Significance of Features from Biomedical Signals in Heart Health Monitoring
Abstract
1. Introduction
2. Background Information
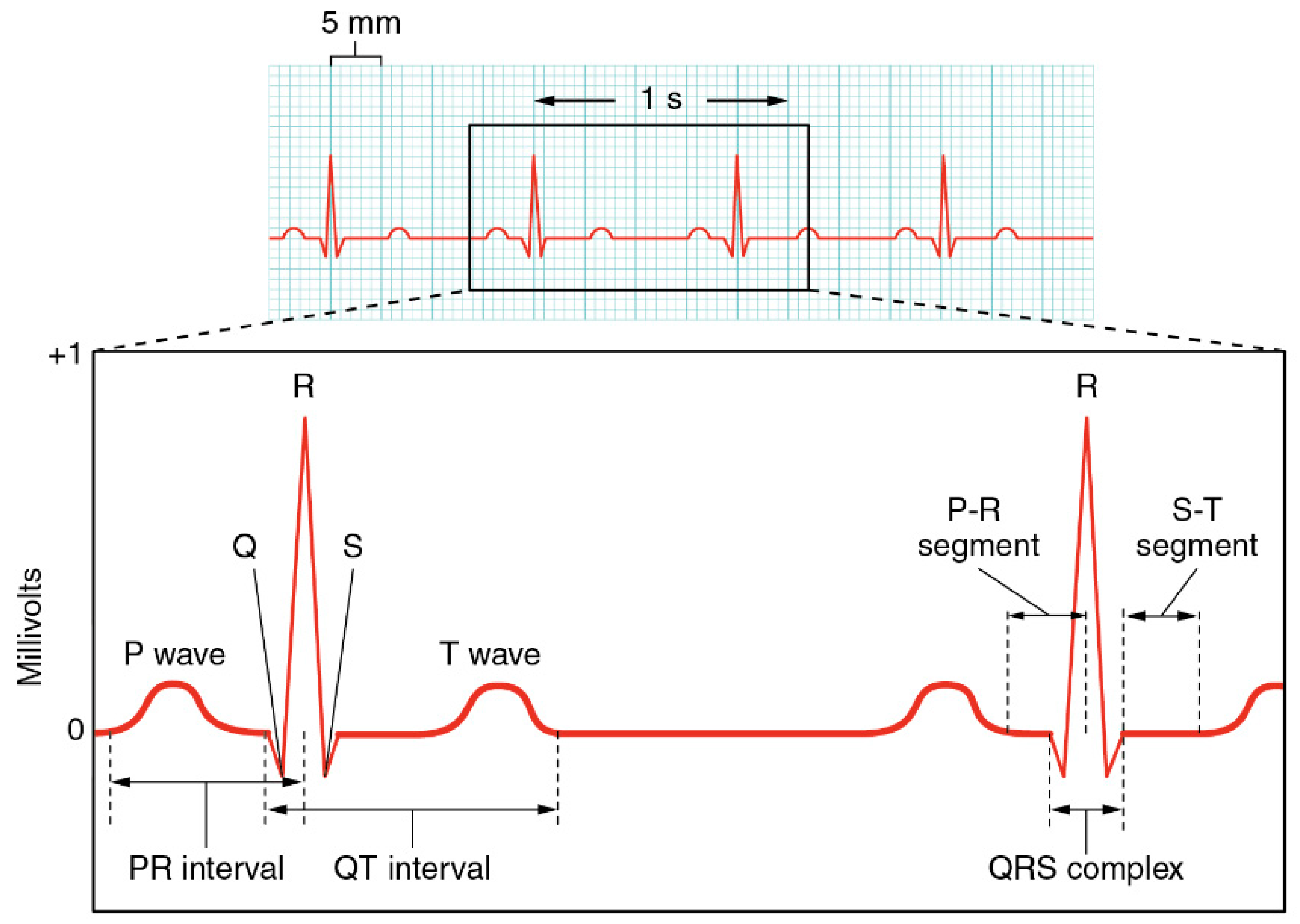
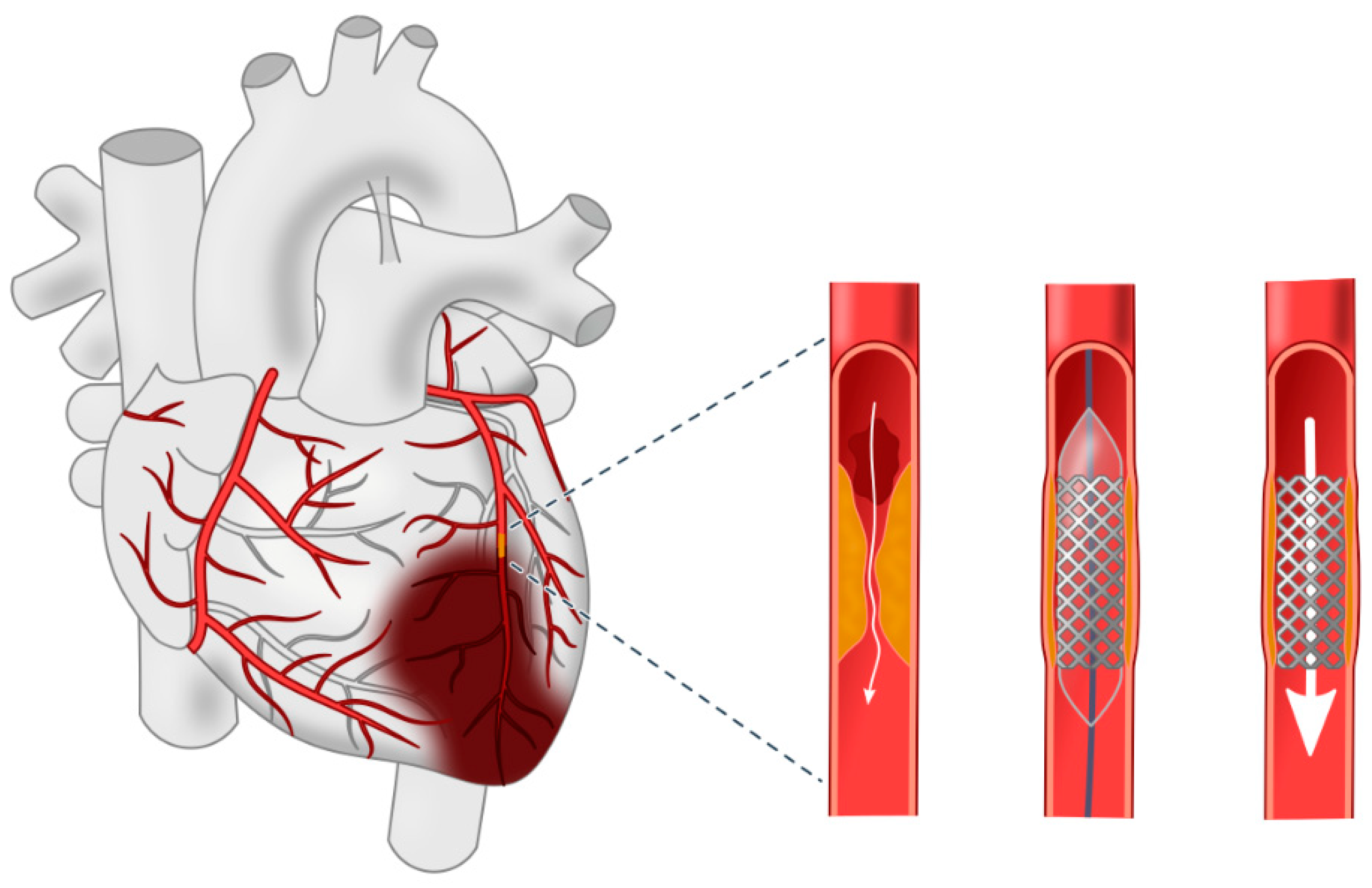
2.1. Functioning of Heart
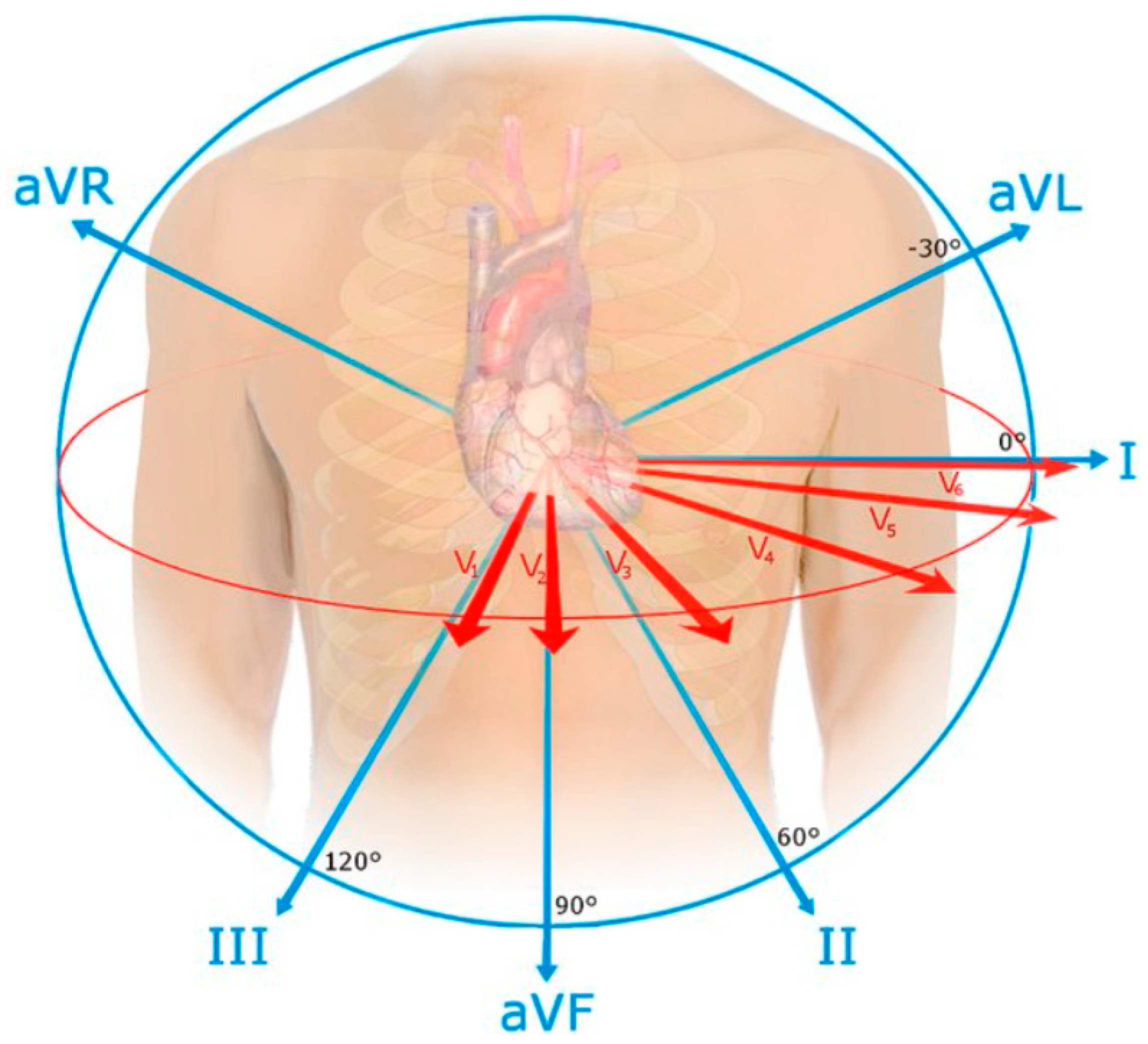
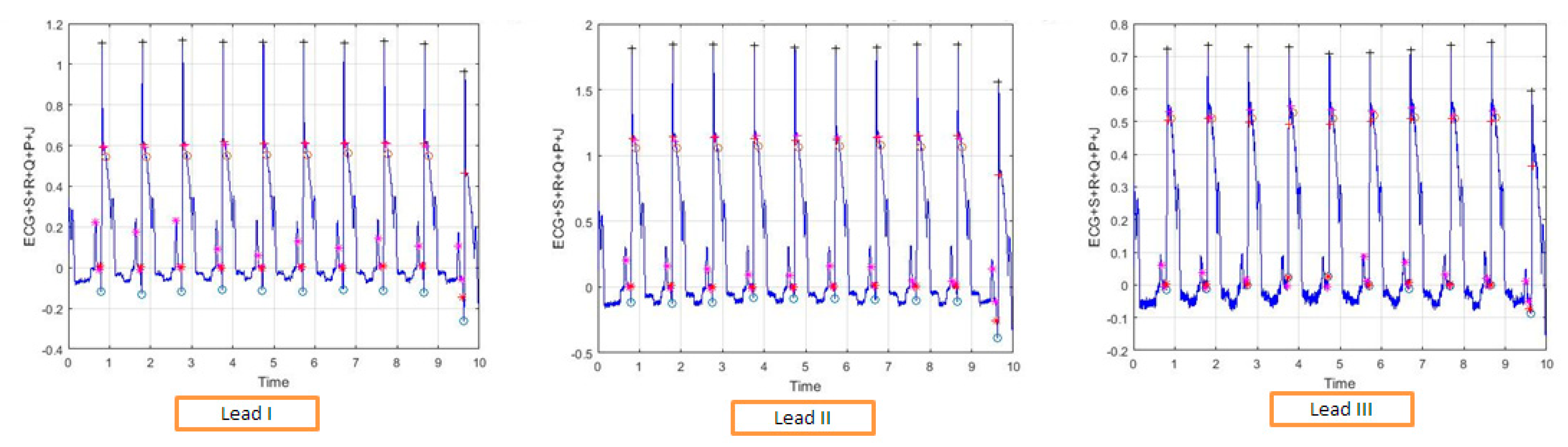
2.2. ECG Leads
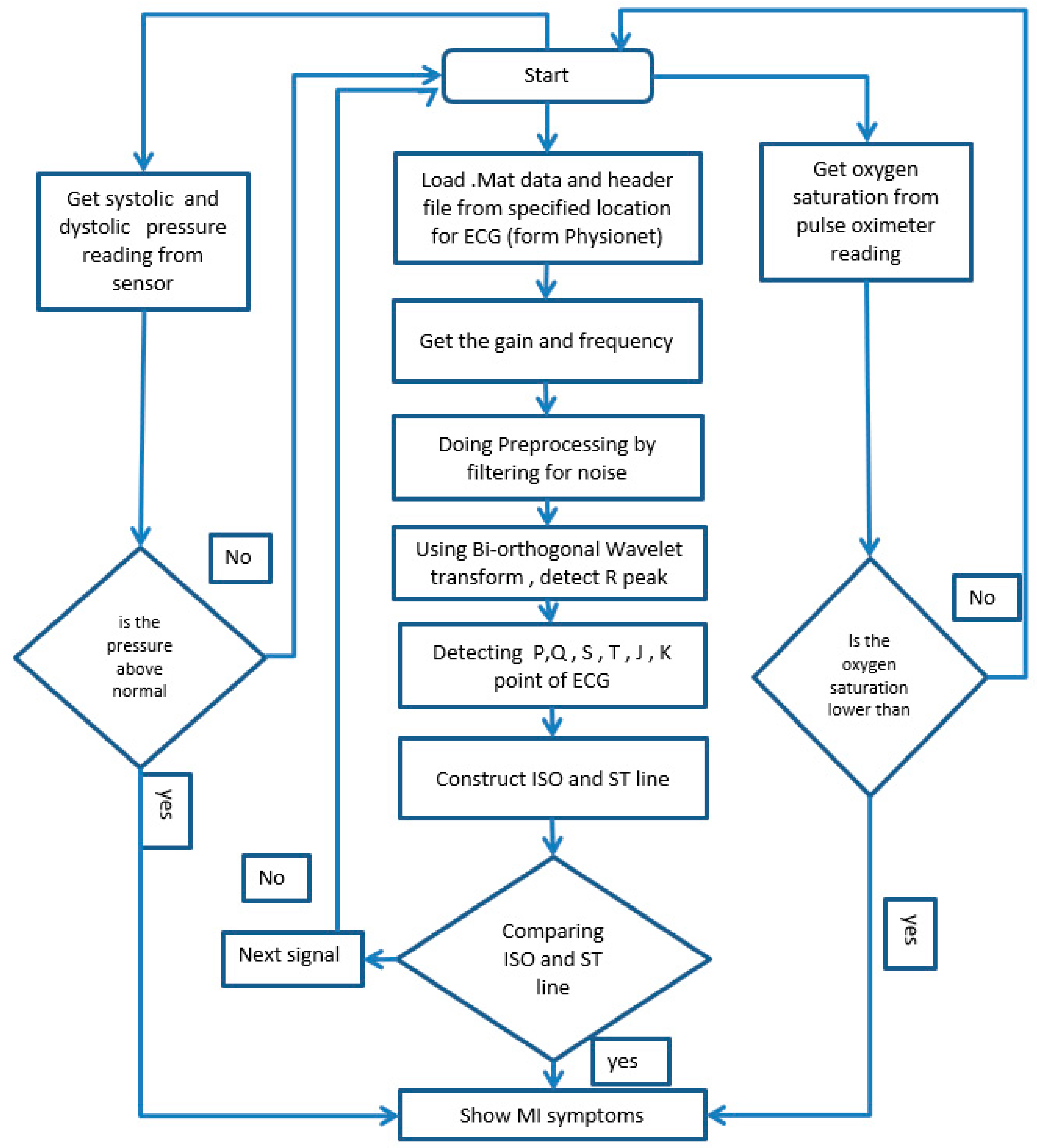
3. Materials and Methods
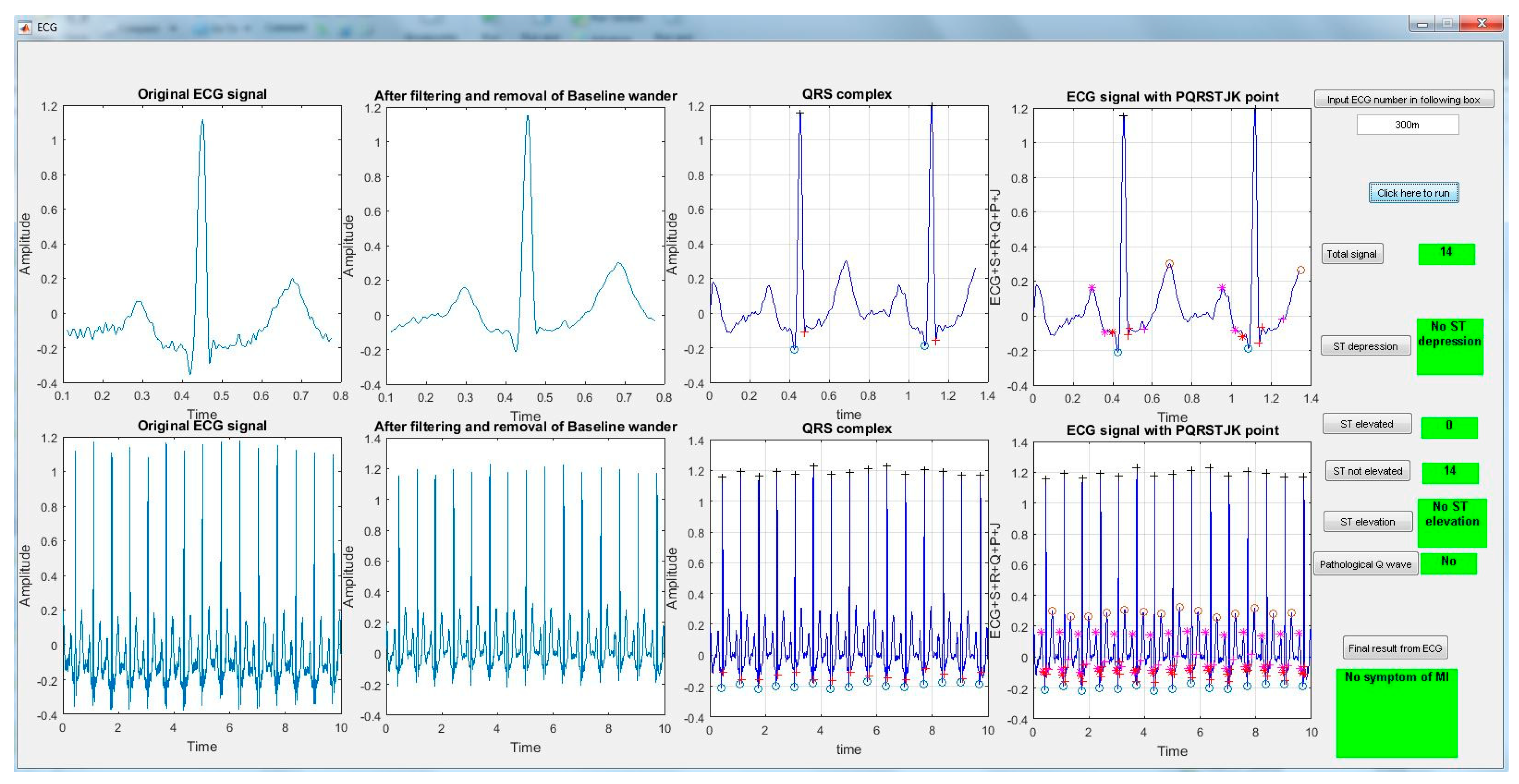
3.1. Dataset and Preprocessing
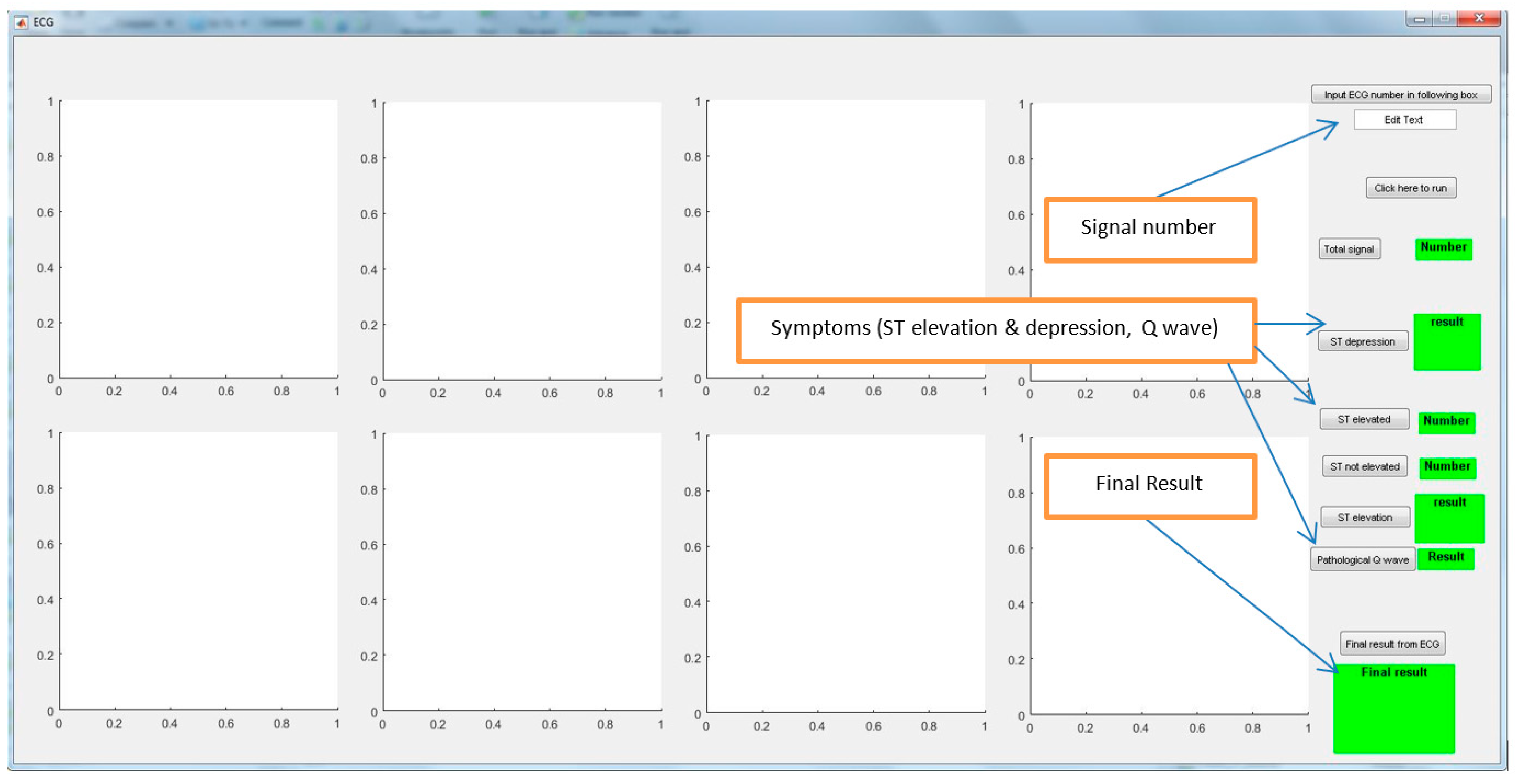
3.2. Decision from the Sensor
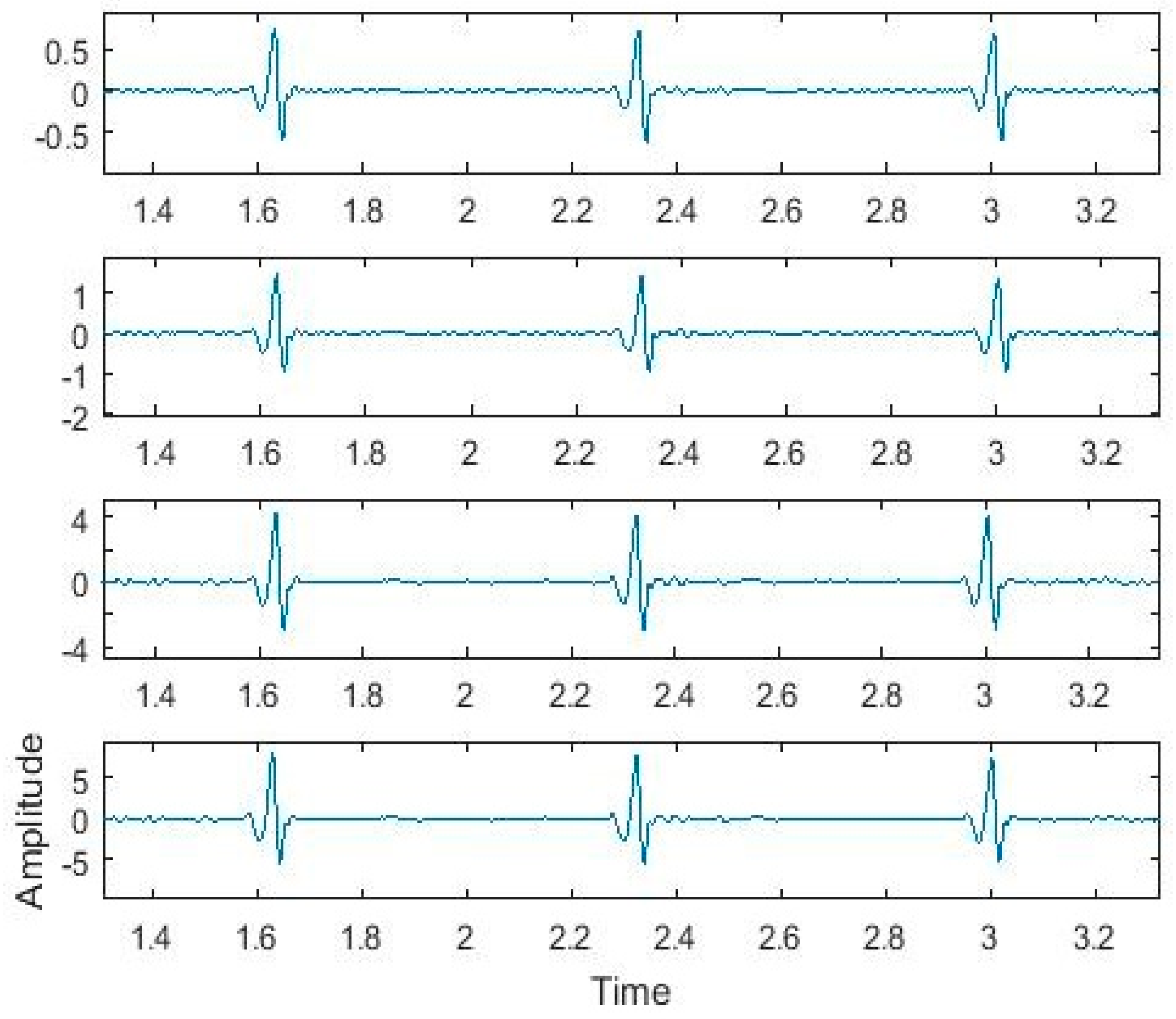
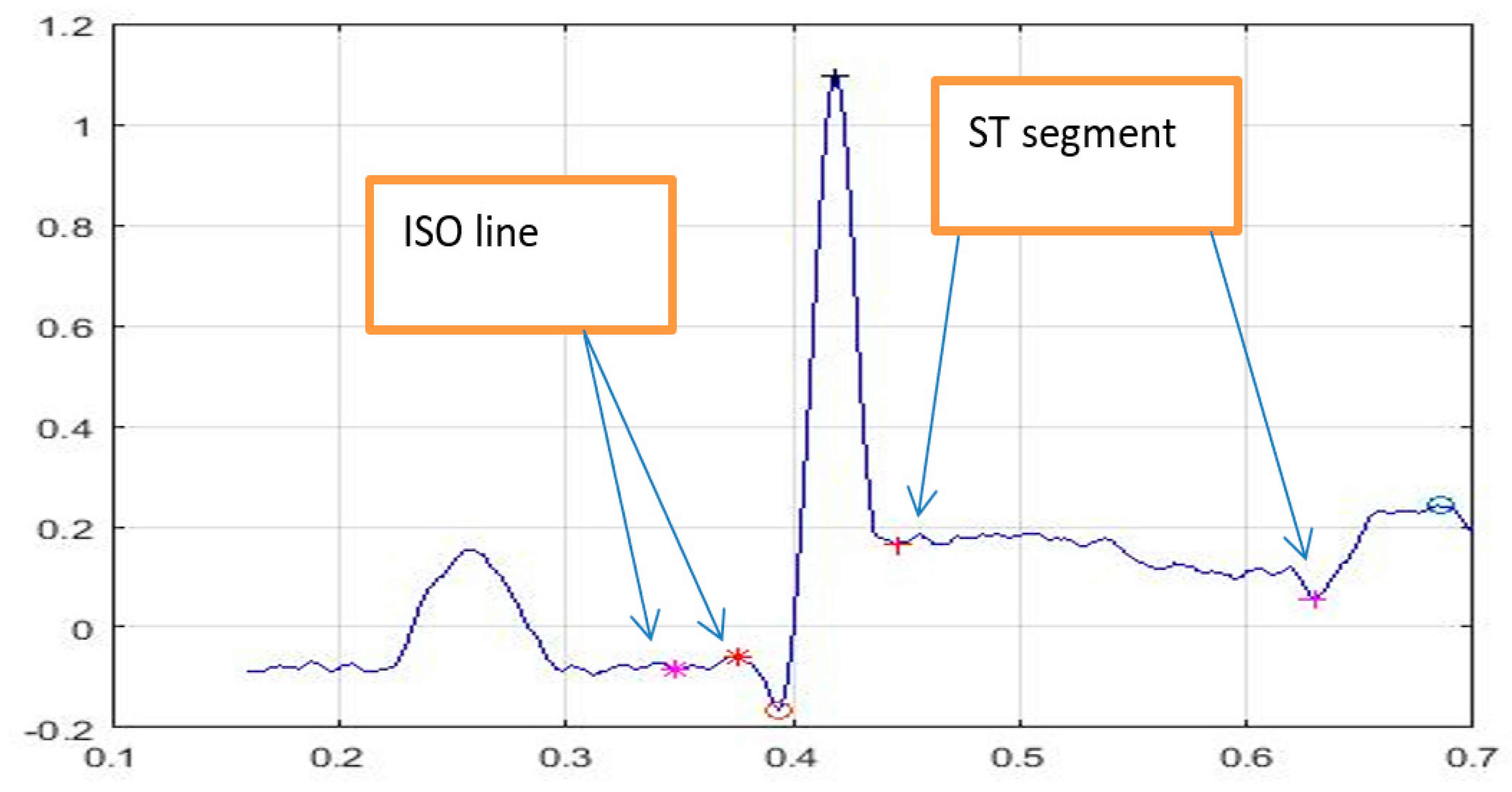
4. Biomedical Data Analysis
5. Performance Evaluation
6. Discussion
7. Conclusions and Future Work
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- De Chazal, P.; O’Dwyer, M.; Reilly, R.B. Automatic classification of heartbeats using ECG morphology and heartbeat interval features. IEEE Trans. Biomed. Eng. 2004, 51, 1196–1206. [Google Scholar] [CrossRef] [PubMed]
- Palaniappan, R.; Krishnan, S.M. Detection of ectopic heart beats using ECG and blood pressure signals. In Proceedings of the 2004 International Conference on Signal Processing and Communications, SPCOM’04, Beijing, China, 31 August–4 September 2004; pp. 573–576. [Google Scholar]
- Lovell, N.H.; Magrabi, F.; Celler, B.G.; Huynh, K.; Garsden, H. Web-Based acquisition, storage, and retrieval of biomedical signals. IEEE Eng. Med. Biol. Mag. 2001, 20, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Asif, I.; Zhao, X. Cloud-ECG for real time ECG monitoring and analysis. Comput. Methods Programs Biomed. 2013, 110, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Mamun, K.; Rahman, M.M.; Alouani, A. Automatic detection of heart diseases using biomedical signals: A literature review of current status and limitations. In Proceedings of the Future of Information and Communication Conference, San Francisco, CA, USA, 3–4 March 2022; pp. 420–440. [Google Scholar]
- Wiharto, W.; Kusnanto, H.; Herianto, H. Intelligence system for diagnosis level of coronary heart disease with K-star algorithm. Healthc. Inform. Res. 2016, 22, 30–38. [Google Scholar] [CrossRef]
- Bashir, S.; Qamar, U.; Khan, F.H. A multicriteria weighted vote-based classifier ensemble for heart disease prediction. Comput. Intell. 2016, 32, 615–645. [Google Scholar] [CrossRef]
- Daraei, A.; Hamidi, H. An Efficient Predictive Model for Myocardial Infarction Using Cost-sensitive J48 Model. Iran. J. Public. Health 2017, 46, 682–692. [Google Scholar]
- Dutta, A.; Batabyal, T.; Basu, M. An efficient convolutional neural network for coronary heart disease prediction. Expert Syst. Appl. 2020, 159, 113408. [Google Scholar] [CrossRef]
- Li, Y.; He, Z.; Wang, H. CraftNet: A deep learning ensemble to diagnose cardiovascular diseases. Biomed. Signal Process. Control 2020, 62, 102091. [Google Scholar] [CrossRef]
- Ahsan, M.M.; Nazim, R.; Siddique, Z.; Huebner, P. Detection of COVID-19 patients from CT scan and chest X-ray data using modified MobileNetV2 and LIME. Healthcare 2021, 9, 1099. [Google Scholar] [CrossRef]
- Ahsan, M.M.; Ahad, M.T.; Soma, F.A. Detecting SARS-CoV-2 from chest X-Ray using artificial intelligence. IEEE Access 2021, 9, 35501–35513. [Google Scholar] [CrossRef]
- Verma, S.; Gupta, A. Effective prediction of heart disease using data mining and machine learning: A review. In Proceedings of the 2021 International Conference on Artificial Intelligence and Smart Systems (ICAIS), Coimbatore, India, 25–27 March 2021; pp. 249–253. [Google Scholar]
- Hoodbhoy, Z.; Jiwani, U.; Sattar, S.; Salam, R.; Hasan, B.; Das, J.K. Diagnostic accuracy of machine learning models to identify congenital heart disease: A meta-analysis. Front. Artif. Intell. 2021, 4, 97. [Google Scholar] [CrossRef] [PubMed]
- Rath, A.; Mishra, D.; Panda, G.; Satapathy, S.C. An exhaustive review of machine and deep learning based diagnosis of heart diseases. Multimed. Tools Appl. 2021, 81, 36069–36127. [Google Scholar] [CrossRef]
- Benhar, H.; Idri, A.; Fernández-Alemán, J. Data preprocessing for heart disease classification: A systematic literature review. Comput. Methods Programs Biomed. 2020, 195, 105635. [Google Scholar] [CrossRef] [PubMed]
- Thakor, N.V.; Zhu, Y. Applications of adaptive filtering to ECG analysis: Noise cancellation and arrhythmia detection. IEEE Trans. Biomed. Eng. 1991, 38, 785–794. [Google Scholar] [CrossRef]
- Axelsson, M.; Dang, Q.; Pitsillides, K.; Munns, S.; Hicks, J.; Kassab, G.S. A novel, fully implantable, multichannel biotelemetry system for measurement of blood flow, pressure, ECG, and temperature. J. Appl. Physiol. 2007, 102, 1220–1228. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.H.; Rautaharju, P.M.; Calhoun, H.P. Selection of a reduced set of parameters for classification of ventricular conduction defects by cluster analysis. In Proceedings of the Computers in Cardiology Conference, London, UK, 5–8 September 1993; pp. 879–882. [Google Scholar]
- Herrero, G.G.; Gotchev, A.; Christov, I.; Egiazarian, K. Feature extraction for heartbeat classification using independent component analysis and matching pursuits. In Proceedings of the (ICASSP’05) IEEE International Conference on Acoustics, Speech, and Signal Processing, Philadelphia, PA, USA, 23 March 2005; Volume 4, pp. iv/725–iv/728. [Google Scholar]
- Gay, V.; Leijdekkers, P. A Health Monitoring System Using Smart Phones and Wearable Sensors. Int. J. Assist. Robot. Mechatron. 2007, 8, 29–36. [Google Scholar]
- Baloglu, U.B.; Talo, M.; Yildirim, O.; San Tan, R.; Acharya, U.R. Classification of myocardial infarction with multi-lead ECG signals and deep CNN. Pattern Recog. Lett. 2019, 122, 23–30. [Google Scholar] [CrossRef]
- Wasimuddin, M.; Elleithy, K.; Abuzneid, A.; Faezipour, M.; Abuzaghleh, O. Multiclass ECG signal analysis using global average-based 2-D convolutional neural network modeling. Electronics 2021, 10, 170. [Google Scholar] [CrossRef]
- Tang, X.; Hu, Q.; Tang, W. A real-time QRS detection system with PR/RT interval and ST segment measurements for wearable ECG sensors using parallel delta modulators. IEEE Trans. Biomed. Circuits Syst. 2018, 12, 751–761. [Google Scholar] [CrossRef]
- Campero Jurado, I.; Fedjajevs, A.; Vanschoren, J.; Brombacher, A. Interpretable Assessment of ST-Segment Deviation in ECG Time Series. Sensors 2022, 22, 4919. [Google Scholar] [CrossRef]
- Jurado, I.C.; Vanschoren, J. Multi-Fidelity optimization method with asynchronous generalized island model for AutoML. In Proceedings of the Genetic and Evolutionary Computation Conference Companion, Boston, MA, USA, 9–13 July 2022; pp. 220–223. [Google Scholar]
- Tao, R.; Zhang, S.; Huang, X. Magnetocardiography-Based ischemic heart disease detection and localization using machine learning methods. IEEE Trans. Biomed. Eng. 2018, 66, 1658–1667. [Google Scholar] [CrossRef] [PubMed]
- Han, C.; Shi, L. Automated interpretable detection of myocardial infarction fusing energy entropy and morphological features. Comput. Methods Programs Biomed. 2019, 175, 9–23. [Google Scholar] [CrossRef] [PubMed]
- Zhang, G.; Si, Y.; Wang, D.; Yang, W.; Sun, Y. Automated detection of myocardial infarction using a gramian angular field and principal component analysis network. IEEE Access 2019, 7, 171570–171583. [Google Scholar] [CrossRef]
- AlZu’bi, S.; Shehab, M.; Al-Ayyoub, M.; Jararweh, Y.; Gupta, B. Parallel implementation for 3d medical volume fuzzy segmentation. Pattern Recog. Lett. 2020, 130, 312–318. [Google Scholar] [CrossRef]
- Zhang, X.; Li, R.; Dai, H.; Liu, Y.; Zhou, B.; Wang, Z. Localization of myocardial infarction with multi-lead bidirectional gated recurrent unit neural network. IEEE Access 2019, 7, 161152–161166. [Google Scholar] [CrossRef]
- Zhang, J.; Lin, F.; Xiong, P. Automated detection and localization of myocardial infarction with staked sparse autoencoder and treebagger. IEEE Access 2019, 7, 70634–70642. [Google Scholar] [CrossRef]
- Wang, H.; Li, Z.; Li, Y.; Gupta, B.B.; Choi, C. Visual saliency guided complex image retrieval. Pattern Recog. Lett. 2020, 130, 64–72. [Google Scholar] [CrossRef]
- Hammad, M.; Alkinani, M.H.; Gupta, B.; El-Latif, A.; Ahmed, A. Myocardial infarction detection based on deep neural network on imbalanced data. Multimed. Syst. 2022, 28, 1373–1385. [Google Scholar] [CrossRef]
- Northern Sami. SVG Human Heart Cross-Section. Available online: https://commons.wikimedia.org/wiki/File:Diagram_of_the_human_heart_(cropped).svg (accessed on 8 August 2022).
- Peterson, E.D.; Shah, B.R.; Parsons, L. Trends in quality of care for patients with acute myocardial infarction in the National Registry of Myocardial Infarction from 1990 to 2006. Am. Heart J. 2008, 156, 1045–1055. [Google Scholar] [CrossRef]
- Hasdai, D.; Behar, S.; Wallentin, L. A prospective survey of the characteristics, treatments and outcomes of patients with acute coronary syndromes in Europe and the Mediterranean basin. The Euro Heart Survey of Acute Coronary Syndromes (Euro Heart Survey ACS). Eur. Heart J. 2002, 23, 1190–1201. [Google Scholar] [CrossRef]
- Thygesen, K.; Alpert, J.S.; White, H.D. Universal definition of myocardial infarction. J. Am. Coll. Cardiol. 2007, 50, 2173–2195. [Google Scholar] [CrossRef] [PubMed]
- Fernández, J.C. New Methodologies for the Development and Validation of Electrophysiological Models. Ph.D. Thesis, Universidad de Zaragoza, Zaragoza, Spain, 2019. [Google Scholar]
- Whitmer, K.H. Assessment of Pulmonary Function. In A Mixed Course-Based Research Approach to Human Physiology; Iowa State University Digital Press: Ames, IA, USA, 2021. [Google Scholar]
- Chobanian, A.V.; Bakris, G.L.; Black, H.R. The seventh report of the joint national committee on prevention, detection, evaluation, and treatment of high blood pressure: The JNC 7 report. JAMA 2003, 289, 2560–2571. [Google Scholar] [CrossRef] [PubMed]
- Myocardial Infarction. Available online: https://commons.wikimedia.org/wiki/File:Myocardial_infarction.svg (accessed on 8 August 2022).
- Atanasova, G.; Marinov, M. The pulse pressure amplitude as a marker of myocardial infarction risk. J. Clin. Exp. Cardiolog. 2013, 4, 2. [Google Scholar] [CrossRef]
- Mamun, M.M.R.K.; Alouani, A.T. Myocardial infarction detection using multi biomedical sensors. In Proceedings of the 10th International Conference on Bioinformatics and Computational Biology, Las Vegas, NV, USA, 19–21 March 2018; pp. 117–122. [Google Scholar]
- PhysioBank. PhysioNet. Available online: https://archive.physionet.org (accessed on 19 April 2017).
- Martin, T.N.; Groenning, B.A.; Murray, H.M. ST-Segment deviation analysis of the admission 12-lead electrocardiogram as an aid to early diagnosis of acute myocardial infarction with a cardiac magnetic resonance imaging gold standard. J. Am. Coll. Cardiol. 2007, 50, 1021–1028. [Google Scholar] [CrossRef]
- Jouck, P. Application of the Wavelet Transform Modulus Maxima Method to T-Wave Detection in Cardiac Signals; Maastricht University, Department of Mathematics and Maastricht Instruments: Maastricht, The Netherlands, 2004; pp. 1–32. [Google Scholar]
- Masip, J.; Gayà, M.; Páez, J.; Betbesé, A.; Vecilla, F.; Manresa, R.; Ruíz, P. Pulse oximetry in the diagnosis of acute heart failure. Rev. Española Cardiol. 2012, 65, 879–884. [Google Scholar] [CrossRef]
- Kannel, W.B.; Levy, D.; Cupples, L.A. Left ventricular hypertrophy and risk of cardiac failure: Insights from the Framingham Study. J. Cardiovasc. Pharmacol. 1987, 10 (Suppl. 6), S135–S140. [Google Scholar] [CrossRef] [PubMed]
- Lip, G. Hypertensive heart disease: A complex syndrome or a hypertensive cardiomyopathy? Eur. Heart J. 2000, 21, 1653–1665. [Google Scholar] [CrossRef]
- Ramirez-Carracedo, R.; Sanmartin, M.; Ten, A.; Hernandez, I.; Tesoro, L.; Diez-Mata, J.; Botana, L.; Ovejero-Paredes, K.; Filice, M.; Alberich-Bayarri, A.; et al. Theranostic contribution of extracellular matrix metalloprotease inducer-paramagnetic nanoparticles against acute myocardial infarction in a pig model of coronary ischemia-reperfusion. Circ. Cardiovasc. Imaging 2022, 15, e013379. [Google Scholar] [CrossRef]
- Milo-Cotter, O.; Cotter, G.; Kaluski, E. Rapid clinical assessment of patients with acute heart failure: First blood pressure and oxygen saturation—Is that all we need? Cardiology 2009, 114, 75–82. [Google Scholar] [CrossRef]
- Wong, N.D.; Levy, D.; Kannel, W.B. Prognostic significance of the electrocardiogram after Q wave myocardial infarction. The Framingham Study. Circulation 1990, 81, 780–789. [Google Scholar] [CrossRef]
- Channer, K.; Morris, F. ABC of clinical electrocardiography: Myocardial ischaemia. BMJ 2002, 324, 1023–1026. [Google Scholar] [CrossRef] [PubMed]
- Candil, J.J.; Luengo, C.M. QT interval and acute myocardial ischemia: Past promises, new evidences. Rev. Esp. Cardiol. 2008, 61, 561–563. [Google Scholar]
- Psaty, B.M.; Furberg, C.D.; Kuller, L.H. Association between blood pressure level and the risk of myocardial infarction, stroke, and total mortality: The cardiovascular health study. Arch. Intern. Med. 2001, 161, 1183–1192. [Google Scholar] [CrossRef] [PubMed]
- Taloba, A.I.; Alanazi, R.; Shahin, O.R.; Elhadad, A.; Abozeid, A.; El-Aziz, A.; Rasha, M. Machine algorithm for heartbeat monitoring and arrhythmia detection based on ECG systems. Comput. Intell. Neurosci. 2021, 2021, 7677568. [Google Scholar] [CrossRef]
- Hakraborty, A.; Chatterjee, S.; Majumder, K.; Shaw, R.N.; Ghosh, A. A comparative study of myocardial infarction detection from ECG data using machine learning. In Advanced Computing and Intelligent Technologies; Springer: Cham, Switzerland, 2022; pp. 257–267. [Google Scholar]
- Wang, J. Advances in ECG-Based Cardiac Ischemia Monitoring—A Review. In Proceedings of the 2021 Computing in Cardiology (CinC), Brno, Czech Republic, 12–15 September 2021; Volume 48, pp. 1–4. [Google Scholar]
- Wang, H.; Zhao, W.; Xu, Y. St segment change classification based on multiple feature extraction using ecg. In Proceedings of the 2018 Computing in Cardiology Conference (CinC), Maastricht, The Netherlands, 23–26 September 2018; pp. 1–4. [Google Scholar]
- Bigler, M.R.; Seiler, C. Detection of myocardial ischemia by intracoronary ECG using convolutional neural networks. PLoS ONE 2021, 16, e0253200. [Google Scholar] [CrossRef]
- Zhao, Y.; Xiong, J.; Hou, Y. Early detection of ST-segment elevated myocardial infarction by artificial intelligence with 12-lead electrocardiogram. Int. J. Cardiol. 2020, 317, 223–230. [Google Scholar] [CrossRef]







| ECG Threshold | ST Elevation | ST Depression | Hyperacute T Wave | Pathological Q Wave | Prolong Q Wave |
|---|---|---|---|---|---|
| single sensor | counter3/counter1 ≥ 0.95 | mean_ST_dvalue > 1 | abs(H_T_peak) > 0.5 | mean_path_c ≤ −0.25 | mean_path_QT > 0.4 |
| combination | counter3/counter1 ≥ 0.95||mean_ST_dvalue > 1 then e = 4 | mean_path_c ≤ −0.25||H_T_peak > 0.5 then f = 1 | mean_path_QT > 0.4 then g = 2 | ||
| need ECG monitoring consistency | (e + f + g) > 0 && (f + g) < 3 && c_ox > 93 && (c_bp ≥ 105 & c_bp ≤ 120) | ||||
| Potential MI scenario 1 | ((e + f + g) > 0 & (e + f + g) < 3) && ((c_bp ≥ 120||c_bp < 105)||H_R > 80||c_ox < 93) | ||||
| Potential MI scenario 2 | ((e + f + g) > 0 & (e + f + g) < 3) && (c_ox > 93||(c_bp ≥ 105 & c_bp ≤ 120)) | ||||
| The initial case of MI | (e + f + g) ≥ 3 && (((c_bp ≥ 120 & c_bp < 140)||(c_bp < 105 & c_bp ≥ 90))||H_R > 80||(c_ox < 93 & c_ox ≥ 88)) | ||||
| Medium case of MI | (e + f + g) ≥ 3 && ((c_bp ≥ 120 & c_bp < 140)||(c_bp < 105 & c_bp ≥ 90))||H_R > 80 && (c_ox < 93 & c_ox ≥ 88) | ||||
| A severe case of MI | (e + f + g) > 3 && (c_bp > 160||c_bp < 90) && c_ox < 88 | ||||
| Arrhythmia detected | H_R > 80 && (e + f + g) == 0 | ||||
| No MI symptoms | all normal | ||||
| MI State | Decision Fusion |
|---|---|
| Needs ECG monitoring consistency | (e + f + g) > 0 && (f + g) < 3 && c_ox > 93 && (c_bp ≥ 105 & c_bp ≤ 120) |
| Potential MI scenario 1 | ((e + f + g) > 0 & (e + f + g) < 3) && ((c_bp ≥ 120||c_bp < 105)||H_R > 80||c_ox < 93) |
| Potential MI scenario 2 | ((e + f + g) > 0 & (e + f + g) < 3) && (c_ox > 93||(c_bp ≥ 105 & c_bp ≤ 120)) |
| The initial case of MI | (e + f + g) ≥ 3 && (((c_bp ≥ 120 & c_bp < 140)||(c_bp < 105 & c_bp ≥ 90))||H_R > 80||(c_ox < 93 & c_ox ≥ 88)) |
| Medium case of MI | (e + f + g) ≥ 3 && ((c_bp ≥ 120 & c_bp < 140)||(c_bp < 105 & c_bp ≥ 90))||H_R > 80 && (c_ox < 93 & c_ox ≥ 88) |
| A severe case of MI | (e + f + g) > 3 && (c_bp > 160||c_bp < 90) && c_ox < 88 |
| Arrhythmia detected | H_R > 80 && (e + f + g) == 0 |
| No MI symptoms | all normal |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mamun, M.M.R.K. Significance of Features from Biomedical Signals in Heart Health Monitoring. BioMed 2022, 2, 391-408. https://doi.org/10.3390/biomed2040031
Mamun MMRK. Significance of Features from Biomedical Signals in Heart Health Monitoring. BioMed. 2022; 2(4):391-408. https://doi.org/10.3390/biomed2040031
Chicago/Turabian StyleMamun, Mohammad Mahbubur Rahman Khan. 2022. "Significance of Features from Biomedical Signals in Heart Health Monitoring" BioMed 2, no. 4: 391-408. https://doi.org/10.3390/biomed2040031
APA StyleMamun, M. M. R. K. (2022). Significance of Features from Biomedical Signals in Heart Health Monitoring. BioMed, 2(4), 391-408. https://doi.org/10.3390/biomed2040031







