; Kings, I.N.; Lee, A.F.; Simmons, M.J.H.; Wilson, K.; Jackson, S.D. Deactivation Study of the Hydrodeoxygenation of P-Methylguaiacol over Silica Supported Rhodium and Platinum Catalysts. Appl. Catal. A Gen. 2017, 539, 29–37. [Google Scholar] [CrossRef]
- Hita, I.; Deuss, P.J.; Bonura, G.; Frusteri, F.; Heeres, H.J. Biobased Chemicals from the Catalytic Depolymerization of Kraft Lignin Using Supported Noble Metal-Based Catalysts. Fuel Process. Technol. 2018, 179, 143–153. [Google Scholar] [CrossRef]
- Cheng, C.; Shen, D.; Gu, S.; Luo, K.H. State-of-the-Art Catalytic Hydrogenolysis of Lignin for the Production of Aromatic Chemicals. Catal. Sci. Technol. 2018, 8, 6275–6296. [Google Scholar] [CrossRef]
- Chatterjee, M.; Chatterjee, A.; Ishizaka, T.; Kawanami, H. Rhodium-Mediated Hydrogenolysis/Hydrolysis of the Aryl Ether Bond in Supercritical Carbon Dioxide/Water: An Experimental and Theoretical Approach. Catal. Sci. Technol. 2015, 5, 1532–1539. [Google Scholar] [CrossRef]
- He, J.; Lu, L.; Zhao, C.; Mei, D.; Lercher, J.A. Mechanisms of Catalytic Cleavage of Benzyl Phenyl Ether in Aqueous and Apolar Phases. J. Catal. 2014, 311, 41–51. [Google Scholar] [CrossRef]
- He, J.; Zhao, C.; Mei, D.; Lercher, J.A. Mechanisms of Selective Cleavage of C-O Bonds in Di-Aryl Ethers in Aqueous Phase. J. Catal. 2014, 309, 280–290. [Google Scholar] [CrossRef]
- Yoshikawa, T.; Shinohara, S.; Yagi, T.; Ryumon, N.; Nakasaka, Y.; Tago, T.; Masuda, T. Production of Phenols from Lignin-Derived Slurry Liquid Using Iron Oxide Catalyst. Appl. Catal. B Environ. 2014, 146, 289–297. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, K.; Li, H.; Xiao, L.-P.; Song, G. Selective Hydrogenolysis of Catechyl Lignin into Propenylcatechol over an Atomically Dispersed Ruthenium Catalyst. Nat. Commun. 2021, 12, 416. [Google Scholar] [CrossRef] [PubMed]
- Lapierre, C.; Pollet, B.; Rolando, C. New Insights into the Molecular Architecture of Hardwood Lignins by Chemical Degradative Methods. Res. Chem. Intermed. 1995, 21, 397–412. [Google Scholar] [CrossRef]
- Kang, S.; Li, X.; Fan, J.; Chang, J. Hydrothermal Conversion of Lignin: A Review. Renew. Sustain. Energy Rev. 2013, 27, 546–558. [Google Scholar] [CrossRef]
- Yuan, Z.; Tymchyshyn, M.; Xu, C. Reductive Depolymerization of Kraft and Organosolv Lignin in Supercritical Acetone for Chemicals and Materials. ChemCatChem 2016, 8, 1968–1976. [Google Scholar] [CrossRef]
- Fang, Z.; Sato, T.; Smith, R.L.; Inomata, H.; Arai, K.; Kozinski, J.A. Reaction Chemistry and Phase Behavior of Lignin in High-Temperature and Supercritical Water. Bioresour. Technol. 2008, 99, 3424–3430. [Google Scholar] [CrossRef]
- Ravenelle, R.M.; Copeland, J.R.; Kim, W.-G.; Crittenden, J.C.; Sievers, C. Structural Changes of γ-Al 2 O 3 -Supported Catalysts in Hot Liquid Water. ACS Catal. 2011, 1, 552–561. [Google Scholar] [CrossRef]
- Sattler, J.J.H.B.; Beale, A.M.; Weckhuysen, B.M. Operando Raman Spectroscopy Study on the Deactivation of Pt/Al2O3 and Pt–Sn/Al2O3 Propane Dehydrogenation Catalysts. Phys. Chem. Chem. Phys. 2013, 15, 12095. [Google Scholar] [CrossRef]
- Sadezky, A.; Muckenhuber, H.; Grothe, H.; Niessner, R.; Pöschl, U. Raman Microspectroscopy of Soot and Related Carbonaceous Materials: Spectral Analysis and Structural Information. Carbon 2005, 43, 1731–1742. [Google Scholar] [CrossRef]




| Elemental Analysis (%) | |
|---|---|
| C | 54.1 |
| H | 5.6 |
| N | 0.9 |
| O | 39.4 |
| Compound Code | Compound Name |
|---|---|
| (1) | 2-methoxyphenol |
| (2) | 4-methyl-2-methoxyphenol |
| (2A) | 4-ethylphenol |
| (3) | 4-ethyl-2-methoxyphenol |
| (4) | 4-propyl-2-methoxyphenol |
| (5) | 1,2-dihydroxybenzene |
| (6) | 4-ethylbenzene-1,2-diol |
| (6A) | 2,6-dimethoxyphenol |
| (7) | 4-(3-hydroxypropyl)-2-methoxyphenol |
| (8) | 4-(3-methoxypropyl)-2-methoxyphenol |
| (9) | 4-methyl-2,6-dimethoxyphenol |
| (10) | 4-(2-hydroxyethyl)-2,6-dimethoxyphenol |
| (11) | 4-ethyl-2,6-dimethoxyphenol |
| (12) | 4-propenyl-2,6-dimethoxyphenol |
| (13) | 4-(2-hydroxyethyl)-2-methoxyphenol |
| (14) | 4-propyl-2,6-dimethoxyphenol |
| (15) | 4-(1-hydroxy-2-methylpent-3-enyl)-2,6-dimethoxyphenol |
| (15A) | 4-(3-hydroxypropyl)-2,6-dimethoxyphenol |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Albuquerque Fragoso, D.M.; Goulart, H.F.; Santana, A.E.G.; Jackson, S.D. Targeted Substituted-Phenol Production by Strategic Hydrogenolysis of Sugar-Cane Lignin. Biomass 2021, 1, 11-28. https://doi.org/10.3390/biomass1010002
de Albuquerque Fragoso DM, Goulart HF, Santana AEG, Jackson SD. Targeted Substituted-Phenol Production by Strategic Hydrogenolysis of Sugar-Cane Lignin. Biomass. 2021; 1(1):11-28. https://doi.org/10.3390/biomass1010002
Chicago/Turabian Stylede Albuquerque Fragoso, Danielle Munick, Henrique Fonseca Goulart, Antonio Euzebio Goulart Santana, and Samuel David Jackson. 2021. "Targeted Substituted-Phenol Production by Strategic Hydrogenolysis of Sugar-Cane Lignin" Biomass 1, no. 1: 11-28. https://doi.org/10.3390/biomass1010002
APA Stylede Albuquerque Fragoso, D. M., Goulart, H. F., Santana, A. E. G., & Jackson, S. D. (2021). Targeted Substituted-Phenol Production by Strategic Hydrogenolysis of Sugar-Cane Lignin. Biomass, 1(1), 11-28. https://doi.org/10.3390/biomass1010002







