The Gasotransmitter Hydrogen Sulfide and the Neuropeptide Oxytocin as Potential Mediators of Beneficial Cardiovascular Effects through Meditation after Traumatic Events
Abstract
1. Introduction
2. Early Life Stress
3. H2S System
4. Oxytocin System
5. Hypothalamic–Pituitary–Adrenal Axis and Brain
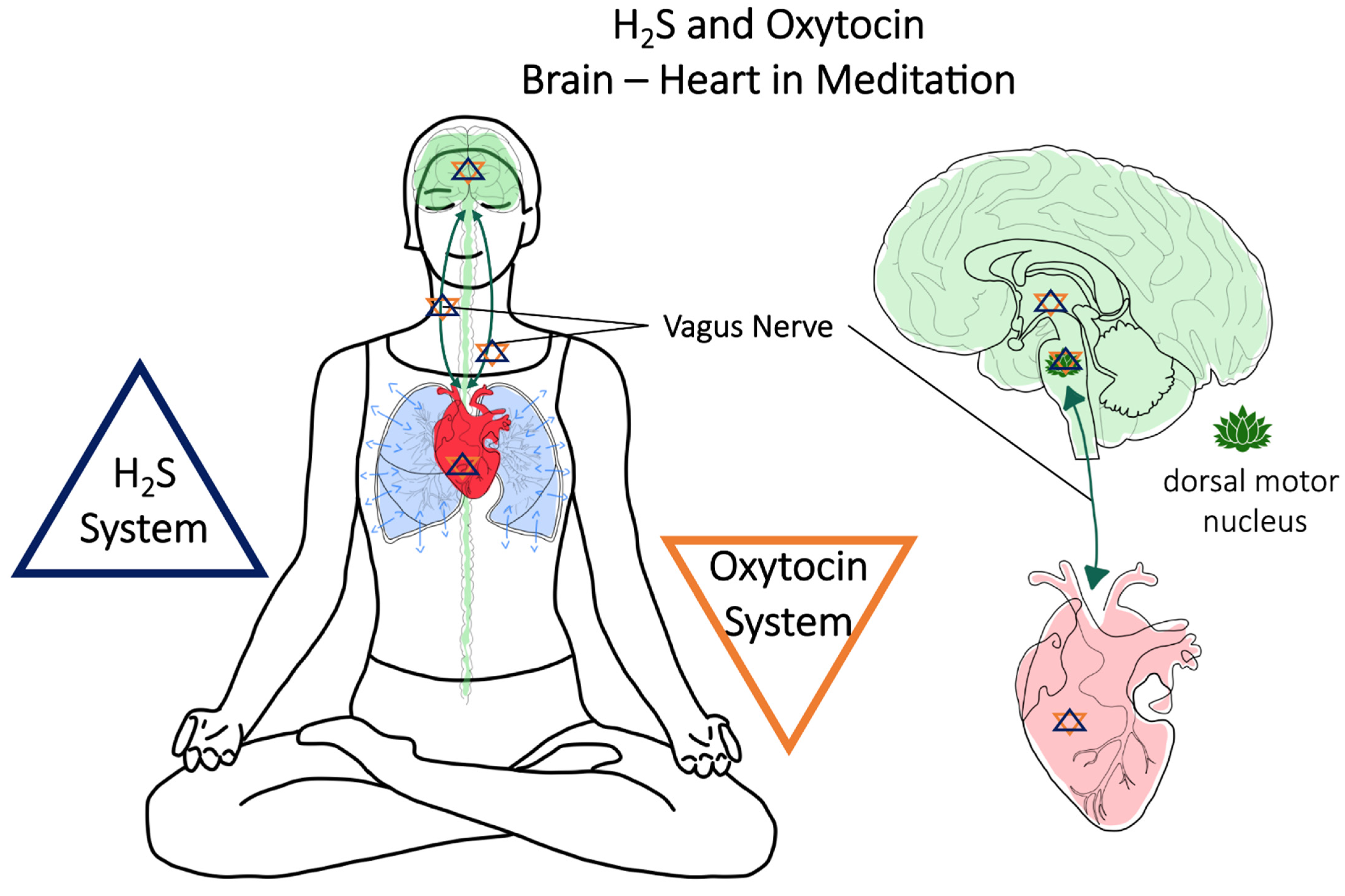
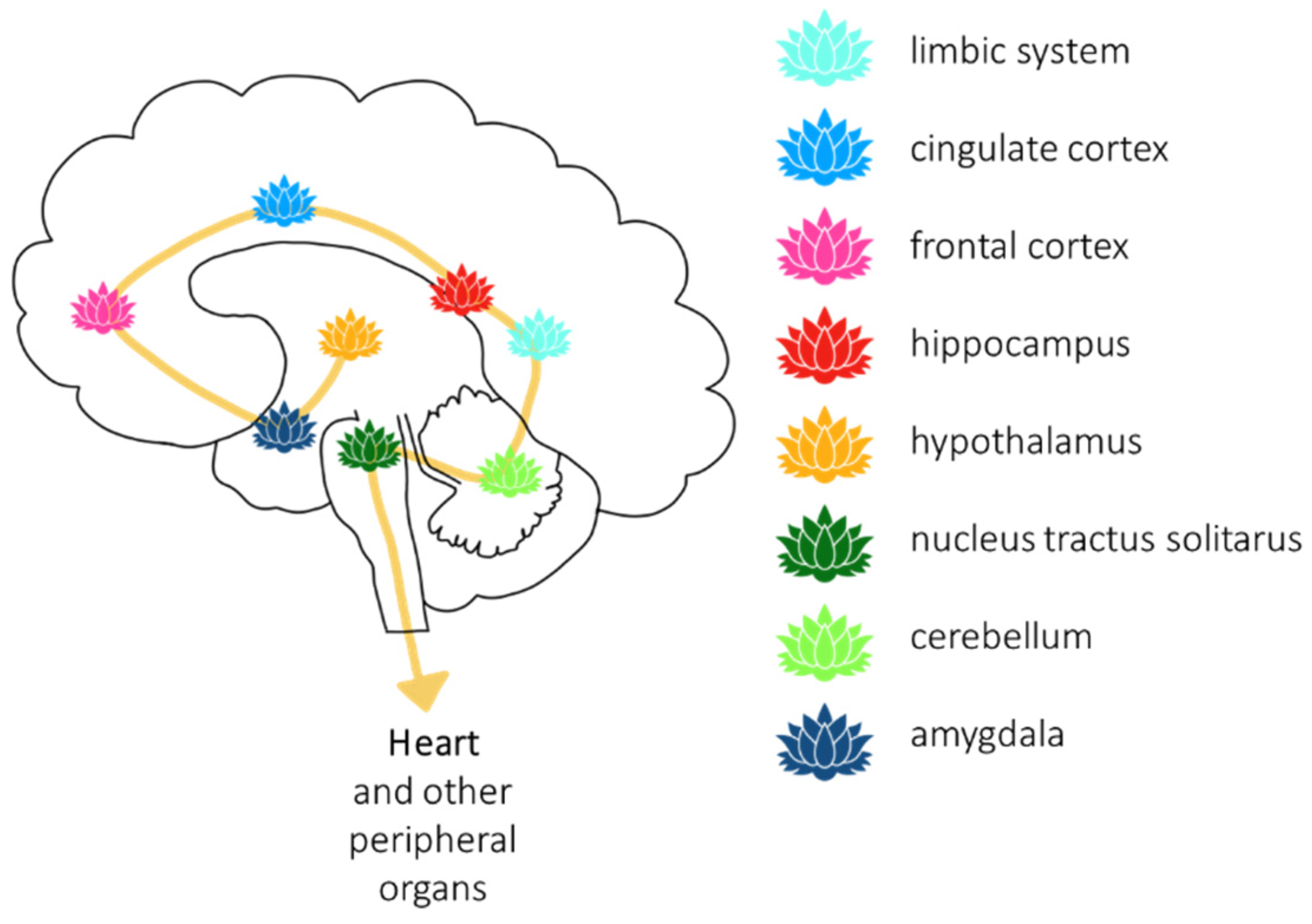
6. Brain and Heart–Vagus Nerve–H2S and Oxytocin
7. Meditation (Breath Control)
8. Conclusions
“This evidence linking the hippocampus and amygdala with oxytocin provides a scientific mechanism liberating downstream ancestral memories catalogued in neuronal networks. Knowing that oxytocin may likewise be boosted during meditation or prayer provides a possible portal to ancestral diaries. Following this logic one step further, the meditating mind struggles to put the knowledge archives into familiar themes, filling in the gaps, and imparting the feelings of a guiding voice or visiting an Akashic Record library. These treasures are unbundled and retrieved under the influence of oxytocin to convey resiliency for survival in life-and death situations.”[116]
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- McCook, O.; Denoix, N.; Radermacher, P.; Waller, C.; Merz, T. H2S and Oxytocin Systems in Early Life Stress and Cardiovascular Disease. J. Clin. Med. 2021, 10, 3484. [Google Scholar] [CrossRef]
- Denoix, N.; McCook, O.; Ecker, S.; Wang, R.; Waller, C.; Radermacher, P.; Merz, T. The Interaction of the Endogenous Hydrogen Sulfide and Oxytocin Systems in Fluid Regulation and the Cardiovascular System. Antioxidants 2020, 9, 748. [Google Scholar] [CrossRef]
- Wang, C.-Y.; Chen, Y.-C.; Chien, T.-H.; Chang, H.-Y.; Chen, Y.-H.; Chien, C.-Y.; Huang, T.-S. Impact of Comorbidities on the Prognoses of Trauma Patients: Analysis of a Hospital-Based Trauma Registry Database. PLoS ONE 2018, 13, e0194749. [Google Scholar] [CrossRef]
- Ferraris, V.A.; Ferraris, S.P.; Saha, S.P. The Relationship Between Mortality and Preexisting Cardiac Disease in 5,971 Trauma Patients. J. Trauma Inj. Infect. Crit. Care 2010, 69, 645–652. [Google Scholar] [CrossRef]
- Trautwein, B.; Merz, T.; Denoix, N.; Szabo, C.; Calzia, E.; Radermacher, P.; McCook, O. DMST and the Regulation of Cardiac CSE and OTR Expression in Trauma and Hemorrhage. Antioxidants 2021, 10, 233. [Google Scholar] [CrossRef]
- McCook, O.; Scheuerle, A.; Denoix, N.; Kapapa, T.; Radermacher, P.; Merz, T. Localization of the Hydrogen Sulfide and Oxytocin Systems at the Depth of the Sulci in a Porcine Model of Acute Subdural Hematoma. Neural Regen. Res. 2021, 16, 2376–2382. [Google Scholar] [CrossRef]
- Denoix, N.; Merz, T.; Unmuth, S.; Hoffmann, A.; Nespoli, E.; Scheuerle, A.; Huber-Lang, M.; Gündel, H.; Waller, C.; Radermacher, P.; et al. Cerebral Immunohistochemical Characterization of the H2S and the Oxytocin Systems in a Porcine Model of Acute Subdural Hematoma. Front. Neurol. 2020, 11, 649. [Google Scholar] [CrossRef]
- Wigger, D.C.; Gröger, N.; Lesse, A.; Krause, S.; Merz, T.; Gündel, H.; Braun, K.; McCook, O.; Radermacher, P.; Bock, J.; et al. Maternal Separation Induces Long-Term Alterations in the Cardiac Oxytocin Receptor and Cystathionine g-Lyase Expression in Mice. Oxid. Med. Cell. Longev. 2020, 2020, 4309605. [Google Scholar] [CrossRef]
- Merz, T.; Lukaschewski, B.; Wigger, D.; Rupprecht, A.; Wepler, M.; Gröger, M.; Hartmann, C.; Whiteman, M.; Szabo, C.; Wang, R.; et al. Interaction of the Hydrogen Sulfide System with the Oxytocin System in the Injured Mouse Heart. Intensive Care Med. Exp. 2018, 6, 41. [Google Scholar] [CrossRef]
- Merz, T.; Denoix, N.; Wigger, D.; Waller, C.; Wepler, M.; Vettorazzi, S.; Tuckermann, J.; Radermacher, P.; McCook, O. The Role of Glucocorticoid Receptor and Oxytocin Receptor in the Septic Heart in a Clinically Relevant, Resuscitated Porcine Model with Underlying Atherosclerosis. Front. Endocrinol. 2020, 11, 299. [Google Scholar] [CrossRef]
- Agorastos, A.; Pervanidou, P.; Chrousos, G.P.; Baker, D.G. Developmental Trajectories of Early Life Stress and Trauma: A Narrative Review on Neurobiological Aspects Beyond Stress System Dysregulation. Front. Psychiatry 2019, 10, 118. [Google Scholar] [CrossRef] [PubMed]
- Akashi, Y.J.; Goldstein, D.S.; Barbaro, G.; Ueyama, T. Takotsubo cardiomyopathy: A new form of acute, reversible heart failure. Circulation 2008, 118, 2754–2762. [Google Scholar] [CrossRef]
- Auxéméry, Y. Post-Traumatic Psychiatric Disorders: PTSD Is Not the Only Diagnosis. Presse Méd. 2018, 47, 423–430. [Google Scholar] [CrossRef]
- Pascoe, M.C.; Thompson, D.R.; Jenkins, Z.M.; Ski, C.F. Mindfulness Mediates the Physiological Markers of Stress: Systematic Review and Meta-Analysis. J. Psychiatr. Res. 2017, 95, 156–178. [Google Scholar] [CrossRef]
- Krittanawong, C.; Kumar, A.; Wang, Z.; Narasimhan, B.; Jneid, H.; Virani, S.S.; Levine, G.N. Meditation and Cardiovascular Health in the US. Am. J. Cardiol. 2020, 131, 23–26. [Google Scholar] [CrossRef]
- Levine, G.N.; Lange, R.A.; Bairey-Merz, C.N.; Davidson, R.J.; Jamerson, K.; Mehta, P.K.; Michos, E.D.; Norris, K.; Ray, I.B.; Saban, K.L.; et al. Meditation and Cardiovascular Risk Reduction: A Scientific Statement From the American Heart Association. J. Am. Heart Assoc. 2017, 6, e002218. [Google Scholar] [CrossRef]
- Albus, C.; Waller, C.; Fritzsche, K.; Gunold, H.; Haass, M.; Hamann, B.; Kindermann, I.; Köllner, V.; Leithäuser, B.; Marx, N.; et al. Significance of Psychosocial Factors in Cardiology: Update 2018: Position Paper of the German Cardiac Society. Clin. Res. Cardiol. 2019, 108, 1175–1196. [Google Scholar] [CrossRef]
- Basu, A.; McLaughlin, K.A.; Misra, S.; Koenen, K.C. Childhood Maltreatment and Health Impact: The Examples of Cardiovascular Disease and Type 2 Diabetes Mellitus in Adults. Clin. Psychol. Sci. Pract. 2017, 24, 125–139. [Google Scholar] [CrossRef]
- Suglia, S.F.; Koenen, K.C.; Boynton-Jarrett, R.; Chan, P.S.; Clark, C.J.; Danese, A.; Faith, M.S.; Goldstein, B.I.; Hayman, L.L.; Isasi, C.R.; et al. Childhood and Adolescent Adversity and Cardiometabolic Outcomes: A Scientific Statement from the American Heart Association. Circulation 2018, 137, e15–e28. [Google Scholar] [CrossRef]
- Chandan, J.S.; Okoth, K.; Gokhale, K.M.; Bandyopadhyay, S.; Taylor, J.; Nirantharakumar, K. Increased Cardiometabolic and Mortality Risk Following Childhood Maltreatment in the United Kingdom. J. Am. Heart Assoc. 2020, 9, e015855. [Google Scholar] [CrossRef]
- Higgins, M.W.; Keller, J.B.; Metzner, H.L.; Moore, F.E.; Ostrander, L.D. Studies of Blood Pressure in Tecumseh, Michigan. II. Antecedents in Childhood of High Blood Pressure in Young Adults. Hypertension 1980, 2, 117–123. [Google Scholar] [CrossRef]
- Hsu, C.-N.; Tain, Y.-L. Preventing Developmental Origins of Cardiovascular Disease: Hydrogen Sulfide as a Potential Target? Antioxidants 2021, 10, 247. [Google Scholar] [CrossRef] [PubMed]
- Thornburg, K.L. The Programming of Cardiovascular Disease. J. Dev. Orig. Health Dis. 2015, 6, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Blackmore, H.L.; Ozanne, S.E. Programming of Cardiovascular Disease across the Life-Course. J. Mol. Cell. Cardiol. 2015, 83, 122–130. [Google Scholar] [CrossRef]
- Tain, Y.-L.; Hsu, C.-N. Interplay between Oxidative Stress and Nutrient Sensing Signaling in the Developmental Origins of Cardiovascular Disease. Int. J. Mol. Sci. 2017, 18, 841. [Google Scholar] [CrossRef]
- Coletti, R.; Almeida-Pereira, G.; Elias, L.L.K.; Antunes-Rodrigues, J. Effects of Hydrogen Sulfide (H2S) on Water Intake and Vasopressin and Oxytocin Secretion Induced by Fluid Deprivation. Horm. Behav. 2015, 67, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Gouin, J.-P.; Carter, C.S.; Pournajafi-Nazarloo, H.; Glaser, R.; Malarkey, W.B.; Loving, T.J.; Stowell, J.; Kiecolt-Glaser, J.K. Marital Behavior, Oxytocin, Vasopressin, and Wound Healing. Psychoneuroendocrinology 2010, 35, 1082–1090. [Google Scholar] [CrossRef] [PubMed]
- Olff, M.; Frijling, J.L.; Kubzansky, L.D.; Bradley, B.; Ellenbogen, M.A.; Cardoso, C.; Bartz, J.A.; Yee, J.R.; van Zuiden, M. The Role of Oxytocin in Social Bonding, Stress Regulation and Mental Health: An Update on the Moderating Effects of Context and Interindividual Differences. Psychoneuroendocrinology 2013, 38, 1883–1894. [Google Scholar] [CrossRef]
- Polhemus, D.J.; Calvert, J.W.; Butler, J.; Lefer, D.J. The Cardioprotective Actions of Hydrogen Sulfide in Acute Myocardial Infarction and Heart Failure. Scientifica 2014, 2014, 768607. [Google Scholar] [CrossRef]
- Szabo, C.; Papapetropoulos, A. International Union of Basic and Clinical Pharmacology. CII: Pharmacological Modulation of H2S Levels: H2S Donors and H2S Biosynthesis Inhibitors. Pharmacol. Rev. 2017, 69, 497–564. [Google Scholar] [CrossRef]
- Wang, R. Physiological Implications of Hydrogen Sulfide: A Whiff Exploration That Blossomed. Physiol. Rev. 2012, 92, 791–896. [Google Scholar] [CrossRef]
- Merz, T.; Stenzel, T.; Nußbaum, B.; Wepler, M.; Szabo, C.; Wang, R.; Radermacher, P.; McCook, O. Cardiovascular Disease and Resuscitated Septic Shock Lead to the Downregulation of the H2S-Producing Enzyme Cystathionine-γ-Lyase in the Porcine Coronary Artery. Intensive Care Med. Exp. 2017, 5, 17. [Google Scholar] [CrossRef]
- Nußbaum, B.L.; McCook, O.; Hartmann, C.; Matallo, J.; Wepler, M.; Antonucci, E.; Kalbitz, M.; Huber-Lang, M.; Georgieff, M.; Calzia, E.; et al. Left Ventricular Function during Porcine-Resuscitated Septic Shock with Pre-Existing Atherosclerosis. Intensive Care Med. Exp. 2016, 4, 14. [Google Scholar] [CrossRef]
- Sun, N.-I.; Xi, Y.; Yang, S.; Ma, Z.; Tang, C. Plasma Hydrogen Sulfide and Homocysteine Levels in Hypertensive Patients with Different Blood Pressure Levels and Complications. Zhonghua Xin Xue Guan Bing Za Zhi 2007, 35, 1145–1148. [Google Scholar]
- Chen, L.; Ingrid, S.; Ding, Y.; Liu, Y.; Qi, J.; Tang, C.; Du, J. Imbalance of Endogenous Homocysteine and Hydrogen Sulfide Metabolic Pathway in Essential Hypertensive Children. Chin. Med. J. 2007, 120, 389–393. [Google Scholar] [CrossRef]
- Wang, P.; Wang, S.C.; Yang, H.; Lv, C.; Jia, S.; Liu, X.; Wang, X.; Meng, D.; Qin, D.; Zhu, H.; et al. Therapeutic Potential of Oxytocin in Atherosclerotic Cardiovascular Disease: Mechanisms and Signaling Pathways. Front. Neurosci. 2019, 13, 454. [Google Scholar] [CrossRef]
- Libby, P.; Buring, J.E.; Badimon, L.; Hansson, G.K.; Deanfield, J.; Bittencourt, M.S.; Tokgözŏglu, L.; Lewis, E.F. Atherosclerosis. Nat. Rev. Dis. Primer 2019, 5, 56. [Google Scholar] [CrossRef] [PubMed]
- Lo Faro, M.L.; Fox, B.; Whatmore, J.L.; Winyard, P.G.; Whiteman, M. Hydrogen Sulfide and Nitric Oxide Interactions in Inflammation. Nitric Oxide 2014, 41, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Jankowski, M.; Broderick, T.L.; Gutkowska, J. Oxytocin and Cardioprotection in Diabetes and Obesity. BMC Endocr. Disord. 2016, 16, 34. [Google Scholar] [CrossRef] [PubMed]
- Całka, J. The Role of Nitric Oxide in the Hypothalamic Control of LHRH and Oxytocin Release, Sexual Behavior and Aging of the LHRH and Oxytocin Neurons. Folia Histochem. Cytobiol. 2006, 44, 3–12. [Google Scholar]
- Corrêa, P.B.F.; Pancoto, J.A.T.; de Oliveira-Pelegrin, G.R.; Cárnio, E.C.; Rocha, M.J.A. Participation of INOS-Derived NO in Hypothalamic Activation and Vasopressin Release during Polymicrobial Sepsis. J. Neuroimmunol. 2007, 183, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Kadekaro, M.; Summy-Long, J.Y. Centrally Produced Nitric Oxide And The Regulation Of Body Fluid And Blood Pressure Homeostases. Clin. Exp. Pharmacol. Physiol. 2000, 27, 450–459. [Google Scholar] [CrossRef]
- Reis, W.L.; Giusti-Paiva, A.; Ventura, R.R.; Margatho, L.O.; Gomes, D.A.; Elias, L.L.K.; Antunes-Rodrigues, J. Central Nitric Oxide Blocks Vasopressin, Oxytocin and Atrial Natriuretic Peptide Release and Antidiuretic and Natriuretic Responses Induced by Central Angiotensin II in Conscious Rats: Central NO Blocks Antidiuresis and Natriuresis. Exp. Physiol. 2007, 92, 903–911. [Google Scholar] [CrossRef]
- Coletti, R.; de Lima, J.B.M.; Vechiato, F.M.V.; de Oliveira, F.L.; Debarba, L.K.; Almeida-Pereira, G.; Elias, L.L.K.; Antunes-Rodrigues, J. Nitric Oxide Acutely Modulates Hypothalamic and Neurohypophyseal Carbon Monoxide and Hydrogen Sulphide Production to Control Vasopressin, Oxytocin and Atrial Natriuretic Peptide Release in Rats. J. Neuroendocrinol. 2019, 31, e12686. [Google Scholar] [CrossRef] [PubMed]
- Ali, I.I.; Al-Salam, S.; Howarth, F.C.; Shmygol, A. Oxytocin Induces Intracellular Ca2+ Release in Cardiac Fibroblasts from Neonatal Rats. Cell Calcium 2019, 84, 102099. [Google Scholar] [CrossRef] [PubMed]
- Gutkowska, J.; Jankowski, M.; Antunes-Rodrigues, J. The Role of Oxytocin in Cardiovascular Regulation. Braz. J. Med. Biol. Res. 2014, 47, 206–214. [Google Scholar] [CrossRef]
- Gutkowska, J.; Jankowski, M.; Lambert, C.; Mukaddam-Daher, S.; Zingg, H.H.; McCann, S.M. Oxytocin Releases Atrial Natriuretic Peptide by Combining with Oxytocin Receptors in the Heart. Proc. Natl. Acad. Sci. USA 1997, 94, 11704–11709. [Google Scholar] [CrossRef]
- Krause, S.; Boeck, C.; Gumpp, A.M.; Rottler, E.; Schury, K.; Karabatsiakis, A.; Buchheim, A.; Gündel, H.; Kolassa, I.-T.; Waller, C. Child Maltreatment Is Associated with a Reduction of the Oxytocin Receptor in Peripheral Blood Mononuclear Cells. Front. Psychol. 2018, 9, 173. [Google Scholar] [CrossRef]
- Jankowski, M.; Hajjar, F.; Kawas, S.A.; Mukaddam-Daher, S.; Hoffman, G.; McCann, S.M.; Gutkowska, J. Rat Heart: A Site of Oxytocin Production and Action. Proc. Natl. Acad. Sci. USA 1998, 95, 14558–14563. [Google Scholar] [CrossRef]
- Jankowski, M.; Wang, D.; Hajjar, F.; Mukaddam-Daher, S.; McCann, S.M.; Gutkowska, J. Oxytocin and Its Receptors Are Synthesized in the Rat Vasculature. Proc. Natl. Acad. Sci. USA 2000, 97, 6207–6211. [Google Scholar] [CrossRef] [PubMed]
- Szeto, A.; Sun-Suslow, N.; Mendez, A.J.; Hernandez, R.I.; Wagner, K.V.; McCabe, P.M. Regulation of the Macrophage Oxytocin Receptor in Response to Inflammation. Am. J. Physiol.-Endocrinol. Metab. 2017, 312, E183–E189. [Google Scholar] [CrossRef]
- Reiss, A.B.; Glass, D.S.; Lam, E.; Glass, A.D.; De Leon, J.; Kasselman, L.J. Oxytocin: Potential to Mitigate Cardiovascular Risk. Peptides 2019, 117, 170089. [Google Scholar] [CrossRef]
- Jankowski, M.; Broderick, T.L.; Gutkowska, J. The Role of Oxytocin in Cardiovascular Protection. Front. Psychol. 2020, 11, 2139. [Google Scholar] [CrossRef]
- Faghihi, M.; Alizadeh, A.M.; Khori, V.; Latifpour, M.; Khodayari, S. The Role of Nitric Oxide, Reactive Oxygen Species, and Protein Kinase C in Oxytocin-Induced Cardioprotection in Ischemic Rat Heart. Peptides 2012, 37, 314–319. [Google Scholar] [CrossRef]
- Gutkowska, J.; Jankowski, M. Oxytocin Revisited: Its Role in Cardiovascular Regulation: Role of OT in Cardiovascular Regulation. J. Neuroendocrinol. 2012, 24, 599–608. [Google Scholar] [CrossRef] [PubMed]
- Kajstura, J.; Hosoda, T.; Bearzi, C.; Rota, M.; Maestroni, S.; Urbanek, K.; Leri, A.; Anversa, P. The Human Heart: A Self-Renewing Organ. Clin. Transl. Sci. 2008, 1, 80–86. [Google Scholar] [CrossRef]
- Mukaddam-Daher, S.; Yin, Y.-L.; Roy, J.; Gutkowska, J.; Cardinal, R. Negative Inotropic and Chronotropic Effects of Oxytocin. Hypertension 2001, 38, 292–296. [Google Scholar] [CrossRef]
- Bernatova, I.; Rigatto, K.V.; Key, M.P.; Morris, M. Stress-Induced Pressor and Corticosterone Responses in Oxytocin-Deficient Mice: Stress Reactivity in Oxytocin-Deficient Mice. Exp. Physiol. 2004, 89, 549–557. [Google Scholar] [CrossRef]
- Holst, S.; Uvnäs-Moberg, K.; Petersson, M. Postnatal Oxytocin Treatment and Postnatal Stroking of Rats Reduce Blood Pressure in Adulthood. Auton. Neurosci. 2002, 99, 85–90. [Google Scholar] [CrossRef]
- Petersson, M.; Uvnäs-Moberg, K. Postnatal Oxytocin Treatment of Spontaneously Hypertensive Male Rats Decreases Blood Pressure and Body Weight in Adulthood. Neurosci. Lett. 2008, 440, 166–169. [Google Scholar] [CrossRef] [PubMed]
- Peters, S.; Slattery, D.A.; Uschold-Schmidt, N.; Reber, S.O.; Neumann, I.D. Dose-Dependent Effects of Chronic Central Infusion of Oxytocin on Anxiety, Oxytocin Receptor Binding and Stress-Related Parameters in Mice. Psychoneuroendocrinology 2014, 42, 225–236. [Google Scholar] [CrossRef]
- Wsol, A.; Cudnoch-Jedrzejewska, A.; Szczepanska-Sadowska, E.; Kowalewski, S.; Puchalska, L. Oxytocin in The Cardiovascular Responses to Stress. J. Physiol. Pharmacol. 2008, 59, 123–127. [Google Scholar]
- Dobruch, J.; Cudnoch-Jedrzejewska, A.; Szczepanska-Sadowska, E. Enhanced Involvement of Brain Vasopressin V1 Receptors in Cardiovascular Responses to Stress in Rats with Myocardial Infarction: Research Report. Stress 2005, 8, 273–284. [Google Scholar] [CrossRef] [PubMed]
- Fleet, R.; Lespérance, F.; Arsenault, A.; Grégoire, J.; Lavoie, K.; Laurin, C.; Harel, F.; Burelle, D.; Lambert, J.; Beitman, B.; et al. Myocardial Perfusion Study of Panic Attacks in Patients with Coronary Artery Disease. Am. J. Cardiol. 2005, 96, 1064–1068. [Google Scholar] [CrossRef]
- Klein, B.Y.; Albers, H.; Anwar, M.; Ludwig, R.J.; Kaidbey, J.H.; Glickstein, S.B.; Welch, M.G. Assessing Cellular Stress and Inflammation in Discrete Oxytocin-Secreting Brain Nuclei in the Neonatal Rat before and after First Colostrum Feeding. J. Vis. Exp. 2018, 141, e58341. [Google Scholar] [CrossRef]
- Carter, C.S.; Kenkel, W.M.; MacLean, E.L.; Wilson, S.R.; Perkeybile, A.M.; Yee, J.R.; Ferris, C.F.; Nazarloo, H.P.; Porges, S.W.; Davis, J.M.; et al. Is Oxytocin “Nature’s Medicine”? Pharmacol. Rev. 2020, 72, 829–861. [Google Scholar] [CrossRef] [PubMed]
- Peleli, M.; Bibli, S.-I.; Li, Z.; Chatzianastasiou, A.; Varela, A.; Katsouda, A.; Zukunft, S.; Bucci, M.; Vellecco, V.; Davos, C.H.; et al. Cardiovascular Phenotype of Mice Lacking 3-Mercaptopyruvate Sulfurtransferase. Biochem. Pharmacol. 2020, 176, 113833. [Google Scholar] [CrossRef] [PubMed]
- Donadon, M.F.; Martin-Santos, R.; Osório, F.d.L. The Associations Between Oxytocin and Trauma in Humans: A Systematic Review. Front. Pharmacol. 2018, 9, 154. [Google Scholar] [CrossRef] [PubMed]
- Heinrichs, M.; Baumgartner, T.; Kirschbaum, C.; Ehlert, U. Social Support and Oxytocin Interact to Suppress Cortisol and Subjective Responses to Psychosocial Stress. Biol. Psychiatry 2003, 54, 1389–1398. [Google Scholar] [CrossRef]
- Juruena, M.F.; Cleare, A.J.; Pariante, C.M. The Hypothalamic Pituitary Adrenal Axis, Glucocorticoid Receptor Function and Relevance to Depression. Rev. Bras. Pisquiatr. 2004, 26, 189–201. [Google Scholar] [CrossRef]
- Boyce, W.T.; Ellis, B.J. Biological Sensitivity to Context: I. An Evolutionary–Developmental Theory of the Origins and Functions of Stress Reactivity. Dev. Psychopathol. 2005, 17, 271–301. [Google Scholar] [CrossRef]
- Neumann, I.D.; Landgraf, R. Balance of Brain Oxytocin and Vasopressin: Implications for Anxiety, Depression, and Social Behaviors. Trends Neurosci. 2012, 35, 649–659. [Google Scholar] [CrossRef] [PubMed]
- Olff, M. Bonding after Trauma: On the Role of Social Support and the Oxytocin System in Traumatic Stress. Eur. J. Psychotraumatol. 2012, 3, 18597. [Google Scholar] [CrossRef]
- Kuhlman, K.R.; Vargas, I.; Geiss, E.G.; Lopez-Duran, N.L. Age of Trauma Onset and HPA Axis Dysregulation Among Trauma-Exposed Youth: Age of First Trauma and HPA Axis Functioning. J. Trauma. Stress 2015, 28, 572–579. [Google Scholar] [CrossRef] [PubMed]
- Kundakovic, M.; Champagne, F.A. Early-Life Experience, Epigenetics, and the Developing Brain. Neuropsychopharmacology 2015, 40, 141–153. [Google Scholar] [CrossRef]
- Lesse, A.; Rether, K.; Gröger, N.; Braun, K.; Bock, J. Chronic Postnatal Stress Induces Depressive-Like Behavior in Male Mice and Programs Second-Hit Stress-Induced Gene Expression Patterns of OxtR and AvpR1a in Adulthood. Mol. Neurobiol. 2017, 54, 4813–4819. [Google Scholar] [CrossRef]
- Ozbay, F.; Fitterling, H.; Charney, D.; Southwick, S. Social Support and Resilience to Stress across the Life Span: A Neurobiologic Framework. Curr. Psychiatry Rep. 2008, 10, 304–310. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.; Jenkins, J.M.; Steiner, M.; Fleming, A.S. The Relation between Early Life Adversity, Cortisol Awakening Response and Diurnal Salivary Cortisol Levels in Postpartum Women. Psychoneuroendocrinology 2009, 34, 76–86. [Google Scholar] [CrossRef]
- Nicolson, N.A.; Davis, M.C.; Kruszewski, D.; Zautra, A.J. Childhood Maltreatment and Diurnal Cortisol Patterns in Women with Chronic Pain. Psychosom. Med. 2010, 72, 471–480. [Google Scholar] [CrossRef]
- Kirsch, P. Oxytocin Modulates Neural Circuitry for Social Cognition and Fear in Humans. J. Neurosci. 2005, 25, 11489–11493. [Google Scholar] [CrossRef]
- Laviola, G.; Busdraghi, L.M.; Meschino, N.; Petrella, C.; Fiore, M. Aberrant Early in Life Stimulation of the Stress-Response System Affects Emotional Contagion and Oxytocin Regulation in Adult Male Mice. Int. J. Mol. Sci. 2021, 22, 5039. [Google Scholar] [CrossRef] [PubMed]
- Yehuda, R.; Bierer, L.M.; Schmeidler, J.; Aferiat, D.H.; Breslau, I.; Dolan, S. Low Cortisol and Risk for PTSD in Adult Offspring of Holocaust Survivors. Am. J. Psychiatry 2000, 157, 1252–1259. [Google Scholar] [CrossRef]
- Brown, C.A.; Cardoso, C.; Ellenbogen, M.A. A Meta-Analytic Review of the Correlation between Peripheral Oxytocin and Cortisol Concentrations. Front. Neuroendocrinol. 2016, 43, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Tan, H.; Zou, W.; Jiang, J.; Tian, Y.; Xiao, Z.; Bi, L.; Zeng, H.; Tang, X. Disturbance of Hippocampal H2S Generation Contributes to CUMS-Induced Depression-like Behavior: Involvement in Endoplasmic Reticulum Stress of Hippocampus. Acta Biochim. Biophys. Sin. 2015, 47, 285–291. [Google Scholar] [CrossRef]
- Liu, S.-Y.; Li, D.; Zeng, H.-Y.; Kan, L.-Y.; Zou, W.; Zhang, P.; Gu, H.-F.; Tang, X.-Q. Hydrogen Sulfide Inhibits Chronic Unpredictable Mild Stress-Induced Depressive-Like Behavior by Upregulation of Sirt-1: Involvement in Suppression of Hippocampal Endoplasmic Reticulum Stress. Int. J. Neuropsychopharmacol. 2017, 20, 867–876. [Google Scholar] [CrossRef]
- Brenner, L.A. Neuropsychological and Neuroimaging Findings in Traumatic Brain Injury and Post-Traumatic Stress Disorder. Dialogues Clin. Neurosci. 2011, 13, 311–323. [Google Scholar] [CrossRef] [PubMed]
- Mauri, M.C.; Paletta, S.; Di Pace, C.; Reggiori, A.; Cirnigliaro, G.; Miserocchi, G. Depression and Post-Traumatic Brain Injury: Clinical and Neuropsychological Characteristics. Dual Diagn. Open Access 2017, 2, 29. [Google Scholar] [CrossRef]
- Howland, R.H. Vagus Nerve Stimulation. Curr. Behav. Neurosci. Rep. 2014, 1, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Tracey, K.J. Physiology and Immunology of the Cholinergic Antiinflammatory Pathway. J. Clin. Investig. 2007, 117, 289–296. [Google Scholar] [CrossRef]
- Lozić, M.; Greenwood, M.; Šarenac, O.; Martin, A.; Hindmarch, C.; Tasić, T.; Paton, J.; Murphy, D.; Japundžić-Žigon, N. Overexpression of Oxytocin Receptors in the Hypothalamic PVN Increases Baroreceptor Reflex Sensitivity and Buffers BP Variability in Conscious Rats: Oxytocin Receptors in Autonomic Circulatory Control. Br. J. Pharmacol. 2014, 171, 4385–4398. [Google Scholar] [CrossRef] [PubMed]
- Garrott, K.; Dyavanapalli, J.; Cauley, E.; Dwyer, M.K.; Kuzmiak-Glancy, S.; Wang, X.; Mendelowitz, D.; Kay, M.W. Chronic Activation of Hypothalamic Oxytocin Neurons Improves Cardiac Function during Left Ventricular Hypertrophy-Induced Heart Failure. Cardiovasc. Res. 2017, 113, 1318–1328. [Google Scholar] [CrossRef]
- Mioni, C.; Bazzani, C.; Giuliani, D.; Altavilla, D.; Leone, S.; Ferrari, A.; Minutoli, L.; Bitto, A.; Marini, H.; Zaffe, D.; et al. Activation of an Efferent Cholinergic Pathway Produces Strong Protection against Myocardial Ischemia/Reperfusion Injury in Rats. Crit. Care Med. 2005, 33, 2621–2628. [Google Scholar] [CrossRef]
- Zhang, X.-Y. Effects of Nuclei Ambiguus and Dorsal Motor Nuclei of Vagus on Gastric H+ and HCO3− Secretion in Rats. World J. Gastroenterol. 2006, 12, 3271. [Google Scholar] [CrossRef] [PubMed]
- Elorza-Ávila, A.R.; Reyes-Lagos, J.J.; Hadamitzky, M.; Peña-Castillo, M.Á.; Echeverría, J.C.; Ortiz-Pedroza, M.d.R.; Lückemann, L.; Schedlowski, M.; Pacheco-López, G. Oxytocin’s Role on the Cardiorespiratory Activity of Endotoxemic Rats. Respir. Physiol. Neurobiol. 2017, 236, 19–22. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.-Z.; Yu, K.-H.; Ai, H.-B. Role of Hydrogen Sulfide within the Dorsal Motor Nucleus of the Vagus in the Control of Gastric Function in Rats. Neurogastroenterol. Motil. 2015, 27, 618–626. [Google Scholar] [CrossRef] [PubMed]
- Gan, X.-B.; Liu, T.-Y.; Xiong, X.-Q.; Chen, W.-W.; Zhou, Y.-B.; Zhu, G.-Q. Hydrogen Sulfide in Paraventricular Nucleus Enhances Sympathetic Activity and Cardiac Sympathetic Afferent Reflex in Chronic Heart Failure Rats. PLoS ONE 2012, 7, e50102. [Google Scholar] [CrossRef]
- Austgen, J.R.; Hermann, G.E.; Dantzler, H.A.; Rogers, R.C.; Kline, D.D. Hydrogen Sulfide Augments Synaptic Neurotransmission in the Nucleus of the Solitary Tract. J. Neurophysiol. 2011, 106, 1822–1832. [Google Scholar] [CrossRef] [PubMed]
- Okazaki, M.; Uozu, S.; Sato, Y.; Matsumoto, M.; Koganezawa, T. Endogenous Hydrogen Sulfide Maintains Eupnea in an in Situ Arterially Perfused Preparation of Rats. Commun. Biol. 2020, 3, 583. [Google Scholar] [CrossRef]
- Makarenko, V.V.; Nanduri, J.; Raghuraman, G.; Fox, A.P.; Gadalla, M.M.; Kumar, G.K.; Snyder, S.H.; Prabhakar, N.R. Endogenous H2S Is Required for Hypoxic Sensing by Carotid Body Glomus Cells. Am. J. Physiol. Cell Physiol. 2012, 303, C916–C923. [Google Scholar] [CrossRef]
- Xiao, L.; Wu, Y.; Wang, R.; Liu, Y.; Wang, F.; He, R. Hydrogen Sulfide Facilitates Carotid Sinus Baroreceptor Activity in Anesthetized Male Rats. Chin. Med. J. 2007, 120, 1343–1347. [Google Scholar] [CrossRef]
- Gerritsen, R.J.S.; Band, G.P.H. Breath of Life: The Respiratory Vagal Stimulation Model of Contemplative Activity. Front. Hum. Neurosci. 2018, 12, 397. [Google Scholar] [CrossRef]
- Dudeja, J.P. Scientific Analysis of Mantra-Based Meditation and Its Beneficial Effects: An Overview. Int. J. Adv. Sci. Technol. Eng. Manag. Sci. 2017, 3, 21–26. [Google Scholar] [CrossRef]
- Dusek, J.A.; Chang, B.H.; Zaki, J.; Lazar, S.; Deykin, A.; Stefano, G.B.; Wohlhueter, A.L.; Hibberd, P.L.; Benson, H. Association between oxygen consumption and nitric oxide production during the relaxation response. Med. Sci. Monit. 2005, 12, CR1–CR10. [Google Scholar] [PubMed]
- Kaliman, P.; Álvarez-López, M.J.; Cosín-Tomás, M.; Rosenkranz, M.A.; Lutz, A.; Davidson, R.J. Rapid Changes in Histone Deacetylases and Inflammatory Gene Expression in Expert Meditators. Psychoneuroendocrinology 2014, 40, 96–107. [Google Scholar] [CrossRef]
- Kabat-Zinn, J. Mindfulness-based stress reduction (MBSR). Constr. HumSci. 2003, 8, 73. [Google Scholar]
- Amarasekera, A.T.; Chang, D. Buddhist meditation for vascular function: A narrative review. Integr. Med. Res. 2019, 8, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Van Cappellen, P.; Way, B.M.; Isgett, S.F.; Fredrickson, B.L. Effects of Oxytocin Administration on Spirituality and Emotional Responses to Meditation. Soc. Cogn. Affect. Neurosci. 2016, 11, 1579–1587. [Google Scholar] [CrossRef]
- Machida, S.; Sunagawa, M.; Takahashi, T. Oxytocin Release during the Meditation of Altruism and Appreciation (Arigato-Zen). Int. J. Neurol. Res. 2018, 4, 364–370. [Google Scholar] [CrossRef]
- Ito, E.; Shima, R.; Yoshioka, T. A Novel Role of Oxytocin: Oxytocin-Induced Well-Being in Humans. Biophys. Physicobiol. 2019, 16, 132–139. [Google Scholar] [CrossRef]
- Kim, D.H.; Moon, Y.S.; Kim, H.S.; Jung, J.S.; Park, H.M.; Suh, H.W.; Kim, Y.H.; Song, D.K. Effect of Zen Meditation on serum nitric oxide activity and lipid peroxidation. Prog. Neuropsychopharmacol. Biol. Psychiatry 2005, 29, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Bellosta-Batalla, M.; Blanco-Gandía, M.; Rodríguez-Arias, M.; Cebolla, A.; Pérez-Blasco, J.; Moya-Albiol, L. Brief Mindfulness Session Improves Mood and Increases Salivary Oxytocin in Psychology Students. Stress Health 2020, 36, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Mayer, S. Studien zur Physiologie des Herzens und der Blutgefaesse 6.Abhandlung: Ueber spontane Blutdruckschwenkungen. [Studies on the physiology of the heart and the blood vessels 6. Discourse on fluctuations in blood pressure]. Sitz Ber Akad Wiss Wien Mathe Naturwiss Kl Anat 1876, 74, 281–307. [Google Scholar]
- Bernardi, L.; Sleight, P.; Bandinelli, G.; Cencetti, S.; Fattorini, L.; Wdowczyc-Szulc, J.; Lagi, A. Effect of Rosary Prayer and Yoga Mantras on Autonomic Cardiovascular Rhythms: Comparative Study. BMJ 2001, 323, 1446–1449. [Google Scholar] [CrossRef] [PubMed]
- Peressutti, C.; Martín-González, J.M.; García-Manso, J.M. Does Mindfulness Meditation Shift the Cardiac Autonomic Nervous System to a Highly Orderly Operational State? Int. J. Cardiol. 2012, 154, 210–212. [Google Scholar] [CrossRef]
- Fox, K.C.R.; Nijeboer, S.; Dixon, M.L.; Floman, J.L.; Ellamil, M.; Rumak, S.P.; Sedlmeier, P.; Christoff, K. Is Meditation Associated with Altered Brain Structure? A Systematic Review and Meta-Analysis of Morphometric Neuroimaging in Meditation Practitioners. Neurosci. Biobehav. Rev. 2014, 43, 48–73. [Google Scholar] [CrossRef] [PubMed]
- Erdman, S.E. Microbial Muses: Threads of Our Inner Wisdom. Challenges 2021, 12, 10. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
McCook, O.; Denoix, N.; Merz, T. The Gasotransmitter Hydrogen Sulfide and the Neuropeptide Oxytocin as Potential Mediators of Beneficial Cardiovascular Effects through Meditation after Traumatic Events. Trauma Care 2021, 1, 183-194. https://doi.org/10.3390/traumacare1030016
McCook O, Denoix N, Merz T. The Gasotransmitter Hydrogen Sulfide and the Neuropeptide Oxytocin as Potential Mediators of Beneficial Cardiovascular Effects through Meditation after Traumatic Events. Trauma Care. 2021; 1(3):183-194. https://doi.org/10.3390/traumacare1030016
Chicago/Turabian StyleMcCook, Oscar, Nicole Denoix, and Tamara Merz. 2021. "The Gasotransmitter Hydrogen Sulfide and the Neuropeptide Oxytocin as Potential Mediators of Beneficial Cardiovascular Effects through Meditation after Traumatic Events" Trauma Care 1, no. 3: 183-194. https://doi.org/10.3390/traumacare1030016
APA StyleMcCook, O., Denoix, N., & Merz, T. (2021). The Gasotransmitter Hydrogen Sulfide and the Neuropeptide Oxytocin as Potential Mediators of Beneficial Cardiovascular Effects through Meditation after Traumatic Events. Trauma Care, 1(3), 183-194. https://doi.org/10.3390/traumacare1030016




