Abstract
Background: To determine risk factors of death in diagnosed patients with COVID-19 who were aged ≥60 years and could not benefit from intubation and mechanical ventilation. Methods: Retrospective multicentre study including all patients with COVID-19 admitted to four medium-stay centres in Catalonia (March-June 2020). At the multivariate level, we calculated hazard ratios (HR) with 95% confidence intervals (CI) to determine risk factors associated with mortality. Results: 683 patients were included, of whom 227 died (case fatality rate of 33%, reaching 42% in patients of more than 90 years). Mean survival was 21.92 (20.98–22.86) days. Factors associated with death were fever (HR:1.5 (1.06–2.13)), malaise (HR:1.4 (1.04–1.99)), dyspnoea (HR:1.98 (1.41–2.79)) and atrial fibrillation (HR:1.45 (1.03–2.05)), while coughing (HR: 0.66 (0.46–0.94)), diarrhoea (HR:0.46 (0.23–0.92)), dyslipidaemia (HR:0.47 (0.28–0.82)), and receiving antithrombotic treatment (HR:0.56 (0.40–0.78)) had a protective effect. The analysis by age group showed that other factors were uniquely associated with each age group, such as chronic obstructive pulmonary disease at 60–74 years and polypharmacy at 75–90 years, among other factors. Conclusions: Case fatality in COVID-19 patients who could not benefit from intubation and mechanical ventilation was exceptional. Clinical manifestations such as fever, malaise, dyspnoea and atrial fibrillation helped to identify patients at higher risk of mortality, while antithrombotic treatment had a protective effect. Although some symptoms are very general regarding COVID-19, in the context of the first wave without vaccination, when not much was known about the disease, such symptoms could be useful.
1. Introduction
The first COVID-19 epidemic wave had a huge impact on a global scale. According to the World Health Organization (WHO) data, by March 2020, SARS-CoV-2 had infected more than 87,137 people, with more than 2977 deaths worldwide [1]. Globally, cases and deaths due COVID-19 have been increasing dramatically, and around of the world, there has been a varying number of epidemic waves.
The pandemic started in Europe with a significant increase in cases in Italy. In March 2020, Italy reported 101,739 cases and 11,591 deaths [1]. The average age of the deceased was 81 years, and more than two thirds of the patients had comorbidities such as diabetes, cardiovascular disease, cancer or a history of being ex-smokers [2].
Spain was one of the first European countries with the highest number of affected patients. By 18 May 2020, 231,350 PCR-positive cases had been detected in Spain, and despite the limited availability of these tests, 27,650 (11.95%) died. Eighty-seven per cent of the deceased were over 70 years of age, and 95 per cent had at least one previous illness. A total of 55,824 patients corresponded to the Autonomous Community of Catalonia where, in addition, a 71% excess in mortality was observed for all ages (March/April 2020), reaching 85.2% in those over 74 years of age [3].
In this first wave of the epidemic, the health system was overwhelmed by the increase in the number of patients, and unfortunately, some fragile patients could not be intubated to offer them mechanical ventilation. Furthermore, as orotracheal intubation and mechanical ventilation are futile and aggressive treatments for fragile patients at an advanced age and high comorbidity, which in some of these patients could make them more vulnerable to death, it was decided to offer them conservative non-invasive treatment in social healthcare centres (intermediate care centres/hospitals) [4]. These centres are an intermediate step between the hospital and the private home or residential centre. Intermediate-care hospitals mainly admit patients referred from acute hospitals for specific therapy with an interdisciplinary approach, as well as patients in need of palliative care and complex symptom management, many of them in the last days of life.
Despite several published studies on factors related to COVID-19 mortality [5,6] many factors have not yet been identified. Therefore, the aim of this study is to identify factors related to mortality in a cohort of COVID-19 patients aged 60 years and older who were neither intubated nor mechanically ventilated and who were admitted to intermediate care hospitals during the first wave of the COVID-19 epidemic.
2. Materials and Methods
2.1. Study Design
Retrospective cohort study.
2.2. Sample
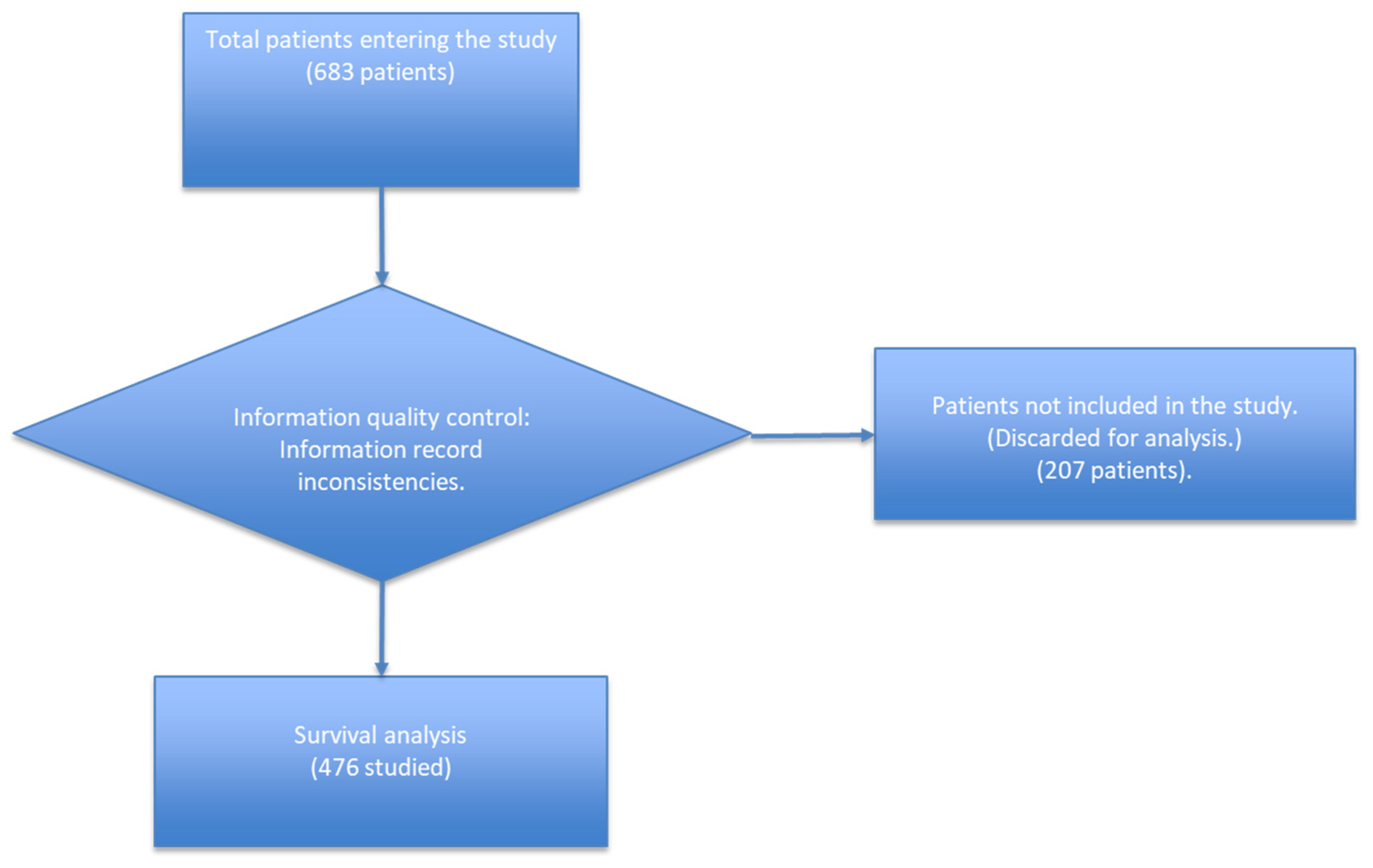
COVID-19 was diagnosed in patients transferred to long-term care facilities from 3 acute hospitals and nursing homes affected by COVID-19 outbreaks in Catalonia, Spain. Between March and June 2020 (Figure 1), there were 683 patients of 60 years and over identified who were ruled out for intubation and mechanical ventilation. A survival analysis was carried out for those allowing for at least 15 days of follow up. Patients who missed the date of the Real-Time Reverse Transcriptase Polymerase Chain Reaction Assay (RT-PCR) or wrong dates of admission or discharge were discarded.
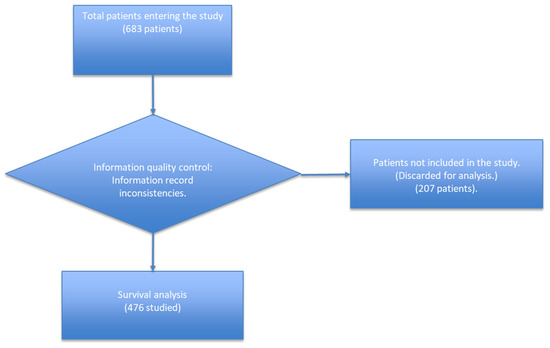
Figure 1.
Mortality study of COVID-19 patients of the Isabel Roig, Dolores Aleu Socio-Health Centres, Saint Joan de Reus Hospital, Centre Fòrum del Parc de Salut Mar, Barcelona; March to June 2020; first wave.
In the general registry, 683 patients were entered. Within the registry, there were cases of patients whose length of stay in the study was not considered, since these had negative or atypical PCR dates.
These patients were considered and evaluated in the general descriptive analysis but were not considered in the Kaplan–Meier survival analysis and Cox regression analysis, because these would produce a bias or error in the final analysis.
2.3. Long-Term Care Facilities
Centro Sociosanitario Isabel Roig. (IR), Centro Sociosanitario Dolors Aleu (DA), Centre Fòrum del Parc de Salut Mar (CF) and Centre Sociosanitari de l’Hospital Universitari Sant Joan de Reus (SJR).
2.4. Variables
According to the Laframboise Epidemiological Model [7].
2.4.1. Human Biology
Sex, Age Band (60–74 years, 75–90, >90) [8], and Comorbidities on Admission
- Hypertension, auricular fibrillation, heart failure, ischemic heart disease, other cardiovascular diseases.
- Asthma, bronchitis, chronic obstructive pulmonary disease (COPD), other respiratory diseases.
- Diabetes, obesity, dyslipidaemia, other metabolic conditions.
- The term dyslipidaemia indicates an elevated concentration of lipids in the blood. There are several categories of this disorder, depending on which lipids are altered. The two most important forms are hypercholesterolaemia and hypertriglyceridaemia, although other disorders can be common, such as hyperchylomicronaemia or decreased HDL-cholesterol [9].
- Ictus, migraine, Parkinson’s disease, Alzheimer’s disease, cognitive impairment, other neurological diseases.
- Other conditions:
- −
- Neoplasia;
- −
- Chronic renal failure;
- −
- Low-frequency digestive pathology;
- −
- Anaemia;
- −
- Other.
- Geriatric syndrome: immobility, recurrent falls, pressure ulcers, malnutrition, incontinence, constipation, dysphagia, polypharmacy (more than 5 drugs), cognitive impairment, depression/insomnia and sensory impairment.
- Initial symptoms: fever, cough, malaise, dyspnoea, myalgia, fatigue, diarrhoea, nausea, other initial symptoms.
- The authors confirmed that all information about the whole medical history of these patients to the time of admission to care was confirmed with the patients’ medical records.
2.4.2. Lifestyles
Smoking (non-ex-smoker, ex-smoker, light smoker (1–9/day), moderate smoker (10–19/day), heavy smoker (20+/day); alcohol intake: non-drinker, trivial <1 units/day, light 1–2 units/day, moderate 3–6 units/day, heavy 7–9 units/day, very heavy >9 units/day. Cocaine, cannabis, heroine and other illicit drugs.
2.4.3. COVID-19 Progression
Respiratory distress onset, PCR date, clinical exacerbation date, radiological confirmation date, time elapsed to negative PCR test, dates of either discharge or death.
2.4.4. Treatments:
- Azithromycin, ceftriaxone, other antibiotics.
- Corticoids and inhalers (INH): methylprednisolone, hydrocortisone, dexamethasone, salbutamol, ipratropium bromide, other inhalers.
- Other treatments: hydroxychloroquine (antimalarial), oxygen, antithrombotics, anti-platelet agents, antiviral and ivermectin.
2.5. Data Analysis
Forms with missing values or with misinterpreted items were returned for completion/correction to health staff. Date consistency was also checked. Randomly, 10% of the completed forms were validated as a quality control.
Analyses were carried out using SPSS 27.0 for descriptive and Cox regressions, as well as R 4.0.4 for the survival plots. We followed the STROBE statement (Strengthening the Reporting of Observational Studies in Epidemiology) to report this study [10].
Descriptive analysis and lethality rates of the sample and by age bands were obtained. Mean and standard deviation was calculated for continuous variables.
The outcome was death. Patients were followed from their admission date for at least 15 days up until they were censored at the earliest date of either being lost to follow-up, death, or 30 June 2020. The survival function of the patients in the groups studied, the risk of occurrence of the COVID-19 death event, as well as the mean survival time were determined.
The probability distribution of survival times in COVID-19 patients was performed using the non-parametric Kaplan–Meier method, followed by Cox regression analysis by specific age groups and the final multivariate analysis in which the variables that were significant (hazard ratio (HR)) were entered, and both protective and risk factors were identified in the total population and in the age groups studied. Kaplan–Meier survival curves were drawn for the death incidence, and differences between age group exposures were compared using the log-rank test. Cox’s proportional hazards models were used to describe the association between death and comorbidities, geriatric syndrome, onset of symptoms and treatments. We assessed the proportional hazards assumption by using log minus log plots. Hazard ratios with 95% confidence intervals were calculated for the sample and by age band.
Ethics statements: Data were treated in a strictly confidential manner according to the ethical principles of the Helsinki Declaration. The study was approved by the statutory clinical research committee of Hospital del Mar (2020/9477) and l’Hospital Universitari Sant Joan de Reus (CEIC 203/2020).
3. Results
We studied 683 patients, of whom 227 died; we identified a 33% lethality rate, and by age group, we observed an upward gradient (60–74: 21%; 75–90: 32%; 90+: 42%). By sex, men showed a higher lethality (42%). By socio-health centres, lethality ranged from 27% to 43% (Table 1). The geriatric syndromes with the highest lethality were dysphagia (42%) and immobility (40%); among cardiovascular diseases, atrial fibrillation (43%) and heart failure (40%); among respiratory diseases, patients with COPD (35%); and among metabolic diseases, patients with hypothyroidism (36%) and diabetes (35%). Neurological diseases included Parkinson’s disease (48%). The initial symptoms associated with the highest lethality were dyspnoea (45%), fever (40%), malaise (40%) and fatigue (40%). In terms of treatment, the highest lethality was observed among patients treated with antiplatelet agents (68%) and salbutamol (51%) (Table 1).

Table 1.
Description of patient mortality by age groups.
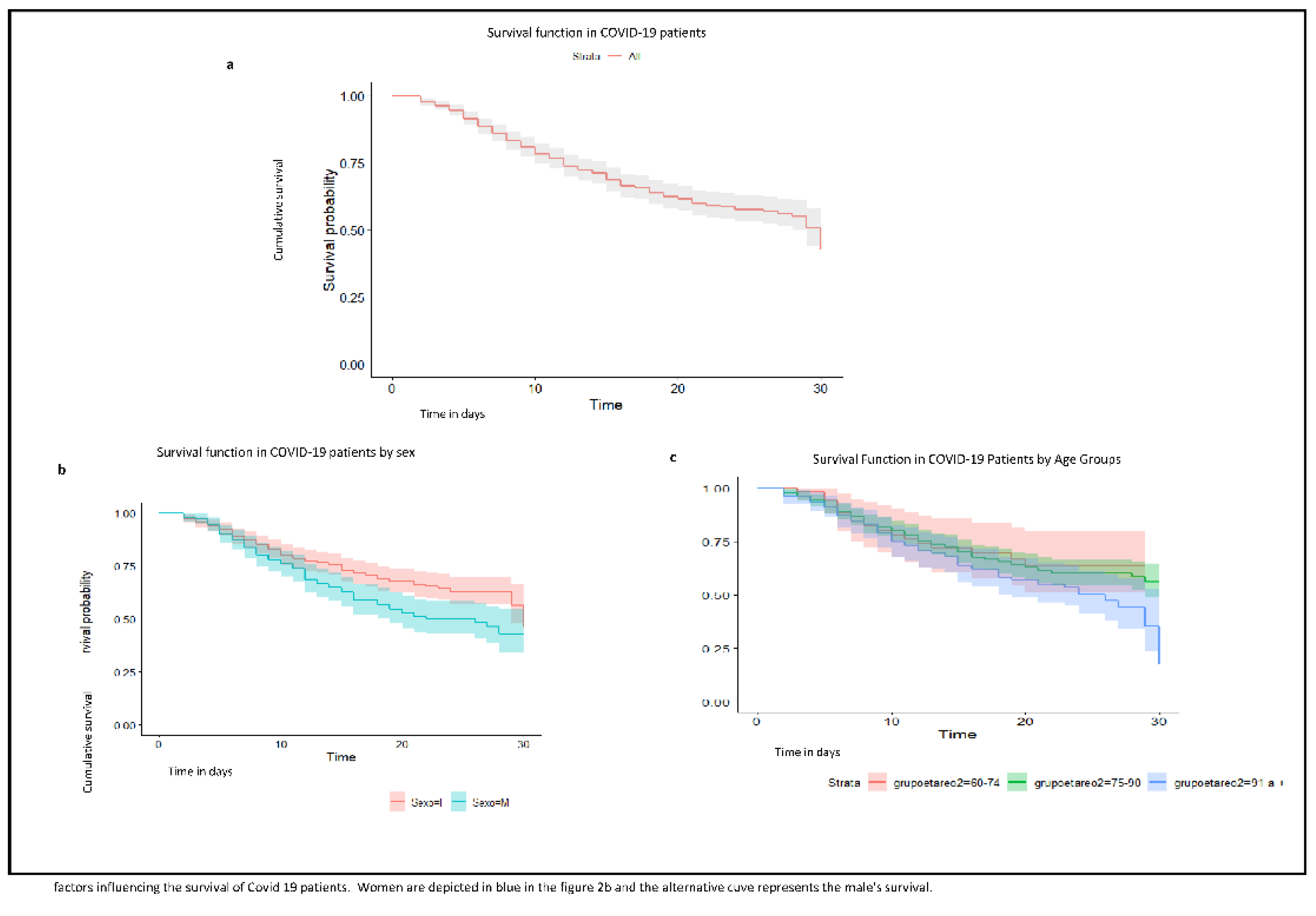
We identified a survival time of 21.9 days. In women, it was 20.37 days (95% CI: 18.9–21.84) and in men 23.01 days (95% CI; 21.81–24.22). By age group, mean survival was 22.20 days for the 60–74 years group, 22.34 days in the 75–90 years group, and 20.63 days in those over 90 years of age (Table 2).

Table 2.
The survival time by gender and age groups.
At the multivariate level, associated variables leading to death were fever HR:1.5 (1.06–2.13), general discomfort HR: 1.4 (1.04–1.99), dyspnoea HR:1.9 (1.41–2.79) and atrial fibrillation HR: 1.45 (1.03–2.05), while cough HR:0.7 (0.46–0.94), diarrhoea HR:0.45 (0.23–0.92), dyslipidaemia HR:0.47 (0.28–0.82) and receiving antithrombotic treatment HR:0.56 (0.40–0.78), had a protective effect. By age, no statistically significant effect was identified (Table 3).

Table 3.
The multivariate level by sex, age, comorbidities and treatments by age group.
When analysed by age group, in the 60–74 years age group, constipation, chronic obstructive pulmonary disease (COPD) and neoplasia were risk factors (RF). Incontinence and migraine presented a very wide CI. Ceftriaxone treatment did not show a protective effect.
In the 75 to 90 years group, RF identified were male sex, polypharmacy, atrial fibrillation, and symptoms such as general discomfort, dyspnoea, fever and methylprednisolone treatment. Protective factors (PF) identified were cough and diarrhoea.
Finally, in the 91 years or older group, dyslipidaemia was confirmed as the PF with a more adjusted HR, as well as the use of azithromycin and antithrombotic agents (See Table 3).
4. Discussion
To the best of our knowledge, this survival study is the first conducted in COVID-19 patients over 60 years of age admitted to social-health centres (HAI) and who could not be treated with intubation and mechanical ventilation for several reasons (saturation of ICU beds, major comorbidities).
A high case mortality rate was observed (33%), which increased as age increased, reaching 42% in the 91 years or more age group. No significant differences were observed between HAI. This lethality rate coincided at a time when in many countries there was a high mortality rate, which worldwide reached 541,310 deaths up to 30 June 2020, although it has been estimated that only a quarter of the deaths were reported. In Spain, the mortality excess was 58% with 120,768 deaths registered as of 29 June 2020 [3].
In order to facilitate reading, we have divided the discussion into several blocks of analysis, which we describe below.
4.1. Demographic Variables
In the survival analysis, it was evident that men had a mean survival time longer than that of women (Table 2). However, in the multivariate regression of the total population, we did not find a statistically significant difference. The only exception was in the 75–90 years age group where male sex was a risk factor; in this regard, a meta-analysis mentions male sex as a mortality RF in the general population [11]. These results are in line with other research in Mexico carried out in people over 50 years of age [12] and India [13], and other studies [14,15,16,17]; however, these studies were carried out in a different population than HAI. One study mentions that there are factors associated with sex, which could increase the higher mortality of the male sex [18], such as the X chromosome and sex hormones, which could play a key role in the innate and adaptive immunity of male patients to the COVID-19 virus [16,18].
It was observed that survival time tends to decrease with age (Figure 2). Although this same trend is observed in the central estimators of the adjusted HRs (Table 3), when the age was adjusted for other variables, it did not appear to play a statistically significant role. This may be related to confusion between age and comorbidity. Thus, we can differentiate between chronological age and “biological age”, and this difference would be the key to why age alone is not associated with mortality, but we must analyse the factors that condition “biological age” [19]. This “biological age” is reflected in the so-called frailty, i.e., the decrease in functional reserve and resistance to stressors [20,21].
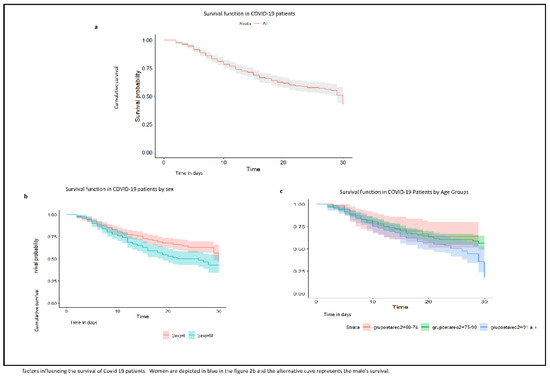
Figure 2.
(a). Graph of survival function in COVID-19 patients from the Isabel Roig; (b). survival function in COVID-19 patients by sex; (c). survival function in COVID-19 patients by age group. Dolores Aleu Socio-Health Centres, Saint Joan de Reus Hospital, Centre Fòrum del Parc de Salut Mar, Barcelona; March to June 2020; first wave.
4.2. Geriatric Syndromes
There are many scientific reports on the association between immobility and the risk of dying from COVID-19 [22,23]. A study carried out on 102 patients showed a tendency towards higher mortality at day 30 in patients with greater functional impairment/immobility, although no statistically significant differences were found [24]. Another study in a residential centre showed that there was a statistically significant relationship between immobility and higher mortality due to COVID-19 [25].
We observed that mortality in patients with immobility increases as age increases, 23% for the group of 60–74, 40% for the group of 75 to 90 and 50% for the group of 91 and over (Table 1). The lethality rate in relation to recurrent falls also increases as age increases; however, the recurrent falls/lethality rates are lower than immobility lethality rate. Recurrent falls could be an indicator of greater patient mobility and better health status compared to immobile patients [25,26]. Despite lethality rate being high among patients with immobility and patients with recurrent falls, none of the variables were associated with a risk of mortality due to COVID-19 in the multivariate analysis (Table 3).
Polypharmacy, however, was a statistically significant RF in the age group of 75 to 90 years, although it was not observed in the general population, as reported in the literature [24,27,28]. Nevertheless, polypharmacy has been found to be associated with a higher risk of mortality among older persons [29] and the reasons for these could be various. Polypharmacy could behave as a surrogate for multi-morbidity, which has been found to be associated with a higher risk of mortality [30]. Conversely, the use of multiple concomitant medications may outweigh individual benefits due to potential ensuing side effects [31].
Finally, the associations observed with incontinence with very large CI and constipation (which has no biological explanation) in the 60–74 age group could be spurious associations.
4.3. Cardiovascular, Respiratory, Metabolic and Neurologic Comorbidities
Dyslipidaemia was associated as PF with mortality. Contrarily, some studies have suggested that dyslipidaemia could be associated with a higher risk of severe COVID-19 infection and mortality [26,32,33,34]. Nevertheless, no relationship between dyslipidaemia and COVID-19 infection has been well established yet.
In this study, older persons with atrial fibrillation had a higher risk of mortality (total population and 75–90 years). Cardiac arrhythmias are among the most common comorbidities in patients with COVID-19 [35], and atrial fibrillation has been found to be associated with a higher 60-day mortality.
Chronic obstructive pulmonary disease (COPD) (Table 3) was associated with a higher risk of mortality in the 60 to 74 years group. A systematic review and meta-analysis found that among individuals with COVID-19, the presence of co-morbidities (both cardiometabolic, COPD and other) is associated with a higher risk of severe COVID-19 and mortality [36]. Furthermore, neoplasia could play a role as another comorbidity. However, migraine due to such a wide CI seems to be a spurious association.
4.4. Initial Symptoms
Fever and dyspnoea were identified as RF of mortality, especially in older subgroups. These findings coincide with several studies, both in the general population and in older age groups [14,15,25,37]. A systematic review found that fever was associated as a mortality RF in almost all the of the studies analysed [38].
Cough and diarrhoea were PF of mortality. A systematic review [17] mentions that digestive symptoms have been associated with the severity of the disease, but are not associated with increased mortality. Furthermore, digestive symptoms were not among the clusters of symptoms mostly associated with mortality risk among patients with COVID-19 as studied [39]. The result of cough as a PF for mortality could be related to the patient’s fragilities. In patients with associated immobility syndrome, the cough reflex is usually reduced or goes away. Therefore, in patients with a better overall status, cough may be associated with a higher survival, evidencing a better respiratory functional capacity (the use of antitussives was not recorded in the patients studied). General discomfort/malaise is a symptom present in most of the patients studied. Malaise was a RF of mortality in the present study. This symptom seems to be related to the response of the body’s immune system to infection. COVID-19 disease results in a massive cytokine storm which results in a variety of symptoms, including malaise [40]. Thus, the presence of general discomfort/malaise may be associated with a more massive cytokine reaction, resulting in a more severe disease with a higher mortality risk [41].
4.5. Treatment
Patients treated with antithrombotic therapy (including both oral anticoagulants and low-molecular-weight heparins (LMWH)) had a lower risk of mortality in this study. These results are in line with the current literature on this issue as well as with current COVID-19 guidelines [42]. Another study suggested that anticoagulation for 7 days or longer may improve outcomes in hospitalized patients [43]. Moreover, in another study, it was observed that a longer duration of anticoagulation was associated with a reduced risk of mortality [44]. Furthermore, another study demonstrated the preventive use of apixaban or enoxaparin with a significant decrease in mortality in patients with D-dimer levels >1 µg/mL. [45]. Several studies have associated greater benefit with therapeutic anticoagulation compared with prophylactic anticoagulation [46]. Similarly, pre-existing antithrombotic treatment with direct acting oral anticoagulants (DOAC) or vitamin K antagonists (VKA) was also associated with improved outcomes [47].
The severe inflammatory response to COVID-19 may predispose patients to thrombotic events [43,48]. Moreover, autopsy data suggest that thrombotic events are directly responsible for up to 30% of deaths [49]. Consequently, the use of anticoagulants with LMWH or oral anticoagulants may be of benefit to these patients [43].
Fewer studies have reported on the effects of anticoagulants specifically among older persons with regard to mortality. Other authors found anticoagulant treatment to be associated with a higher chance of survival among older persons [50], and in another study, the treatment with LMWH was associated with better survival [51].
Receiving a treatment with methylprednisolone did not affect mortality among the overall sample of older people. Similar results were reported in other studies [52].
In the subgroup analysis (75 to 90 years), a higher risk of mortality was observed among those who received treatment with methylprednisolone. In the same line, another study found that the administration of high doses of corticosteroids was associated with an increased risk of mortality [53].
Contrarily, several studies have reported different results [54,55]. In the subgroup analysis of patients over 60 years old, those patients in the methylprednisolone group had a lower mortality rate at day 28 [54]. Furthermore, intravenous methylprednisolone administration has been found to be associated with a reduced risk of death among patients with moderate to severe COVID-19 [56]. The fact that neither data on COVID-19 severity nor data on the dose and duration of treatment with methylprednisolone were analysed in the present study may limit the interpretation of our results.
The safety profile of azithromycin used as an antibacterial agent is well established [57]. However, in the case of COVID-19, its role is controversial; thus, the results should be assessed with caution.
Although treatment with ceftriaxone was identified as a probable RF, the CI in the total population is very low and as the 60–74 years age group is a very small population (n = 55), we believe that the association presented could be spurious.
This study is one of the few studies carried out in a population over 60 years of age who could not benefit from orotracheal intubation and mechanical ventilation due to ICU bed saturation and/or suffering from many comorbidities and who were admitted to intermediate care hospitals (social-health centres (HAI)) in Spain.
5. Limitations
In terms of limitations, the retrospective methodology of the study may have limited the quality of the information collected. As regards information on drug use, no information is available on treatment times or doses. At the same time, this study has the limitation of retrospective studies. This study does not allow us to identify cause–effect relationships such that the associations observed must be interpreted in terms of associations with the survival analysed.
In addition, as this is an exploratory analysis of field observations, the sample size is small, especially in the 60–74 age groups and in those over 91 years of age, which could affect the results, as it limits the statistical power. Therefore, results by age group should be treated with caution.
Regarding the selection of the study variables, some of the parameters studied are considered as generalised conditions that could hide a different disorder. General discomfort/malaise, for example, cannot be objectively defined, while many conditions can lead to this situation. In addition, the sensation of dyspnoea is a subjective response, while oxygen saturation or PaO 2, and even more, the ratio to inspired oxygen fraction (GiO 2) are considered as valuable and objective factors; thus, results related to symptoms should be taken with caution.
6. Conclusions
This study is one of the few studies describing the main characteristics and analysing factors potentially related to mortality in a sample of unvaccinated elderly persons admitted to intermediate care hospitals (social-health centres (HAI)) with COVID-19.
Mortality due to COVID-19 in this population was relevant. Clinical manifestations such as initial fever, malaise, dyspnoea and atrial fibrillation were associated with an increased risk of death. These findings may help identify those older patients with COVID-19 who may specially benefit from a closer surveillance by health care professionals, especially in countries that do not yet have good vaccination coverage and health infrastructure.
The use of antithrombotic agents (including both oral anticoagulants and low-molecular-weight heparins) was associated with a lower risk of mortality in this population, suggesting that treatment protocols for the management of COVID-19 in this population should consider their use.
This study may help to improve the prognosis of older COVID-19 patients in countries that do not yet have good vaccination coverage and health infrastructure as well as good monitoring programmes.
Author Contributions
Conceptualization, D.R.C., J.O.S., E.D.C.-V., A.R.-G., E.R., S.M.M.T., P.R. and A.L.A.T.; data curation, D.R.C., J.O.S., E.D.C.-V., A.R.-G., E.R., S.M.M.T., P.R., A.L.A.T., I.M., L.C.-G., M.H.-T., P.J.-M., M.M.-C., R.A.S.-G., C.R., M.d.C.M., G.D.F., J.A.L.-B., M.N.-M., C.G. and J.A.C.; formal analysis, D.R.C., J.O.S., E.D.C.-V., A.R.-G., E.R., S.M.M.T., P.R., J.A.L.-B., M.N.-M., C.G. and J.A.C.; resources, D.R.C., J.O.S., E.D.C.-V., A.R.-G., E.R., S.M.M.T., P.R. and A.L.A.T.; software, D.R.C., J.A.L.-B., M.N.-M., C.G. and J.A.C.; supervision, D.R.C., J.O.S., E.D.C.-V., A.R.-G., E.R., S.M.M.T., P.R. and A.L.A.T.; validation, D.R.C., J.O.S., E.D.C.-V., A.R.-G., E.R., S.M.M.T., P.R. and A.L.A.T.; visualization, D.R.C., J.O.S., E.D.C.-V., A.R.-G., E.R., S.M.M.T., P.R., A.L.A.T., I.M., L.C.-G., M.H.-T., P.J.-M., M.M.-C., R.A.S.-G., C.R., M.d.C.M., G.D.F., J.A.L.-B., M.N.-M., C.G. and J.A.C.; writing—original draft, D.R.C., J.O.S., E.D.C.-V., A.R.-G., E.R., S.M.M.T., P.R., A.L.A.T., I.M., L.C.-G., M.H.-T., P.J.-M., M.M.-C., R.A.S.-G., C.R., M.d.C.M., G.D.F., J.A.L.-B., M.N.-M., C.G. and J.A.C.; writing—review and editing, D.R.C., J.O.S., E.D.C.-V., A.R.-G., E.R., S.M.M.T., P.R., A.L.A.T., I.M., L.C.-G., M.H.-T., P.J.-M., M.M.-C., R.A.S.-G., C.R., M.d.C.M., G.D.F., J.A.L.-B., M.N.-M., C.G. and J.A.C. (WGSO). All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
All authors have endorsed the submitted version of this manuscript. Data were treated in a strictly confidential manner according to the ethical principles of the Helsinki Declaration. The study was approved by the statutory clinical research committee of Hospital del Mar (2020/9477) and l’Hospital Universitari Sant Joan de Reus (CEIC 203/2020). Due to the emergency situation, the researchers asked to exclude the patient consent, and this was authorised by the ethics committee of the Hospital del Mar.
Informed Consent Statement
This study was exempted from informed consent by the ethics committee due to the emergency situation during the first wave. The study was approved by the statutory clinical research committee of Hospital del Mar (2020/9477) and l’Hospital Universitari Sant Joan de Reus (CEIC 203/2020).
Data Availability Statement
The data of this study are not accessible in open access in order to preserve the confidentiality rights of the patients included in the study. Further enquiries can be directed to the corresponding author.
Acknowledgments
The authors acknowledge the support of the Centro Sociosanitario Isabel Roig, Centro Sociosanitario Dolors Aleu, Centre Fòrum del Parc de Salut Mar, and Centre Sociosanitari de l’Hospital Universitari Sant Joan de Reus. To the working Group about Survival in Old COVID-19 Patients: nurses: Montserrat Lago, Ruben Palacios, Zaraida Moreno, Mónica Ferreira, Celso Silva Fernandez, Zoila Alcalde, Geraldine Velez Vergara, Rene Flores Gonzales, María Elena Lopez Salguero, Sharalyn García, Noelia Hermoso, and Noelia Buitrago, and to the doctors: Victor Reyes, Sonia Fernandez, Luis A. Nania, Luis Arrarian, Cristina Garzón, Marita Saldarriaga, Jose Teixidor, and Andreu Garrigos. Staff of the Hospital Sant Joan de Reus Noemi Gamez Fernandez and Pilar Garcia. Staff of H.del Mar. To Alicia Padrón of the National School of Health—Carlos III Health Institute of Madrid.
Conflicts of Interest
All authors declare that they have no conflict of interest. Thus, the results and conclusions were freely arrived, and the authors’ views do not necessarily coincide with the official statements and declarations of their institutions. Regarding funding, this work has been carried out without grants or sponsors.
References
- WHO. Coronavirus Disease 2019 (COVID-19) Situation Report-41; WHO: Geneva, Switzerland, 2020. [Google Scholar]
- Cesari, M.; Proietti, M. COVID-19 in Italy: Ageism and Decision Making in a Pandemic. J. Am. Med. Dir. Assoc. 2020, 21, 576–577. [Google Scholar] [CrossRef] [PubMed]
- Diaz, L.F. Vigilancia De Los Excesos De Mortalidad Por Todas Las Causas. MoMo. Available online: https://www.isciii.es/QueHacemos/Servicios/VigilanciaSaludPublicaRENAVE/EnfermedadesTransmisibles/MoMo/Documents/InformesMoMo2022/MoMo_Situacion%20a%202%20de%20febrero_CNE.pdf (accessed on 27 June 2022).
- Sezgin, D.; O’Caoimh, R.; Liew, A.; O’Donovan, M.R.; Illario, M.; Salem, M.A.; Kennelly, S.; Carriazo, A.M.; Lopez-Samaniego, L.; Carda, C.A.; et al. The effectiveness of intermediate care including transitional care interventions on function, healthcare utilisation and costs: A scoping review. Eur. Geriatr. Med. 2020, 11, 961–974. [Google Scholar] [CrossRef]
- Mena-Vázquez, N.; Manrique Arija, S.; Rojas-Giménez, M.; Raya-Álvarez, E.; Velloso-Feijoó, M.L.; López-Medina, C.; Ramos-Giraldez, C.; Godoy-Navarrete, F.J.; Redondo-Rodríguez, R.; Cabezas-Lucena, A.M.; et al. Hospitalization and Mortality from COVID-19 of Patients with Rheumatic Inflammatory Diseases in Andalusia. Reumatol. Clin. 2021, in press. [CrossRef] [PubMed]
- Gutiérrez Rodríguez, J.; Montero Muñoz, J.; Jiménez Muela, F.; Guirola García-Prendes, C.; Martínez Rivera, M.; Gómez Armas, L. Variables associated with mortality in a selected sample of patients older than 80 years and with some degree of functional dependence hospitalized for COVID-19 in a Geriatrics Service. Rev. Esp. Geriatr. Gerontol. 2020, 55, 317–325. [Google Scholar] [CrossRef]
- Dever, A.G.E. Epidemiología Y Administración De Servicios De Salud; Organización Panamericana de la Salud/Organización Mundial de la Salud: Washington, DC, USA, 1991. [Google Scholar]
- Ouchi, Y.; Rakugi, H.; Arai, H.; Akishita, M.; Ito, H.; Toba, K.; Kai, I. Redefining the elderly as aged 75 years and older: Proposal from the Joint Committee of Japan Gerontological Society and the Japan Geriatrics Society. Geriatr. Gerontol. Int. 2017, 17, 1045–1047. [Google Scholar] [CrossRef]
- Lozano, J.A. Dislipidemias. Offarm 2005, 24, 100–108. [Google Scholar]
- Cuschieri, S. The STROBE guidelines. Saudi J. Anaesth. 2019, 13, S31–S34. [Google Scholar] [CrossRef] [PubMed]
- Peckham, H.; de Gruijter, N.M.; Raine, C.; Radziszewska, A.; Ciurtin, C.; Wedderburn, L.R.; Rosser, E.C.; Webb, K.; Deakin, C.T. Male sex identified by global COVID-19 meta-analysis as a risk factor for death and ITU admission. Nat. Commun. 2020, 11, 6317. [Google Scholar] [CrossRef]
- Salinas-Escudero, G.; Carrillo-Vega, M.F.; Granados-García, V.; Martínez-Valverde, S.; Toledano-Toledano, F.; Garduño-Espinosa, J. A survival analysis of COVID-19 in the Mexican population. BMC Public Health 2020, 20, 1616. [Google Scholar]
- Kundu, S.; Chauhan, K.; Mandal, D. Survival analysis of patients with COVID-19 in india by demographic factors: Quantitative study. JMIR Form. Res. 2021, 5, e23251. [Google Scholar] [CrossRef]
- Iftimie, S.; López-Azcona, A.F.; Vicente-Miralles, M.; Descarrega-Reina, R.; Hernández-Aguilera, A.; Riu, F.; Simó, J.M.; Garrido, P.; Joven, J.; Camps, J.; et al. Risk factors associated with mortality in hospitalized patients with SARS-CoV-2 infection. A prospective, longitudinal, unicenter study in Reus, Spain. PLoS ONE 2020, 15, e0234452. [Google Scholar] [CrossRef] [PubMed]
- Bertsimas, D.; Lukin, G.; Mingardi, L.; Nohadani, O.; Orfanoudaki, A.; Stellato, B.; Wiberg, H.; Gonzalez-Garcia, S.; Parra-Calderón, C.L.; Robinson, K.; et al. COVID-19 mortality risk assessment: An international multi-center study. PLoS ONE 2020, 15, e0243262. [Google Scholar] [CrossRef] [PubMed]
- Petrilli, C.M.; Jones, S.A.; Yang, J.; Rajagopalan, H.; O’Donnell, L.; Chernyak, Y.; Tobin, K.A.; Cerfolio, R.J.; Francois, F.; Horwitz, L.I. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: Prospective cohort study. BMJ 2020, 369, m1966. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Ding, M.; Dong, X.; Zhang, J.; Kursat Azkur, A.; Azkur, D.; Gan, H.; Sun, Y.; Fu, W.; Li, W.; et al. Risk factors for severe and critically ill COVID-19 patients: A review. Allergy 2021, 76, 428–455. [Google Scholar] [CrossRef]
- Chen, N.; Zhou, M.; Dong, X.; Qu, J.; Gong, F.; Han, Y.; Qiu, Y.; Wang, J.; Liu, Y.; Wei, Y.; et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: A descriptive study. Lancet 2020, 395, 507–513. [Google Scholar] [CrossRef]
- Searle, S.D.; Mitnitski, A.; Gahbauer, E.A.; Gill, T.M.; Rockwood, K. A standard procedure for creating a frailty index. BMC Geriatr. 2008, 8, 24. [Google Scholar] [CrossRef]
- Sociedad Española de Geriatría y Gerontología Tratado de Geriatría. Available online: https://www.segg.es/tratadogeriatria/main.html (accessed on 23 July 2022).
- Rubenstein, L.Z. Geriatric assessment: An overview of its impacts. Clin. Geriatr. Med. 1987, 3, 1–15. [Google Scholar] [CrossRef]
- Salgado Alba, A.; Guillem Llera, F.; Ruipérez Cantera, I. Manual De Geriatría; Masson: Barcelona, Spain, 2003. [Google Scholar]
- Aprahamian, I.; Cesari, M. Geriatric Syndromes and SARS-CoV-2: More than Just Being Old. J. Frailty Aging 2020, 9, 127–129. [Google Scholar] [CrossRef] [PubMed]
- Karlsson, L.K.; Jakobsen, L.H.; Hollensberg, L.; Ryg, J.; Midttun, M.; Frederiksen, H.; Glenthøj, A.; Kodahl, A.R.; Secher-Johnsen, J.; Nielsen, L.K.; et al. Clinical presentation and mortality in hospitalized patients aged 80+ years with COVID-19—A retrospective cohort study. Arch. Gerontol. Geriatr. 2021, 94, 104335. [Google Scholar] [CrossRef]
- Knopp, P.; Miles, A.; Webb, T.E.; Mcloughlin, B.C.; Mannan, I.; Raja, N.; Wan, B.; Davis, D. Presenting features of COVID-19 in older people: Relationships with frailty, inflammation and mortality. Eur. Geriatr. Med. 2020, 11, 1089–1094. [Google Scholar] [CrossRef] [PubMed]
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; Bacon, S.; Bates, C.; Morton, C.E.; Curtis, H.J.; Mehrkar, A.; Evans, D.; Inglesby, P.; et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef]
- Trecarichi, E.M.; Mazzitelli, M.; Serapide, F.; Pelle, M.C.; Tassone, B.; Arrighi, E.; Perri, G.; Fusco, P.; Scaglione, V.; Davoli, C.; et al. Clinical characteristics and predictors of mortality associated with COVID-19 in elderly patients from a long-term care facility. Sci. Rep. 2020, 10, 20834. [Google Scholar] [CrossRef] [PubMed]
- De Smet, R.; Mellaerts, B.; Vandewinckele, H.; Lybeert, P.; Frans, E.; Ombelet, S.; Lemahieu, W.; Symons, R.; Ho, E.; Frans, J.; et al. Frailty and Mortality in Hospitalized Older Adults With COVID-19: Retrospective Observational Study. J. Am. Med. Dir. Assoc. 2020, 21, 928–932.e1. [Google Scholar] [CrossRef]
- Chang, T.I.; Park, H.; Kim, D.W.; Jeon, E.K.; Rhee, C.M.; Kalantar-Zadeh, K.; Kang, E.W.; Kang, S.W.; Han, S.H. Polypharmacy, hospitalization, and mortality risk: A nationwide cohort study. Sci. Rep. 2020, 10, 18964. [Google Scholar] [CrossRef] [PubMed]
- Iaccarino, G.; Grassi, G.; Borghi, C.; Ferri, C.; Salvetti, M.; Volpe Massimo, M. Age and multimorbidity predict death among COVID-19 Patients: Results of the SARS-RAS study of the Italian society of hypertension. Hypertension 2020, 76, 366–372. [Google Scholar] [CrossRef] [PubMed]
- Turgeon, J.; Michaud, V.; Steffen, L. The dangers of polypharmacy in elderly patients. JAMA Intern. Med. 2017, 177, 1544. [Google Scholar] [CrossRef] [PubMed]
- Izcovich, A.; Ragusa, M.A.; Tortosa, F.; Marzio, M.A.L.; Agnoletti, C.; Bengolea, A.; Ceirano, A.; Espinosa, F.; Saavedra, E.; Sanguine, V.; et al. Prognostic factors for severity and mortality in patients infected with COVID-19: A systematic review. PLoS ONE 2020, 15, e0241955. [Google Scholar] [CrossRef]
- Zheng, Z.; Peng, F.; Xu, B.; Zhao, J.; Liu, H.; Peng, J.; Li, Q.; Jiang, C.; Zhou, Y.; Liu, S.; et al. Risk factors of critical & mortal COVID-19 cases: A systematic literature review and meta-analysis. J. Infect. 2020, 81, e16–e25. [Google Scholar] [PubMed]
- Deng, G.; Yin, M.; Chen, X.; Zeng, F. Clinical determinants for fatality of 44,672 patients with COVID-Crit. Care 2020, 24, 179. [Google Scholar]
- Sanchis-Gomar, F.; Perez-Quilis, C.; Lavie, C.J. Should atrial fibrillation be considered a cardiovascular risk factor for a worse prognosis in COVID-19 patients? Eur. Heart J. 2020, 41, 3092–3093. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Gillies, C.L.; Singh, R.; Singh, A.; Chudasama, Y.; Coles, B.; Seidu, S.; Zaccardi, F.; Davies, M.J.; Khunti, K. Prevalence of co-morbidities and their association with mortality in patients with COVID-19: A systematic review and meta-analysis. Diabetes Obes. Metab. 2020, 22, 1915–1924. [Google Scholar] [CrossRef] [PubMed]
- Iftimie, S.; López-Azcona, A.F.; Vallverdú, I.; Hernández-Flix, S.; de Febrer, G.; Parra, S.; Hernández-Aguilera, A.; Riu, F.; Joven, J.; Andreychuk, N.; et al. First and second waves of coronavirus disease-19: A comparative study in hospitalized patients in Reus, Spain. PLoS ONE 2021, 16, e0248029. [Google Scholar] [CrossRef] [PubMed]
- Cangiano, B.; Fatti, L.M.; Danesi, L.; Gazzano, G.; Croci, M.; Vitale, G.; Gilardini, L.; Bonadonna, S.; Chiodini, I.; Caparello, C.F.; et al. Mortality in an Italian nursing home during COVID-19 pandemic: Correlation with gender, age, ADL, vitamin D supplementation, and limitations of the diagnostic tests. Aging 2020, 12, 24522–24534. [Google Scholar] [CrossRef]
- Rubio-Rivas, M.; Corbella, X.; Mora-Luján, J.M.; Loureiro-Amigo, J.; López Sampalo, A.; Yera Bergua, C.; Esteve Atiénzar, P.J.; Díez García, L.F.; Gonzalez Ferrer, R.; Plaza Canteli, S.; et al. Predicting Clinical Outcome with Phenotypic Clusters in COVID-19 Pneumonia: An Analysis of 12,066 Hospitalized Patients from the Spanish Registry SEMI-COVID-19. J. Clin. Med. 2020, 9, 3488. [Google Scholar] [CrossRef] [PubMed]
- Lippi, G.; Plebani, M. Laboratory abnormalities in patients with COVID-2019 infection. Clin. Chem. Lab. Med. 2020, 58, 1131–1134. [Google Scholar] [CrossRef] [PubMed]
- Rabaan, A.A.; Al-Ahmed, S.H.; Muhammad, J.; Khan, A.; Sule, A.A.; Tirupathi, R.; Al Mutair, A.; Alhumaid, S.; Al-Omari, A.; Dhawan, M.; et al. Role of Inflammatory Cytokines in COVID-19 Patients: A Review on Molecular Mechanisms, Immune Functions, Immunopathology and Immunomodulatory Drugs to Counter Cytokine Storm. Vaccines 2021, 9, 436. [Google Scholar] [CrossRef] [PubMed]
- Matli, K.; Farah, R.; Maalouf, M.; Chamoun, N.; Costanian, C.; Ghanem, G. Role of combining anticoagulant and antiplatelet agents in COVID-19 treatment: A rapid review. Open Heart 2021, 8, 1628. [Google Scholar] [CrossRef]
- Tang, N.; Bai, H.; Chen, X.; Gong, J.; Li, D.; Sun, Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. J. Thromb. Haemost. 2020, 18, 1094–1099. [Google Scholar] [CrossRef] [PubMed]
- Paranjpe, I.; Fuster, V.; Lala, A.; Russak, A.J.; Glicksberg, B.S.; Levin, M.A.; Charney, A.W.; Narula, J.; Fayad, Z.A.; Bagiella, E.; et al. Association of Treatment Dose Anticoagulation with In-Hospital Survival among Hospitalized Patients with COVID-19. J. Am. Coll. Cardiol. 2020, 76, 122–124. [Google Scholar] [CrossRef] [PubMed]
- Billett, H.H.; Reyes-Gil, M.; Szymanski, J.; Ikemura, K.; Stahl, L.R.; Lo, Y.; Rahman, S.; Gonzalez-Lugo, J.D.; Kushnir, M.; Barouqa, M.; et al. Anticoagulation in COVID-19: Effect of Enoxaparin, Heparin, and Apixaban on Mortality. Thromb. Haemost. 2020, 120, 1691–1699. [Google Scholar] [PubMed]
- Hanif, A.; Khan, S.; Mantri, N.; Hanif, S.; Saleh, M.; Alla, Y.; Chinta, S.; Shrestha, N.; Ji, W.; Attwood, K.; et al. Thrombotic complications and anticoagulation in COVID-19 pneumonia: A New York City hospital experience. Ann. Hematol. 2020, 99, 2323–2328. [Google Scholar] [CrossRef]
- Fröhlich, G.M.; Jeschke, E.; Eichler, U.; Thiele, H.; Alhariri, L.; Reinthaler, M.; Kastrati, A.; Leistner, D.M.; Skurk, C.; Landmesser, U.; et al. Impact of oral anticoagulation on clinical outcomes of COVID-19: A nationwide cohort study of hospitalized patients in Germany. Clin. Res. Cardiol. 2021, 110, 1041–1050. [Google Scholar] [CrossRef] [PubMed]
- Bikdeli, B.; Madhavan, M.V.; Jimenez, D.; Chuich, T.; Dreyfus, I.; Driggin, E.; Der Nigoghossian, C.; Ageno, W.; Madjid, M.; Guo, Y.; et al. COVID-19 and Thrombotic or Thromboembolic Disease: Implications for Prevention, Antithrombotic Therapy, and Follow-Up: JACC State-of-the-Art Review. J. Am. Coll. Cardiol. 2020, 75, 2950–2973. [Google Scholar] [CrossRef] [PubMed]
- Corrochano, M.; Acosta-Isaac, R.; Mojal, S.; Miqueleiz, S.; Rodriguez, D.; Quijada-Manuitt, M.Á.; Fraga, E.; Castillo-Ocaña, M.; Amaro-Hosey, K.; Albiol, N.; et al. Impact of pre-admission antithrombotic therapy on disease severity and mortality in patients hospitalized for COVID-19. J. Thromb. Thrombolysis 2022, 53, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Rossi, R.; Coppi, F.; Talarico, M.; Boriani, G. Protective role of chronic treatment with direct oral anticoagulants in elderly patients affected by interstitial pneumonia in COVID-19 era. Eur. J. Intern. Med. 2020, 77, 158–160. [Google Scholar] [CrossRef] [PubMed]
- Saifi, E.S.; Giorgi-Pierfranceschi, M.; Salvetti, M.; Maninetti, L.; Cavalli, I.; Muiesan, M.L. Factors associated with survival in older patients affected by COVID-19: A retrospective cohort study. Arch. Gerontol. Geriatr. 2021, 94, 104349. [Google Scholar] [CrossRef] [PubMed]
- Albani, F.; Fusina, F.; Granato, E.; Capotosto, C.; Ceracchi, C.; Gargaruti, R.; Santangelo, G.; Schiavone, L.; Taranto, M.S.; Tosati, C.; et al. Corticosteroid treatment has no effect on hospital mortality in COVID-19 patients. Sci. Rep. 2021, 11, 1015. [Google Scholar] [CrossRef]
- Monreal, E.; Sainz de la Maza, S.; Natera-Villalba, E.; Beltrán-Corbellini, Á.; Rodríguez-Jorge, F.; Fernández-Velasco, J.I.; Walo-Delgado, P.; Muriel, A.; Zamora, J.; Alonso-Canovas, A.; et al. High versus standard doses of corticosteroids in severe COVID-19: A retrospective cohort study. Eur. J. Clin. Microbiol. Infect. Dis. 2021, 40, 761–769. [Google Scholar] [CrossRef]
- Jeronimo, C.M.P.; Farias, M.E.L.; Val, F.F.A.; Sampaio, V.S.; Alexandre, M.A.A.; Melo, G.C.; Safe, I.P.; Borba, M.G.S.; Netto, R.L.A.; Maciel, A.B.S.; et al. Methylprednisolone as Adjunctive Therapy for Patients Hospitalized With Coronavirus Disease 2019 (COVID-19; Metcovid): A Randomized, Double-blind, Phase IIb, Placebo-controlled Trial. Clin. Infect. Dis. 2021, 72, E373–E381. [Google Scholar] [CrossRef]
- Piniella-Ruiz, E.; Bellver-Álvarez, M.T.; Mestre-Gómez, B.; Escolano-Fernández, B.; Vinat-Prado, S.; Cabezas-Olea, R.; Acedo-Gutiérrez, M.S.; Akasbi-Montalvo, M.; Ryan-Murua, P.; Bustamante-Fermosel, A.; et al. Impact of Systemic Corticosteroids on Mortality in Older Adults with Critical COVID-19 Pneumonia. J. Gerontol.-Ser. A Biol. Sci. Med. Sci. 2021, 76, E127–E132. [Google Scholar] [CrossRef]
- Fadel, R.; Morrison, A.R.; Vahia, A.; Smith, Z.R.; Chaudhry, Z.; Bhargava, P.; Miller, J.; Kenney, R.M.; Alangaden, G.; Ramesh, M.S.; et al. Early Short-Course Corticosteroids in Hospitalized Patients with COVID-19. Clin. Infect. Dis. 2020, 71, 2114–2120. [Google Scholar] [CrossRef] [PubMed]
- Pfizer Zithromax. Available online: https://www.pfizer.com/products/product-detail/zithromax (accessed on 23 July 2022).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).