Exploring the Impact of Herbal Therapies on COVID-19 and Influenza: Investigating Novel Delivery Mechanisms for Emerging Interventions
Abstract
1. Introduction
2. Results
3. Discussion
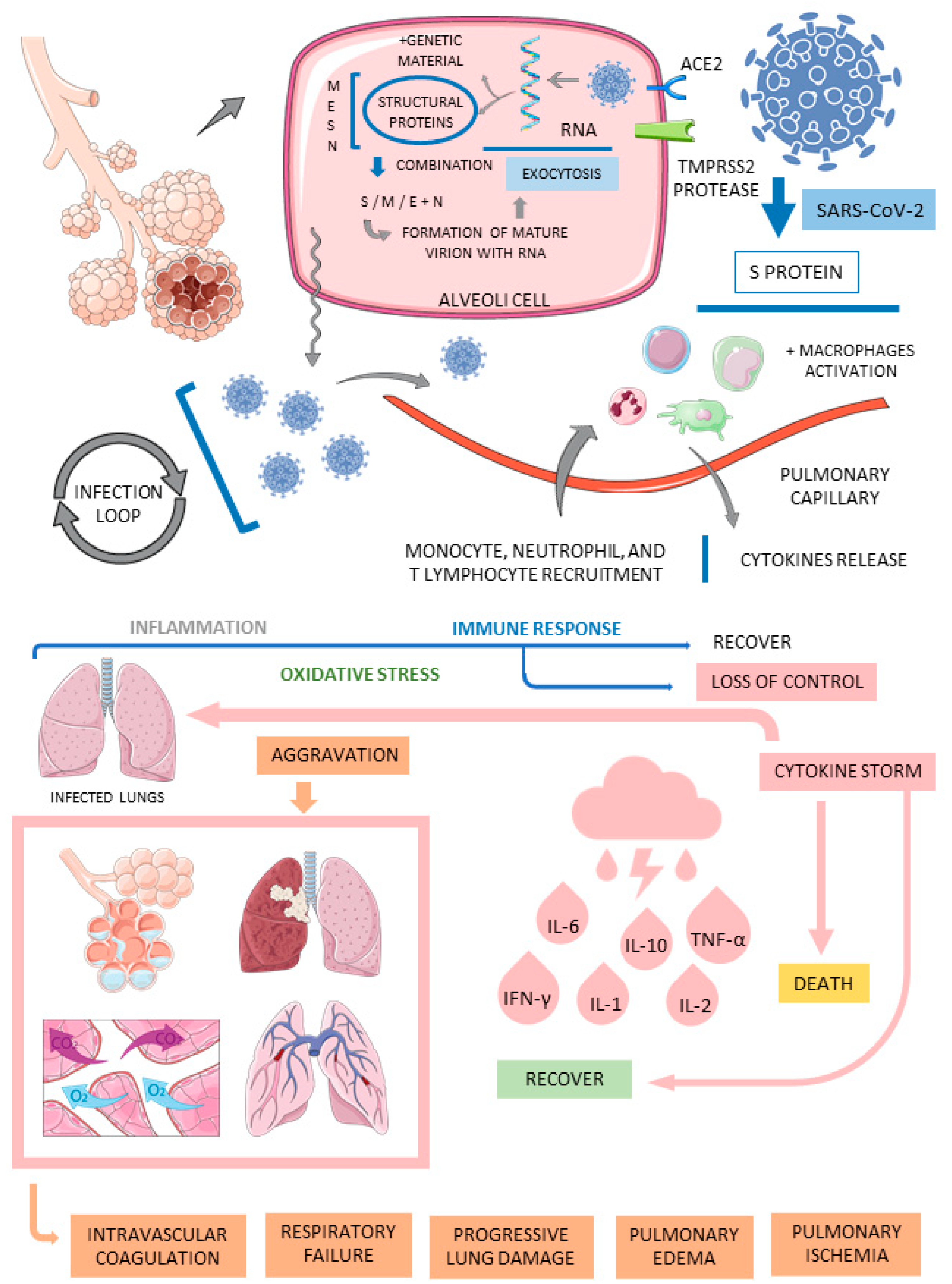
3.1. COVID-19
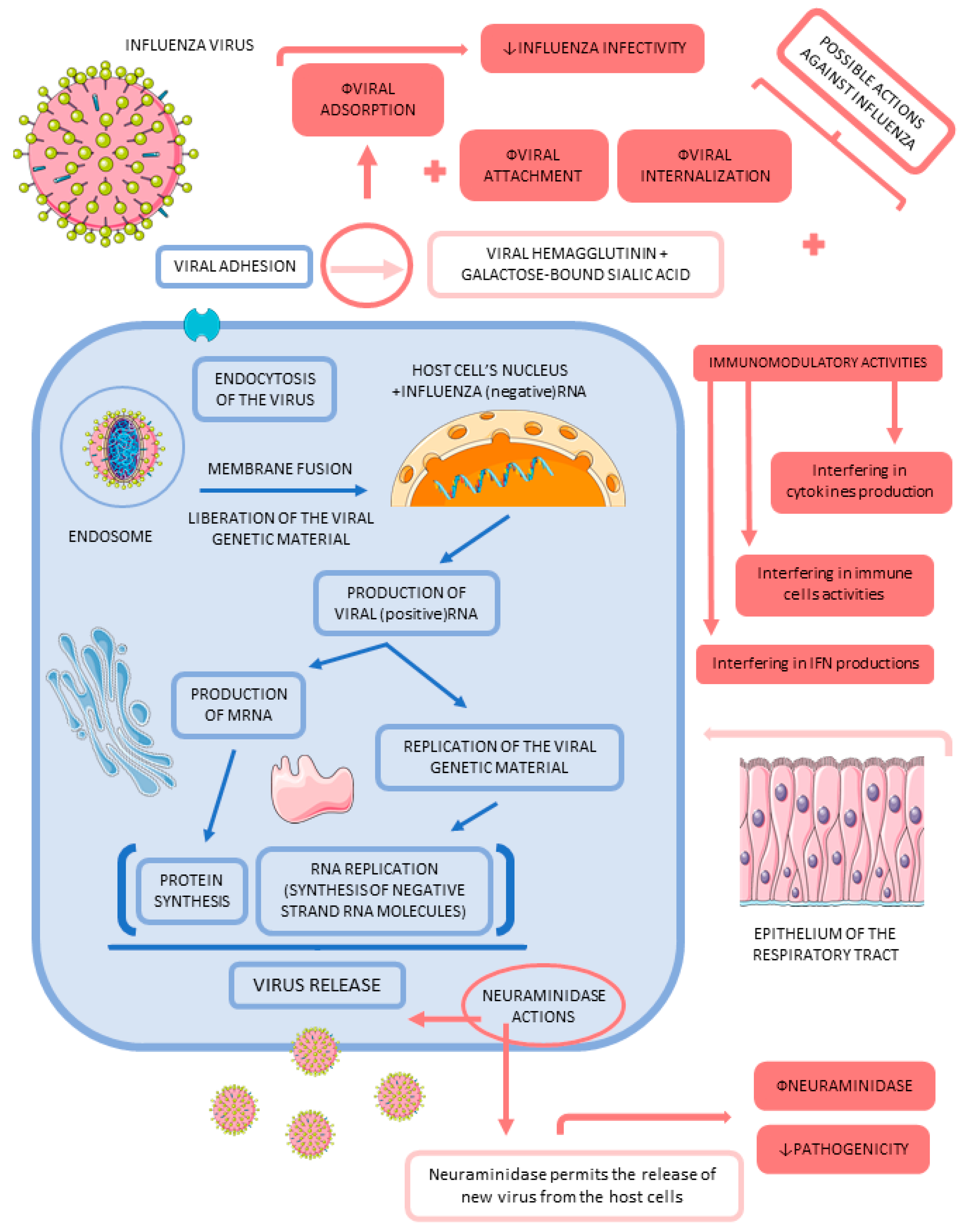
3.2. Influenza
3.3. The Role of Medicinal Plants on COVID-19
3.4. The Role of Medicinal Plants on Influenza
3.5. Medicinal Plants, COVID-19, and Influenza
3.6. Delivery Systems for Medicinal Plants and Their Derivatives against COVID-19 and Influenza
4. Materials and Methods
4.1. Focal Question
4.2. Language
4.3. Databases
4.4. Study Selection
4.5. Data Extraction
4.6. Quality Assessment
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Flato, U.A.P.; Biteli, P.; Reina, D.O.B.R.; Reina, F.T.R.; Araújo, A.C.; de Souza, G.A.; dos Santos Campanari, G.; Matias, J.N.; Marinho, V.; Lima, T.L.Z.; et al. Myocarditis as a serious complication of COVID-19. Int. J. Adv. Eng. Res. Sci. 2021, 8, 3. [Google Scholar]
- Vallejos, J.; Zoni, R.; Bangher, M.; Villamandos, S.; Bobadilla, A.; Plano, F.; Campias, C.; Chaparro Campias, E.; Medina, M.F.; Achinelli, F.; et al. Ivermectin to prevent hospitalizations in patients with COVID-19 (IVERCOR-COVID19) a randomized, double-blind, placebo-controlled trial. BMC Infect. Dis. 2021, 21, 635. [Google Scholar] [CrossRef] [PubMed]
- Yanai, H. Metabolic Syndrome and COVID-19. Cardiol. Res. 2020, 11, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Deressa, W.; Worku, A.; Abebe, W.; Gizaw, M.; Amogne, W. Risk perceptions and preventive practices of COVID-19 among healthcare professionals in public hospitals in Addis Ababa, Ethiopia. PLoS ONE 2021, 16, e0242471. [Google Scholar] [CrossRef] [PubMed]
- Durstenfeld, M.S.; Peluso, M.J.; Peyser, N.D.; Lin, F.; Knight, S.J.; Djibo, A.; Khatib, R.; Kitzman, H.; O’Brien, E.; Williams, N.; et al. Factors Associated with Long COVID Symptoms in an Online Cohort Study. Open. Forum Infect. Dis. 2023, 10, ofad047. [Google Scholar] [CrossRef] [PubMed]
- Rahier, J.F.; Papay, P.; Salleron, J.; Sebastian, S.; Ellul, P.; Teich, N.; Fiorino, G.; Blaha, B.; Garcia-Sanchez, V.; Haas, T.; et al. Influenza A (H1N1)v infection in patients with inflammatory bowel disease: A case series. Aliment. Pharmacol. Ther. 2011, 33, 499–500. [Google Scholar] [CrossRef]
- Aghaali, M.; Kavousi, A.; Shahsavani, A.; Hashemi Nazari, S.S. Performance of Bayesian Outbreak Detection Algorithm in the Syndromic Surveillance of Influenza-Like Illness in Small Region. Transbound. Emerg. Dis. 2020. ahead of print. [Google Scholar] [CrossRef]
- Cantan, B.; Luyt, C.E.; Martin-Loeches, I. Influenza Infections and Emergent Viral Infections in Intensive Care Unit. Semin. Respir. Crit. Care Med. 2019, 40, 488–497. [Google Scholar] [CrossRef]
- Caldera, F.; Hillman, L.; Saha, S.; Wald, A.; Grimes, I.; Zhang, Y.; Sharpe, A.R.; Reichelderfer, M.; Hayney, M.S. Immunogenicity of High Dose Influenza Vaccine for Patients with Inflammatory Bowel Disease on Anti-TNF Monotherapy: A Randomized Clinical Trial. Inflamm. Bowel Dis. 2020, 26, 593–602. [Google Scholar] [CrossRef]
- Berg, S.H.; O’Hara, J.K.; Shortt, M.T.; Thune, H.; Brønnick, K.K.; Lungu, D.A.; Røislien, J.; Wiig, S. Health authorities’ health risk communication with the public during pandemics: A rapid scoping review. BMC Public Health 2021, 21, 1401. [Google Scholar] [CrossRef]
- Grohskopf, L.A.; Alyanak, E.; Broder, K.R.; Blanton, L.H.; Fry, A.M.; Jernigan, D.B.; Atmar, R.L. Prevention and Control of Seasonal Influenza with Vaccines: Recommendations of the Advisory Committee on Immunization Practices—United States, 2020–2021 Influenza Season. MMWR Recomm. Rep. 2020, 69, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Duvvuri, V.R.; Hicks, J.T.; Damodaran, L.; Grunnill, M.; Braukmann, T.; Wu, J.; Gubbay, J.B.; Patel, S.N.; Bahl, J. Comparing the transmission potential from sequence and surveillance data of 2009 North American influenza pandemic waves. Infect. Dis. Model. 2023, 8, 240–252. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Chen, J.; Cheng, L.; Xu, K.; Yang, Y.; Su, X. Deficiency of HIF-1α enhances influenza A virus replication by promoting autophagy in alveolar type II epithelial cells. Emerg. Microbes Infect. 2020, 9, 691–706. [Google Scholar] [CrossRef] [PubMed]
- Barbalho, S.M.; Matias, J.N.; Flato, U.A.P.; Pilon, J.P.G.; Bitelli, P.; Junior, M.A.P.; de Carvalho, A.C.A.; dos Santos Haber, J.F.; Reis, C.H.B.; de Alvares Goulart, R.J.G.R. What do influenza and COVID-19 represent for patients with inflammatory bowel disease? Gastroenterol. Res. 2021, 14, 1. [Google Scholar] [CrossRef]
- Jagadesh, A.; Salam, A.A.; Mudgal, P.P.; Arunkumar, G. Influenza virus neuraminidase (NA): A target for antivirals and vaccines. Arch. Virol. 2016, 161, 2087–2094. [Google Scholar] [CrossRef]
- Mullender, C.; da Costa, K.A.S.; Alrubayyi, A.; Pett, S.L.; Peppa, D. SARS-CoV-2 immunity and vaccine strategies in people with HIV. Oxf. Open. Immunol. 2022, 3, iqac005. [Google Scholar] [CrossRef]
- Haunhorst, S.; Bloch, W.; Wagner, H.; Ellert, C.; Krüger, K.; Vilser, D.C.; Finke, K.; Reuken, P.; Pletz, M.W.; Stallmach, A.; et al. Long COVID: A narrative review of the clinical aftermaths of COVID-19 with a focus on the putative pathophysiology and aspects of physical activity. Oxf. Open. Immunol. 2022, 3, iqac006. [Google Scholar] [CrossRef]
- Torres, A.R.; Gómez, V.; Kislaya, I.; Rodrigues, A.P.; Fernandes Tavares, M.; Pereira, A.C.; Pereira, D.; Côrte-Real, R.; Flores, C.H.; Verdasca, N.; et al. Monitoring COVID-19 and Influenza: The Added Value of a Severe Acute Respiratory Infection Surveillance System in Portugal. Can. J. Infect. Dis. Med. Microbiol. 2023, 2023, 6590011. [Google Scholar] [CrossRef]
- Bartoli, A.; Gabrielli, F.; Alicandro, T.; Nascimbeni, F.; Andreone, P. COVID-19 treatment options: A difficult journey between failed attempts and experimental drugs. Intern. Emerg. Med. 2021, 16, 281–308. [Google Scholar] [CrossRef]
- Caricchio, R.; Abbate, A.; Gordeev, I.; Meng, J.; Hsue, P.Y.; Neogi, T.; Arduino, R.; Fomina, D.; Bogdanov, R.; Stepanenko, T.; et al. Effect of Canakinumab vs Placebo on Survival Without Invasive Mechanical Ventilation in Patients Hospitalized with Severe COVID-19: A Randomized Clinical Trial. JAMA 2021, 326, 230–239. [Google Scholar] [CrossRef]
- Bian, H.; Zheng, Z.H.; Wei, D.; Wen, A.; Zhang, Z.; Lian, J.Q.; Kang, W.Z.; Hao, C.Q.; Wang, J.; Xie, R.H.; et al. Safety and efficacy of meplazumab in healthy volunteers and COVID-19 patients: A randomized phase 1 and an exploratory phase 2 trial. Signal. Transduct. Target. Ther. 2021, 6, 194. [Google Scholar] [CrossRef]
- Abiri, R.; Abdul-Hamid, H.; Sytar, O.; Abiri, R.; Bezerra de Almeida, E., Jr.; Sharma, S.K.; Bulgakov, V.P.; Arroo, R.R.J.; Malik, S. A Brief Overview of Potential Treatments for Viral Diseases Using Natural Plant Compounds: The Case of SARS-CoV. Molecules 2021, 26, 3868. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, D.; Chen, X.; Liu, X.; Xiao, W.; Feng, L. The effect of nirmatrelvir-ritonavir on viral clearance and length of hospital stay in patients infected with SARS-CoV-2 omicron variants. Influenza Other Respir. Viruses 2023, 17, e13095. [Google Scholar] [CrossRef]
- Xia, L.; Shi, Y.; Su, J.; Friedemann, T.; Tao, Z.; Lu, Y.; Ling, Y.; Lv, Y.; Zhao, R.; Geng, Z.; et al. Shufeng Jiedu, a promising herbal therapy for moderate COVID-19: Antiviral and anti-inflammatory properties, pathways of bioactive compounds, and a clinical real-world pragmatic study. Phytomedicine Int. J. Phytother. Phytopharm. 2021, 85, 153390. [Google Scholar] [CrossRef]
- Sun, Z.; He, G.; Huang, N.; Thilakavathy, K.; Lim, J.C.W.; Kumar, S.S.; Xiong, C. Glycyrrhizic Acid: A Natural Plant Ingredient as a Drug Candidate to Treat COVID-19. Front. Pharmacol. 2021, 12, 707205. [Google Scholar] [CrossRef] [PubMed]
- Rolta, R.; Salaria, D.; Sharma, P.; Sharma, B.; Kumar, V.; Rathi, B.; Verma, M.; Sourirajan, A.; Baumler, D.J.; Dev, K. Phytocompounds of Rheum emodi, Thymus serpyllum, and Artemisia annua Inhibit Spike Protein of SARS-CoV-2 Binding to ACE2 Receptor: In Silico Approach. Curr. Pharmacol. Rep. 2021, 7, 135–149. [Google Scholar] [CrossRef]
- Tegen, D.; Dessie, K.; Damtie, D. Candidate Anti-COVID-19 Medicinal Plants from Ethiopia: A Review of Plants Traditionally Used to Treat Viral Diseases. Evid. Based Complement. Altern. Med. 2021, 2021, 6622410. [Google Scholar] [CrossRef] [PubMed]
- Tao, Z.; Meng, X.; Han, Y.-q.; Xue, M.-m.; Wu, S.; Wu, P.; Yuan, Y.; Zhu, Q.; Zhang, T.-J.; Wong, C.C.J. Therapeutic mechanistic studies of ShuFengJieDu capsule in an acute lung injury animal model using quantitative proteomics technology. J. Proteome Res. 2017, 16, 4009–4019. [Google Scholar] [CrossRef] [PubMed]
- Jiang, F.; Xu, N.; Zhou, Y.; Song, J.; Liu, J.; Zhu, H.; Jiang, J.; Xu, Y.; Li, R. Contribution of traditional Chinese medicine combined with conventional western medicine treatment for the novel coronavirus disease (COVID-19), current evidence with systematic review and meta-analysis. Phytother. Res. 2021, 35, 5992–6009. [Google Scholar] [CrossRef]
- Yu, C.; Huang, Y.; Ren, X.; Sun, L. Plant-derived Ren’s oligopeptide has antiviral effects on influenza virus and SARS-CoV-2. Front. Vet. Sci. 2022, 9, 1090372. [Google Scholar] [CrossRef]
- Houeze, E.A.; Wang, Y.; Zhou, Q.; Zhang, H.; Wang, X. Comparison study of Beninese and Chinese herbal medicines in treating COVID-19. J. Ethnopharmacol. 2023, 308, 116172. [Google Scholar] [CrossRef] [PubMed]
- Abbas, H.S.; Abd-Elhakeem, M.M.; Abd El Galil, R.M.; Reyad, O.A.; Mohamed, H.A.; Ismail, S.E.S.; Nabil, M.A. Natural Immunomodulators Treat the Cytokine Storm in SARS-CoV-2. Adv. Pharm. Bull. 2023, 13, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Chojnacka, K.; Witek-Krowiak, A.; Skrzypczak, D.; Mikula, K.; Młynarz, P. Phytochemicals containing biologically active polyphenols as an effective agent against COVID-19-inducing coronavirus. J. Funct. Foods 2020, 73, 104146. [Google Scholar] [CrossRef] [PubMed]
- Zeng, N.; Chen, X.; Liu, Z. Natural Products and Nanotechnology Against Coronavirus Disease 2019. Front. Chem. 2022, 10, 819969. [Google Scholar] [CrossRef] [PubMed]
- Kaur, M.; Devi, G.; Nagpal, M.; Singh, M.; Dhingra, G.A.; Aggarwal, G. Antiviral Essential Oils Incorporated in Nanocarriers: Strategy for Prevention from COVID-19 and Future Infectious Pandemics. Pharm. Nanotechnol. 2020, 8, 437–451. [Google Scholar] [CrossRef]
- Adel Mehraban, M.S.; Shirzad, M.; Mohammad Taghizadeh Kashani, L.; Ahmadian-Attari, M.M.; Safari, A.A.; Ansari, N.; Hatami, H.; Kamalinejad, M. Efficacy and safety of add-on Viola odorata L. in the treatment of COVID-19: A randomized double-blind controlled trial. J. Ethnopharmacol. 2023, 304, 116058. [Google Scholar] [CrossRef]
- Forouzanfar, F.; Ahmadpoor, M.; Farahi, M.M.; Hadianfar, A.; Sahebkar, A.; Esmaily, H.; Nematy, M.; Rakhshandeh, H. The Effect of Pomegranate Juice and Sumac Consumption in the Treatment of Outpatients with COVID-19. Mediat. Inflamm. 2022, 2022, 6850342. [Google Scholar] [CrossRef]
- Hasanpour, M.; Safari, H.; Mohammadpour, A.H.; Iranshahy, M.; Dehghan Nayyeri, M.J.; Farhadi, F.; Emami, B.; Iranshahi, M. Efficacy of Covexir® (Ferula foetida oleo-gum) treatment in symptomatic improvement of patients with mild to moderate COVID-19: A randomized, double-blind, placebo-controlled trial. Phytother. Res. 2022, 36, 4504–4515. [Google Scholar] [CrossRef]
- Borujerdi, R.; Adeli, S.H.; Mohammadbeigi, A.; Aliasl, F.; Asghari, A.; Hormati, A.; Dehnavi, H.M.; Hoseini, F.; Asghari, M. Effects of Iranian Polyherbal Syrup (Zufa syrup) on oxygen saturation and clinical symptoms in suspected patients with COVID-19: A triple-blinded, randomized, placebo-controlled trial. Med. Gas. Res. 2022, 12, 44–50. [Google Scholar] [CrossRef]
- Karimi, M.; Zarei, A.; Soleymani, S.; Jamalimoghadamsiahkali, S.; Asadi, A.; Shati, M.; Jafari, M.; Rezadoost, H.; Kordafshar, G.; Naghizadeh, A.; et al. Efficacy of Persian medicine herbal formulations (capsules and decoction) compared to standard care in patients with COVID-19, a multicenter open-labeled, randomized, controlled clinical trial. Phytother. Res. 2021, 35, 6295–6309. [Google Scholar] [CrossRef]
- Devpura, G.; Tomar, B.S.; Nathiya, D.; Sharma, A.; Bhandari, D.; Haldar, S.; Balkrishna, A.; Varshney, A. Randomized placebo-controlled pilot clinical trial on the efficacy of ayurvedic treatment regime on COVID-19 positive patients. Phytomedicine Int. J. Phytother. Phytopharm. 2021, 84, 153494. [Google Scholar] [CrossRef] [PubMed]
- Margolin, L.; Luchins, J.; Margolin, D.; Margolin, M.; Lefkowitz, S. 20-Week Study of Clinical Outcomes of Over-the-Counter COVID-19 Prophylaxis and Treatment. J. Evid. Based Integr. Med. 2021, 26, 2515690X211026193. [Google Scholar] [CrossRef] [PubMed]
- Vidal, K.; Bucheli, P.; Gao, Q.; Moulin, J.; Shen, L.S.; Wang, J.; Blum, S.; Benyacoub, J. Immunomodulatory effects of dietary supplementation with a milk-based wolfberry formulation in healthy elderly: A randomized, double-blind, placebo-controlled trial. Rejuvenation Res. 2012, 15, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Nantz, M.P.; Rowe, C.A.; Muller, C.E.; Creasy, R.A.; Stanilka, J.M.; Percival, S.S. Supplementation with aged garlic extract improves both NK and γδ-T cell function and reduces the severity of cold and flu symptoms: A randomized, double-blind, placebo-controlled nutrition intervention. Clin. Nutr. 2012, 31, 337–344. [Google Scholar] [CrossRef]
- Nabeshima, S.; Kashiwagi, K.; Ajisaka, K.; Masui, S.; Takeoka, H.; Ikematsu, H.; Kashiwagi, S. A randomized, controlled trial comparing traditional herbal medicine and neuraminidase inhibitors in the treatment of seasonal influenza. J. Infect. Chemother. Off. J. Jpn. Soc. Chemother. 2012, 18, 534–543. [Google Scholar] [CrossRef]
- Lee, C.S.; Lee, J.H.; Oh, M.; Choi, K.M.; Jeong, M.R.; Park, J.D.; Kwon, D.Y.; Ha, K.C.; Park, E.O.; Lee, N.; et al. Preventive effect of Korean red ginseng for acute respiratory illness: A randomized and double-blind clinical trial. J. Korean Med. Sci. 2012, 27, 1472–1478. [Google Scholar] [CrossRef]
- Müller, L.; Meyer, M.; Bauer, R.N.; Zhou, H.; Zhang, H.; Jones, S.; Robinette, C.; Noah, T.L.; Jaspers, I. Effect of Broccoli Sprouts and Live Attenuated Influenza Virus on Peripheral Blood Natural Killer Cells: A Randomized, Double-Blind Study. PLoS ONE 2016, 11, e0147742. [Google Scholar] [CrossRef]
- Noah, T.L.; Zhang, H.; Zhou, H.; Glista-Baker, E.; Müller, L.; Bauer, R.N.; Meyer, M.; Murphy, P.C.; Jones, S.; Letang, B.; et al. Effect of broccoli sprouts on nasal response to live attenuated influenza virus in smokers: A randomized, double-blind study. PLoS ONE 2014, 9, e98671. [Google Scholar] [CrossRef]
- Yao, J.; Zhang, H.; Ma, L.; Mu, X.; Wang, Y.; Lu, Y.; Yu, P.; Dai, H. Effect of traditional Chinese medicine Bupleurum in the treatment of influenza A (H1N1). Pak. J. Pharm. Sci. 2018, 31, 1713–1717. [Google Scholar]
- Macknin, M.; Wolski, K.; Negrey, J.; Mace, S. Elderberry Extract Outpatient Influenza Treatment for Emergency Room Patients Ages 5 and Above: A Randomized, Double-Blind, Placebo-Controlled Trial. J. Gen. Intern. Med. 2020, 35, 3271–3277. [Google Scholar] [CrossRef]
- Vanderbeke, L.; Janssen, N.A.F.; Bergmans, D.; Bourgeois, M.; Buil, J.B.; Debaveye, Y.; Depuydt, P.; Feys, S.; Hermans, G.; Hoiting, O.; et al. Posaconazole for prevention of invasive pulmonary aspergillosis in critically ill influenza patients (POSA-FLU): A randomised, open-label, proof-of-concept trial. Intensive Care Med. 2021, 47, 674–686. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, Z.; Goswami, S.; Paneerselvam, A.; Kabilan, K.; Chowdhury, H.; Roy, A.; Guleria, R.; Soni, K.D.; Baruah, U.; Das, C.J. Imaging of Coronavirus Disease 2019 Infection from Head to Toe: A Primer for the Radiologist. Curr. Probl. Diagn. Radiol. 2021, 50, 842–855. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.P.; Pritam, M.; Pandey, B.; Yadav, T.P. Microstructure, pathophysiology, and potential therapeutics of COVID-19: A comprehensive review. J. Med. Virol. 2021, 93, 275–299. [Google Scholar] [CrossRef] [PubMed]
- Wiersinga, W.J.; Rhodes, A.; Cheng, A.C.; Peacock, S.J.; Prescott, H.C. Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19): A Review. JAMA 2020, 324, 782–793. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Huang, X.; Sun, J.; Xie, T.; Lei, Y.; Muhammad, J.; Li, X.; Zeng, X.; Zhou, F.; Qin, H.; et al. Clinical Characteristics and Immune Injury Mechanisms in 71 Patients with COVID-19. mSphere 2020, 5, e00362-20. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Roche, L.; Mesta, F. Oxidative Stress as Key Player in Severe Acute Respiratory Syndrome Coronavirus (SARS-CoV) Infection. Arch. Med. Res. 2020, 51, 384–387. [Google Scholar] [CrossRef]
- Fajgenbaum, D.C.; June, C.H. Cytokine Storm. N. Engl. J. Med. 2020, 383, 2255–2273. [Google Scholar] [CrossRef]
- Fara, A.; Mitrev, Z.; Rosalia, R.A.; Assas, B.M. Cytokine storm and COVID-19: A chronicle of pro-inflammatory cytokines. Open. Biol. 2020, 10, 200160. [Google Scholar] [CrossRef]
- Gustine, J.N.; Jones, D. Immunopathology of Hyperinflammation in COVID-19. Am. J. Pathol. 2021, 191, 4–17. [Google Scholar] [CrossRef]
- Kim, I.J.; Lee, Y.H.; Khalid, M.M.; Chen, I.P.; Zhang, Y.; Ott, M.; Verdin, E. SARS-CoV-2 protein ORF8 limits expression levels of Spike antigen and facilitates immune evasion of infected host cells. J Biol Chem 2023, 104955. [Google Scholar] [CrossRef]
- Tay, M.Z.; Poh, C.M.; Rénia, L.; MacAry, P.A.; Ng, L.F.P. The trinity of COVID-19: Immunity, inflammation and intervention. Nat. Rev. Immunol. 2020, 20, 363–374. [Google Scholar] [CrossRef] [PubMed]
- Leven, Y.; Bösel, J. Neurological manifestations of COVID-19—An approach to categories of pathology. Neurol. Res. Pract. 2021, 3, 39. [Google Scholar] [CrossRef] [PubMed]
- Feddema, J.J.; Fernald, K.D.S.; Schikan, H.; van de Burgwal, L.H.M. Upscaling vaccine manufacturing capacity—Kkey bottlenecks and lessons learned. Vaccine 2023. ahead of print. [Google Scholar] [CrossRef]
- Li, T.; Fu, J.; Cheng, J.; Elfiky, A.A.; Wei, C.; Fu, J. New progresses on cell surface protein HSPA5/BiP/GRP78 in cancers and COVID-19. Front. Immunol. 2023, 14, 1166680. [Google Scholar] [CrossRef] [PubMed]
- Gaitonde, D.Y.; Moore, F.C.; Morgan, M.K. Influenza: Diagnosis and Treatment. Am. Fam. Physician 2019, 100, 751–758. [Google Scholar] [PubMed]
- Kalil, A.C.; Thomas, P.G. Influenza virus-related critical illness: Pathophysiology and epidemiology. Crit. Care 2019, 23, 258. [Google Scholar] [CrossRef]
- Park, J.E.; Ryu, Y. Transmissibility and severity of influenza virus by subtype. Infect. Genet. Evol. 2018, 65, 288–292. [Google Scholar] [CrossRef]
- Webster, R.G.; Govorkova, E.A. Continuing challenges in influenza. Ann. N. Y. Acad. Sci. 2014, 1323, 115–139. [Google Scholar] [CrossRef]
- Barbachano-Guerrero, A.; Perez, D.R.; Sawyer, S.L. How avian influenza viruses spill over to mammals. Elife 2023, 12, e86051. [Google Scholar] [CrossRef]
- Labella, A.M.; Merel, S.E. Influenza. Med. Clin. N. Am. 2013, 97, 621–645. [Google Scholar] [CrossRef]
- Shim, J.M.; Kim, J.; Tenson, T.; Min, J.-Y.; Kainov, D.E. Influenza Virus Infection, Interferon Response, Viral Counter-Response, and Apoptosis. Viruses 2017, 9, 223. [Google Scholar] [CrossRef] [PubMed]
- Kehagia, E.; Papakyriakopoulou, P.; Valsami, G. Advances in intranasal vaccine delivery: A promising non-invasive route of immunization. Vaccine 2023, 41, 3589–3603. [Google Scholar] [CrossRef] [PubMed]
- Escuret, V.; Terrier, O. Co-infection of the respiratory epithelium, scene of complex functional interactions between viral, bacterial, and human neuraminidases. Front. Microbiol. 2023, 14, 1137336. [Google Scholar] [CrossRef] [PubMed]
- Adhikari, B.; Marasini, B.P.; Rayamajhee, B.; Bhattarai, B.R.; Lamichhane, G.; Khadayat, K.; Adhikari, A.; Khanal, S.; Parajuli, N. Potential roles of medicinal plants for the treatment of viral diseases focusing on COVID-19: A review. Phytother. Res. 2021, 35, 1298–1312. [Google Scholar] [CrossRef]
- Asif, M.; Saleem, M.; Saadullah, M.; Yaseen, H.S.; Al Zarzour, R. COVID-19 and therapy with essential oils having antiviral, anti-inflammatory, and immunomodulatory properties. Inflammopharmacology 2020, 28, 1153–1161. [Google Scholar] [CrossRef]
- Ben-Shabat, S.; Yarmolinsky, L.; Porat, D.; Dahan, A. Antiviral effect of phytochemicals from medicinal plants: Applications and drug delivery strategies. Drug. Deliv. Transl. Res. 2020, 10, 354–367. [Google Scholar] [CrossRef]
- Benarba, B.; Pandiella, A. Medicinal Plants as Sources of Active Molecules Against COVID-19. Front. Pharmacol. 2020, 11, 1189. [Google Scholar] [CrossRef]
- Bhuiyan, F.R.; Howlader, S.; Raihan, T.; Hasan, M. Plants Metabolites: Possibility of Natural Therapeutics Against the COVID-19 Pandemic. Front. Med. 2020, 7, 444. [Google Scholar] [CrossRef]
- Shahzad, F.; Anderson, D.; Najafzadeh, M. The Antiviral, Anti-Inflammatory Effects of Natural Medicinal Herbs and Mushrooms and SARS-CoV-2 Infection. Nutrients 2020, 12, 2573. [Google Scholar] [CrossRef]
- Jalali, A.; Dabaghian, F.; Akbrialiabad, H.; Foroughinia, F.; Zarshenas, M.M. A pharmacology-based comprehensive review on medicinal plants and phytoactive constituents possibly effective in the management of COVID-19. Phytother. Res. 2021, 35, 1925–1938. [Google Scholar] [CrossRef]
- Lim, X.Y.; Teh, B.P.; Tan, T.Y.C. Medicinal Plants in COVID-19: Potential and Limitations. Front. Pharmacol. 2021, 12, 611408. [Google Scholar] [CrossRef] [PubMed]
- Balachandar, V.; Mahalaxmi, I.; Kaavya, J.; Vivekanandhan, G.; Ajithkumar, S.; Arul, N.; Singaravelu, G.; Senthil Kumar, N.; Mohana Dev, S. COVID-19: Emerging protective measures. Eur. Rev. Med. Pharm. Sci. 2020, 24, 3422–3425. [Google Scholar] [CrossRef]
- Khan, S.A.; Al-Balushi, K. Combating COVID-19: The role of drug repurposing and medicinal plants. J. Infect. Public Health 2021, 14, 495–503. [Google Scholar] [CrossRef] [PubMed]
- Wong, L.Y.; Lui, P.Y.; Jin, D.Y. A molecular arms race between host innate antiviral response and emerging human coronaviruses. Virol. Sin. 2016, 31, 12–23. [Google Scholar] [CrossRef]
- Zhang, L.; Lin, D.; Sun, X.; Curth, U.; Drosten, C.; Sauerhering, L.; Becker, S.; Rox, K.; Hilgenfeld, R. Crystal structure of SARS-CoV-2 main protease provides a basis for design of improved α-ketoamide inhibitors. Science 2020, 368, 409–412. [Google Scholar] [CrossRef]
- Xia, H.; Cao, Z.; Xie, X.; Zhang, X.; Chen, J.Y.; Wang, H.; Menachery, V.D.; Rajsbaum, R.; Shi, P.Y. Evasion of Type I Interferon by SARS-CoV-2. Cell. Rep. 2020, 33, 108234. [Google Scholar] [CrossRef]
- Chen, J.; Malone, B.; Llewellyn, E.; Grasso, M.; Shelton, P.M.M.; Olinares, P.D.B.; Maruthi, K.; Eng, E.; Vatandaslar, H.; Chait, B.T.; et al. Structural basis for helicase-polymerase coupling in the SARS-CoV-2 replication-transcription complex. bioRxiv 2020. 2020.2007.2008.194084. [Google Scholar] [CrossRef]
- Habtemariam, S.; Nabavi, S.F.; Banach, M.; Berindan-Neagoe, I.; Sarkar, K.; Sil, P.C.; Nabavi, S.M. Should We Try SARS-CoV-2 Helicase Inhibitors for COVID-19 Therapy? Arch. Med. Res. 2020, 51, 733–735. [Google Scholar] [CrossRef]
- Khare, P.; Sahu, U.; Pandey, S.C.; Samant, M. Current approaches for target-specific drug discovery using natural compounds against SARS-CoV-2 infection. Virus Res. 2020, 290, 198169. [Google Scholar] [CrossRef]
- Romano, M.; Ruggiero, A.; Squeglia, F.; Maga, G.; Berisio, R. A Structural View of SARS-CoV-2 RNA Replication Machinery: RNA Synthesis, Proofreading and Final Capping. Cells 2020, 9, 1267. [Google Scholar] [CrossRef]
- Vandelli, A.; Monti, M.; Milanetti, E.; Armaos, A.; Rupert, J.; Zacco, E.; Bechara, E.; Delli Ponti, R.; Tartaglia, G.G. Structural analysis of SARS-CoV-2 genome and predictions of the human interactome. Nucleic Acids Res. 2020, 48, 11270–11283. [Google Scholar] [CrossRef] [PubMed]
- Alhazmi, H.A.; Najmi, A.; Javed, S.A.; Sultana, S.; Al Bratty, M.; Makeen, H.A.; Meraya, A.M.; Ahsan, W.; Mohan, S.; Taha, M.M.E.; et al. Medicinal Plants and Isolated Molecules Demonstrating Immunomodulation Activity as Potential Alternative Therapies for Viral Diseases Including COVID-19. Front. Immunol. 2021, 12, 637553. [Google Scholar] [CrossRef] [PubMed]
- Khan, T.; Khan, M.A.; Mashwani, Z.U.; Ullah, N.; Nadhman, A. Therapeutic potential of medicinal plants against COVID-19: The role of antiviral medicinal metabolites. Biocatal. Agric. Biotechnol. 2021, 31, 101890. [Google Scholar] [CrossRef]
- Bhardwaj, V.K.; Singh, R.; Sharma, J.; Rajendran, V.; Purohit, R.; Kumar, S. Identification of bioactive molecules from tea plant as SARS-CoV-2 main protease inhibitors. J. Biomol. Struct. Dyn. 2021, 39, 3449–3458. [Google Scholar] [CrossRef]
- Solnier, J.; Fladerer, J.-P. Flavonoids: A complementary approach to conventional therapy of COVID-19? Phytochem. Rev. 2020, 20, 773–795. [Google Scholar] [CrossRef]
- Hafez Ghoran, S.; El-Shazly, M.; Sekeroglu, N.; Kijjoa, A. Natural Products from Medicinal Plants with Anti-Human Coronavirus Activities. Molecules 2021, 26, 1754. [Google Scholar] [CrossRef] [PubMed]
- Sargin, S.A. Potential anti-influenza effective plants used in Turkish folk medicine: A review. J. Ethnopharmacol. 2021, 265, 113319. [Google Scholar] [CrossRef]
- Brochot, A.; Guilbot, A.; Haddioui, L.; Roques, C. Antibacterial, antifungal, and antiviral effects of three essential oil blends. MicrobiologyOpen 2017, 6, e00459. [Google Scholar] [CrossRef]
- Hamauzu, Y.; Yasui, H.; Inno, T.; Kume, C.; Omanyuda, M. Phenolic Profile, Antioxidant Property, and Anti-influenza Viral Activity of Chinese Quince (Pseudocydonia sinensis Schneid.), Quince (Cydonia oblonga Mill.), and Apple (Malus domestica Mill.) Fruits. J. Agric. Food Chem. 2005, 53, 928–934. [Google Scholar] [CrossRef]
- Ding, Y.; Cao, Z.; Cao, L.; Ding, G.; Wang, Z.; Xiao, W. Antiviral activity of chlorogenic acid against influenza A (H1N1/H3N2) virus and its inhibition of neuraminidase. Sci. Rep. 2017, 7, 45723. [Google Scholar] [CrossRef]
- Lee, I.-K.; Hwang, B.S.; Kim, D.-W.; Kim, J.-Y.; Woo, E.E.; Lee, Y.-J.; Choi, H.J.; Yun, B.-S. Characterization of Neuraminidase Inhibitors in Korean Papaver rhoeas Bee Pollen Contributing to Anti-Influenza Activities In Vitro. Planta Med. 2016, 82, 524–529. [Google Scholar] [CrossRef] [PubMed]
- Chandan, S.; Mohan, B.P.; Chandan, O.C.; Ahmad, R.; Challa, A.; Tummala, H.; Singh, S.; Dhawan, P.; Ponnada, S.; Singh, A.B.; et al. Curcumin use in ulcerative colitis: Is it ready for prime time? A systematic review and meta-analysis of clinical trials. Ann. Gastroenterol. 2020, 33, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Fiore, C.; Eisenhut, M.; Krausse, R.; Ragazzi, E.; Pellati, D.; Armanini, D.; Bielenberg, J. Antiviral effects of Glycyrrhiza species. Phytother. Res. 2008, 22, 141–148. [Google Scholar] [CrossRef]
- Hu, Z.; Lin, J.; Chen, J.; Cai, T.; Xia, L.; Liu, Y.; Song, X.; He, Z. Overview of Viral Pneumonia Associated with Influenza Virus, Respiratory Syncytial Virus, and Coronavirus, and Therapeutics Based on Natural Products of Medicinal Plants. Front. Pharmacol. 2021, 12, 630834. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Du, R.; Chen, Z.; Wang, Y.; Zhan, P.; Liu, X.; Kang, D.; Chen, Z.; Zhao, X.; Wang, L.; et al. Punicalagin is a neuraminidase inhibitor of influenza viruses. J. Med. Virol. 2021, 93, 3465–3472. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Jia, W.; Zhao, A.; Wang, X. Anti-influenza agents from plants and traditional Chinese medicine. Phytother. Res. 2006, 20, 335–341. [Google Scholar] [CrossRef]
- Akram, M.; Tahir, I.M.; Shah, S.M.A.; Mahmood, Z.; Altaf, A.; Ahmad, K.; Munir, N.; Daniyal, M.; Nasir, S.; Mehboob, H. Antiviral potential of medicinal plants against HIV, HSV, influenza, hepatitis, and coxsackievirus: A systematic review. Phytother. Res. 2018, 32, 811–822. [Google Scholar] [CrossRef]
- Bahramsoltani, R.; Sodagari, H.R.; Farzaei, M.H.; Abdolghaffari, A.H.; Gooshe, M.; Rezaei, N. The preventive and therapeutic potential of natural polyphenols on influenza. Expert. Rev. Anti Infect. Ther. 2016, 14, 57–80. [Google Scholar] [CrossRef]
- Mehrbod, P.; Abdalla, M.A.; Njoya, E.M.; Ahmed, A.S.; Fotouhi, F.; Farahmand, B.; Gado, D.A.; Tabatabaian, M.; Fasanmi, O.G.; Eloff, J.N.; et al. South African medicinal plant extracts active against influenza A virus. BMC Complement. Altern. Med. 2018, 18, 112. [Google Scholar] [CrossRef]
- Zhang, Z.; Morris-Natschke, S.; Yung-Yi, C.; Lee, K.-H.; Li, R.T. Development of anti-influenza agents from natural products. Med. Res. Rev. 2020, 40, 2290–2338. [Google Scholar] [CrossRef]
- Kültür, S. Medicinal plants used in Kirklareli Province (Turkey). J. Ethnopharmacol. 2007, 111, 341–364. [Google Scholar] [CrossRef] [PubMed]
- Maria John, K.M.; Enkhtaivan, G.; Ayyanar, M.; Jin, K.; Yeon, J.B.; Kim, D.H. Screening of ethnic medicinal plants of South India against influenza (H1N1) and their antioxidant activity. Saudi J. Biol. Sci. 2015, 22, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Mohan, S.; Elhassan Taha, M.M.; Makeen, H.A.; Alhazmi, H.A.; Al Bratty, M.; Sultana, S.; Ahsan, W.; Najmi, A.; Khalid, A. Bioactive Natural Antivirals: An Updated Review of the Available Plants and Isolated Molecules. Molecules 2020, 25, 4878. [Google Scholar] [CrossRef] [PubMed]
- Siew, Y.Y.; Zareisedehizadeh, S.; Seetoh, W.G.; Neo, S.Y.; Tan, C.H.; Koh, H.L. Ethnobotanical survey of usage of fresh medicinal plants in Singapore. J. Ethnopharmacol. 2014, 155, 1450–1466. [Google Scholar] [CrossRef]
- Chen, F.; Yang, L.; Huang, Y.; Chen, Y.; Sang, H.; Duan, W.; Yang, J. Isocorilagin, isolated from Canarium album (Lour.) Raeusch, as a potent neuraminidase inhibitor against influenza A virus. Biochem. Biophys. Res. Commun. 2020, 523, 183–189. [Google Scholar] [CrossRef]
- Wolkerstorfer, A.; Kurz, H.; Bachhofner, N.; Szolar, O.H. Glycyrrhizin inhibits influenza A virus uptake into the cell. Antivir. Res. 2009, 83, 171–178. [Google Scholar] [CrossRef]
- Huang, C.T.; Hung, C.Y.; Hseih, Y.C.; Chang, C.S.; Velu, A.B.; He, Y.C.; Huang, Y.L.; Chen, T.A.; Chen, T.C.; Lin, C.Y.; et al. Effect of aloin on viral neuraminidase and hemagglutinin-specific T cell immunity in acute influenza. Phytomedicine 2019, 64, 152904. [Google Scholar] [CrossRef]
- Yu, S.; Zhu, Y.; Xu, J.; Yao, G.; Zhang, P.; Wang, M.; Zhao, Y.; Lin, G.; Chen, H.; Chen, L.; et al. Glycyrrhizic acid exerts inhibitory activity against the spike protein of SARS-CoV-2. Phytomedicine 2021, 85, 153364. [Google Scholar] [CrossRef]
- Park, J.-Y.; Jeong, H.J.; Kim, J.H.; Kim, Y.M.; Park, S.-J.; Kim, D.; Park, K.H.; Lee, W.S.; Ryu, Y.B. Diarylheptanoids from Alnus japonica Inhibit Papain-Like Protease of Severe Acute Respiratory Syndrome Coronavirus. Biol. Pharm. Bull. 2012, 35, 2036–2042. [Google Scholar] [CrossRef]
- Laurindo, L.F.; de Carvalho, G.M.; de Oliveira Zanuso, B.; Figueira, M.E.; Direito, R.; de Alvares Goulart, R.; Buglio, D.S.; Barbalho, S.M. Curcumin-Based Nanomedicines in the Treatment of Inflammatory and Immunomodulated Diseases: An Evidence-Based Comprehensive Review. Pharmaceutics 2023, 15, 229. [Google Scholar] [CrossRef]
- Prajapati, S.K.; Malaiya, A.; Mishra, G.; Jain, D.; Kesharwani, P.; Mody, N.; Ahmadi, A.; Paliwal, R.; Jain, A. An exhaustive comprehension of the role of herbal medicines in Pre- and Post-COVID manifestations. J. Ethnopharmacol. 2022, 296, 115420. [Google Scholar] [CrossRef] [PubMed]
- Derakhshan, M.A.; Amani, A.; Faridi-Majidi, R. State-of-the-Art of Nanodiagnostics and Nanotherapeutics against SARS-CoV-2. ACS Appl. Mater. Interfaces 2021, 13, 14816–14843. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.K.; Prateeksha; Singh, S.P.; Singh, B.N.; Rao, C.V.; Barik, S.K. Nanocurcumin Potently Inhibits SARS-CoV-2 Spike Protein-Induced Cytokine Storm by Deactivation of MAPK/NF-κB Signaling in Epithelial Cells. ACS Appl. Bio Mater. 2022, 5, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Pourhajibagher, M.; Azimi, M.; Haddadi-Asl, V.; Ahmadi, H.; Gholamzad, M.; Ghorbanpour, S.; Bahador, A. Robust antimicrobial photodynamic therapy with curcumin-poly (lactic-co-glycolic acid) nanoparticles against COVID-19: A preliminary in vitro study in Vero cell line as a model. Photodiagnosis Photodyn. Ther. 2021, 34, 102286. [Google Scholar] [CrossRef]
- AbouAitah, K.; Allayh, A.K.; Wojnarowicz, J.; Shaker, Y.M.; Swiderska-Sroda, A.; Lojkowski, W. Nanoformulation Composed of Ellagic Acid and Functionalized Zinc Oxide Nanoparticles Inactivates DNA and RNA Viruses. Pharmaceutics 2021, 13, 2174. [Google Scholar] [CrossRef]
- Pilaquinga, F.; Bosch, R.; Morey, J.; Bastidas-Caldes, C.; Torres, M.; Toscano, F.; Debut, A.; Pazmiño-Viteri, K.; Nieves Piña, M.L. Highin vitroactivity of gold and silver nanoparticles from Solanum mammosum L. against SARS-CoV-2 surrogate Phi6 and viral model PhiX174. Nanotechnology 2023, 34, 175705. [Google Scholar] [CrossRef]
- Loutfy, S.A.; Abdel-Salam, A.I.; Moatasim, Y.; Gomaa, M.R.; Abdel Fattah, N.F.; Emam, M.H.; Ali, F.; ElShehaby, H.A.; Ragab, E.A.; Alam El-Din, H.M.; et al. Antiviral activity of chitosan nanoparticles encapsulating silymarin (Sil-CNPs) against SARS-CoV-2 (in silico and in vitro study). RSC Adv. 2022, 12, 15775–15786. [Google Scholar] [CrossRef]
- Saadh, M.J.; Aldalaen, S.M. Inhibitory effects of epigallocatechin gallate (EGCG) combined with zinc sulfate and silver nanoparticles on avian influenza A virus subtype H5N1. Eur. Rev. Med. Pharm. Sci. 2021, 25, 2630–2636. [Google Scholar] [CrossRef]
- Saadh, M.J.; Aggag, M.M.; Alboghdadly, A.; Kharshid, A.M.; Aldalaen, S.M.; Abdelrazek, M.A. Silver nanoparticles with epigallocatechingallate and zinc sulphate significantly inhibits avian influenza A virus H9N2. Microb. Pathog. 2021, 158, 105071. [Google Scholar] [CrossRef]
- Higgins, J.P.; Thomas, J.; Chandler, J.; Cumpston, M.; Li, T.; Page, M.J.; Welch, V.A. Cochrane Handbook for Systematic Reviews of Interventions; John Wiley & Sons: New York, NY, USA, 2019. [Google Scholar]

| Reference | Local | Model and Patients | Intervention | Outcomes | Adverse Effects |
|---|---|---|---|---|---|
| COVID-19 | |||||
| [36] | Iran | Placebo-controlled randomized, double-blind clinical trial with 108 COVID-19 outpatients, 55 male; 53 female; ≥30 y. | 10 mL of violet syrup (Viola odorata L. aqueous extract) thrice daily for 7 days. | Patients who received violet syrup ameliorated faster and had lower mean severity scores regarding cough, myalgia, headache, and diarrhea. This shows that violet syrup effectively controlled prevalent COVID-19 manifestations. | No serious adverse events were reported during the trial. |
| [37] | Iran | Placebo-controlled randomized, single-blind clinical trial with 178 COVID-19 outpatients, 120 male; 58 female; ≤60 y. | 200 mL of pomegranate juice thrice daily and 1.5 g of SUMAC (composed of tannins, flavonoids, anthocyanins, isoflavones, terpenoids, and diterpenes) twice daily. Time of intervention was not elucidated. | Patents who received pomegranate and SUMAC interventions had significant decreases in fever, weakness, cough, chills, smell and taste disorders, diarrhea, shortness of breath, abdominal pain, and vomiting compared with patients who did not undergo this treatment. | Not reported. |
| [38] | Iran | Placebo-controlled randomized, double-blind clinical trial with 50 mild to moderate COVID-19 patients, 34 male; 16 female; ≤80 y. | Covexir (Ferula foetida oleo-gum) twice daily for 7 days. | Covexir inhibited cough and diminished the severity of anorexia, sense of taste, anosmia, and myalgia between the intervention and placebo groups. | No adverse events were reported during the trial. |
| [39] | Iran | Placebo-controlled randomized triple-blind clinical trial with 116 mild to moderate COVID-19, 57 men; 59 females; 20–70 y. | 7.5 mL of Zufa syrup (Nepeta bracteata, Ziziphus jujube, Glycyrrhizaglabra, Ficuscarica, Cordia myxa, Papaver somniferum, Fennel, Adiantumcapillus veneris, Viola, Viper’s-buglosses, Lavender, and Iris) every 4 h for 10 days. | There were no significant differences between placebo and intervention groups in cough, dyspnea, anxiety, anorexia, insomnia, myalgia, and oxygen saturation decline occurrence. | No serious adverse effects were reported during the trial. |
| [40] | Iran | Multicenter open-labeled, randomized, controlled clinical trial with 358 mild to moderate COVID-19 patients, 197 male; 161 female; ≤75 y. | Patients received a polyherbal decoction (one sachet of the following per day respecting the order: Matricaria chamomilla L., Zataria multiflora Boiss., Glycyrrhiza glabra L., Ziziphus jujuba Mill., Ficus carica L., Urtica dioica L., Althaea officinalis L., and Nepeta bracteata Benth) every 8 h and two herbal capsules (Rheum palmatum L. rizhome, Glycyrrhiza glabra root, Punica granatum L. fruit peel, and Rheum palmatum for capsule one and Nigella sativa L. for capsule two) every 12 h for 7 days. | Patients who received the intervention had significantly lower dyspnea periods, as well as accelerated clinical improvement of dry cough, headache, muscle pain, vertigo chills, fatigue, anorexia, sputum cough, and runny nose. | Gastrointestinal adverse effects like nausea and diarrhea were observed. |
| [41] | India | Placebo-controlled randomized, double-blind pilot clinical trial with 95 patients who had no or mild symptoms of COVID-19 and were positive on RT-PCR, 77 males; 18 females; 15- 80 y. | 1 g of giloy, 2 g of swasari ras, 0.5 g each of ashwagandha, and tulsi ghanvati were given orally to the patients in the treatment group twice per day for 7 days. | Ayurvedic treatment can expedite virological clearance, help rapid recovery, and reduce the risk of viral dissemination and inflammation markers (suggesting a lower severity of SARS-CoV-2 infection in the treated group). | There were no side effects. |
| [42] | USA | Controlled clinical trial with 114 multiply exposed adults, 60 female, 40 males; ≥30 y. | Patients in the treatment group received a daily dose of OTC for 20 weeks, while the control group did not receive any placebo as they refused the study regimen. | Just under 4% of the compliant test group presented flu-like symptoms, but none of the test group was COVID-positive; whereas 20% of the non-compliant control group presented flu-like symptoms, three-quarters of whom (15% of the overall control group) were COVID-positive. | Not reported. |
| Influenza | |||||
| [43] | China | Randomized, double-blinded, placebo-controlled study with 150 healthy community-dwelling Chinese elderly, 75 male and 75 female; 65–70 y. | The treated group received a single-dose sachet containing 13.7 g/day lacto-wolfberry (wolfberry fruit (530 mg/gram), bovine skimmed milk (290 mg/gram), and maltodextrin, 180 mg/g). The placebo group received the same sachet but with a skimmed bovine formulation of milk, maltodextrin, sucrose, and colorants/92 d. | The treated subjects showed significantly higher immunoglobulin G levels post-vaccination and in seroconversion (between days 30 and 90, compared with the placebo group). | No serious adverse effects were reported during the trial. |
| [44] | USA | Randomized, double-blind, placebo-controlled with 120 healthy men (55) and women (65), 21–50 y with BMI 18–30 kg/m2. | Each patient consumed four capsules/day of either AGE (2.56 g per day) or a placebo for 90 days. | The use of aged garlic extract enhanced immune cell function (possibly responsible for the reduction of cold and flu severity.) | Not reported. |
| [45] | Japan | Open-labeled, randomized controlled trial with 33 patients, 14 male; 14 female; 20–64 y, presented within 48 h of onset of flu symptoms, including fever, and were positive by quick diagnostic test kit for influenza virus antigens from nasal swabs. | Patients were randomized into three groups to receive Maoto orally at 2.5 g TID, or Oseltamivir orally 75 mg BID, or Zanamivir by inhalation of 20 mg BID for 5 days. | The administration of oral Maoto granules to healthy adults with seasonal influenza was well tolerated and associated with equivalent clinical and virological efficacy to neuraminidase inhibitors. | One patient in the Maoto group and one in the Oseltamivir group showed a mildly elevated serum aminotransferase level after treatment. |
| [46] | Korea | Randomized, double-blinded, placebo-controlled trial in 100 healthy volunteers, 38 male; 62 female, 30–70 y. | The treatment group received concentrated red ginseng 1.0 g three times a day (3.0 g/day)/12 w. Placebo was similar in taste and appearance but with no principal ingredients. | KRG is effective in protecting subjects against ARI and may decrease the duration and scores of ARI symptoms. | There were no specific clinical and laboratory side effects. |
| [48] | USA | A randomized, double-blind, placebo-controlled trial with 69 healthy young adult smokers and nonsmokers, 26 female; 25 male; 18–40 y. | Smokers and nonsmokers ingested one daily dose 200 g of BSH or placebo (ASH) for 4 days. On day 0 they received a standard vaccine dose of LAIV intranasally. | In smokers, short-term intake of BSH appears to significantly reduce some markers of inflammation, such as IL-6, and reduce the amount of the influenza virus. | No patients reported intolerable taste or side effects. |
| [47] | USA | Randomized, double- blinded, placebo-controlled study with 42 healthy volunteers, 19 female; 10 male; 25–28 y. | Subjects received BSH or placebo (ASH) for 4 consecutive days. A daily portion of BSH shake was about 200 g. On day 0 they received a standard vaccine dose of LAIV intranasally. | BSH increases virus-induced peripheral blood NK cell granzyme B production, an effect that may be important for enhanced antiviral defense responses. | No subject reported intolerable taste or side effects. |
| [49] | China | Randomized clinical trial with 120 subjects who have mild influenza A (H1N1). Including 62 males and 58 females, 14–65 y. | The treated group received chima qingwen decoction two times a day for 5 days (children received half the dose). The antiviral group took oral Oseltamivir every 75 mg (50 mg children), two times a day, one course of 5 days. | The overall effective rate was 93.3%. A combination of therapy (Chinese and Western medicine) is effective for mild cases of influenza A (H1N1). | No adverse effects occurred. |
| [50] | USA | Randomized, double-blind, placebo-controlled trial with eighty-seven patients, ≥4, with less than 48 h of at least two moderate-severity symptoms of influenza and positive polymerase chain reaction influenza test, 49 males; 38 females. | Participants from age 5 to 12 y received a placebo or 15 mL (5.7 g) elderberry extract orally 2× d for 5 d; those > 12 years received 15 mL 4× d for 5 d. Patients were permitted to choose to also receive the standard dosage of Oseltamivir. | No evidence was found that the elderberry benefits the duration or severity of the flu. | Dry mouth, constipation, rash, and bad taste. There were no significant differences between the elderberry and placebo. |
| [51] | Belgium, The Netherlands, and France | Randomized, open-label, proof-of-concept trial with 88 critically ill influenza patients, 41 male; 32 female, ≥18 y. | Participants submitted to the prophylaxis arm received the first dose of POS prophylaxis within 48 h of admission to the ICU, starting with a loading dose of 300 mg 2× d on day 1, followed by a 1× d of 300 mg from day 2 onwards for 7 days. The other group received the standard of care only. | The higher-than-expected incidence of early IAPA precluded any definitive conclusions about POS prophylaxis. After 48 h, still, 11% of patients developed IAPA. | Not reported. |
| Study | Question Focus | Appropriate Randomization | Allocation Blinding | Double-Blind | Losses (<20%) | Prognostics or Demographic Characteristics | Outcomes | Intention to Treat Analysis | Sample Calculation | Adequate Follow-Up |
|---|---|---|---|---|---|---|---|---|---|---|
| [36] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [37] | Yes | No | Yes | No | Yes | No | Yes | No | Yes | NR |
| [38] | Yes | No | Yes | Yes | No | Yes | Yes | No | No | Yes |
| [39] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | No | Yes | Yes |
| [40] | Yes | Yes | No | No | Yes | Yes | Yes | Yes | Yes | Yes |
| [41] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | N | No | Yes |
| [42] | Yes | No | No | No | Yes | NR | Yes | No | Yes | Yes |
| [43] | Yes | Yes | Yes | Yes | Yes | NR | Yes | Yes | Yes | Yes |
| [44] | Yes | Yes | Yes | Yes | Yes | NR | Yes | Yes | NR | Yes |
| [45] | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes | Yes |
| [46] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | NR | Yes |
| [48] | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes |
| [47] | Yes | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | Yes |
| [49] | Yes | NR | NR | NR | NR | Yes | Yes | NR | Yes | Yes |
| [50] | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| [51] | Yes | Yes | Yes | No | Yes | Yes | Yes | Yes | No | Yes |
| Bioactive Compound | Molecular Structure | Antiviral Function (s) | References |
|---|---|---|---|
| Bioactive Compounds of Medicinal Plants That Mainly Affect SARS-CoV-2 | |||
| Casticin | 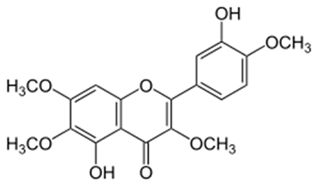 |
| [82] |
| Emodin |  |
| [77,78,89] |
| Dieckol | 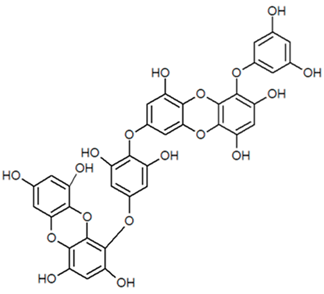 |
| [77] |
| Curcumin |  |
| [78,92] |
| Myricetin |  |
| [77,89] |
| Hirsutenone | 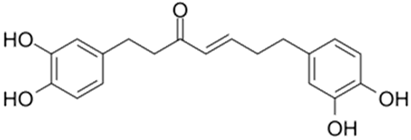 |
| [77] |
| Scutellarein | 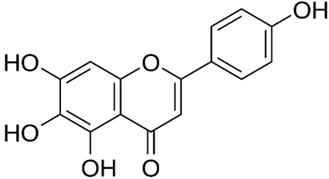 |
| [77,89] |
| Quercetin | 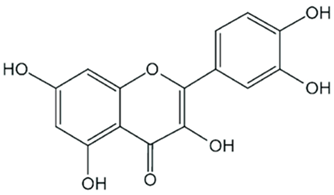 |
| [77,78,92,93] |
| Sulforaphane |  |
| [77] |
| Resveratrol |  |
| [92] |
| Ginsenoside | 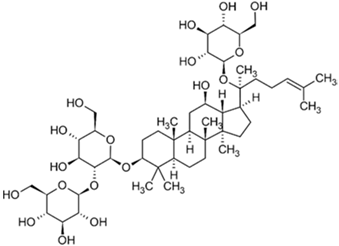 |
| [92] |
| Kaempferol | 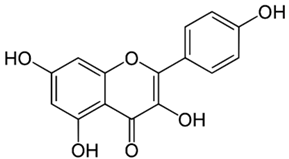 |
| [77,93] |
| Caffeic Acid | 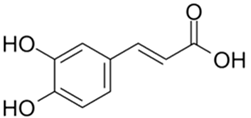 |
| [77,78] |
| Desmethoxyreserpine | --- |
| [78,94] |
| Luteolin | 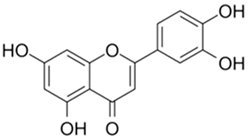 |
| [77,78,89,93] |
| Betulinic Acid |  |
| [78] |
| Xanthoangelol E |  |
| [95] |
| Hyoscyamine |  |
| [82] |
| Cryptotanshinone | 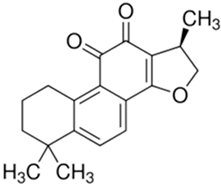 |
| [77,78] |
| Allicin | 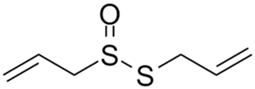 |
| [82,92] |
| Dihydrotanshinone-1 |  |
| [77,78,94] |
| Tartaric Acid | 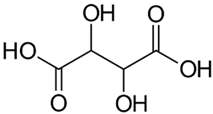 |
| [82] |
| Andrographolide | 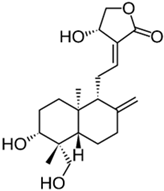 |
| [80] |
| Amentoflavone |  |
| [77] |
| Nicotianamine |  |
| [89] |
| Glycyrrhizin | 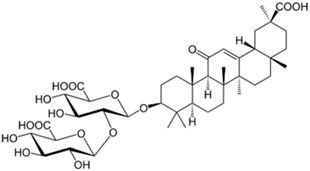 |
| [89] |
| Bilobetin | 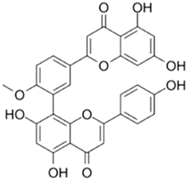 |
| [96] |
| Sciadopitysin | 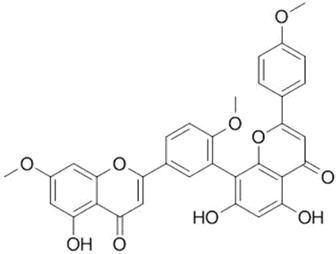 |
| [96] |
| Bioactive Compounds of Medicinal Plants That Mainly Affect Influenza. | |||
| Allicin |  |
| [82,92,97] |
| Eucalyptol |  |
| [75,98] |
| Pentagalloylglucose(Polyphenol) |  |
| [74] |
| Coumarin | 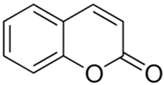 |
| [74] |
| Ellagic Acid | 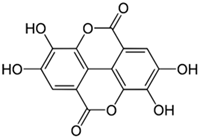 |
| [82] |
| Chlorogenic Acid | 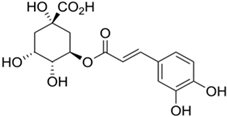 |
| [97,99,100] |
| Isoquercetin | 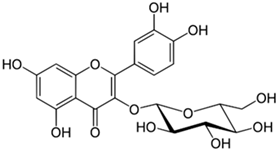 |
| [97] |
| Luteolin |  |
| [97,101,102] |
| Glycyrrhizin | 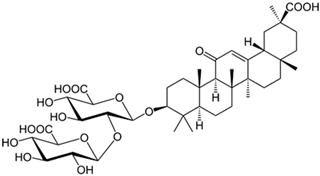 |
| [103] |
| Glycyrrhizin Acid | 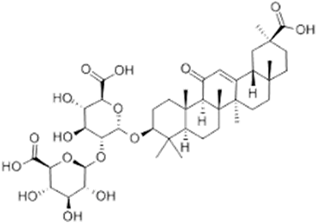 |
| [103] |
| Aloin |  |
| [104] |
| Quercetin-3-sophoroside | 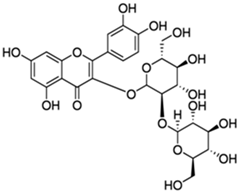 |
| [97,101] |
| 4-Methoxycinnamaldehyde | 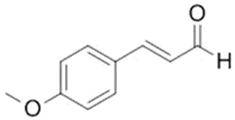 |
| [104] |
| Punicalagin |  |
| [105] |
| Agathisflavone | 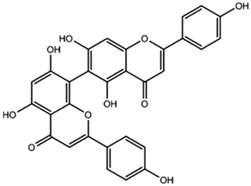 |
| [97] |
| Guggulsterone |  |
| [104] |
| Chinonin |  |
| [106] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fornari Laurindo, L.; Taynara Marton, L.; Minniti, G.; Dogani Rodrigues, V.; Buzinaro Suzuki, R.; Maria Cavallari Strozze Catharin, V.; Joshi, R.K.; Barbalho, S.M. Exploring the Impact of Herbal Therapies on COVID-19 and Influenza: Investigating Novel Delivery Mechanisms for Emerging Interventions. Biologics 2023, 3, 158-186. https://doi.org/10.3390/biologics3030009
Fornari Laurindo L, Taynara Marton L, Minniti G, Dogani Rodrigues V, Buzinaro Suzuki R, Maria Cavallari Strozze Catharin V, Joshi RK, Barbalho SM. Exploring the Impact of Herbal Therapies on COVID-19 and Influenza: Investigating Novel Delivery Mechanisms for Emerging Interventions. Biologics. 2023; 3(3):158-186. https://doi.org/10.3390/biologics3030009
Chicago/Turabian StyleFornari Laurindo, Lucas, Ledyane Taynara Marton, Giulia Minniti, Victória Dogani Rodrigues, Rodrigo Buzinaro Suzuki, Virgínia Maria Cavallari Strozze Catharin, Rakesh Kumar Joshi, and Sandra Maria Barbalho. 2023. "Exploring the Impact of Herbal Therapies on COVID-19 and Influenza: Investigating Novel Delivery Mechanisms for Emerging Interventions" Biologics 3, no. 3: 158-186. https://doi.org/10.3390/biologics3030009
APA StyleFornari Laurindo, L., Taynara Marton, L., Minniti, G., Dogani Rodrigues, V., Buzinaro Suzuki, R., Maria Cavallari Strozze Catharin, V., Joshi, R. K., & Barbalho, S. M. (2023). Exploring the Impact of Herbal Therapies on COVID-19 and Influenza: Investigating Novel Delivery Mechanisms for Emerging Interventions. Biologics, 3(3), 158-186. https://doi.org/10.3390/biologics3030009








